All things thrombosis Julie Wang Thrombosis Fellow VTE

All things thrombosis Julie Wang Thrombosis Fellow

VTE is chronic disease • Recurrence rate after initial VTE up to 24. 6% after 5 years • Clinical need to identify individuals at increased risk of recurrent VTE

VTE terminology • Provoked – surgery – 3% recurrence at 5 years • Minimally provoked – non‐surgical transient risk factor – oestrogen, pregnancy, flight>8 hrs – 15% recurrent at 5 years • Unprovoked – 30% recurrence at 5 years • Cancer‐associated – 15% annualized risk recurrence
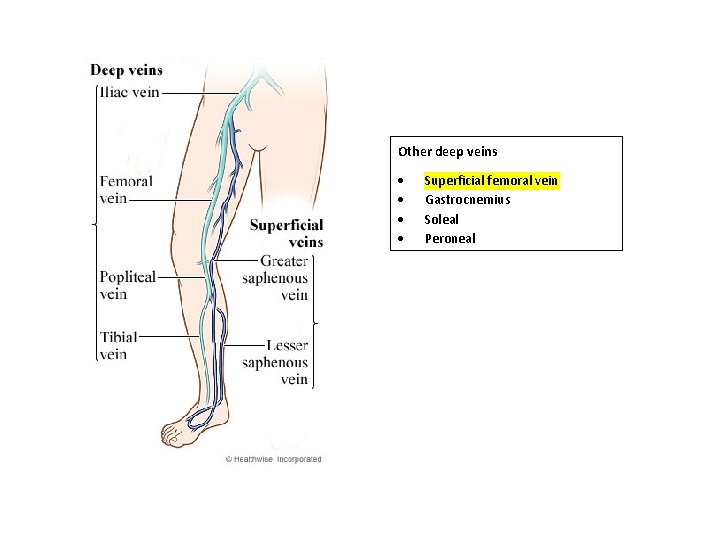
Other deep veins Superficial femoral vein Gastrocnemius Soleal Peroneal

Duration of anticoagulation ‐ proximal DVT and PE • Proximal = Above or involving popliteal vein • Minimum period is 3 months for all patients • After 3 months for unprovoked DVT/PE – Assess risk/benefit of indefinite therapy

Risk factors for recurrent VTE Ref 3
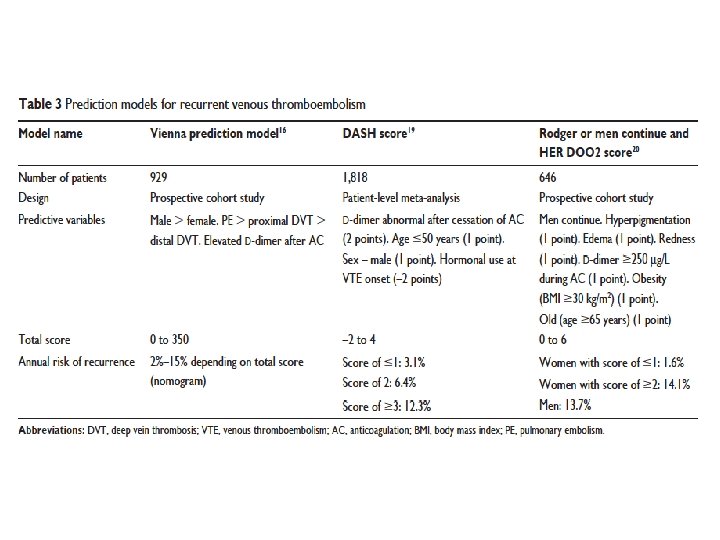
Ref 2
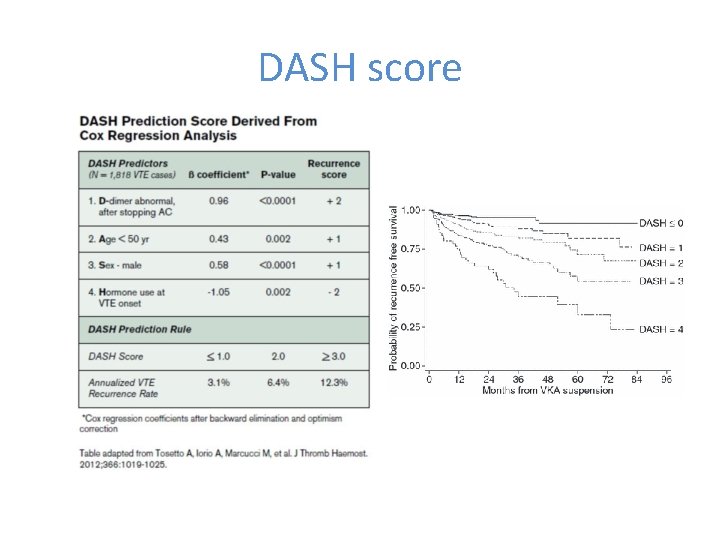
DASH score

