AMAZING WORLD OF CARBON COMPOUNDS 1 HYDROCARBONSTHE FUNDAMENTAL

AMAZING WORLD OF CARBON COMPOUNDS 1 HYDROCARBONS-THE FUNDAMENTAL ORGANIC COMPOUNDS 2 SATURATED AND UNSATURATED HYDROCARBONS 3 FUNCTIONAL GROUPS IN ORGANIC COMPOUNDS 4 NOMENCLATURE OF ORGANIC COMPOUNDS 5 CHEMICAL PROPERTIES OF ORGANIC COMPOUNDS 6 SOAPS AND DETERGENTS

Presented By…. Mr. Dhumal A. N. (Asst. Teacher, Janata English School, Sanvatsar)

SPECIFIC OBJECTIVES • To recall previous year knowledge about carbon and its allotropes. • To inform about hydrocarbons as source of fuel. • To show carbon can form large number of compounds. • To appreciate how the thousands of organic compounds are classified and specifically named. • To understand how chemical and physical properties of some organic compounds are useful to mankind.

LEARNING OUTPUT • Students will be able to understand importance of organic compounds in day to day life. • Students will get information regarding classification of organic compounds. • Students will acquire knowledge and importance of naming organic compounds systematically. • Students will understand the use of chemical reactions to produce useful products.

INTRODUCTION OUTLINE OF CONTENT ( in short what we are going to learn in this topic) USEFUL PRODUCTS IN DAY- TO-DAY LIFE FUEL, FOOD, SOAPS & DETERGENTS, COSMETICS • Fuel 1) COAL 2) HYDROCARBONS

FUEL • TYPES OF FUELS • SOURCES: CONVENTIONAL & NON CONVENTIONAL CARBON AND ITS DERIVATIVES AS MAIN SOURCE OF FUEL HYDROCARBON AS BETTER SOURCE OF FUEL WE WILL REVISE CARBON

CARBON CONTENT • ALLOTROPES OF CARBON 1 DIAMOND 2 GRAPHITE 3 COAL • ADDITIONAL: 8 FORMS OF CARBON
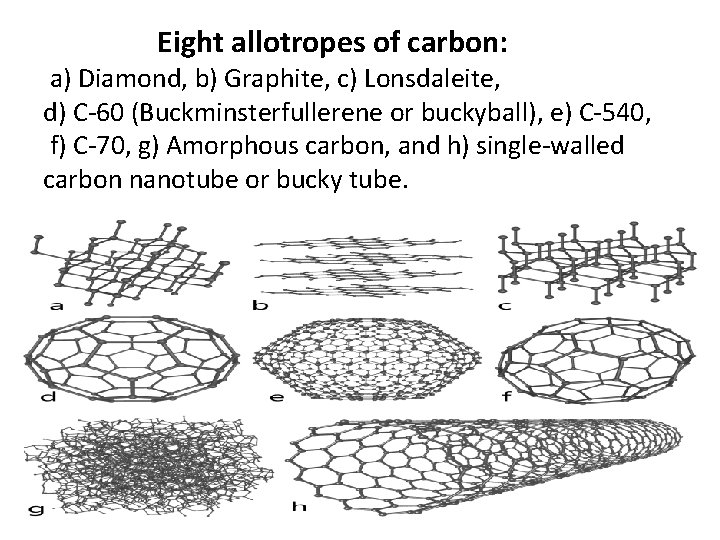
Eight allotropes of carbon: a) Diamond, b) Graphite, c) Lonsdaleite, d) C-60 (Buckminsterfullerene or buckyball), e) C-540, f) C-70, g) Amorphous carbon, and h) single-walled carbon nanotube or bucky tube.

