ALSAC Eric LADEN Amandine LAIDI Dborah LEMAIRE Laetitia

ALSAC Eric, LADEN Amandine, LAIDIÉ Déborah, LEMAIRE Laetitia March, 20 th 20091 University LILLE 2

Safe Harbor This is an independent study performed by students from the Faculté des Sciences Pharmaceutiques of Lille. The opinions expressed are our own and not necessarily those of Addex pharmaceuticals. 2

INTRODUCTION 3

ADDEX Pharmaceuticals IDENTITY CARD Headquarters : Plans-les-Ouates (Geneve) Switzerland Total employees ( March 2009) : 135 Business : Allosteric Modulators for blockbuster indications Disease focuses : CNS, Metabolic & Inflammation Clinical Status : Phase 2 (ADX 10059 in GERD & Migraine) Corporate Partner : Johnson&Johnson and Merck & Co 4

Addex management team Dr. Vincent MUTEL Mr. Tim DYER Mr. Andre MULLER Chief executive officer, Chief financial officer Chairman Vice Chaiman & Co-founder Dr Charlotte KEYWOOD Chief Medical Officer Dr Sonia POLI Head of non clinical development Dr Emmanuel LE POUL Head of SNC business unit Dr Laurent GALIBERT Head of inflammation businesse unit Mr Laurent MASSUYAU Head of Business Development Dr Jean-Philippe ROCHER Head of Core Chemistry Dr Robert LÜTJENS Head of Core Biology Dr Olivier LOGET Head of Non-Clinical Safety Mr Chris MAGGOS Head of Investor Relations & Communications 5

PART 1 From an Allosteric modulation idea … … to the Addex creation 6

A company initially focused on addiction § F. Conquet, from GSK § Enginereed m. Glu. R 5 KO mice: unresponsive to cocaine! § MPEP reduces cocaine demand by 50% § Market judged too small for GSK § F. Conquet started Addex in 2002 7

From addiction to allosteric modulators § Focus on addiction : cocaine, alcohol, nicotine § Cocaine addiction is a difficult indication with marginal return potential § 2003 : focus on allosteric modulation of m. Glu. R 8
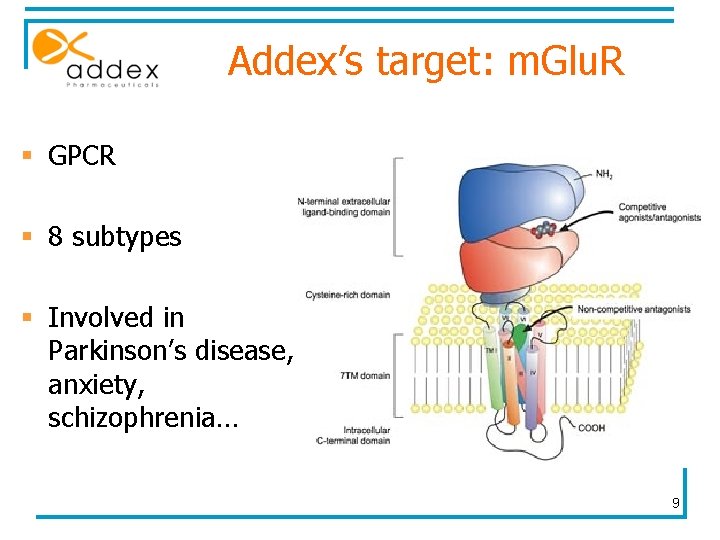
Addex’s target: m. Glu. R § GPCR § 8 subtypes § Involved in Parkinson’s disease, anxiety, schizophrenia… 9

ALLOSTERIC MODULATION AND ITS EFFECTS 10
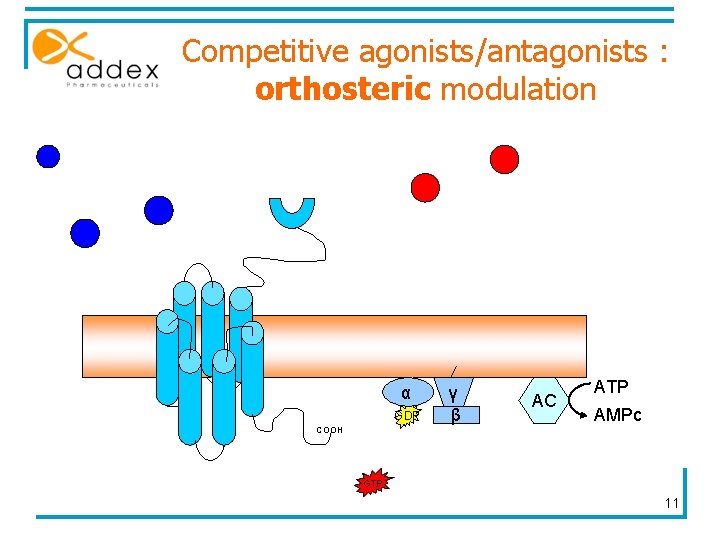
Competitive agonists/antagonists : orthosteric modulation α GDP COOH γ β AC ATP AMPc GTP 11