Methods • Retrospective analysis of medical records at single institution • Patients with discharge diagnosis of DVT/PE between January 2013 and June 2015 • D‐dimer assay Innovance D‐dimer; Normal <500 ng/m. L • Presence of residual thrombus defined by reporting radiologist
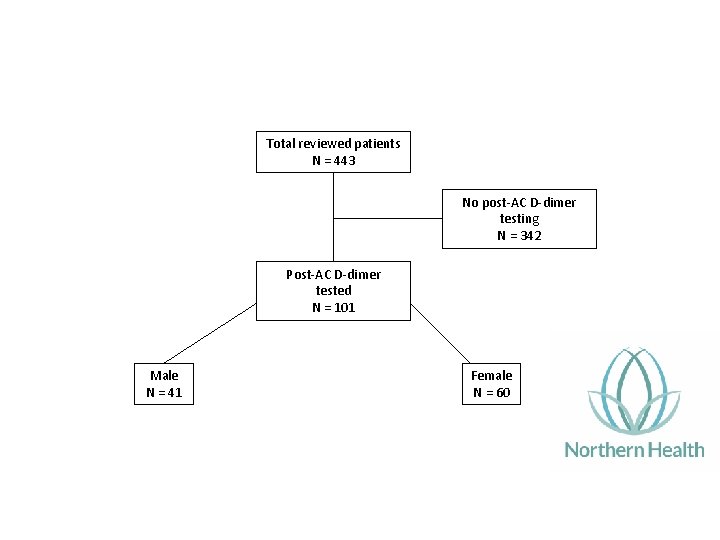
Total reviewed patients N = 443 No post‐AC D‐dimer testing N = 342 Post‐AC D‐dimer tested N = 101 Male N = 41 Female N = 60

Results • Median follow up 10 months (0 – 50 months) • Median time to D‐dimer testing 45 days after stopping anticoagulation (6‐ 318 days)
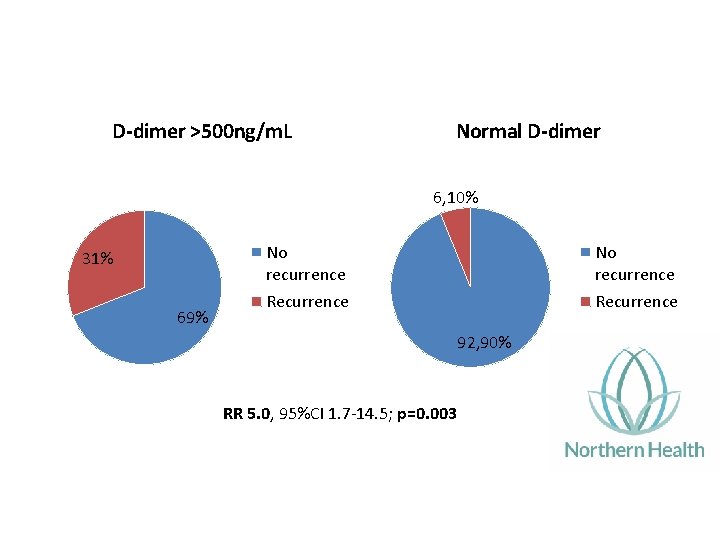
D-dimer >500 ng/m. L Normal D-dimer 6, 10% 31% 69% No recurrence Recurrence 92, 90% RR 5. 0, 95%CI 1. 7‐ 14. 5; p=0. 003

• Anticoagulation was recommenced for 11 patients with elevated D‐dimer • >1000 ng/m. L = 8 patients • 500‐ 1000 ng/m. L = 3 patients • Patients with recurrent VTE = 0 • Bleeding complications = 0

Table 1 – VTE recurrence stratified by D‐dimer Pts without anticoagulation recommenced Relative risk (compared to patients with normal Ddimer) D-dimer (ng/m. L) Pts with recurrent VTE >1000 4 4 100% 16. 3 (95%CI 6. 3 -42; p<0. 0001) 500 -1000 7 21 33. 3% 5. 4 (95%CI 1. 8 -16. 7; p=0. 0033) <500 4 65 6. 2% Recurrence rate
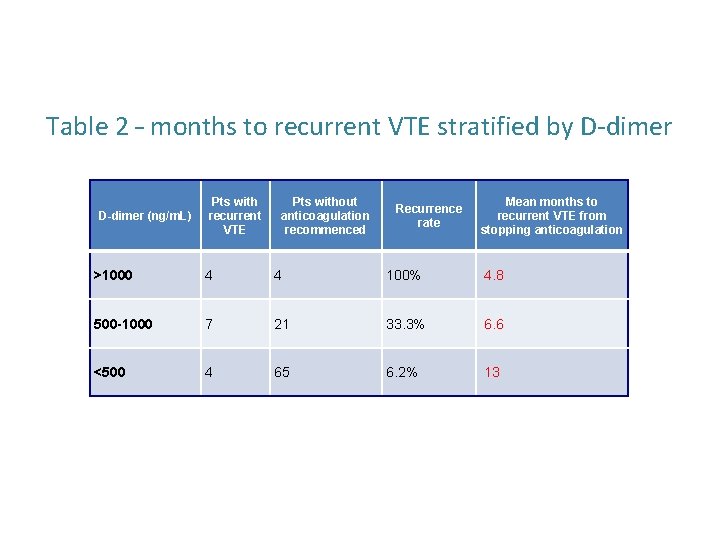
Table 2 – months to recurrent VTE stratified by D‐dimer D-dimer (ng/m. L) Pts with recurrent VTE Pts without anticoagulation recommenced Recurrence rate Mean months to recurrent VTE from stopping anticoagulation >1000 4 4 100% 4. 8 500 -1000 7 21 33. 3% 6. 6 <500 4 65 6. 2% 13
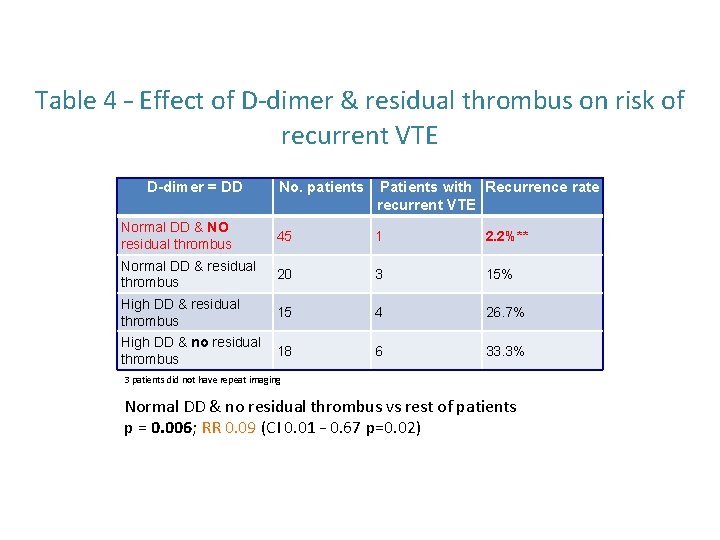
Table 4 – Effect of D‐dimer & residual thrombus on risk of recurrent VTE D-dimer = DD No. patients Patients with Recurrence rate recurrent VTE Normal DD & NO residual thrombus 45 1 2. 2%** Normal DD & residual thrombus 20 3 15% High DD & residual thrombus 15 4 26. 7% High DD & no residual thrombus 18 6 33. 3% 3 patients did not have repeat imaging Normal DD & no residual thrombus vs rest of patients p = 0. 006; RR 0. 09 (CI 0. 01 – 0. 67 p=0. 02)