CARBON COMPOUNDS • FORMS COMPOUNDS WITH MANY ELEMENTS • OXYGEN (CO 2), HYDROGEN (CH 4 ) Metals (Ca. C 2 ) • WHY CARBON COMPOUNDS ARE NUMEROUS IN NUMBER? CATENATION VALENCY (COVALENT BOND)
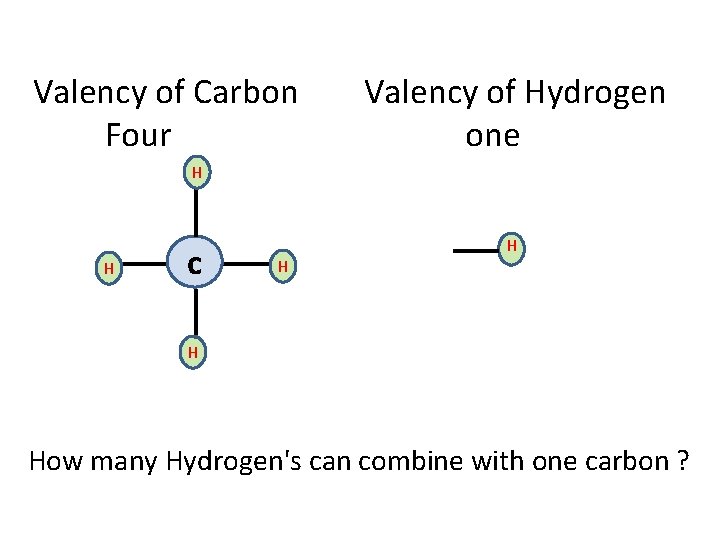
Valency of Carbon Four Valency of Hydrogen one H H c H How many Hydrogen's can combine with one carbon ?

SIMPLEST ORGANIC COMPOUNDS • HYDROCARBONS • SOURCES LPG, CNG, PETROLEUM, BIO GAS • MARSH GAS METHANE
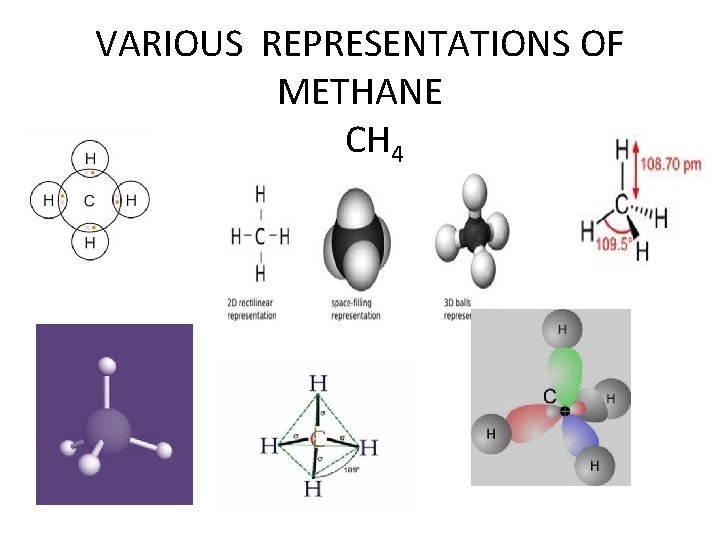
VARIOUS REPRESENTATIONS OF METHANE CH 4
C 2 H 6
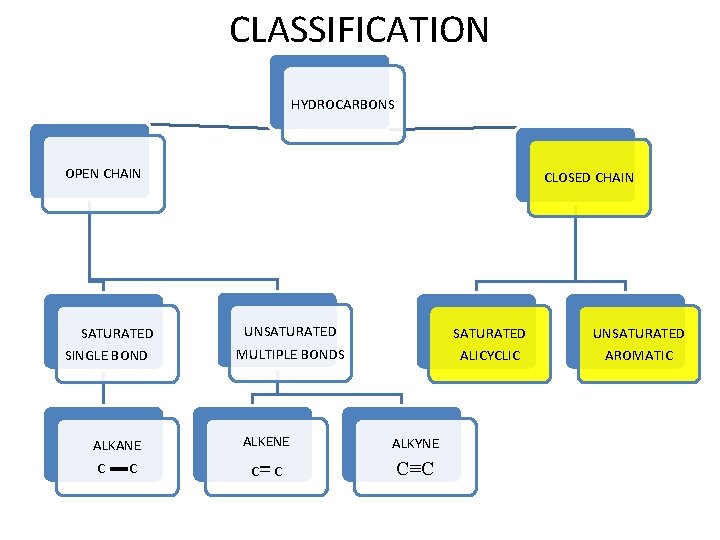
CLASSIFICATION HYDROCARBONS OPEN CHAIN SATURATED SINGLE BOND ALKANE C C CLOSED CHAIN UNSATURATED MULTIPLE BONDS SATURATED ALICYCLIC ALKENE ALKYNE =C C≡C C UNSATURATED AROMATIC