Competitive agonists/antagonists : limits § Large number of competitive agonists and antagonists identified § Therapeutic use limited by : § Poor bioavailability § Poor CNS penetration § Lack of selectivity among m. Glu. R subtypes 12
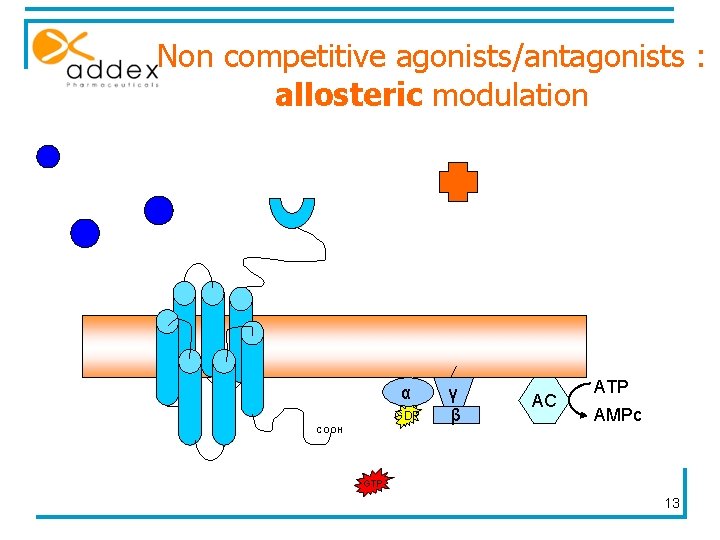
Non competitive agonists/antagonists : allosteric modulation α GDP COOH γ β AC ATP AMPc GTP 13

Allosteric modulators § Interact with sites topographically distinct from the binding site of endogenous ligand § Do not activate receptor § Modulates the binding and/or signalling properties of the orthosteric site § Affinity: increases association rate § Efficacy: change in signalling capacity § Enhance the natural physiological activity of the receptor 14

Allosteric modulators : two types § PAM : § No intrinsic agonism § Increases the effectiveness of the agonist § Increases the affinity of endogenous ligand for the receptor § NAM : § Non competitive antagonists 15

Allosteric modulation : advantages § No competition with endogenous ligands § No activity in the absence of endogenous ligands § Higher sequence difference in allosteric sites : greater selectivity § Ceiling on the effect : less toxicity § Possibility to combine allosteric modulators with orthosteric drug 16
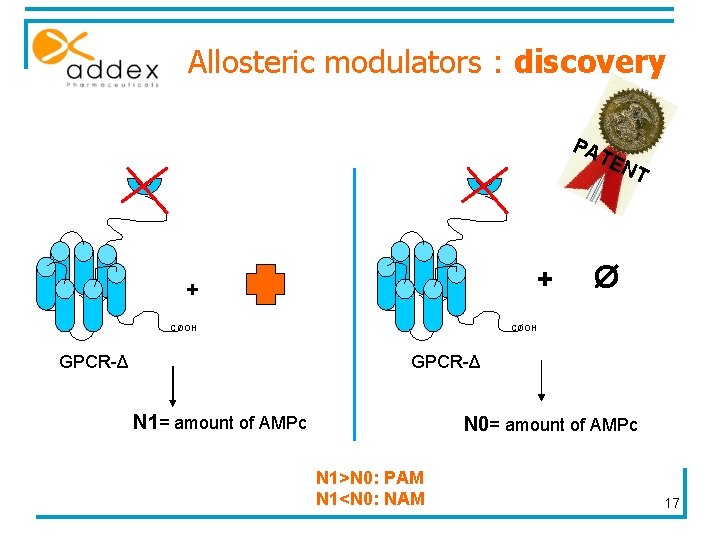
Allosteric modulators : discovery PA TE NT + + COOH GPCR-Δ Ø COOH GPCR-Δ N 1= amount of AMPc N 0= amount of AMPc N 1>N 0: PAM N 1<N 0: NAM 17
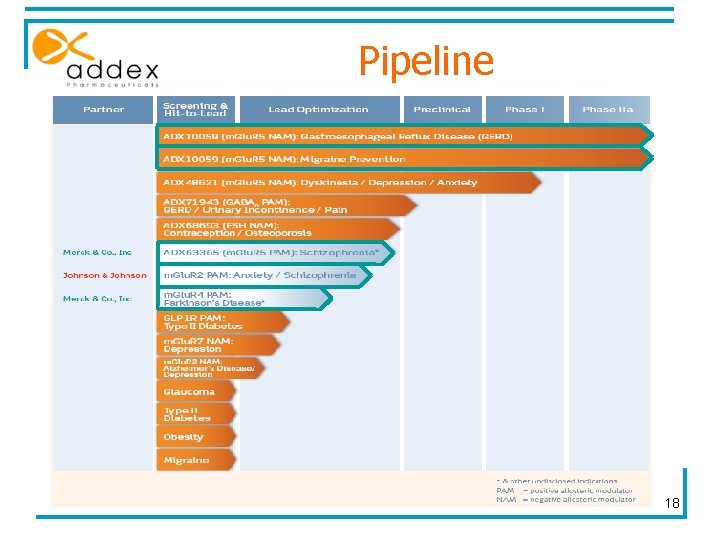
Pipeline 18

HOW WAS THE COMPANY BUILT ? 19
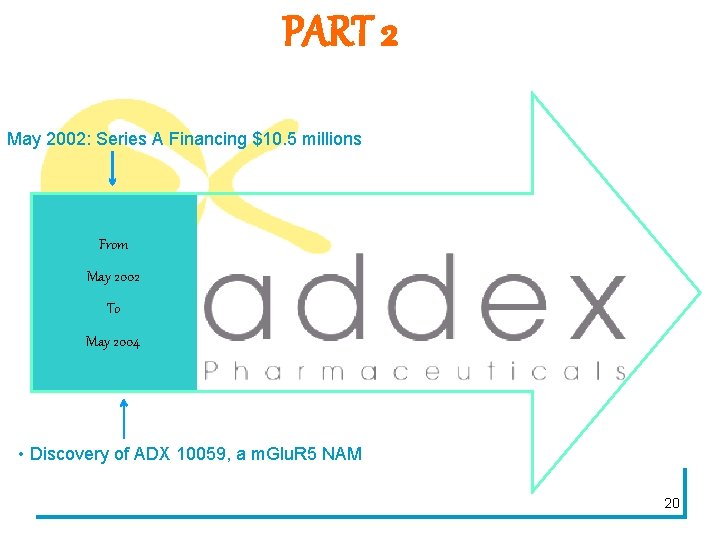
PART 2 May 2002: Series A Financing $10. 5 millions From May 2002 To May 2004 • Discovery of ADX 10059, a m. Glu. R 5 NAM 20

