Alpha Beta Gamma Lesson Contents 1 2 3

Alpha Beta Gamma

Lesson Contents 1. 2. 3. 4. 5. What is Radioactivity? Physical properties of a, b and g Penetrating power of a, b and g N v Z graphs (for fun) Decay laws

Early Pioneers in Radioactivity Rutherford: Villard: Roentgen: Discoverer Discovery of Alpha and Beta gamma rays 1898 rays-1900 1897 The Curies: Discoverers of Radium and Polonium 19001908 Discoverer of Xrays 1895 Becquerel: Discoverer of Radioactivity 1896

What do we mean by Radioactivity? Radioactive decay is the process in which an unstable atomic nucleus loses energy by emitting radiation in the form of particles or electromagnetic waves. The general idea: An unstable nucleus releases energy to become more stable

Radioactivity – Is it a Health Problem? • The Alpha, Beta and Gamma particles all add energy to the body’s tissues. It can alter DNA.
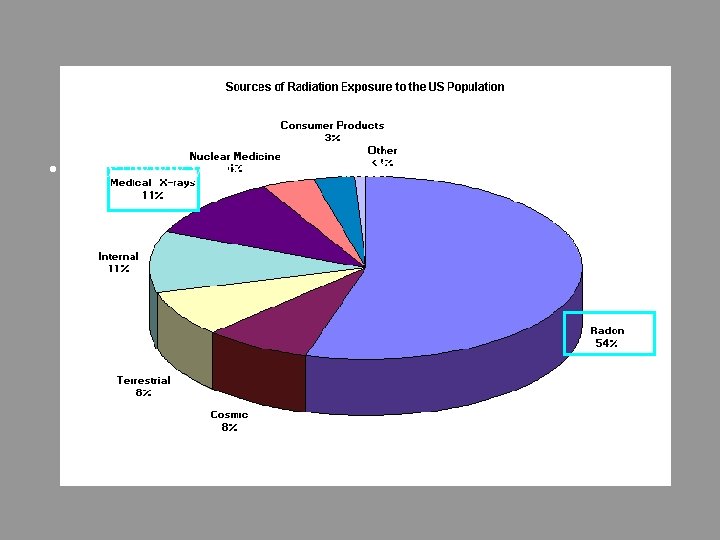
• http: //www. youtube. com/watch? v=5 o. Uago. F_vi. Q
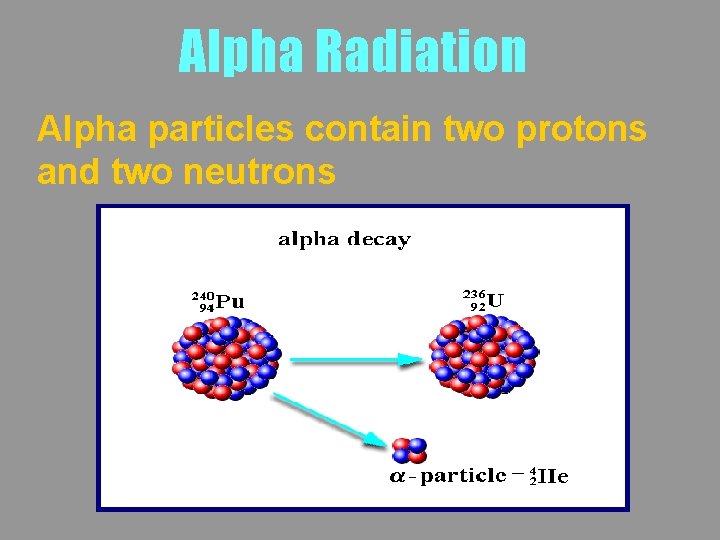
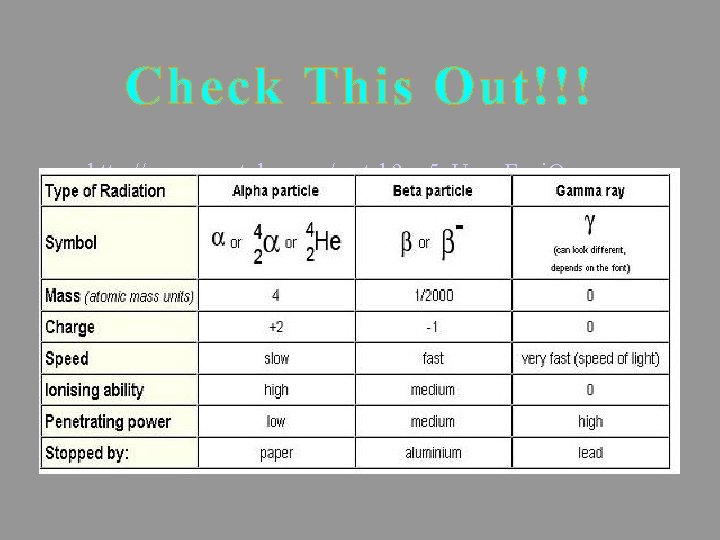
Alpha Radiation Alpha particles contain two protons and two neutrons

