Alopecia Areata Dr Paul Farrant Consultant Dermatologist Brighton

Alopecia Areata Dr Paul Farrant Consultant Dermatologist Brighton & Sussex University Hospitals Trust, UK

Skin: From immune compromise to autoimmunity Alopecia Areata Review advances in the understanding of skin immunity Gain knowledge in: • Pathogenesis • Presentation • Management

Skin: From immune compromise to autoimmunity Alopecia Areata Review advances in the understanding of skin immunity Gain knowledge in: • Presentation • Pathogenesis • Management

Skin: From immune compromise to autoimmunity Alopecia Areata Review advances in the understanding of skin immunity Gain knowledge in: • Presentation • Pathogenesis • Pathobiology & immune privilege collapse • Genetics • Specific cells and cytokines • Management

Alopecia Areata • Hippocrates created the term alōpekia, which translates as fox mange (alopex= fox) • Areata described in AD 30 by Cornelius Celsus (total loss, and ophiasis = snake pattern) • 1800 s – two thoughts – parasitic and nervous • Later ideas were nerve irritation from defective teeth or eye strain • 20 century – links with endocrine disorders (thyroid), syphilis, poisons

Presentation
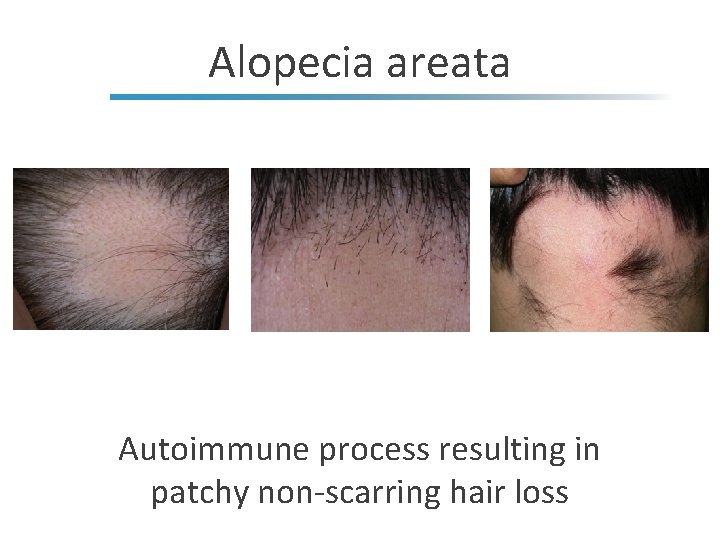
Alopecia areata Autoimmune process resulting in patchy non-scarring hair loss
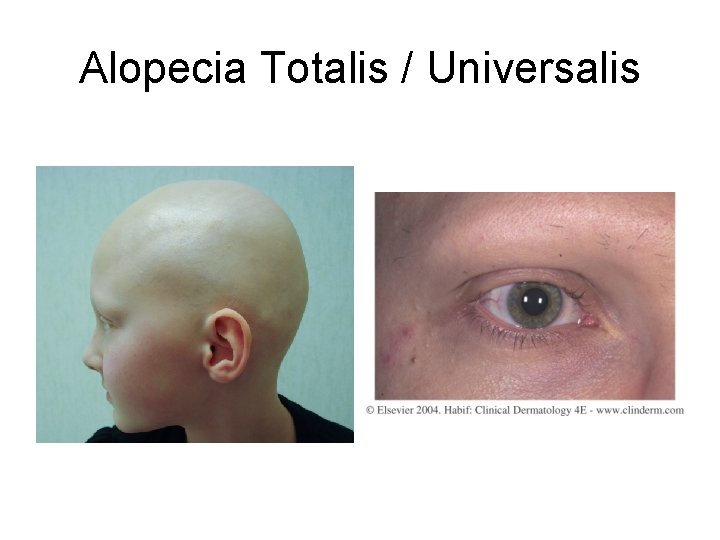
Alopecia Totalis / Universalis
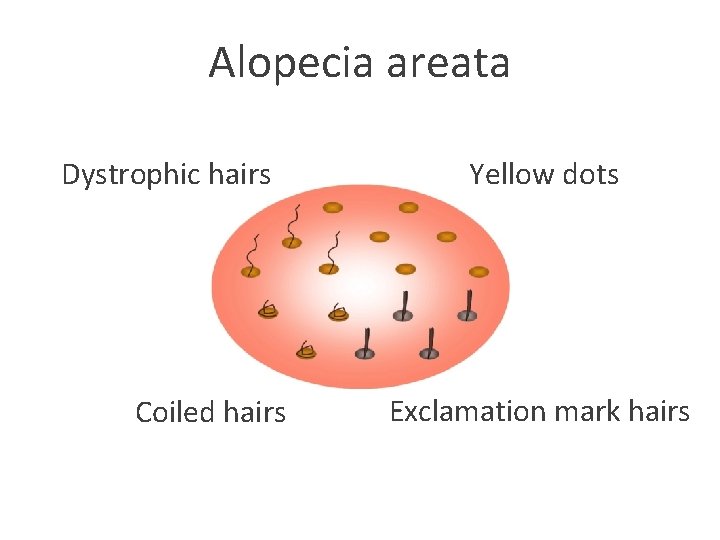
Alopecia areata Dystrophic hairs Coiled hairs Yellow dots Exclamation mark hairs
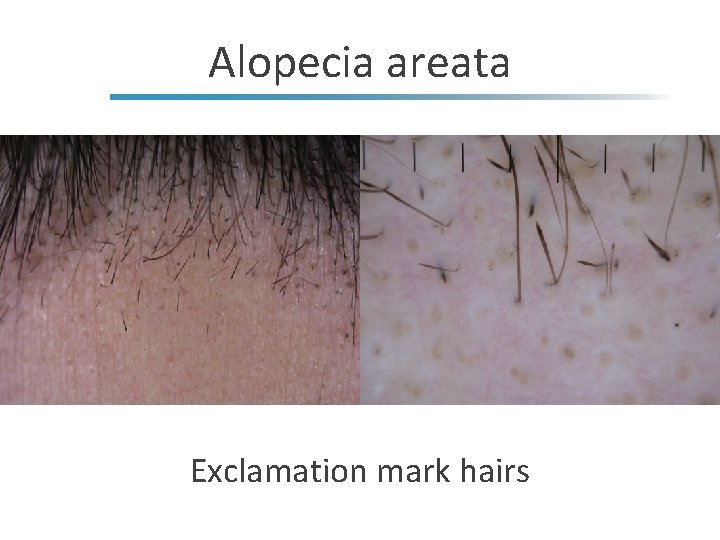
Alopecia areata Exclamation mark hairs