The first round… § July 11, 2002: closing of a CHF 15. 75 million first round (€ 10. 7 million) § Major investors: Index Ventures, Sofinnova Partners, TVM, BCV Initiative Capital and individuals investors § "Addiction is a large under-served market and significant public health issue. Addex Pharmaceuticals is targeting this market with a focus on nicotine, alcohol, cocaine and opiate dependence, " remarked Francois Conquet, CEO of Addex Pharmaceuticals 21 Addexpharma/press-releases-old/news-releases/11 -july-2002

About Index Ventures and Sofinnova Partners § Index Ventures : _ a leading pan-European venture capital. _ Investments in High Technology and Life Science § Sofinnova Partners : _ a leading European venture capital firm. _ In the life science, they have been focused on biopharmaceutical and medical device companies 22
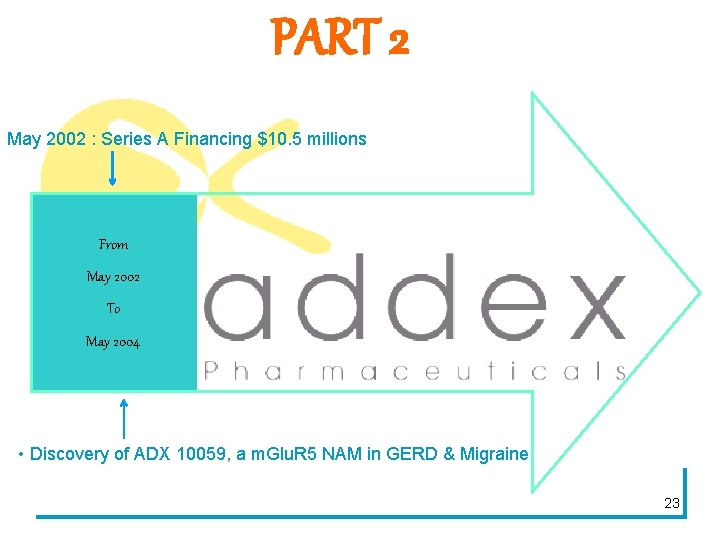
PART 2 May 2002 : Series A Financing $10. 5 millions From May 2002 To May 2004 • Discovery of ADX 10059, a m. Glu. R 5 NAM in GERD & Migraine 23
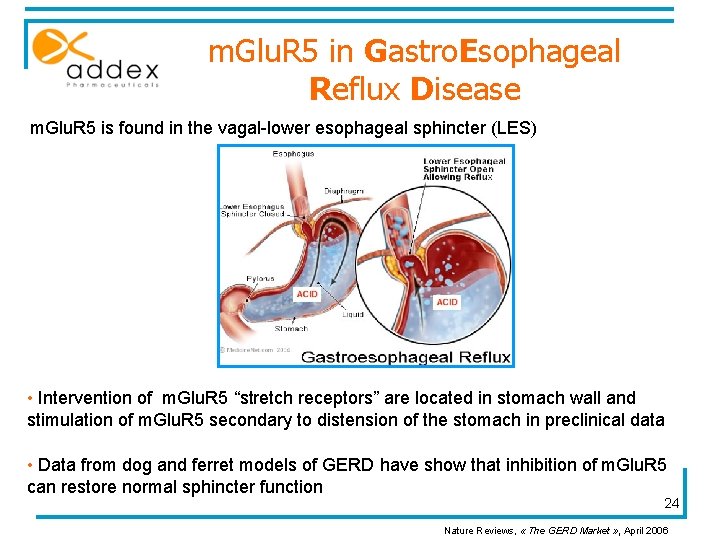
m. Glu. R 5 in Gastro. Esophageal Reflux Disease m. Glu. R 5 is found in the vagal-lower esophageal sphincter (LES) • Intervention of m. Glu. R 5 “stretch receptors” are located in stomach wall and stimulation of m. Glu. R 5 secondary to distension of the stomach in preclinical data • Data from dog and ferret models of GERD have show that inhibition of m. Glu. R 5 can restore normal sphincter function 24 Nature Reviews, « The GERD Market » , April 2006

GERD market § Prevalence in the US: about 15 % of adult population. § GERD market is already in place with: • Antiacids • Histamine Receptor Antagonists • Proton Pump Inhibitors (PPIs) § The unmet medical need in GERD market could still exceed $ 1 billion in the US. 25

WHY MIGRAINE ? • Glutamate believed to important neurotransmitter in migraine circuit. • Blocking m. Glu. R 5 may prevent propagation of signal in the migraine pathway and hence may have utility in migraine prophylaxis. BUT No prior existing animal or human data on the role of m. Glu. R 5 inhibition in migraine. Study objective: to demonstrate whether m. Glu. R 5 inhibition can impact upon the migraine circuit and hence have potential in migraine management. 26
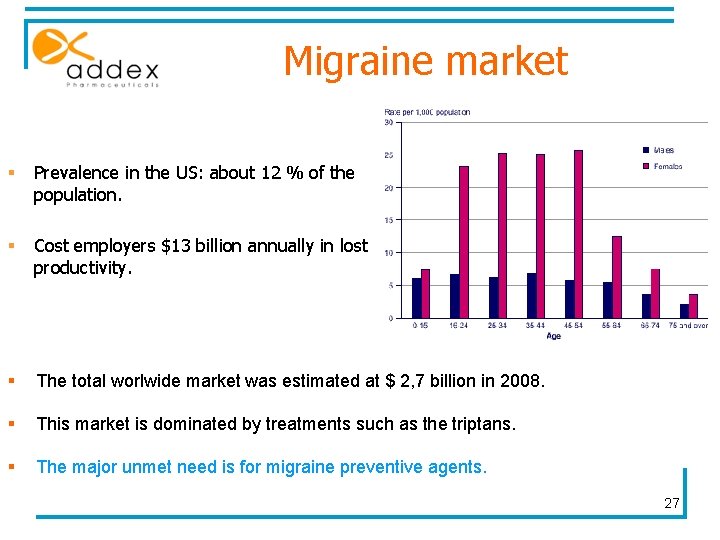
Migraine market § Prevalence in the US: about 12 % of the population. § Cost employers $13 billion annually in lost productivity. § The total worlwide market was estimated at $ 2, 7 billion in 2008. § This market is dominated by treatments such as the triptans. § The major unmet need is for migraine preventive agents. 27
PART 3 May 2004: Series B Financing $39, 0 millions From May 2002 May 2004 To To May 2004 Sept 2006 • Ortho-Mc. Neil (J&J) signs m. Glu. R 2 deal • ADX 10059 Phase 1 trials 28