HOMOLOGOUS SERIES • • 1) General formula 2) Similar chemical properties 3) Difference of - CH 24) Gradual changes in physical properties

4 Molecules made of Hydrogen and Carbon 4 Carbon forms four bonds, hydrogen forms one bond 4 Hydrocarbons come in three different homologous series: – Alkanes (single bond between C’s, saturated) – Alkenes (1 double bond between 2 C’s, unsaturated) – Alkynes (1 triple bond between 2 C’s, unsaturated) 4 These are called aliphatic, or open-chain, hydrocarbons

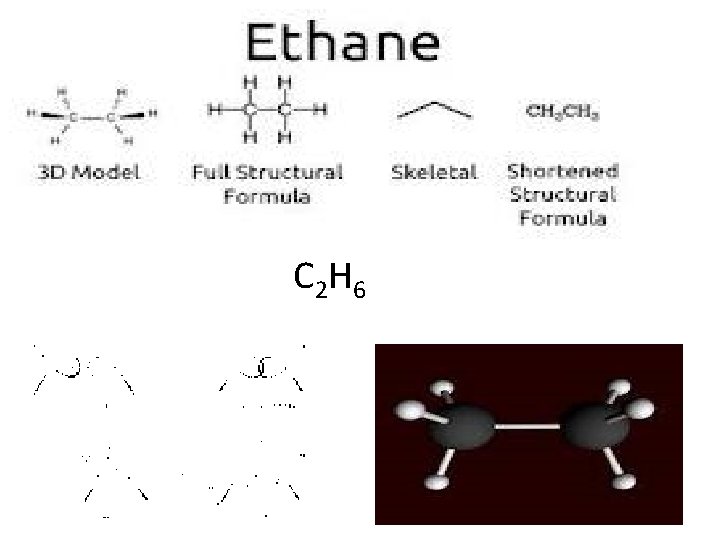
Alkane: names end in ane C n H 2 n+ 2 CH 4 = Methane C 2 H 6 = Ethane C 3 H 8 = Propane C 4 H 10 = Butane C 5 H 12 = Pentane Difference of CH 2 To find the number of hydrogen's, double the number of carbons and add 2
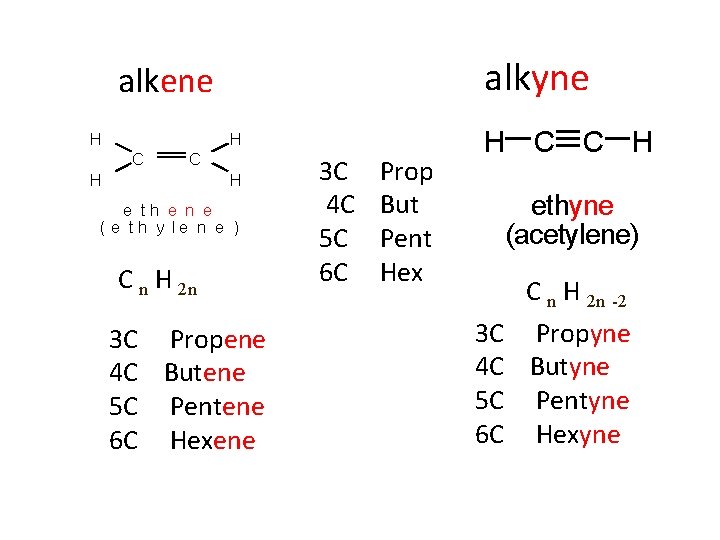
alkyne alkene H H C C H H e th e n e ( e t h y le n e ) C n H 2 n 3 C Propene 4 C Butene 5 C Pentene 6 C Hexene 3 C 4 C 5 C 6 C Prop But Pent Hex H C C H ethyne (acetylene) 3 C 4 C 5 C 6 C C n H 2 n -2 Propyne Butyne Pentyne Hexyne