Alopecia areata Cadaverised Hairs

Alopecia Observations • Small patches spread outwards • Can stop when reach a certain size • Attack pigmented hair and spares white > Going white overnight

Alopecia Observations • Worse in Atopics • Ophiasis, Totalis, Universalis, FHx all have worse prognosis • Worse with time over the years • Link with other autoimmune conditions – particularly thyroid • Less than 10% have any obvious trigger

Pathogenesis
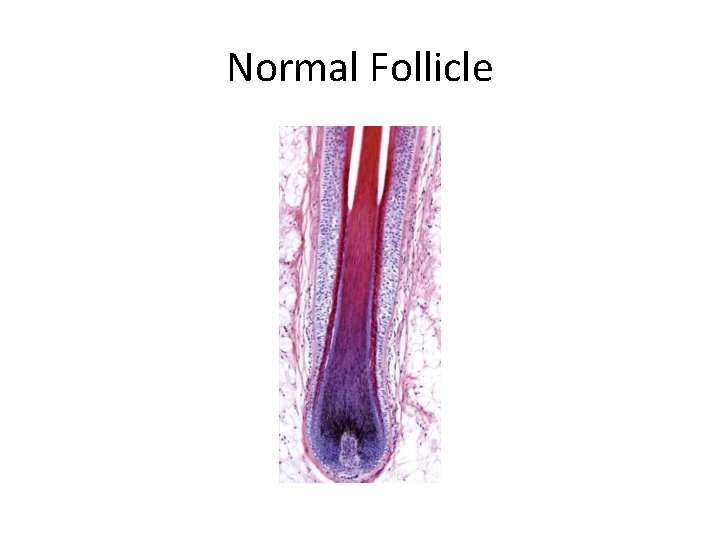
Normal Follicle
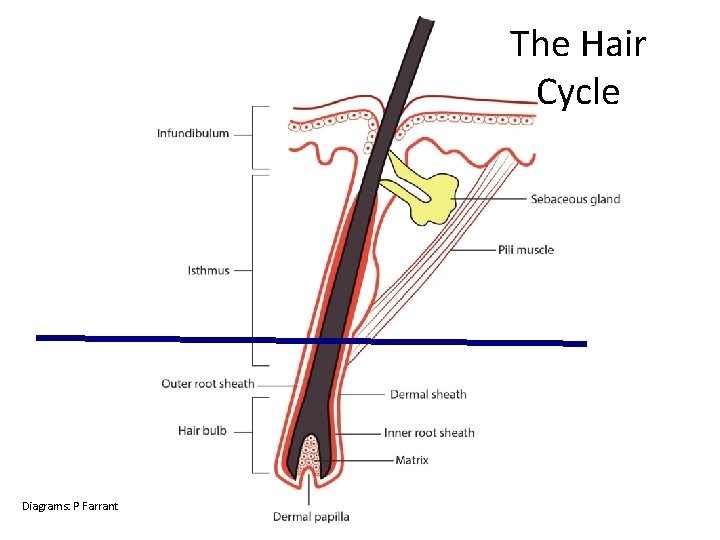
The Hair Cycle Diagrams: P Farrant

Immune Privilege • Suppression of surface molecules required for presenting autoantigens to CD 8 T Cells (MHC class 1 a) • Immunoinhibitory signaling • Decreased MHC 1 puts follicle at risk of NK cells • Down regulation of ligands for activation of NK cell receptors (NKG 2 D) • + factors that inhibit NK and T cells eg TGFb, �MSH, macrophage migration inhibitory factor
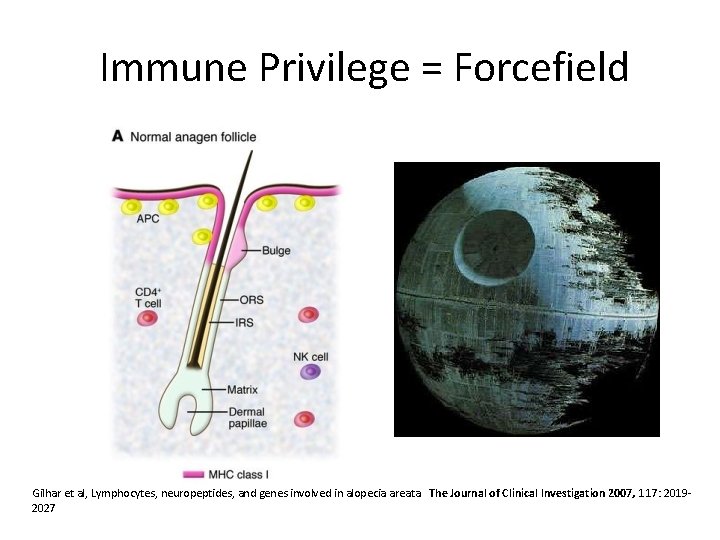
Immune Privilege = Forcefield Gilhar et al, Lymphocytes, neuropeptides, and genes involved in alopecia areata. The Journal of Clinical Investigation 2007, 117: 20192027

