ALTOFORNO The largest BFs at present are about
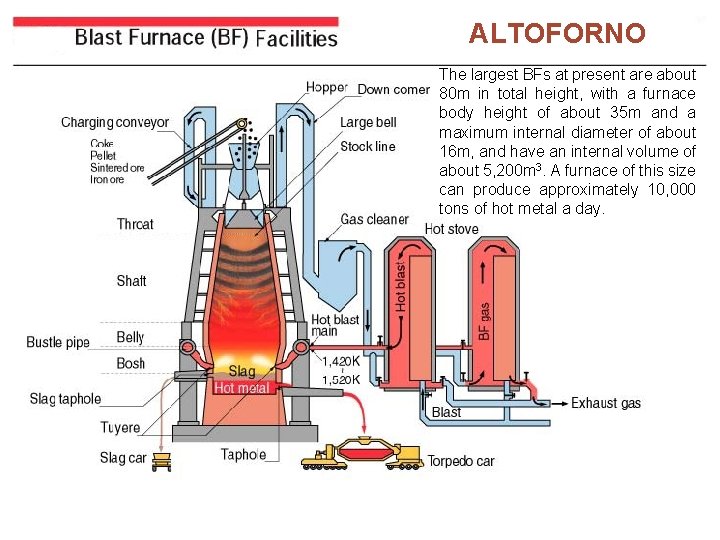
ALTOFORNO The largest BFs at present are about 80 m in total height, with a furnace body height of about 35 m and a maximum internal diameter of about 16 m, and have an internal volume of about 5, 200 m 3. A furnace of this size can produce approximately 10, 000 tons of hot metal a day.

CARICA A total of about 1, 600 kg/ton-hot metal of such iron-bearing materials as sintered ore, lump ore and pellets, and about 380 kg/ton-hot metal of coke as the reductant are charged in alternate layers from the top of the BF. It has recently become common practice to inject usually 90 -120 kg/tonhot metal of pulverized coal as part of the reductant from the tuyeres in the lower part of the furnace. The final raw material in the ironmaking process is limestone used as blast furnace flux . This flux can be pure high calcium limestone, dolomitic limestone containing magnesia or a blend of the two types of limestone. Approximately 1, 000 Nm 3/ton-hot metal of hot blast is also blown through the tuyeres after preheating to 1, 150 -1, 250 C at the hot stoves.
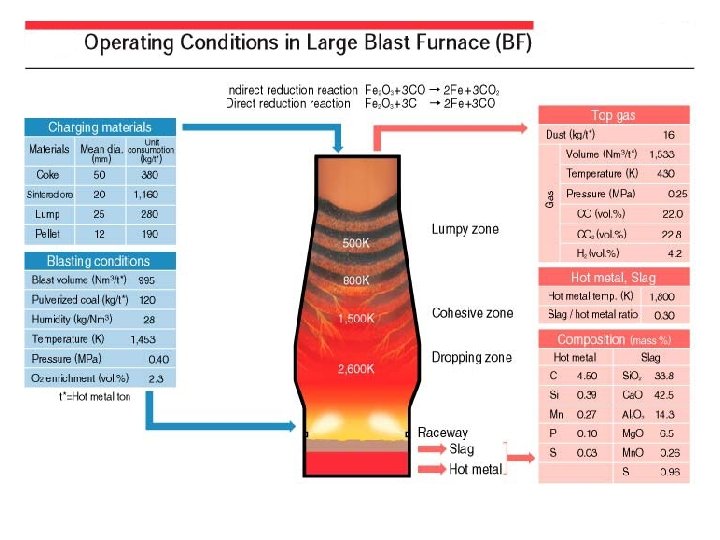

REACTIONS IN BF 1) 3 Fe 2 O 3 + CO = CO 2 + 2 Fe 3 O 4 2) Fe 3 O 4 + CO = CO 2 + 3 Fe. O 3) Fe. O + CO = CO 2 + Fe 4) Fe. O + C = CO +Fe Begins at 450 C Begins at 600 C Begins at 700 C 5)The coke is ignited by hot blast and immediately reacts to generate heat as follows: 6)C + O 2 = CO 2 + Heat 7)Since the reaction takes place in the presence of excess carbon at a high temperature the carbon dioxide is reduced to carbon monoxide as follows: 8)CO 2+ C = 2 CO 9)The product of this reaction, carbon monoxide, is necessary to reduce the iron ore as seen in the previous iron oxide reactions.