The second round… § May 17, 2004 : closing of a CHF 50. 5 million (€ 34. 5 million) § Major investors: TVM, Poly. Technos Venture-Partners, Bio*One Capital Pte Ltd, Vinci Capital - Renaissance PME, Initiative Capital, Fulcrum Pharma Development Ltd § "We are delighted to have successfully completed this financing round and view the support of both our new and current investors as a strong endorsement of the team, our strategy and technology. We are now looking forward to developing and commercializing our product portfolio. “, commented Vincent Mutel , CEO of Addex 29 www. addexpharma. com/press-releases-old/news-releases/17 -may-2004/
About Techno Venture Management, Poly. Technos Venture-Partners and Bio*One Capital § TVM: _ a German – U. S. venture capital firm _ funds have made investments in more than 200 technology companies in Europe and the United States § Poly. Technos Venture-Partners: _ an independent European venture capital firm _ focused on early to expansion stage, technology and life science investments § Bio*One Capital: _ the dedicated biomedical investment arm of the Singapore Economic Development Board 30
PART 3 May 2004: Series B Financing $39, 0 millions From May 2002 May 2004 To To May 2004 Sept 2006 • Ortho-Mc. Neil (J&J) signs m. Glu. R 2 deal • ADX 10059 Phase 1 trials 31

Deal with Ortho-Mc. Neil § December , 31 2004: Collaboration and licence agreement with Ortho -Mc. Neil, a Johnson & Johnson company. § Ortho-Mc. Neil acquire an exclusive worldwide license to develop m. Glu. R 2 PAM compounds. § Aim: Discovering and developing Positive Allosteric Modulator (PAM) of metabotropic glutamate receptor 2 (m. Glu. R 2). § The m. Glu. R 2 PAM approach is a clinically validated strategy in both anxiety and schizophrenia. 32
PART 4 Sept 2006: Series C Financing $32, 0 millions From May 2002 May 2004 Sept 2006 To To To May 2004 Sept 2006 May 2007 • ADX 10059 Ph IIa trials for GERD and migraine 33

The third round… § September 19, 2006: closing of a CHF 40. 0 million (€ 27 million) § Major investors: SR One (USA and UK), the venture capital arm of GSK; Roche Venture Fund § "We are now looking forward to driving the commercialisation of our current CNS clinical product portfolio while at the same time exploring selected discovery opportunities more broadly using our expertise in allosteric modulation - an area of drug discovery expertise which is clearly of increasing interest to the major pharmaceutical companies. “ commented Vincent Mutel 34 www. addexpharma. com/press-releases-old/news-releases/19 -september-2006/

About SR One and the Roche Venture Fund § SR One: _ Glaxo. Smith. Kline's corporate healthcare venture capital fund _ invests globally in emerging biotechnology companies that are engaged in drug discovery, development § Roche Venture Fund : _ support innovative technologies and medicines _ manages a portfolio of over 25 companies in 10 countries 35

ADX 10059 IN GERD: PHASE IIA Two-day study, Single-blind (patient), placebo-controlled trial Effect of two doses of ADX 10059 (metabotropic glutamate receptor 5 (m. Glu. R 5) negative allosteric modulator (NAM) ) on 24 hour esophageal p. H in patients with GERD n = 24 Placebo 1 st day ADX 10059 2 nd day Measures of : R E S U L T S • continuous 24 -hour p. H in the lower esophagus • the occurrence of patientrecorded clinical symptoms ½ hr after 1/2 hr after breakfast lunch dinner Data presentation : 17 april 2007 comparison 36
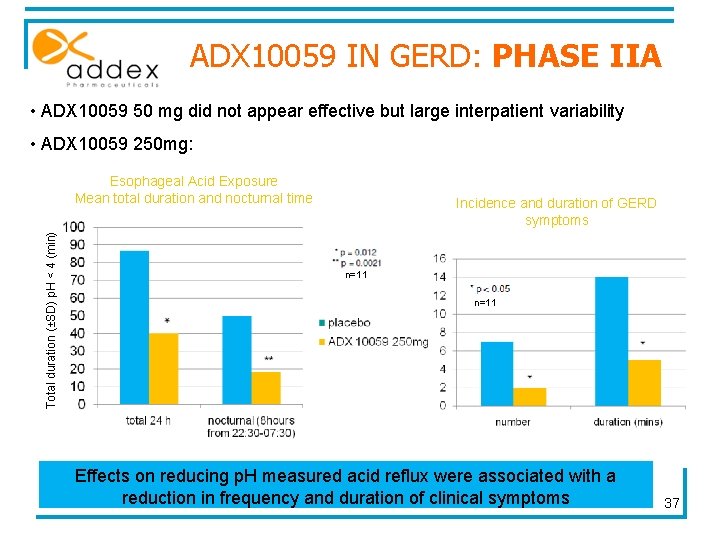
ADX 10059 IN GERD: PHASE IIA • ADX 10059 50 mg did not appear effective but large interpatient variability • ADX 10059 250 mg: Total duration (±SD) p. H < 4 (min) Esophageal Acid Exposure Mean total duration and nocturnal time Incidence and duration of GERD symptoms n=11 Effects on reducing p. H measured acid reflux were associated with a reduction in frequency and duration of clinical symptoms 37