Areata – swarm of bees

Alopecia Areata - Pathobiology • Hair cycling disorder • Immune attack around the bulb of Anagen VI follicles leads to exit of anagen to catagen and then telogen • Regression of the follicle • Dystrophy of the follicle – hair is no longer anchored and is shed
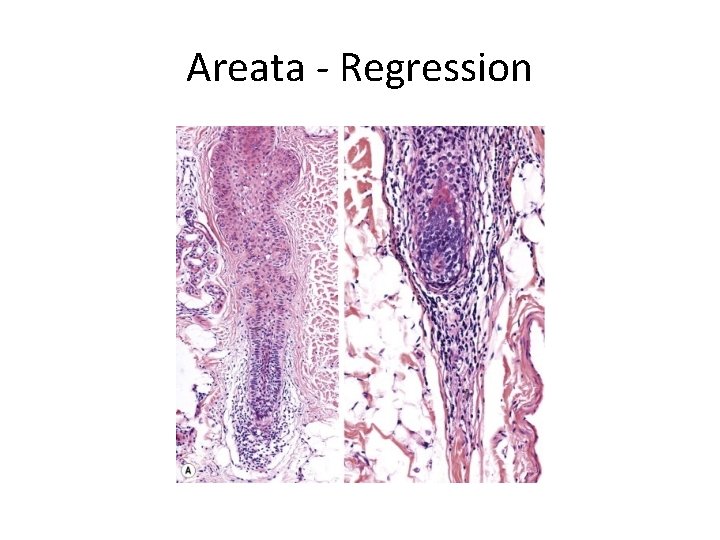
Areata - Regression

Pathobiology • Hair follicle retains potential to regrow and continue cycling • Like other autoimmune conditions – chronic relapsing inflammatory disorder • Suggests cyclic recurrence of disease promoting events
![Immune Privilege collapse • [Decrease in immune privilege guardians] • Pro-inflammatory signals eg IFN Immune Privilege collapse • [Decrease in immune privilege guardians] • Pro-inflammatory signals eg IFN](http://slidetodoc.com/presentation_image_h/bf7d43bec1f8a9d1f28e4919a259285d/image-23.jpg)
Immune Privilege collapse • [Decrease in immune privilege guardians] • Pro-inflammatory signals eg IFN g, Substance P • Upregulations of MHC 1 • Expose follicle associated autoantigens* to CD 8+ T cells • Co-stimulation from CD 4 and Mast cells leads to attack on hair follicle bulb * Autoantigens generated and/or presented only in anagen (eg melanogenesis associated peptides) • Upregulation of MICA (intrafollicular), a NKG 2 D agonist > IFN g • Increase ULBP 3 (peri-follicular), another ligand for NKG 2 D > IFN g • NKG 2 D mediated signalling • Increase in CD 56 and NKG 2 D+ NK cells
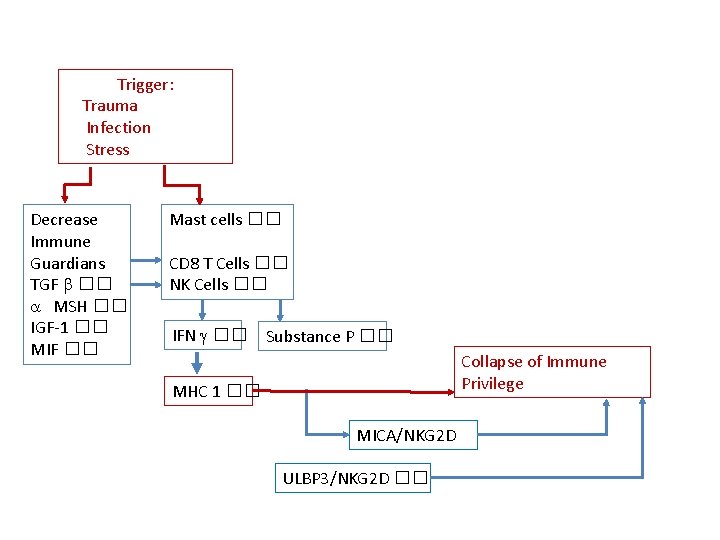
Trigger: Trauma Infection Stress Decrease Immune Guardians TGF b �� a MSH �� IGF-1 �� MIF �� Mast cells �� CD 8 T Cells �� NK Cells �� IFN g �� Substance P �� Collapse of Immune Privilege MHC 1 �� MICA/NKG 2 D ULBP 3/NKG 2 D ��
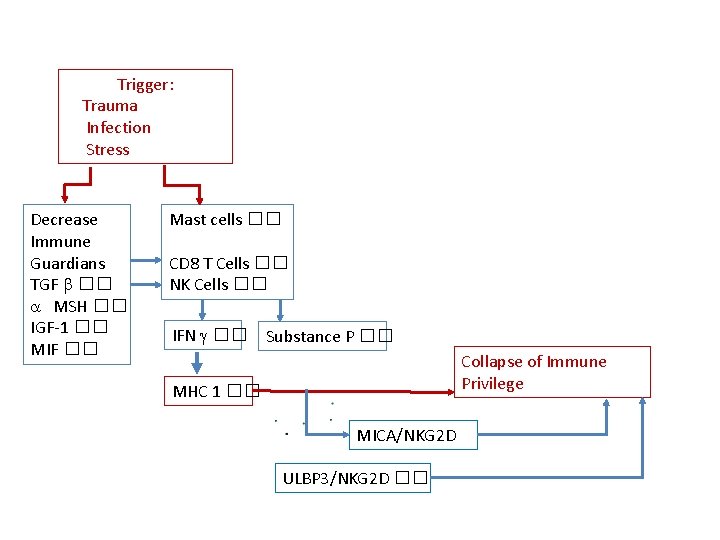
Trigger: Trauma Infection Stress Decrease Immune Guardians TGF b �� a MSH �� IGF-1 �� MIF �� Mast cells �� CD 8 T Cells �� NK Cells �� IFN g �� Substance P �� Collapse of Immune Privilege MHC 1 �� MICA/NKG 2 D ULBP 3/NKG 2 D ��
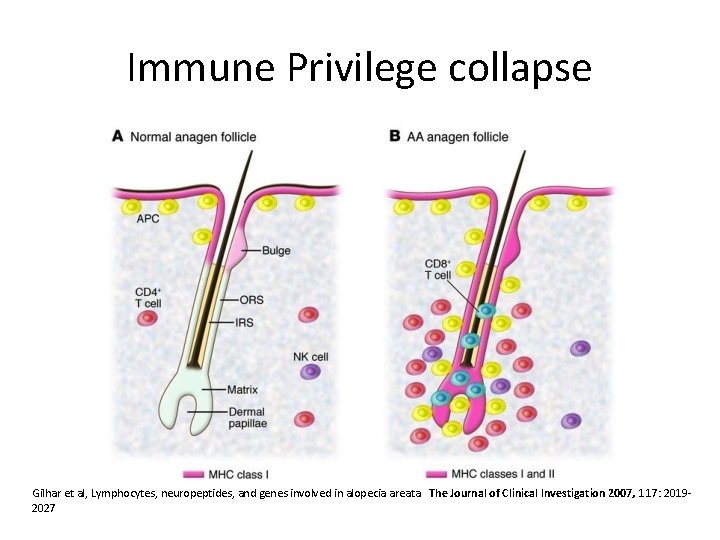
Immune Privilege collapse Gilhar et al, Lymphocytes, neuropeptides, and genes involved in alopecia areata. The Journal of Clinical Investigation 2007, 117: 20192027