The limestone descends in the blast furnace and remains a solid while going through its first reaction as follows: Ca. CO 3 = Ca. O + CO 2 This reaction requires energy and starts at about 870 C. The Ca. O formed from this reaction is used to remove sulfur from the iron which is necessary before the hot metal becomes steel. This sulfur removing reaction is: Fe. S + Ca. O + C = Ca. S + Fe. O + CO The Ca. S becomes part of the slag. The slag is also formed from any remaining Silica (Si. O 2), Alumina (Al 2 O 3), Magnesia (Mg. O) or Calcia (Ca. O) that entered with the iron ore, pellets, sinter or coke. The liquid slag then trickles through the coke bed to the bottom of the furnace where it floats on top of the liquid iron since it is less dense.

In summary, the blast furnace is a counter-current realtor where solids descend and gases ascend. A typical hot metal chemistry follows: Iron (Fe) Silicon (Si) Sulfur (S) Manganese (Mn) Phosphorus (P) Titanium (Ti) Carbon (C) = 93. 5 - 95. 0% = 0. 30 - 0. 90% = 0. 025 - 0. 050% = 0. 55 - 0. 75% = 0. 03 - 0. 09% = 0. 02 - 0. 06% = 4. 1 - 4. 4%
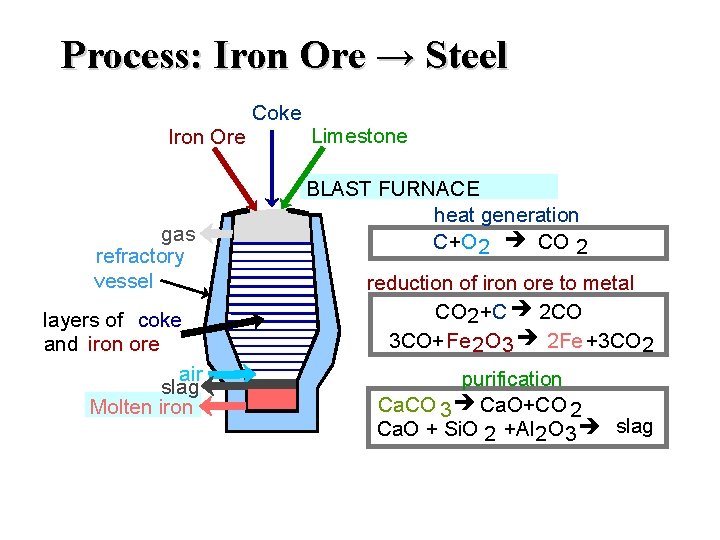
Process: Iron Ore → Steel Coke Iron Ore gas refractory vessel layers of coke and iron ore air slag Molten iron Limestone BLAST FURNACE heat generation C+O 2 CO 2 reduction of iron ore to metal CO 2 +C 2 CO 3 CO+Fe 2 O 3 2 Fe +3 CO 2 purification Ca. CO 3 Ca. O+CO 2 Ca. O + Si. O 2 +Al 2 O 3 slag

Direct-reduced iron (DRI) DRI is obtained when fine ore and lump ore are reduced in a solid state at the relatively low temperature of about 1, 000 C using reformed natural gas. The methods now used include: • FIOR, FINMET and CIRCORED processes and IRON CARBIDE process, all of which reduce fine ore in a fluidized bed; • the HYL-I process, and HYL-II process, which use a retort bed, • the Midrex process and the HYL-III process, which use a countercurrent shaft furnace to reduce pellets and lump ore, and others. Of these, the Midrex, HYL-I and HYL-III processes have been successfully industrialized in large scale production. The Midrex and HYL-III processes are now most commonly used for direct reduction Production of DRI totaled 31 million tons in 1995.
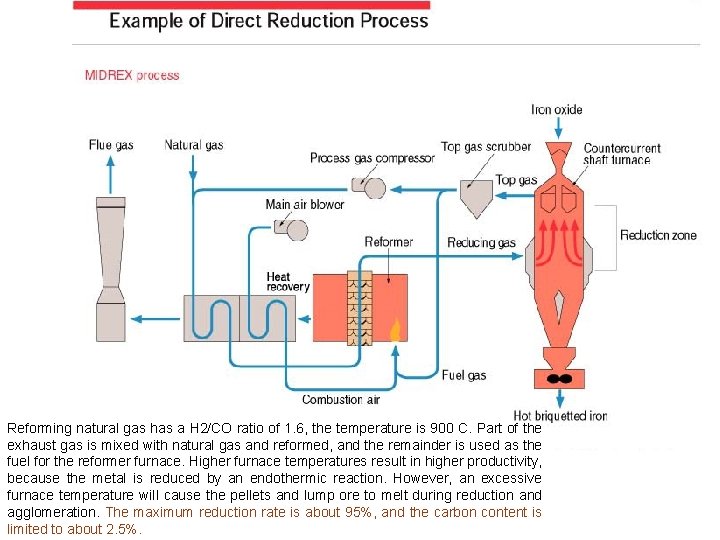
Reforming natural gas has a H 2/CO ratio of 1. 6, the temperature is 900 C. Part of the exhaust gas is mixed with natural gas and reformed, and the remainder is used as the fuel for the reformer furnace. Higher furnace temperatures result in higher productivity, because the metal is reduced by an endothermic reaction. However, an excessive furnace temperature will cause the pellets and lump ore to melt during reduction and agglomeration. The maximum reduction rate is about 95%, and the carbon content is limited to about 2. 5%.