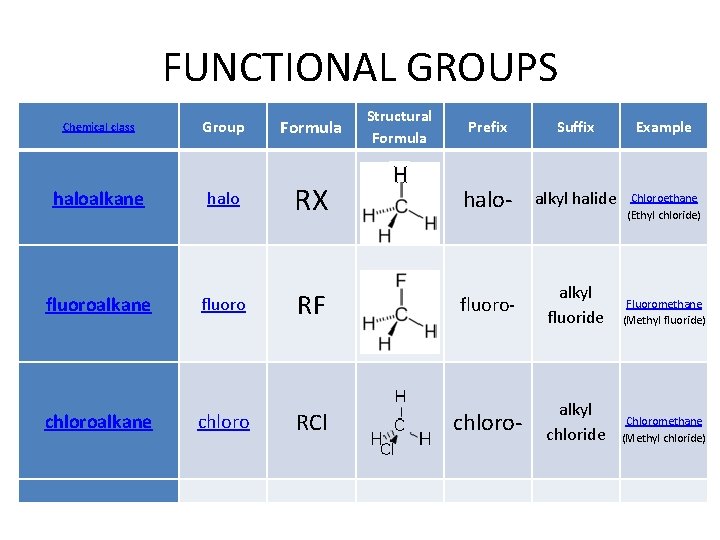
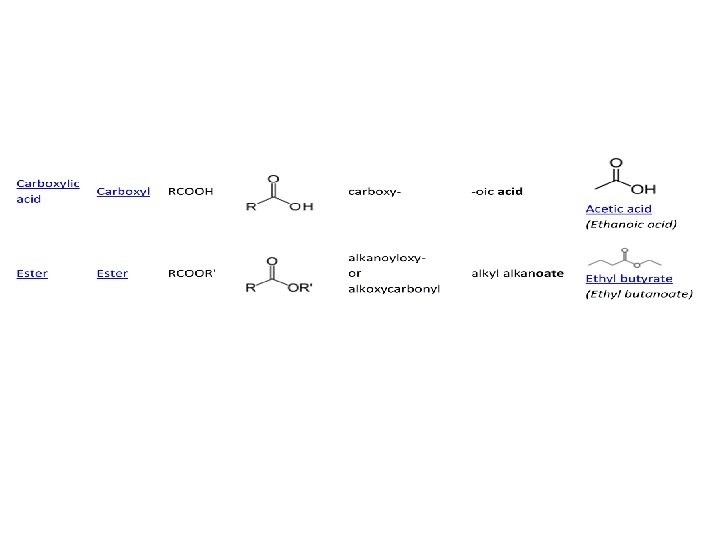
FUNCTIONAL GROUPS Chemical class Group Formula haloalkane halo RX fluoroalkane fluoro RF chloroalkane chloro RCl Structural Formula H Prefix Suffix Example halo- alkyl halide Chloroethane (Ethyl chloride) fluoro- alkyl fluoride Fluoromethane (Methyl fluoride) chloro- alkyl chloride Chloromethane (Methyl chloride)
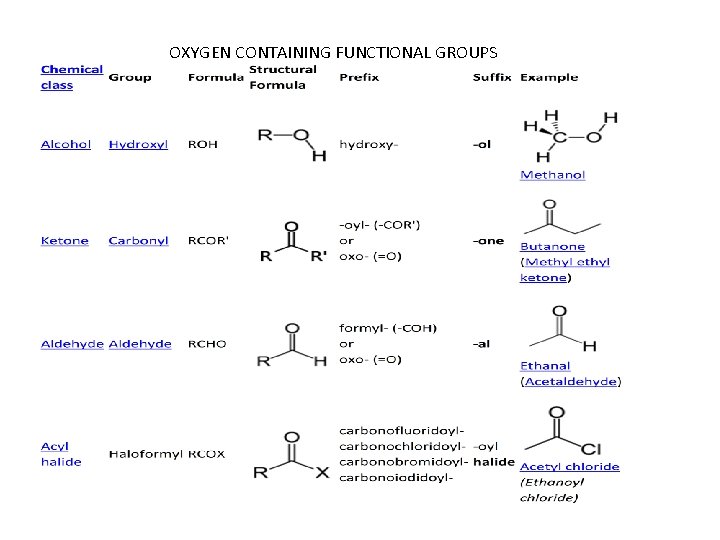
OXYGEN CONTAINING FUNCTIONAL GROUPS
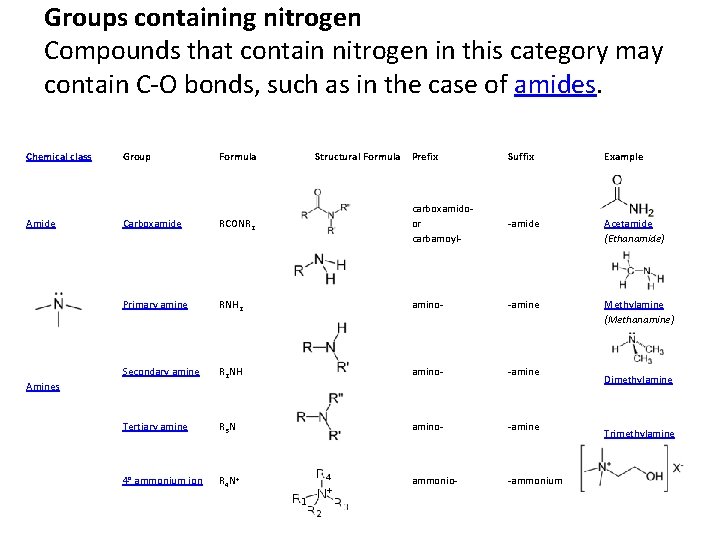
Groups containing nitrogen Compounds that contain nitrogen in this category may contain C-O bonds, such as in the case of amides. Chemical class Group Formula Amide Carboxamide RCONR 2 Primary amine Suffix Example carboxamidoor carbamoyl- -amide Acetamide (Ethanamide) RNH 2 amino- -amine Methylamine (Methanamine) Secondary amine R 2 NH amino- -amine Tertiary amine R 3 N amino- -amine 4° ammonium ion R 4 N+ ammonio- -ammonium Amines Structural Formula Prefix Dimethylamine Trimethylamine Choline