Genetic Component in Alopecia Areata • Many patients have family history of AA • High concordance between monozygotic twins (55%) • Fhx of Atopy • Down’s syndrome • Autoimmune polyendocrinpathy-candidiasisectodermal dyrstophy syndrome • Other autoimmune diseases • Ethnic variations

Genetic Component in Alopecia Areata GWAS of of 20 families by Matinez-Mir 2007 showed: • 4 susceptibility loci, Chr 6, 10, 16, 18 • Chr 6 corresponded to the HLA locus • Chr 16 overlapped with susceptibility locus for Crohn’s disease • Chr 18 contains psoriasis-susceptibility locus Martinez-Mir A et al. Genome wide scan for linkage reveals evidence of several susceptibility loci for alopecia areata. Am J Hum Genet 2007; 80: 316 -28

Genetic Component in Alopecia Areata Larger GWAS of 1054 patients, 3278 controls by Petukhova et al • 139 single-nucleotide polymorphisms associated with areata • Reconfirmed Chr 6 p (HLA) • Genes controlling T Cell activation and proliferation – CTLA 4 – IL 2/21 and IL 2 Receptor A • Genes for ULBP which encode ligands for activating NKG 2 D Petukhova et al 2010 Genome-wide association study in alopecia areata implicates both innate and adaptive immunity. Nature 466, 113– 117
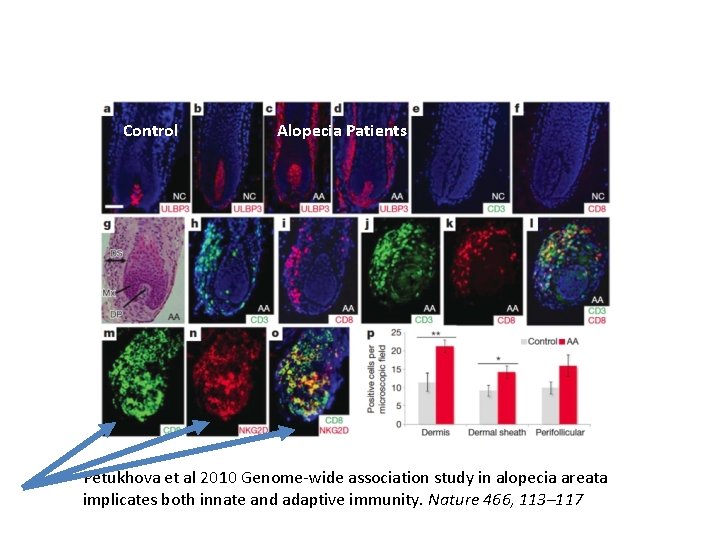
Control Alopecia Patients Petukhova et al 2010 Genome-wide association study in alopecia areata implicates both innate and adaptive immunity. Nature 466, 113– 117

Pathobiology & Genetics combined • Decrease in immunosuppressive signaling, “immune privilege guardians” • Loss of immune privilege, “trigger” in genetically susceptible individuals • Increase in MHC 1 a, presentation of “antigen” • Increase MICA, ULBP 3 and expression of NKG 2 D • CD 8, CD 4, NK cells, Mast cell influx “swarm of bees” • Immune attack leading to hair cycling from anagen to catagen