Alpha Radiation a has the same constitution as a Helium nucleus Alpha particles may be written as
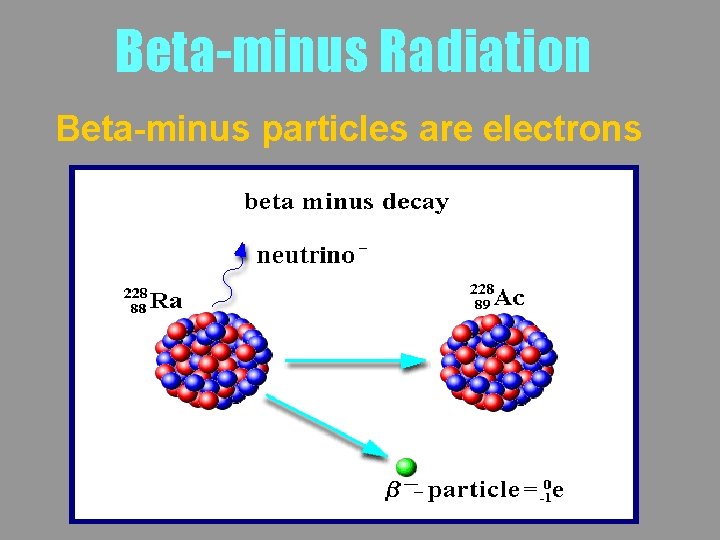
Beta-minus Radiation Beta-minus particles are electrons

Beta-minus Radiation b- is produced when a neutron decays Beta-minus particles may be written as

Beta-minus Radiation b- is produced when a neutron decays The surplus mass is released as kinetic energy in the b- and as an antineutrino