ADX 10059 IN MIGRAINE : PHASE IIA Treatment of a single moderate or severe migraine headache multi-centre, double-blind, placebo-controlled comparison of a single dose of ADX 10059 with placebo sites in the UK and Germany n= 129 ADX 10059 250 mg placebo Data presentation : 20 april 2007 R E S U L T S Measure of : n = 63 n = 66 • the proportion of patients pain-free 2 hours after dosing comparison 38
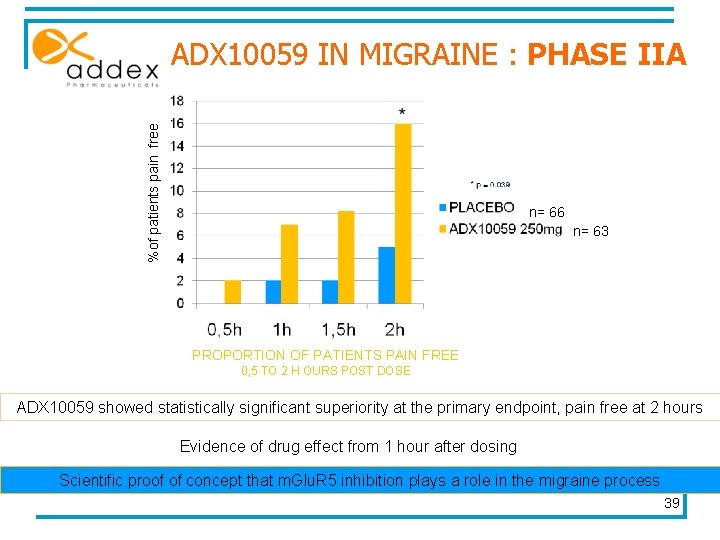
%of patients pain free ADX 10059 IN MIGRAINE : PHASE IIA n= 66 n= 63 PROPORTION OF PATIENTS PAIN FREE 0, 5 TO 2 H OURS POST DOSE ADX 10059 showed statistically significant superiority at the primary endpoint, pain free at 2 hours Evidence of drug effect from 1 hour after dosing Scientific proof of concept that m. Glu. R 5 inhibition plays a role in the migraine process 39

PART 5 • May 2007: Initial Public Offering Swiss Exchange $111 millions From May 2002 May 2004 Sept 2006 May 2007 To To May 2004 Sept 2006 May 2007 Date • ADX 10059 Phase IIB trial for GERD and migraine prevention • Nov. 2007 & Jan. 2008 : License agreement with Merck & Co 40

Initial Public Offering… § May 21, 2007: Addex will raise CHF 136. 9 million (€ 93. 5 million) without the 15% overallotment option § It is one of the most important biotech IPOs worlwide § The shares priced at CHF 73 § Market capitalization: CHF 428. 0 million (€ 292. 3 million) § “This capital increase allows us to pursue our expansion strategy and continue building a sustainable pharmaceutical business around our allosteric modulator platform. ” commented Tim Dyer, CFO and co-founder of Addex 41 Press release May 21 2007

ADX 10059 IN GERD : PHASE IIB 17 December 2008 : Addex announced the start of a Phase IIb trial of ADX 10059 monotherapy double-blind, placebo-controlled, multi-center European n= 90 ADX 10059 120 mg placebo TWICE A DAY 2 weeks Data are expected in 2009 2 weeks R E S U L T S Measure of : • lower esophageal sphincter (LES) function • acid reflux events comparison 42
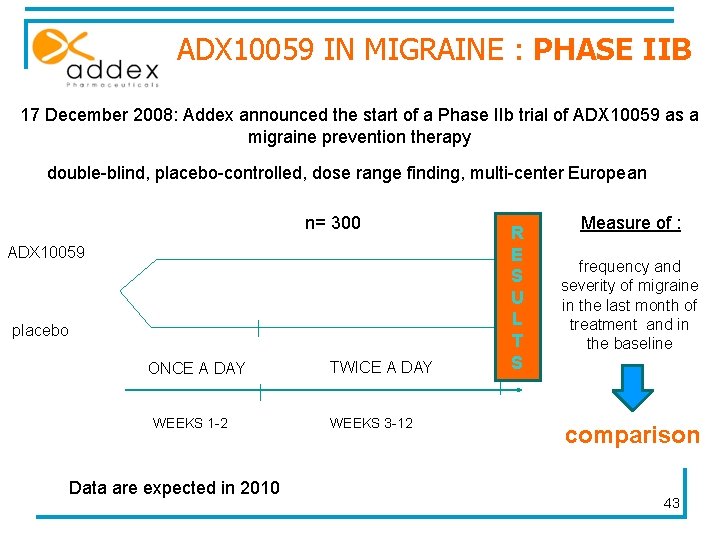
ADX 10059 IN MIGRAINE : PHASE IIB 17 December 2008: Addex announced the start of a Phase IIb trial of ADX 10059 as a migraine prevention therapy double-blind, placebo-controlled, dose range finding, multi-center European n= 300 ADX 10059 placebo ONCE A DAY WEEKS 1 -2 Data are expected in 2010 TWICE A DAY WEEKS 3 -12 R E S U L T S Measure of : frequency and severity of migraine in the last month of treatment and in the baseline comparison 43
PART 5 • May 2007: Initial Public Offering Swiss Exchange $111 millions From May 2002 May 2004 Sept 2006 May 2007 To To May 2004 Sept 2006 May 2007 Date • ADX 10059 Phase IIB trial for GERD and migraine prevention • Nov. 2007 & Jan. 2008 : License agreement with Merck & Co 44

