Alkyl Halides RX X F Cl Br I

Alkyl Halides R-X (X = F, Cl, Br, I) Classification of alkyl halides according to the class of the carbon that the halogen is attached to. RCH 2 -X 1 o R 2 CH-X 2 o R 3 C-X 3 o

Nomenclature: common names: “alkyl halide” (fluoride, chloride, bromide, iodide) IUPAC names: use rules for alkanes halogen = halo (fluoro, chloro, bromo, iodo) Cl CH 3 CH 2 CH 2 -Br CH 3 CHCH 3 n-butyl bromide isopropyl chloride 1 -bromobutane 2 -chloropropane 1 o 2 o
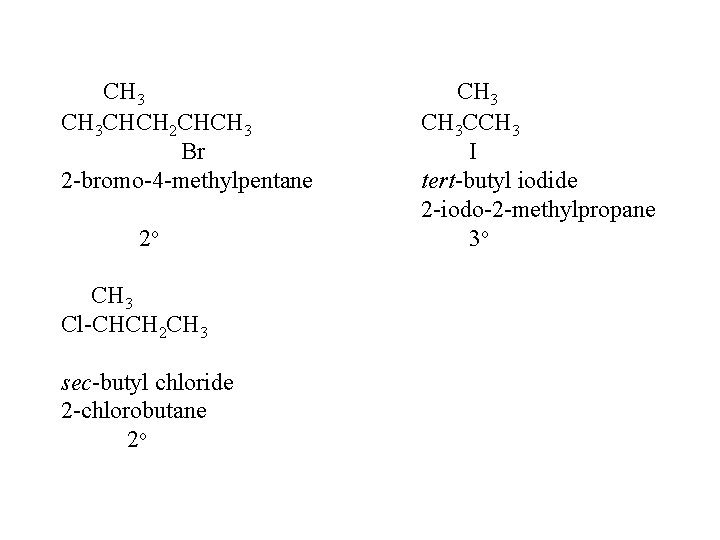
CH 3 CHCH 2 CHCH 3 Br 2 -bromo-4 -methylpentane 2 o CH 3 Cl-CHCH 2 CH 3 sec-butyl chloride 2 -chlorobutane 2 o CH 3 CCH 3 I tert-butyl iodide 2 -iodo-2 -methylpropane 3 o

Physical properties: polar + no hydrogen bonding => moderate boiling/melting points water insoluble Uses: pesticides, refrigerants (freons), solvents, synthetic intermediates. CH 3 Br CCl. F 3 CCl 4

Synthesis of alkyl halides: 1. From alcohols a) HX b) PX 3 2. Halogenation of certain hydrocarbons 3. 3. (later) 4. 5. 4. (later) 6. 5. Halide exchange for iodide

1. From alcohols. #1 synthesis! a) With HX b) R-OH c) i) HX = HCl, HBr, HI d) ii) may be acid catalyzed (H+) e) iii) ROH: 3 o > 2 o > CH 3 > 1 o f) + HX R-X + H 2 O iv) rearrangements are possible except with most 1 o ROH
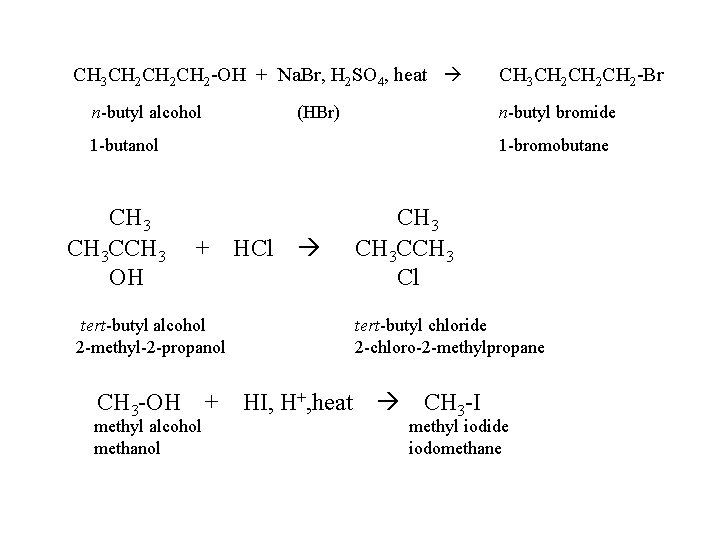
CH 3 CH 2 CH 2 -OH + Na. Br, H 2 SO 4, heat n-butyl alcohol (HBr) n-butyl bromide 1 -butanol CH 3 CCH 3 OH CH 3 CH 2 CH 2 -Br 1 -bromobutane + HCl tert-butyl alcohol 2 -methyl-2 -propanol CH 3 -OH + methyl alcohol methanol CH 3 CCH 3 Cl tert-butyl chloride 2 -chloro-2 -methylpropane HI, H+, heat CH 3 -I methyl iodide iodomethane