Hulk SMASH!!!! • http: //www. youtube. com/watch? v=jv 95 T 5 b. SYk

Gamma Radiation Gamma rays are a form of electromagnetic radiation

Gamma Radiation g release is often associated with a or b decay Gamma rays remove energy from an unstable nucleus. Has no mass and no charge. O O
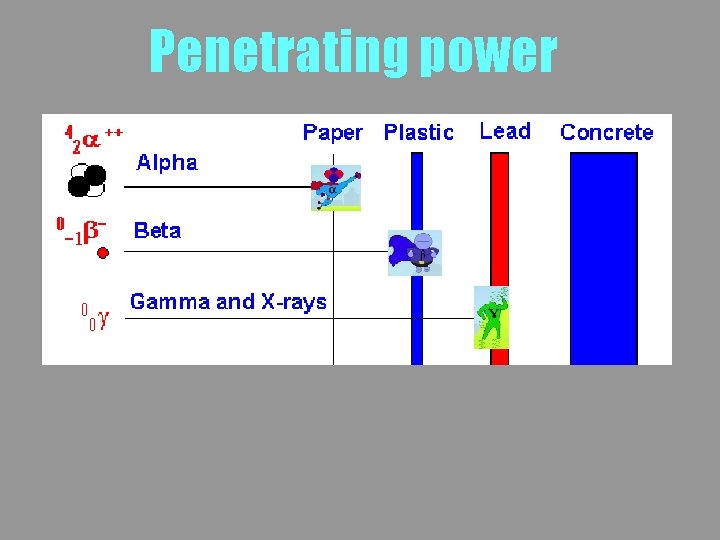
Penetrating power
Check This Out!!! • http: //www. youtube. com/watch? v=5 o. Uago. F_vi. Q
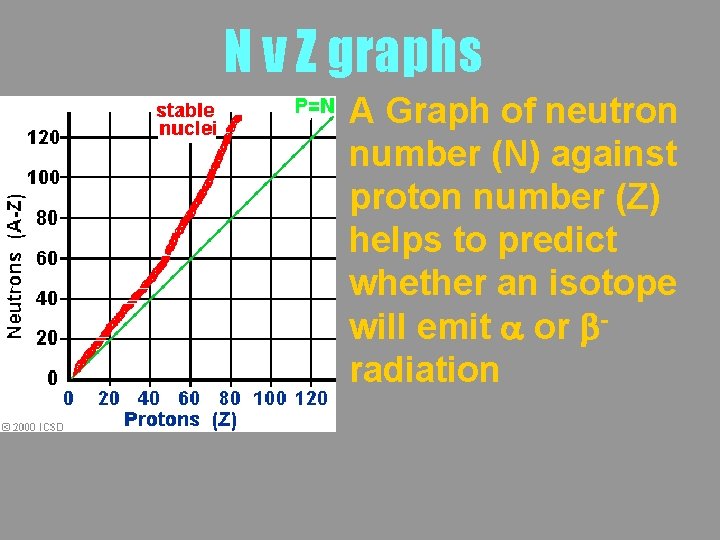
N v Z graphs A Graph of neutron number (N) against proton number (Z) helps to predict whether an isotope will emit a or bradiation
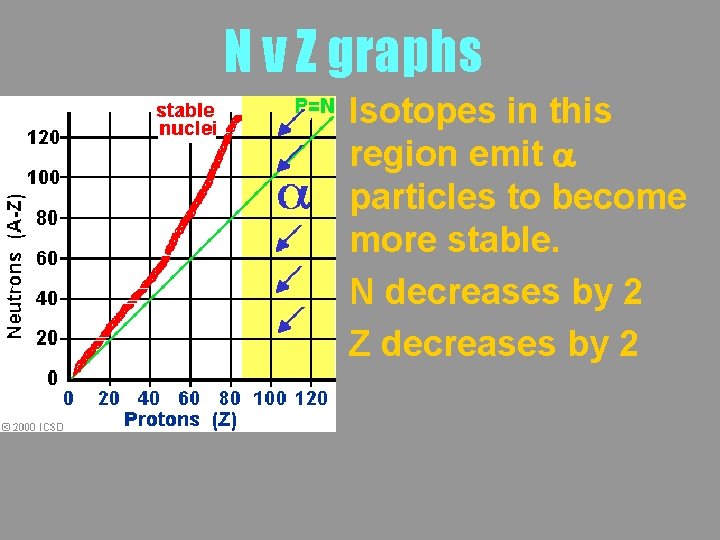
N v Z graphs Isotopes in this region emit a particles to become more stable. N decreases by 2 Z decreases by 2
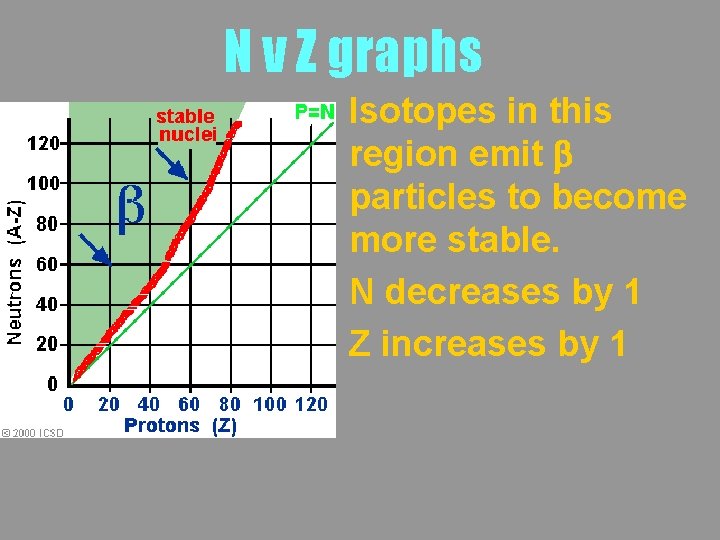
N v Z graphs Isotopes in this region emit b particles to become more stable. N decreases by 1 Z increases by 1