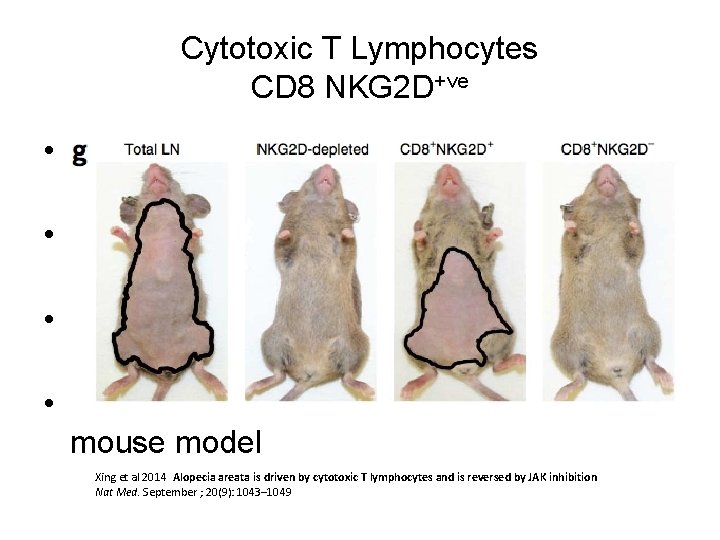
Cytotoxic T Lymphocytes CD 8 NKG 2 D+ve • Subset of T Cells in infiltrate – CD 8 NKG 2 D+ • Upregulation of danger signals ULBP 3 & MICA (two NKG 2 D ligands) by follicle • These cells express high levels of IFN and exhibit cytotoxicity of dermal sheath cells • Transfer of these cells induces alopecia in mouse model Xing et al 2014 Alopecia areata is driven by cytotoxic T lymphocytes and is reversed by JAK inhibition Nat Med. September ; 20(9): 1043– 1049
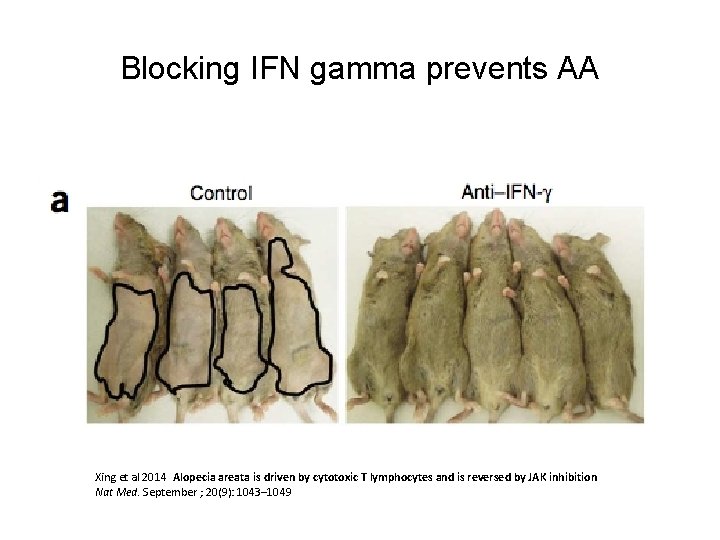
Blocking IFN gamma prevents AA Xing et al 2014 Alopecia areata is driven by cytotoxic T lymphocytes and is reversed by JAK inhibition Nat Med. September ; 20(9): 1043– 1049
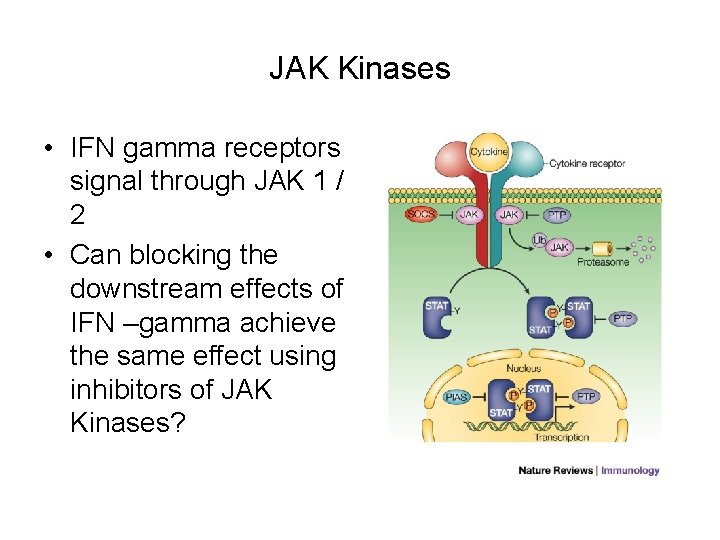
JAK Kinases • IFN gamma receptors signal through JAK 1 / 2 • Can blocking the downstream effects of IFN –gamma achieve the same effect using inhibitors of JAK Kinases?
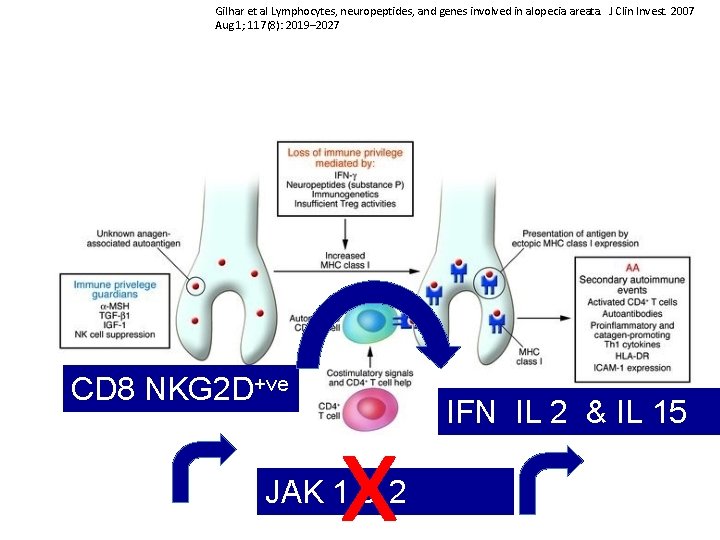
Gilhar et al Lymphocytes, neuropeptides, and genes involved in alopecia areata. J Clin Invest. 2007 Aug 1; 117(8): 2019– 2027 CD 8 NKG 2 D+ve x JAK 1 & 2 IFN IL 2 & IL 15
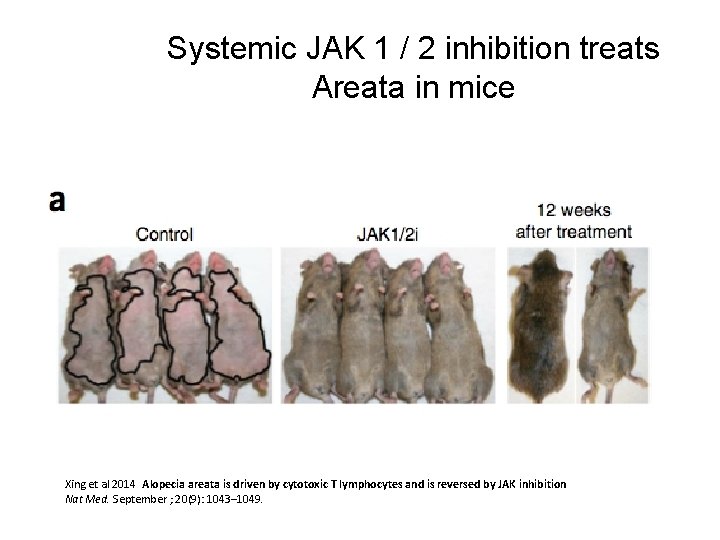