DEALS WITH MERCK PARKINSON’S DISEASE SCHIZOPHRENIA Collaboration to discover and develop m. Glu. R 4 PAM * Collaboration to develop m. Glu. R 5 PAM * : ADX 63365 Merck is responsible for pre-clinical and clinical development Merck is responsible for further development *metabotropic glutamate receptor (m. Glu. R) positive allosteric modulators (PAM) 45

Financial aspects § Up front : $ 3 million § Up front : $22 million § $106. 5 million in milestones - 1 st product in multiple indications § $455 million in milestones - 1 st product in two indications § Additional $61 million milestones - For a 2 nd and 3 rd product § Additional $225 million milestones - For a 2 nd product in two indications § Undisclosed royalties § Option to co-promote in certain EU countries Parkinson’s disease Schizophrenia 46

Why collaborations ? Advantages for MERCK & Co. • Pioneer in research on m. Glu. R receptors and the metabatropic glutamatergic system for multiple indications. • Preclinical validation for m. Glu. R 4 in Parkinson’s disease • Preclinical validation for m. Glu. R 5 in schizophrenia • Y T I IC F I C PE S F KO C A L Clinical development expertise with glutamate receptor modulators Done at Merck 47

Result of collaboration • Glu. R 4 PAM in PD : - m. Glu. R 4 activation triggers a compensatory mechanism, mediated by glutamate, that may spare and/or potentiate the use of dopamine receptor activators; - m. Glu. R 4 activation may have neuroprotective effects that help preserve the brain’s dopaminergic neurons, thus delaying progression of the disease. Nature Reviews Neuroscience, Vol 6, Oct. 2005 • Glu. R 5 PAM in Schizophrenia : Preclinical research shows that activation of m. Glu. R 5 using positive allosteric modulators can act as an antipsychotic and reverse cognitive dysfunction of schizophrenia. Journal of Pharmacology and Experimental Therapeutics, 2005 -Neuroscience 142, 2006 -Psychopharmacology 2004 48

Why collaborations ? Advantages for MERCK & Co. • Pioneer in research on m. Glu. R receptors and the metabatropic glutamatergic system for multiple indications. • Preclinical validation for m. Glu. R 4 in Parkinson’s disease • Preclinical validation for m. Glu. R 5 in schizophrenia • Clinical development expertise with glutamate receptor modulators Done at Merck Aim for ADDEX: mitigate risk through collaborations Schizophrenia and Parkinson’s disease each require large investments & specialized expertise early in development Going it alone significant financial risk for Addex Going it alone slower progress & “learning on the job” 49
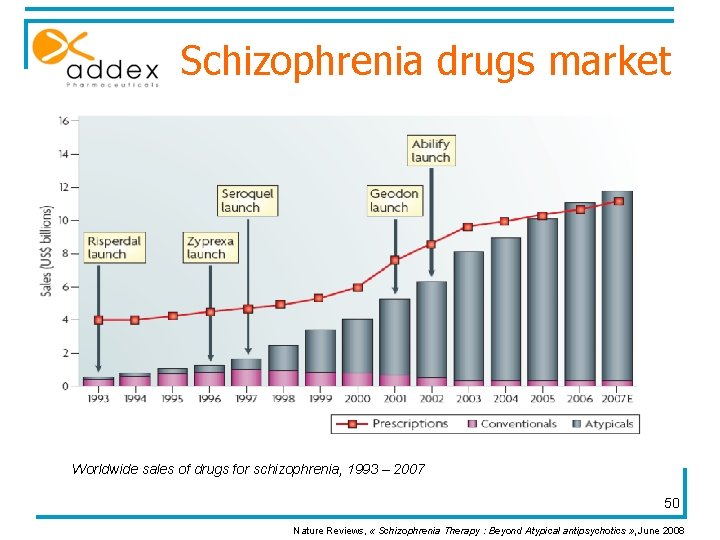
Schizophrenia drugs market Worldwide sales of drugs for schizophrenia, 1993 – 2007 50 Nature Reviews, « Schizophrenia Therapy : Beyond Atypical antipsychotics » , June 2008
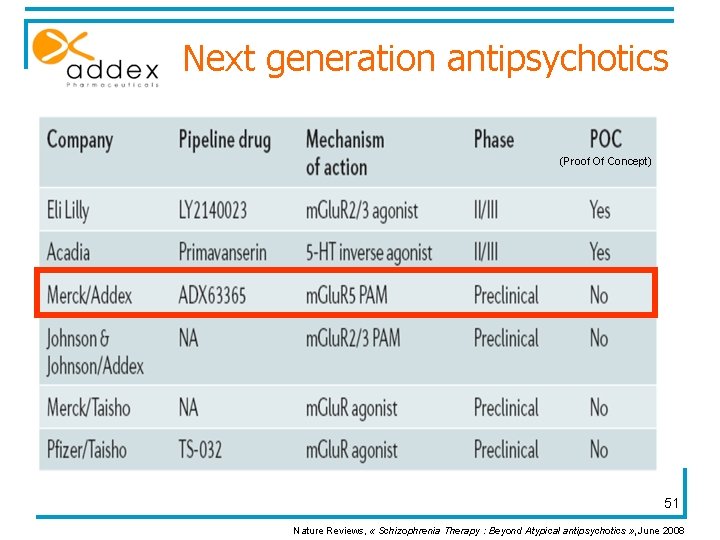
Next generation antipsychotics (Proof Of Concept) 51 Nature Reviews, « Schizophrenia Therapy : Beyond Atypical antipsychotics » , June 2008
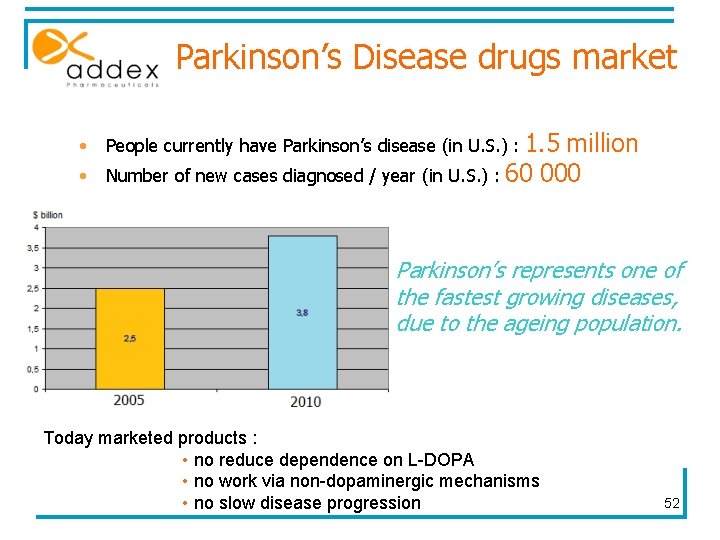
Parkinson’s Disease drugs market • • People currently have Parkinson’s disease (in U. S. ) : 1. 5 million Number of new cases diagnosed / year (in U. S. ) : 60 000 Parkinson’s represents one of the fastest growing diseases, due to the ageing population. Today marketed products : • no reduce dependence on L-DOPA • no work via non-dopaminergic mechanisms • no slow disease progression 52