Conclusions • Highest risk of VTE recurrence in those with D‐ dimer >1000 ng/ml – long term anticoagulation should be considered. • D‐dimer 500‐ 1000 ng/ml confers intermediate risk – age‐adjustment may further risk stratify. • Patients with normal D‐dimer <500 ng/ml without any residual thrombus have significantly lower risk of thrombus recurrence.
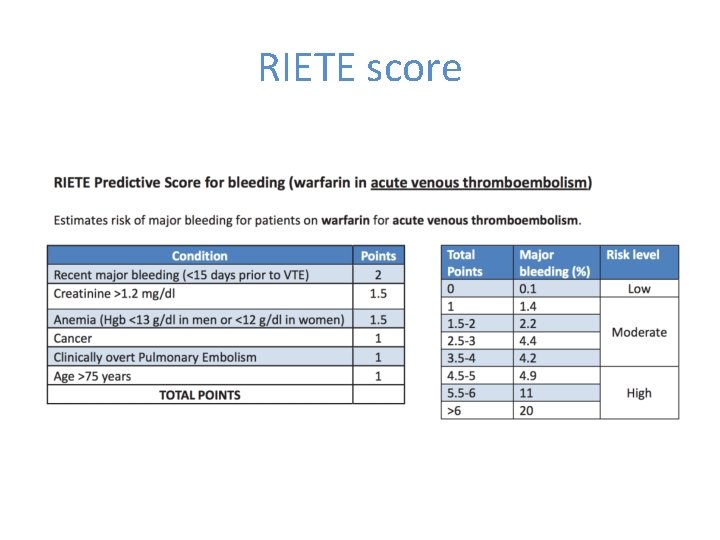
RIETE score