Systemic JAK 1 / 2 inhibition treats Areata in mice Xing et al 2014 Alopecia areata is driven by cytotoxic T lymphocytes and is reversed by JAK inhibition Nat Med. September ; 20(9): 1043– 1049.
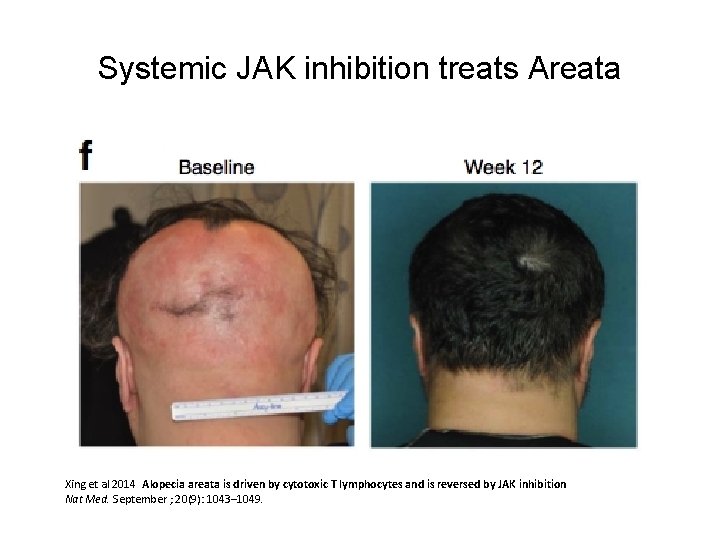
Systemic JAK inhibition treats Areata Xing et al 2014 Alopecia areata is driven by cytotoxic T lymphocytes and is reversed by JAK inhibition Nat Med. September ; 20(9): 1043– 1049.
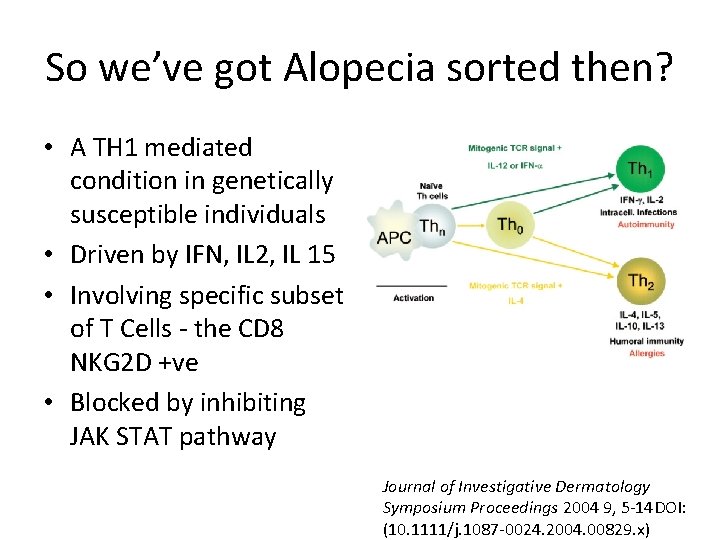
So we’ve got Alopecia sorted then? • A TH 1 mediated condition in genetically susceptible individuals • Driven by IFN, IL 2, IL 15 • Involving specific subset of T Cells - the CD 8 NKG 2 D +ve • Blocked by inhibiting JAK STAT pathway Journal of Investigative Dermatology Symposium Proceedings 2004 9, 5 -14 DOI: (10. 1111/j. 1087 -0024. 2004. 00829. x)
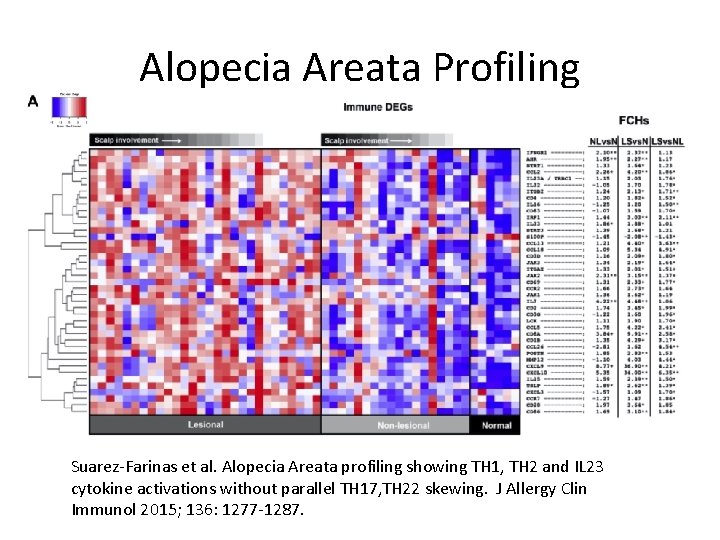
Alopecia Areata Profiling • Molecular profiling comparing of lesion and non-lesional biopsy specimens • TH 1 (IFN, CXCL 9, CXCL 10) • TH 2 (IL 13, CCL 26) • IL 23 • IL 16 & 32 • Without TH 17/TH 12 Suarez-Farinas et al. Alopecia Areata profiling showing TH 1, TH 2 and IL 23 cytokine activations without parallel TH 17, TH 22 skewing. J Allergy Clin Immunol 2015; 136: 1277 -1287.