FINANCIAL ANALYSIS 53
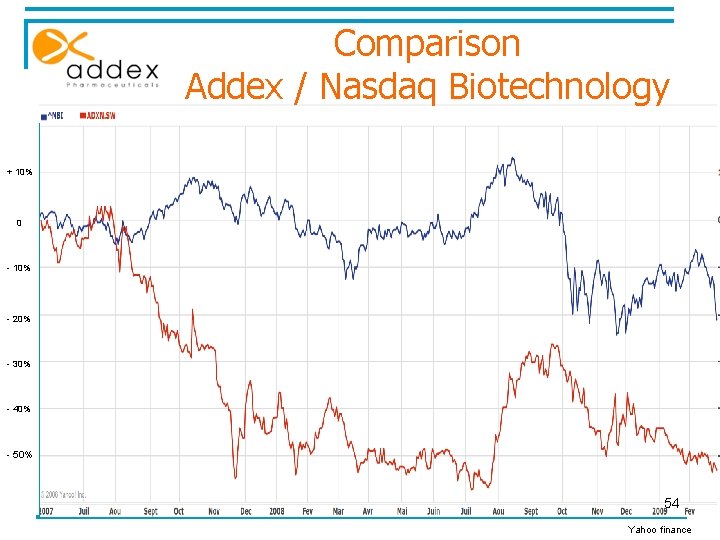
Comparison Addex / Nasdaq Biotechnology + 10% 0 - 10% - 20% - 30% - 40% - 50% 54 Yahoo finance
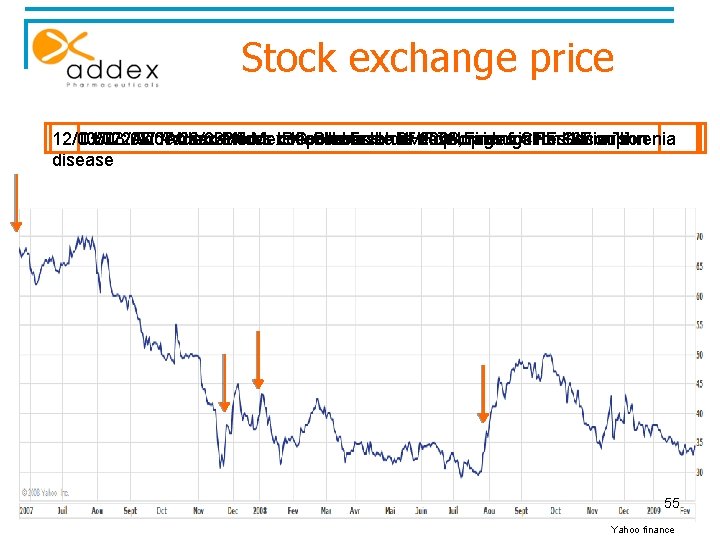
Stock exchange price 12/03/07: 01/03/08: 05/22/07: Addex 07/28/08: Addex andand Prices Merck Addex Merck IPO collaborate reports collaborate Shares first to athalf develop CHF to develop 2008 73, drugs Financial raises drugs for CHF Parkinson’s for results 137 Schizophrenia million disease 55 Yahoo finance

The first half 2008 results § 07/28/08 : Addex reported its first profit in the first half 2008 thanks to a partnership agreement with merck and co § Net profit of CHF 2, 51 million (€ 1, 7 million) § Shares in Addex, listed in Zurich exchange stock, were up to: § § 6, 3% the 28 th July 39, 1% between the 28 th July and the 12 th August. 56
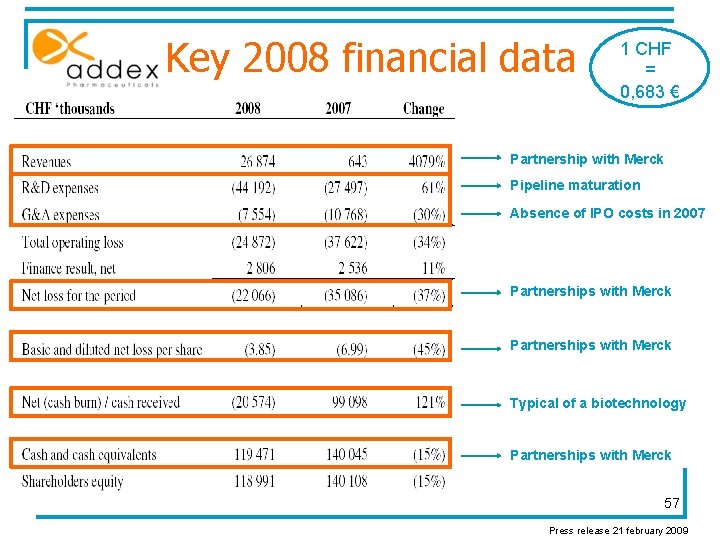
Key 2008 financial data 1 CHF = 0, 683 € Partnership with Merck Pipeline maturation Absence of IPO costs in 2007 Partnerships with Merck Typical of a biotechnology Partnerships with Merck 57 Press release 21 february 2009