Plant locations have been confined to places where natural gas is available. Furthermore, the large specific area of the active surface of spongy DRI makes it sensitive to re oxidation and ignition when it comes into contact with air and water, especially sea water. Handling and transportation were therefore difficult and potentially hazardous, making large-volume export unprofitable. As a result, the production of DRI has failed to reach expectations. To overcome this difficulty, a hot-briquetting facility to minimize the specific area by compaction was developed and industrialized, and has been installed in the lower part of the countercurrent shaft furnaces since 1984. According to the statistics of the International Iron and Steel Institute (IISI), the world's DRI production more than tripled from 9. 1 millon tons in 1984 to 31 millon tons in 1995. During this period, the world's hot metal production leveled off at approximately 500 million tons. Consequently, the ratio of DRI production to world hot metal production has increased from 2% to 6%.

Canada Mexico Trinidad and Tobago Argentina Peru Venezuela Egypt Libya South Africa Iran Qatar Saudi Arabia India Questi paesi nel 2012 hanno prodotto l’ 82% di tutto il DRI e’ usato per ora (10 -15%) nella carica dei forni elettrici, costa 110 -135$/ton

The DRI is intrinsically more energy efficient than the blast furnace because it operates at a lower temperature, and there are several other factors which make it economical: (1) DRI has about the same iron content as Pig Iron, typically 90 – 94% total iron, so it is an excellent feedstock for the electric furnaces (2) Hot-briquetted iron (HBI) is a compacted form of DRI designed for ease of shipping, handling, and storage. (3) The direct reduction process can use natural gas contaminated with inert gases, avoiding the need to remove these gases for other use. (4) In most cases the DRI plant is located near natural gas source as it is more cost effective to ship the ore rather than the gas.

After they have been obtained by reduction, the iron pellets can re-oxidize very rapidly. This re-oxidation occurs if they are heated by air above a certain temperature or if they come into contact with water, including the ambient humidity. The porous structure of the product helps to boost the oxidation. In contact with water, the DRI changes into Fe 2 O 3 by absorbing the O 2 from the water and liberating the hydrogen. The water is the oxidizing agent. The reaction is exothermic and the hydrogen evolved represents a risk of explosion. The ignition temperature is about 125°C. When the temperature in the hold is sufficiently high and the hydrogen content is above its lower explosive limit of about 4% an explosion may occur in the hold of the vessel. The heat given off by the reaction itself can, in turn, stimulate the re-oxidation of the dry pellets and thus set up a chain reaction throughout the cargo which can lead to temperatures, in the open air, of more than 1000°C, high enough to affect the strength of steel.

Problems DRI is highly susceptible to oxidation and rusting if left unprotected, and is normally quickly processed further to steel. The bulk iron can also catch fire since it is pyrophoric. Sponge iron is not useful by itself, but can be processed to create wrought iron. At least two serious casualties involving ships loaded with DRI, both resulting in loss of the vessels and one with loss of lives, have occurred in the last decade; the Ythan (2004) and the deliberate sinking by the French authorities of the Adamandas (2003) which was deliberately with cargo and bunkers onboard
- Slides: 14