So we’ve got Alopecia sorted then? • Current treatments (and majority of IPs in research) are non-specific • Systemic and intralesional treatments are associated side effects • High rates of relapse off treatment • No long-term restoration of immune privilege

New understanding gives new potential targets for “specific”treatments Protecting Immune Privilege • Anti-inflammatory growth factor, r. TGF b 1 • Alpha-melanocyte stimulating hormone a. MSH (Melanotan, Scenesse®� - afamelanotide) Antagonise putative trigger • Interferon g antagonists • Substance P receptor antagonists

New understanding gives new potential targets for “specific”treatments Down regulate MHC 1 • FK 506 – Tacrolimus • Effective in mouse models Target Cells and Ligands • NK • CD 8+ NKG 2 D • NKG 2 D CTLA 4 • Monoclonal antibodies against CTLA 4 effective in mouse models

Other novel targets • • IL 23 (new psoriasis biologics) IL 16 IL 32 IL 13 (IL 4 & 13 Dupilimumab, IL 13 Tralokinumab, phase 3 eczema by Leo)

Management

Management of Alopecia Areata • Counselling, support groups • No treatment • Corticosteroids • Contact Immunotherapy • Photochemotherapy • Minoxidil • Dithranol Corticosteroids: • Topical steroids – Super-potent 3 months – 6 weeks on and off for children • Intralesional steroids – 2. 5 -10 mg/ml • Systemic corticosteroids British Association of Dermatologists’ guidelines for the management of alopecia areata 2012. Messenger et al. Br J Dermatol 2012; 166: 916– 926

Systemic Immunosuppression • • JAK inhibitors Biologic Therapies Methotrexate Ciclosporin
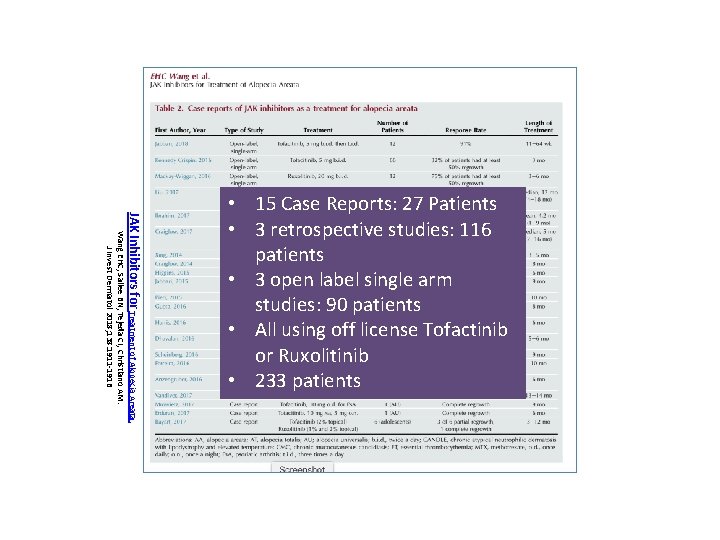
JAK Inhibitors for Treatment of Alopecia Areata. Wang EHC, Sallee BN, Tejeda CI, Christiano AM. J Invest Dermatol 2018; 138: 1911 -1916 • 15 Case Reports: 27 Patients • 3 retrospective studies: 116 patients • 3 open label single arm studies: 90 patients • All using off license Tofactinib or Ruxolitinib • 233 patients
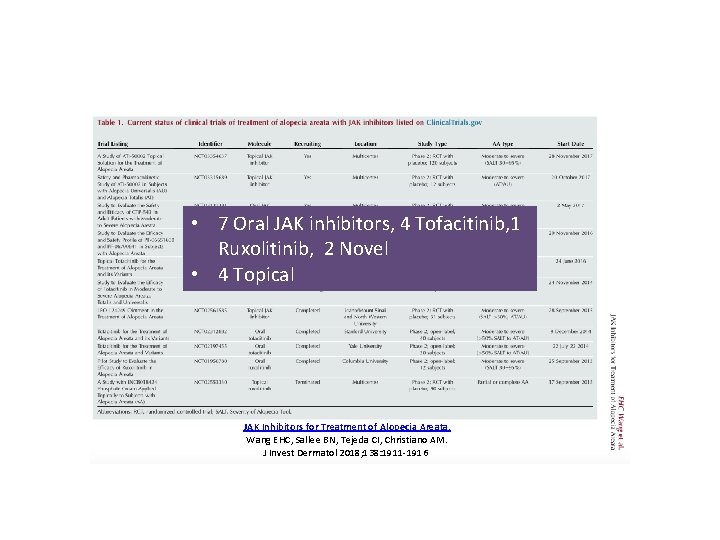
• 7 Oral JAK inhibitors, 4 Tofacitinib, 1 Ruxolitinib, 2 Novel • 4 Topical JAK Inhibitors for Treatment of Alopecia Areata. Wang EHC, Sallee BN, Tejeda CI, Christiano AM. J Invest Dermatol 2018; 138: 1911 -1916