Choice of anticoagulant • NOAC is now preferred over warfarin as: – Efficacy is non‐inferior to warfarin in multiple large RCTs – Lower risk of bleeding, esp intracranial – Convenient to take • No direct comparison between NOACs but indirectly, efficacy appears similar
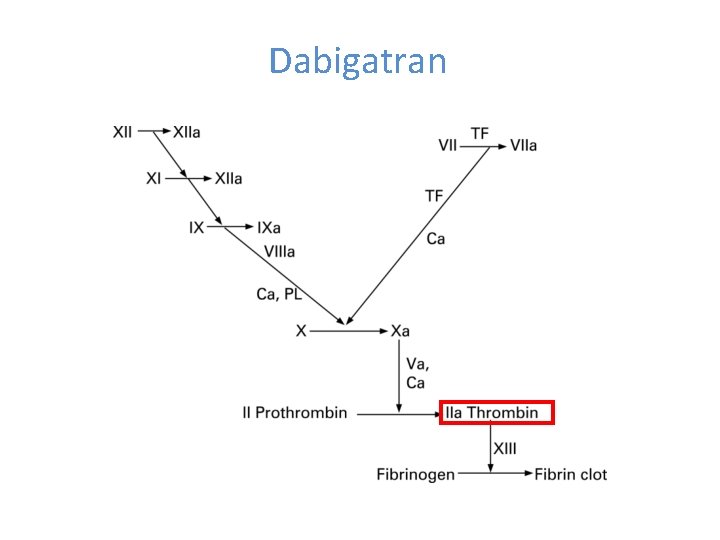
Dabigatran
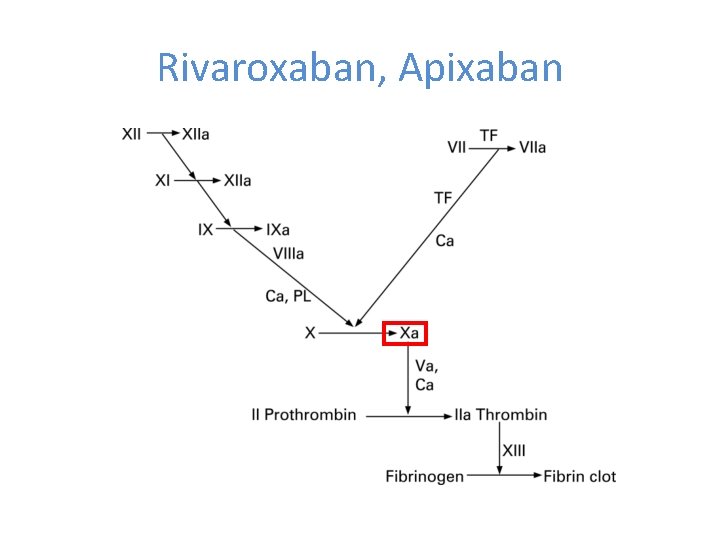
Rivaroxaban, Apixaban
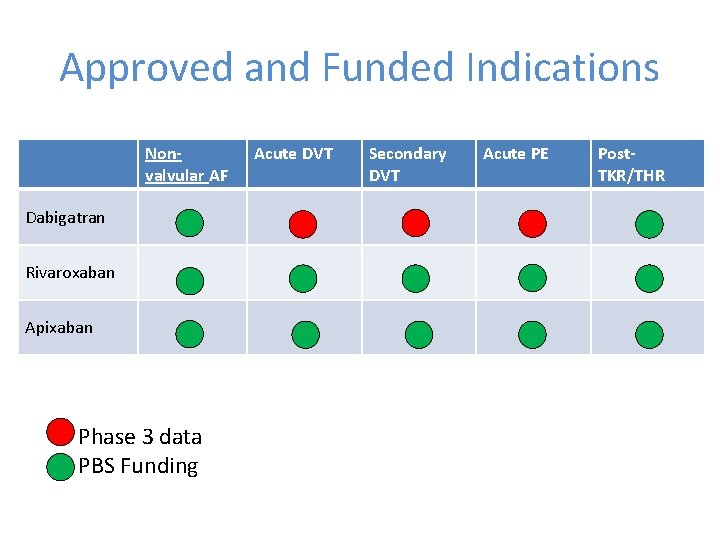
Approved and Funded Indications Nonvalvular AF Dabigatran Rivaroxaban Apixaban Phase 3 data PBS Funding Acute DVT Secondary DVT Acute PE Post. TKR/THR
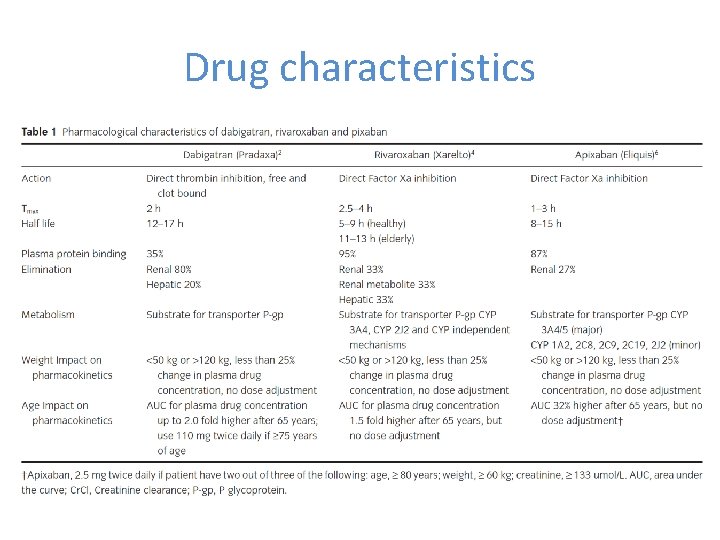
Drug characteristics
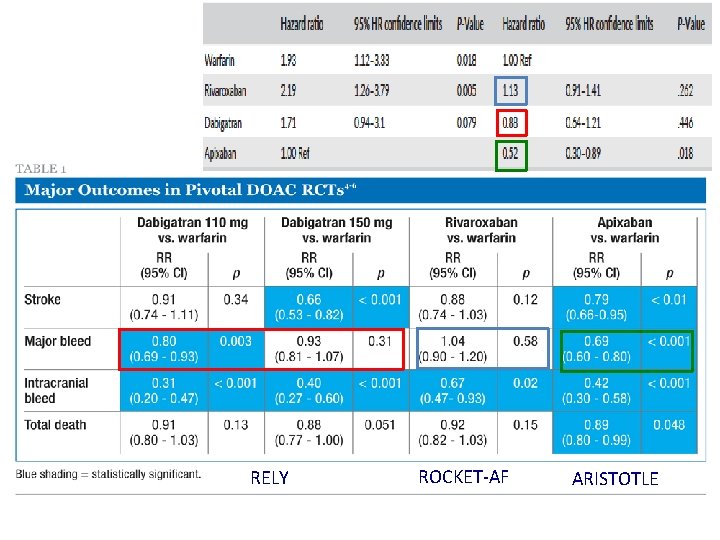
RELY ROCKET‐AF ARISTOTLE
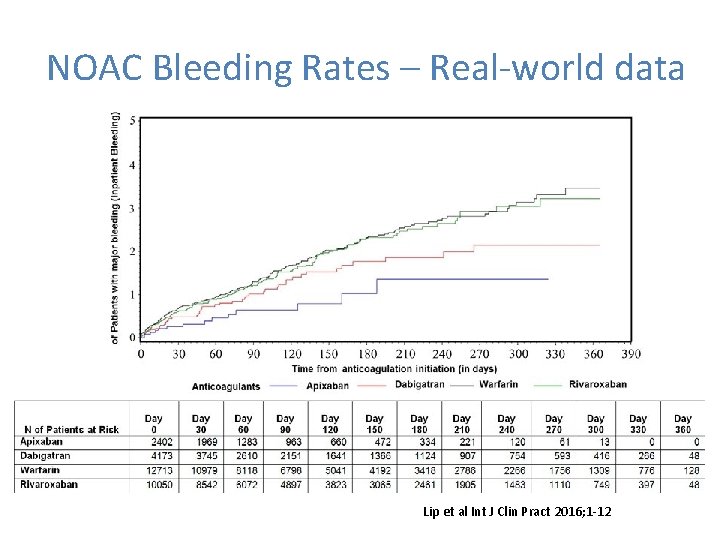
NOAC Bleeding Rates – Real‐world data Lip et al Int J Clin Pract 2016; 1‐ 12