2008 financial results “We have CHF 119. 5 million of cash, which should allow us to drive forward our business plan until early 2012 without seeking additional capital. In light of current market conditions, we have undergone a healthy process of project prioritization and revised our 2010 & 2011 growth to give ourselves additional financial flexibility. ” Commented Tim Dyer, CFO “After nearly doubling the size of our company to 135 employees, and considering current market conditions, we believe it is prudent to slow our growth and control costs. While this will reduce the number of new discovery programs that we plan to put into development, it will not compromise the speed or quality of our discovery and development projects, including ADX 10059, ADX 48621 and ADX 71943. ” Commented Vincent Mutel, CEO 58 Press release 21 february 2009

Work or not at Addex ? 59
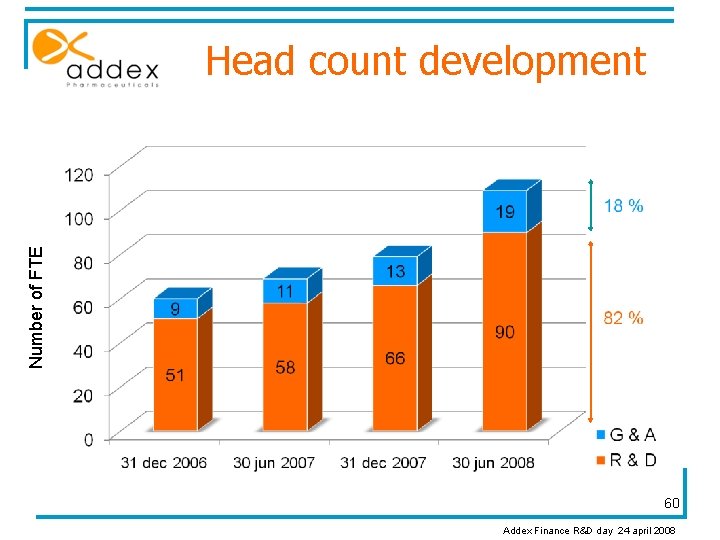
Number of FTE Head count development 60 Addex Finance R&D day 24 april 2008
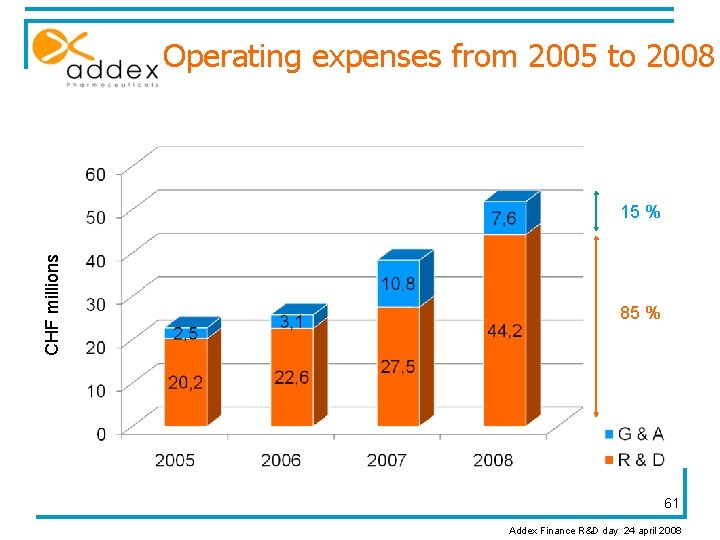
Operating expenses from 2005 to 2008 CHF millions 15 % 85 % 61 Addex Finance R&D day 24 april 2008
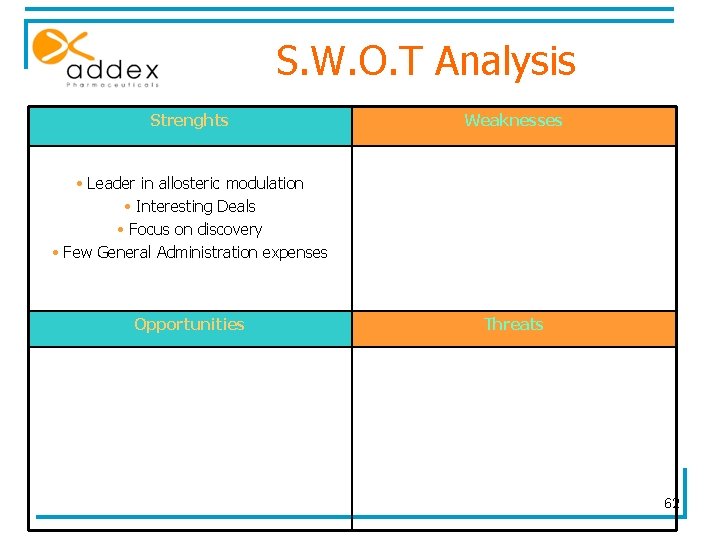
S. W. O. T Analysis Strenghts Weaknesses • Leader in allosteric modulation • Interesting Deals • Focus on discovery • Few General Administration expenses • Only one product for several indications • Future depends on ADX 10059 ph 2 results • Lack of capital Opportunities Threats • First in class • Billion $ markets • Develop new efficient and safe products (Risk aversion) • Big markets • Purchase by a big pharma 62

THANK YOU FOR YOUR ATTENTION 63
- Slides: 63