• • Systematic Review of JAK Inhibitors 2019 196 Studies from 6 database searches > 30 4 cohort studies and 26 case reports 50% treated with oral Tofacitinib; 27% topical Tofactatinib, 18. 5% Ruxolitinib Phan et al. JEADV 2019; 33: 850 -862
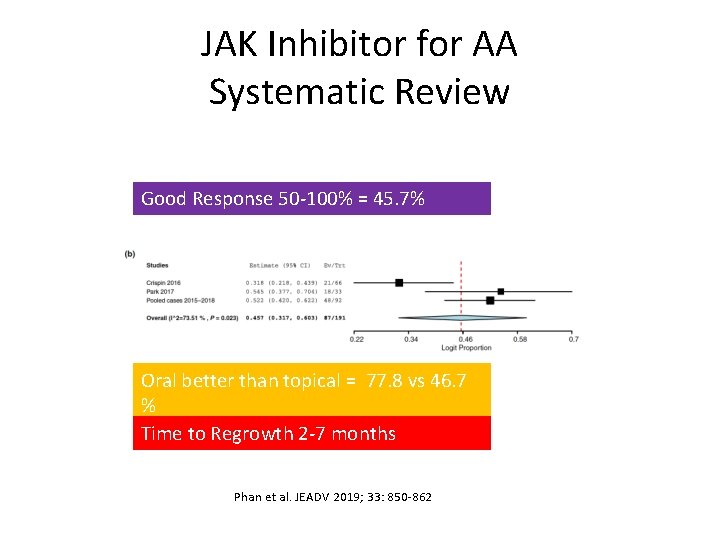
JAK Inhibitor for AA Systematic Review Good Response 50 -100% = 45. 7% Oral better than topical = 77. 8 vs 46. 7 % Time to Regrowth 2 -7 months Phan et al. JEADV 2019; 33: 850 -862

Clinical Trials. Gov • 9 Trial presently recruiting • JAK inhibitors – Tofacitinib – Baricitinib – Jaktinib – PF 06651600 – CTP 543 • Dupilimumab • BNZ 1: PEGylated peptide inhibitor of IL-2, IL-9, and IL-15
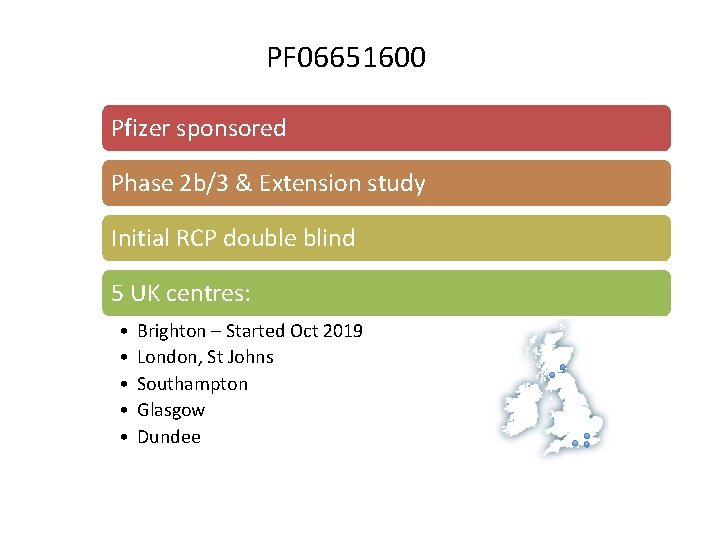
PF 06651600 Pfizer sponsored Phase 2 b/3 & Extension study PF Initial RCP double blind 066516 5 UK centres: 00 • Brighton – Started Oct 2019 • • London, St Johns Southampton Glasgow Dundee
JAK Inhibitors – Do they work and will heath authorities pay? • • • Too early to say 70% respond, 50% achieve SALT 50 What is acceptable treatment? Need to be continued to maintain benefit Likely to be expensive and restricted to severe disease, reflecting trial recruitment

Other Biologics • Dupilumab • PEGylated peptide inhibitor of IL-2, IL-9, and IL -15 • IL 23 • IL 13 Tralokinumab

Old Therapies Revisited METHOTREXAT E CICLOSPORIN
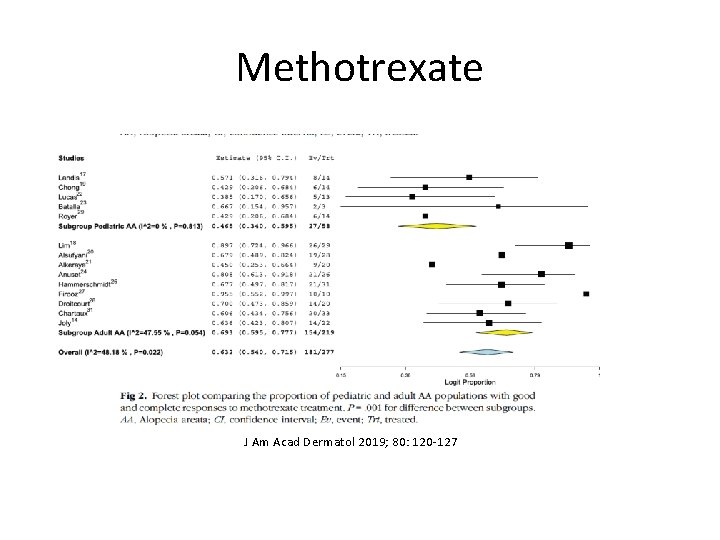
Methotrexate • 2006 Pascal Joly, Rouen – J Am Acad Dermatol 2006; 632 -6 • 2019 Systematic review – 63% Response – 36% Complete Response J Am Acad Dermatol 2019; 80: 120 -127

• Works best in combination with long term prednisolone • High doses required 20 -25 mg weekly • Delayed response 3/12 to see regrowth and 612 months to achieve complete regrowth J Am Acad Dermatol 2019; 80: 120 -127

Ciclosporin J Am Acad Dermal 2019; 81: 694 -701

Ciclosporin • Very small sample size 32 (16 drug vs 16 placebo) • 4 mg/kg • 50% reduction in SALT score in 31% vs 6% at week 12 • P=0. 07

Summary • • Common autoimmune condition Genetic predisposition Triggers poorly understood New understanding is leading to new treatments

Downloadable copy available: www. drpaulfarrant. co. uk Questions: Paul. Farrant 1@nhs. net
- Slides: 62