ISOMERISM • DEFINITION • POSSIBLE SKELETON 1) -c 2) -c-c 3) -c-c-c 4) -c-c-c 5) -c-c-c-
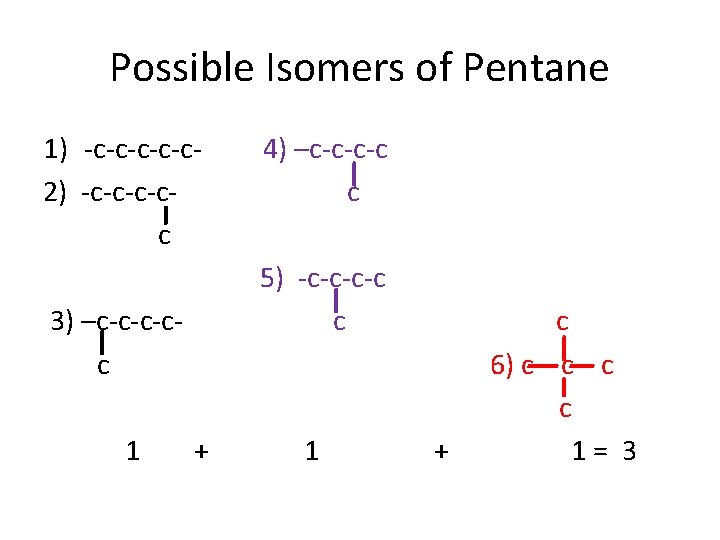
Possible Isomers of Pentane 1) -c-c-c 2) -c-cc 5) -c-c c 3) –c-c-c-cc 1 4) –c-c-c-c c + 1 + c 6) c c 1= 3