Decay laws - alpha When an isotope emits an a particle v Its nucleon number decreases by 4 v Its proton number decreases by 2 For example:

Decay laws – beta-minus When an isotope emits a b- particle v Its nucleon number is unchanged v Its proton number increases by 1 For example:

Decay laws Try writing the nuclear equations for the decay of these isotopes

Decay laws Answers

Magic Numbers 2, 8, 20, 28, 50, 82, and 126

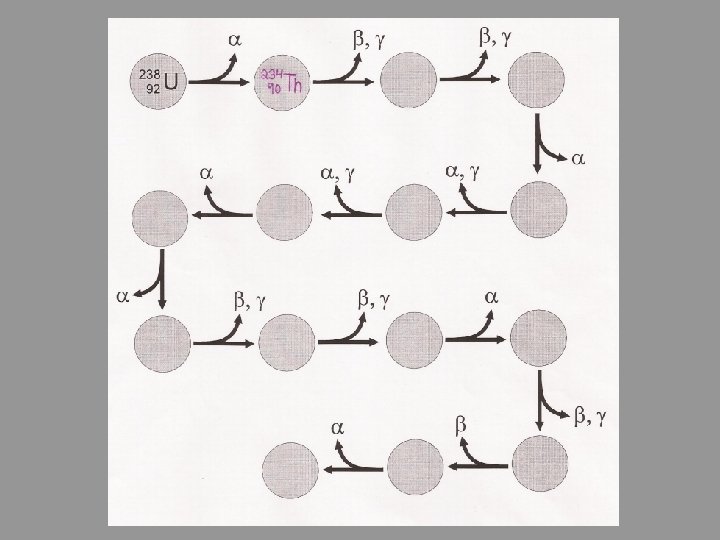
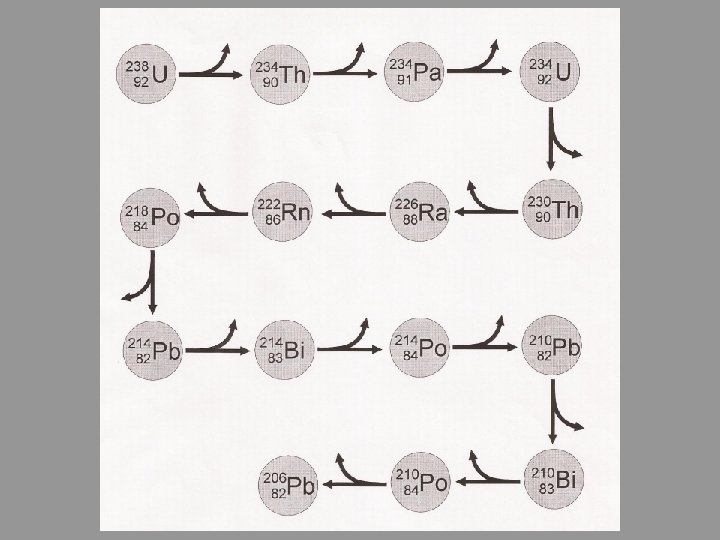
Decay Series • Stability is not usually achieved in a single step • When a radioactive nucleus reaches a stable state by a series of steps, we call it a decay series

Hulk sad the Power. Point is over, Hulk no wanna leave.
- Slides: 28