Lab Assays • No routinely available assays for degree of anticoagulation. • Currently available coagulation testing imperfect. – No clinical correlation yet. • Indications for testing: – Bleeding or overdose – Before surgery or invasive procedure ‐ esp if drug taken in previous 24 hr or renal failure, or thrombolysis for stroke. – Renal failure – +/‐ Adherence • Time of last drug dose important
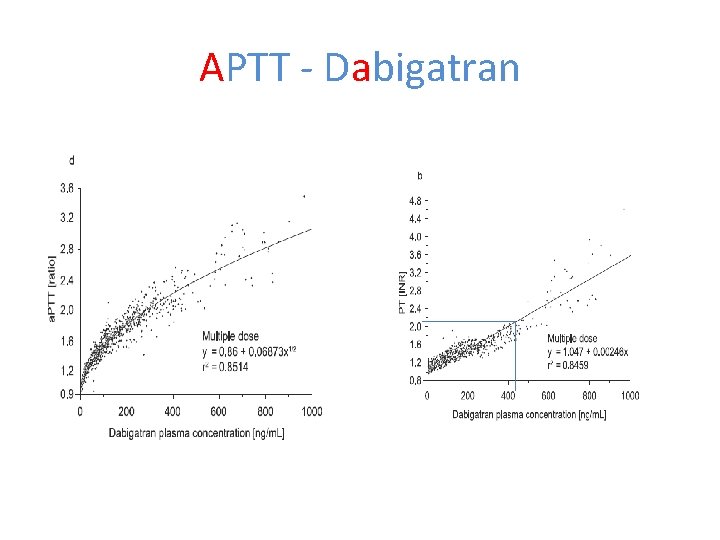
APTT ‐ Dabigatran
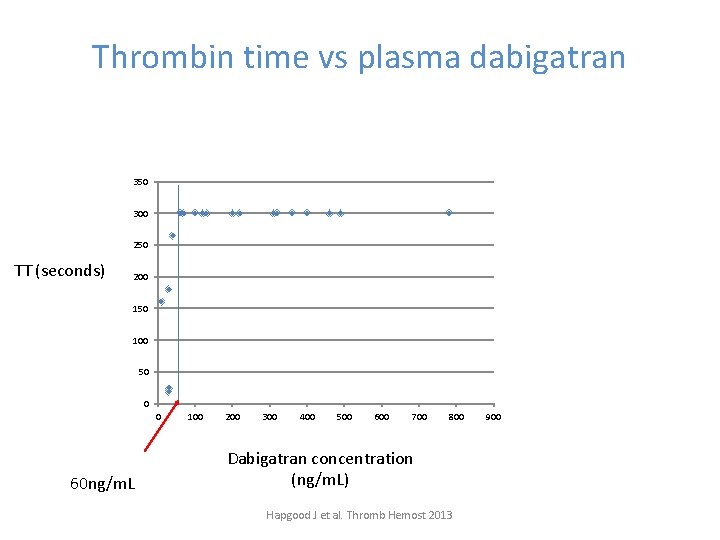
Thrombin time vs plasma dabigatran TCT (seconds) >300 350 300 250 TT (seconds)) 200 TCT (seconds) >300 150 100 50 0 0 60 ng/m. L 100 200 300 400 500 600 700 800 Dabigatran concentration (ng/m. L) Hapgood J et al. Thromb Hemost 2013 900
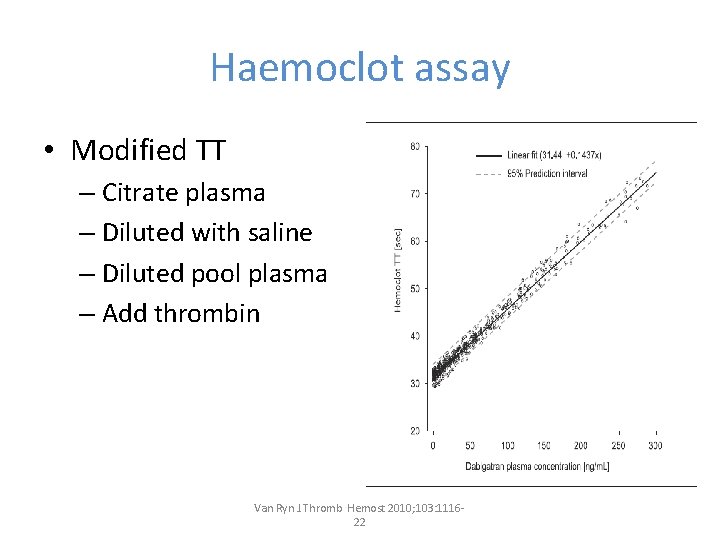
Haemoclot assay • Modified TT – Citrate plasma – Diluted with saline – Diluted pool plasma – Add thrombin Van Ryn J. Thromb Hemost 2010; 103: 1116‐ 22

Assessing Dabigatran levels • TCT (thrombin clotting time) – available at TNH 24/7 • Haemoclot (dilute TCT) = drug level - Not done here – turnaround time ~24 h • PT and INR – not useful APTT normal TCT normal = minimal/no drug APTT normal TCT prolonged = likely low drug levels Both prolonged = significant drug activity