NOMENCLATURE 1. Selection of Longest Carbon Chain 2. lowest possible number to side chains

FOR PRACTICE • What are the possible isomers of Hexane? • Write their structures and IUPAC names.

CHEMICAL PROPERTIES OF HYDROCARBONS • SUBSTITUTION • ADDITION • COMBUSTION
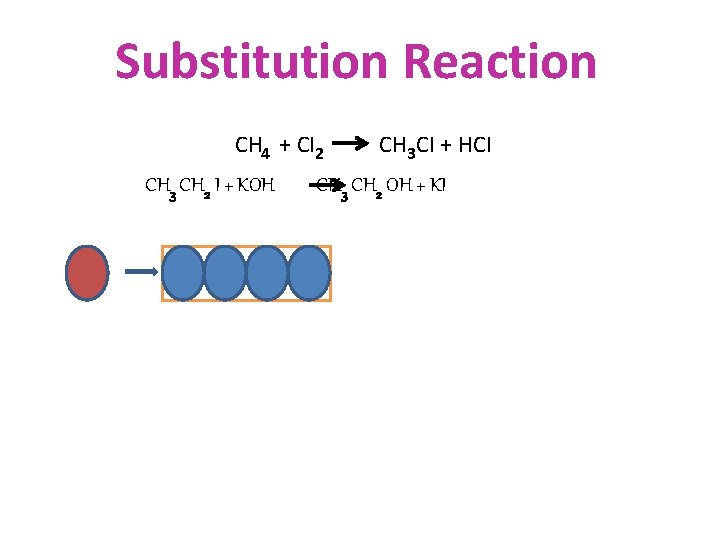
Substitution Reaction CH 4 + CI 2 CH 3 CH 2 I + KOH CH 3 CI + HCI CH 3 CH 2 OH + KI
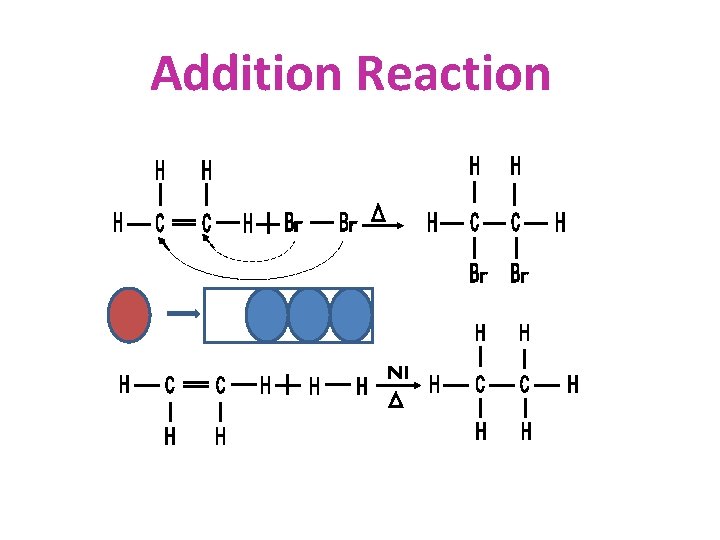
Addition Reaction
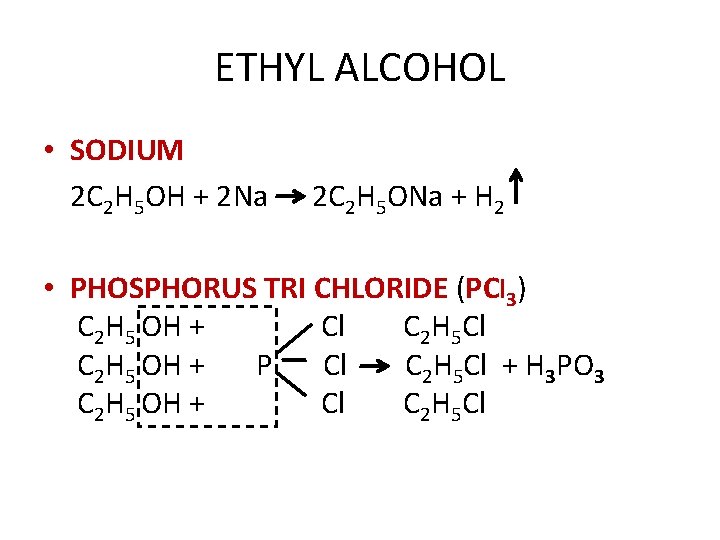
ETHYL ALCOHOL • SODIUM 2 C 2 H 5 OH + 2 Na 2 C 2 H 5 ONa + H 2 • PHOSPHORUS TRI CHLORIDE (PCl 3) C 2 H 5 OH + Cl C 2 H 5 OH + P Cl C 2 H 5 Cl + H 3 PO 3 C 2 H 5 OH + Cl C 2 H 5 Cl