Lab Assays • Rivaroxaban – Prolongs the PT if high levels of drug. • Normal PT ≠ no rivaroxaban. • PT on rivaroxaban ≠ PT on warfarin. – Ignore the INR. – Rivaroxaban anti- Xa = drug level – APTT not useful. • Apixaban – Apixaban anti- Xa = drug level – PT, INR, APTT not useful
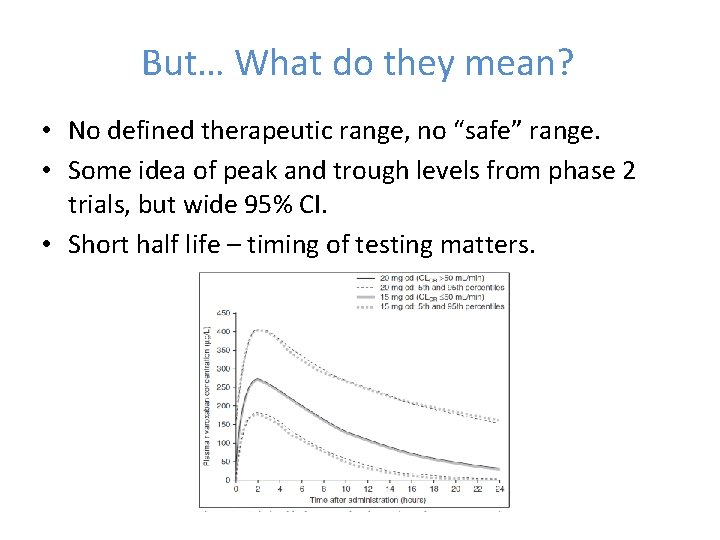
But… What do they mean? • No defined therapeutic range, no “safe” range. • Some idea of peak and trough levels from phase 2 trials, but wide 95% CI. • Short half life – timing of testing matters.
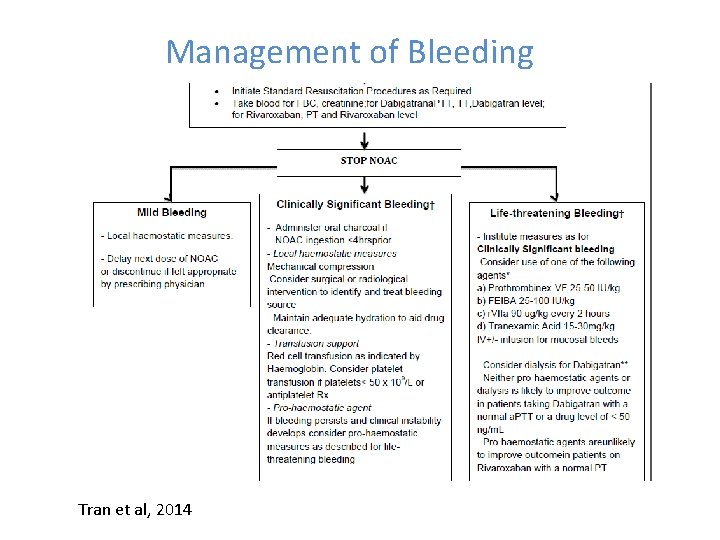
Management of Bleeding Tran et al, 2014

Idarucizumab • Antidote to the DTI – monoclonal antibody which binds to dabigatran with a 350 x affinity • Neutralises activity of free and thrombin‐bound dabigatran • Rapid, complete and sustained reversal • Idarucizumab produced immediate and complete reversal of anticoagulant effects of dabigatran without procoagulant effects in: – Young, healthy volunteers with normal renal function – Elderly volunteers 65 – 80 years of age – Volunteers aged 45 – 80 years with mild or moderate renal impairment • 2 x 2. 5 g bolus IV repeated 15 minutes apart

On the horizon… • ANNEXA‐A – andexanet alfa – Recombinant inactivated factor Xa – decoy with a high affinity for factor Xa inhibitors – Phase III – bolus, followed by infusion • Rapid reduction in anti‐Xa activity and sustained as long as the infusion was continued • Aripazine (PER 977) – Mode of action unclear – Inhibits LMWH, fondaparinux, direct Xa inhibitors and dabigatran
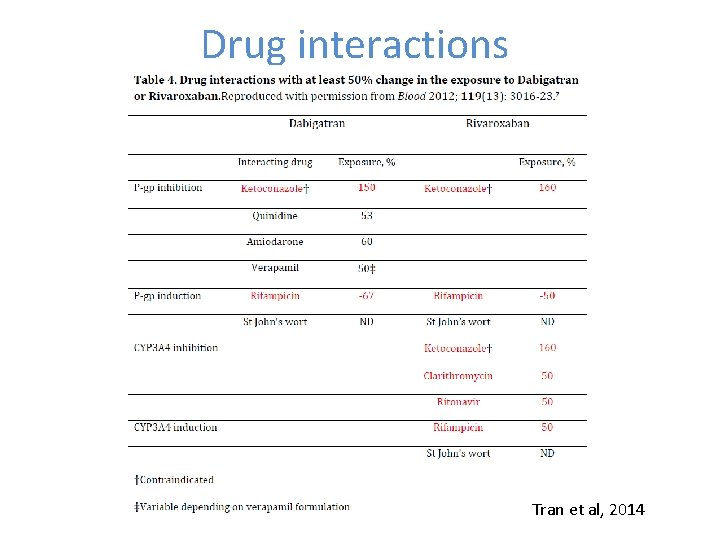
Drug interactions Tran et al, 2014
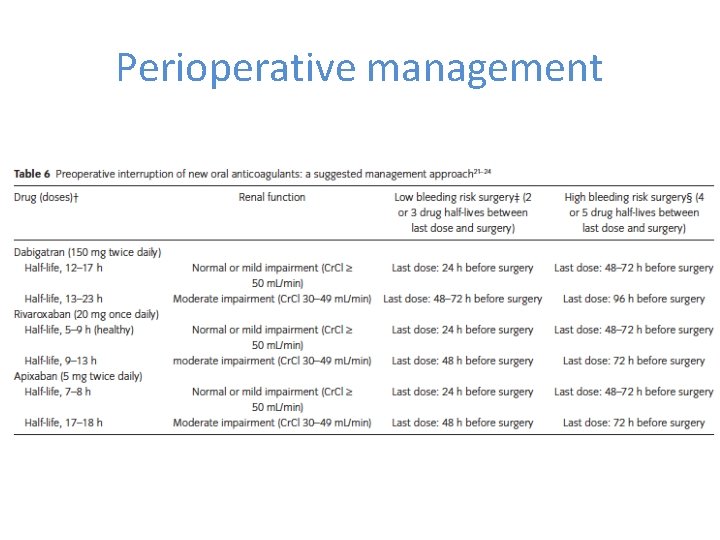
Perioperative management

Who I would put on a NOAC… • • • Outpatients. Cr. Cl >30 m. L/min. LFTs not significantly deranged ? 3 x. ULN Few other comorbidities. Adherent. Difficult warfarin control (<65% TTR? ).

Who I wouldn’t put on a NOAC… • Absolute contraindicated – – – Cr. Cl <30 (very cautious if <50) Mechanical heart valve Antiphospholipid syndrome Pregnant/Lactating Mod‐severe liver disease • Relative contraindications – Extremes of age and weight – Active malignancy – lack of powered RCTs individual case based assessment + involvement with haematologist • Non‐adherent with warfarin. “Falls risk” or anyone “unsafe” for warfarin.

Key Points for NOACs 1. NOAC is non‐inferior to warfarin in non‐valvular AF and VTE 2. It is not appropriate to use NOACs in patients with renal impairment, or in the very elderly or very frail. 3. There are no antidotes at present for Xa inhibitors – these are very early in clinical development. No effective reversal agents. 4. Normal coagulation parameters (PT/APTT) may not be affected by NOACs. Drug levels can be tested, but interpretation is difficult. 5. Adherence is extremely important – rapid offset and loss of anticoagulation if renal function normal.

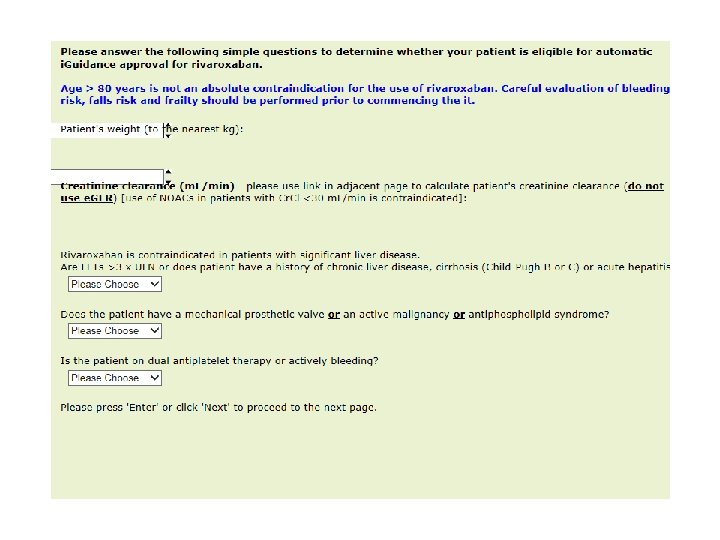
NOAC i. Guidance

Isolated distal DVT • Not great evidence and unclear guidelines • 33% of symptomatic if untreated will extend into popliteal vein; risk extension lower in muscular veins (soleus, gastroc) • Risk of PE in distal DVT 50% of proximal DVT • Risk of recurrence 50% of proximal DVT/PE

Isolated distal DVT • Suggested risk factors for extension – Positive D‐Dimer – >5 cm length, multiple veins involved – Close to proximal veins – Unprovoked – Active cancer – History of VTE – Inpatient status

Isolated distal DVT • If elect to anticoagulate – full dose anticoagulation as per proximal DVT • Duration is uncertain – 6 weeks? 3 months?

NOACs not PBS approved for SVT

VTE and cancer • LMWH – RCTs have shown LMWH more effective than warfarin in active cancer – More reliable if difficulty with oral therapy eg vomiting – Easier to withold if thrombocytopenia or invasive procedures
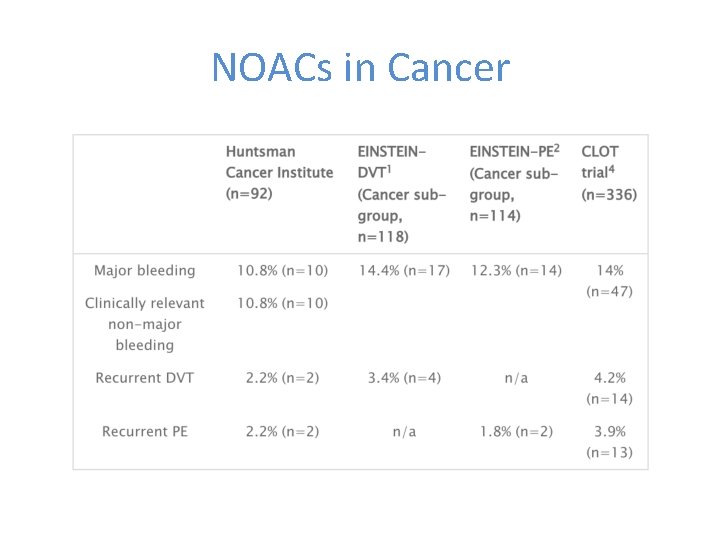
NOACs in Cancer