ETHANOIC ACID • HALOGEN CH 3 COOH + CI 2 CH 2 CICOOH + HCI monochloro acetic acid • CH 2 Cl. COOH + CI 2 CHCI 2 COOH + HCI dichloro acetic acid • CHCI 2 COOH + CI 2 CCI 3 COOH + HCI Trichloro acetic acid • METAL 2 CH 3 COOH + 2 Na • ALCOHOL CH 3 COOH + C 2 H 5 OH 2 CH 3 COONa + H 2 Zn. Cl 2 CH 3 COOC 2 H 5 + H 2 O
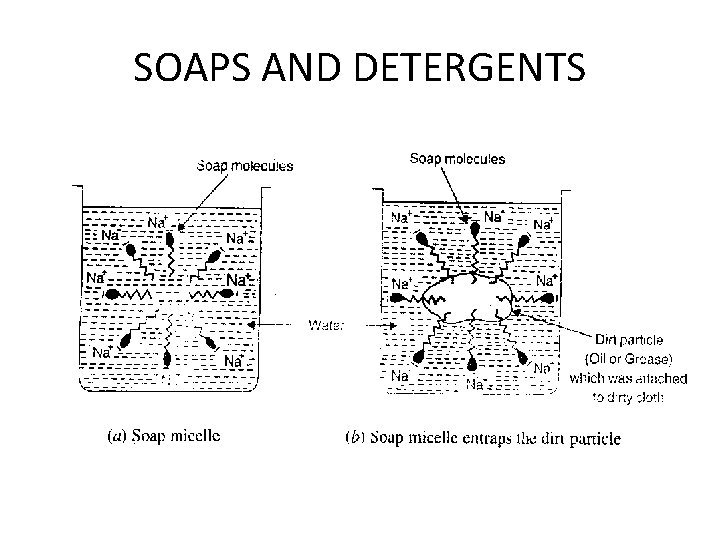
SOAPS AND DETERGENTS
- Slides: 33