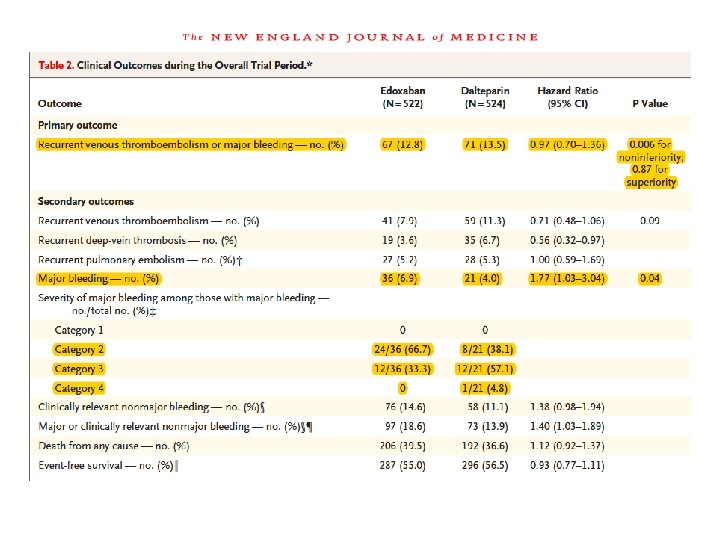
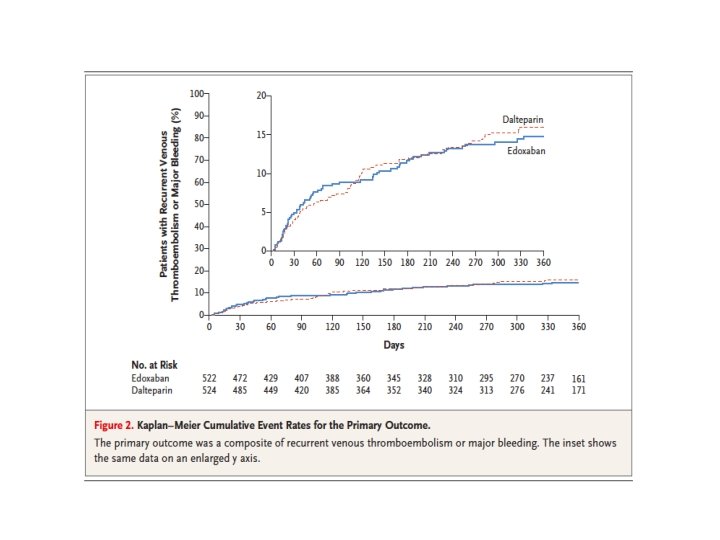

Recurrent VTEs whilst on anticoagulation • Is there really a recurrent thrombus? – compare closely current and previous ultrasounds. ? post-thrombotic syndrome • Potential causes – – – Adherence to therapy Subtherapeutic INR? NOAC interacting with another drug? Cancer Antiphospholipid syndrome Oestrogen therapy • Switch to LMWH if on oral therapy • If already on LMWH consider higher dose
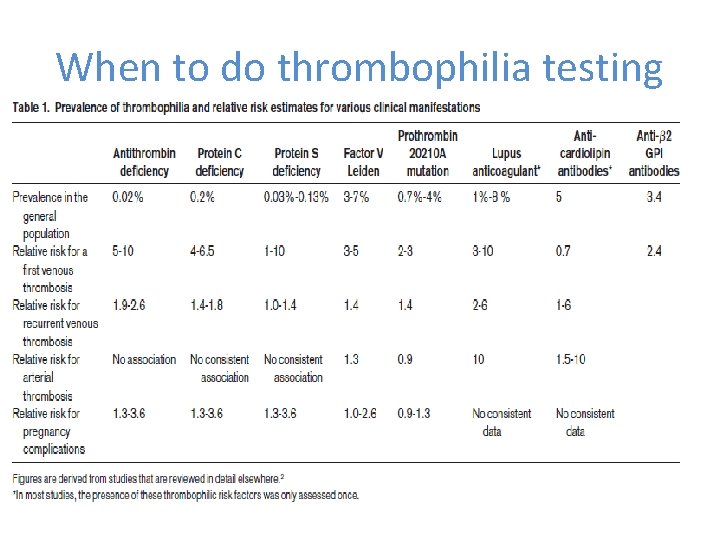
When to do thrombophilia testing • Give estimated risk factors for hereditary/acquired factors • Changed in era of NOAC?

RCPA Recommendations • RCPA recommends to follow the recommendations of the College of American Pathologists and perform specific thrombophilia testing in the patient groups • Additionally, testing for antiphospholipid antibodies is considered appropriate for patients with VTE, particularly if the VTE is idiopathic, associated with autoimmune disease or in the absence of a family history of venous thrombosis

When to test? • FVL and PT 20102 A mutation are PCR based – can test anytime. • Other tests ‐ Not during acute thrombosis or pregnancy – wait at least 6 weeks. • Heparin – lowers AT activity and antigen levels – Affects LAC (clot‐based) assay – delay 5 days after heparin is stopped • Warfarin – reduces the vitamin K–dependent factors, (Protein C and S. ) ‐ may take 6 weeks to normalise. – rarely, may increase AT levels (in deficient pt)

A general approach • Only test if it will change management (eg deciding on duration of anticoagulation, planning for future prophylaxis, family testing in the case of inherited thrombophilias). • Testing for inherited thrombophilia (FVL, PT mutation, AT deficiency, PC and PS deficiencies): for patients with an unprovoked or recurrent clots and family history of VTE. • Testing for acquired thrombophilia (lupus anticoagulant, anticardiolipin antibodies, B 2 G 1 antibodies): for patients with an unprovoked or recurrent clots, young females, clot during pregnancy, or history of miscarriages/ pregnancy complications. • Patients with unusual clots (cerebral sinus thrombosis, splachnic vein thrombosis): test for all of the above, and others eg PNH screen, JAK 2.

Useful references • Antithrombotic therapy for VTE disease: CHEST guideline, CHEST (2016) • Tran et al, New oral anticoagulants – a practical guide on prescription, laboratory testing and peri‐procedural/bleeding management. IMJ; 44: 2014.
- Slides: 58