…from alcohols: b) PX 3 i) PX 3 = PCl 3, PBr 3, P + I 2 ii) ROH: CH 3 > 1 o > 2 o iii) no rearragements CH 3 CH 2 -OH + P, I 2 CH 3 CH 2 -I ethyl alcohol ethyl iodide ethanol iodoethane CH 3 CHCH 2 -OH isobutyl alcohol 2 -methyl-1 -propanol + PBr 3 CH 3 CHCH 2 -Br isobutyl bromide 1 -bromo-2 -methylpropane

2. Halogenation of certain hydrocarbons. 3. R-H 4. (requires Δ or hν; Cl 2 > Br 2 (I 2 NR); 3 o>2 o>1 o) 5. + X 2, Δ or hν R-X + HX yields mixtures! In syntheses, limited to those hydrocarbons that yield only one monohalogenated product. 6. CH 3 7. CH 3 CCH 3 + Cl 2, heat CH 3 CCH 2 -Cl 8. CH 3 9. 10. neopentane 2, 2 -dimethylpropane neopentyl chloride 1 -chloro-2, 2 -dimethylpropane
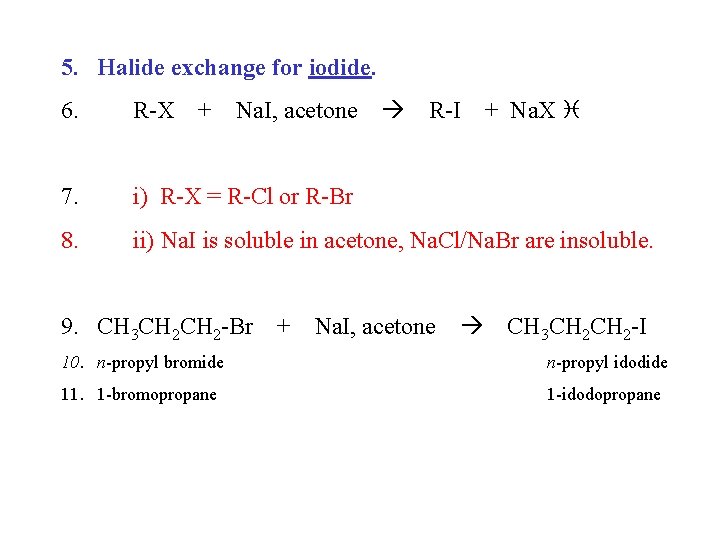
5. Halide exchange for iodide. 6. R-X 7. i) R-X = R-Cl or R-Br 8. ii) Na. I is soluble in acetone, Na. Cl/Na. Br are insoluble. + Na. I, acetone R-I + Na. X 9. CH 3 CH 2 -Br + Na. I, acetone CH 3 CH 2 -I 10. n-propyl bromide n-propyl idodide 11. 1 -bromopropane 1 -idodopropane
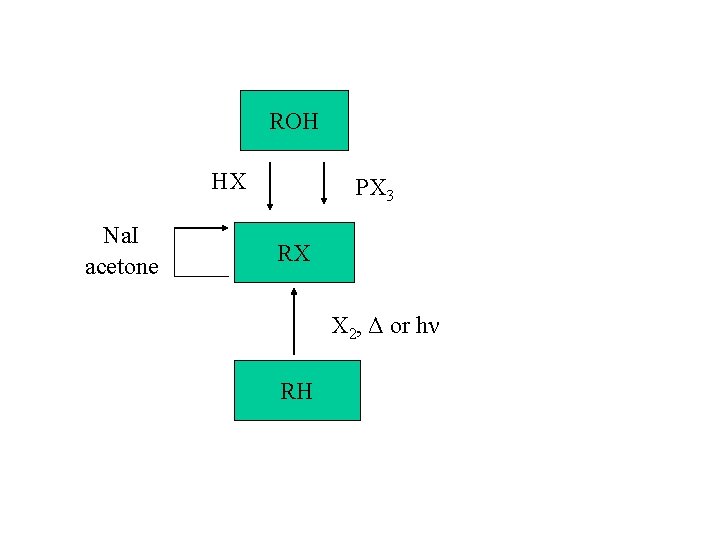
ROH HX Na. I acetone PX 3 RX X 2, Δ or hν RH

Outline a possible laboratory synthesis for each of the following alkyl halides using a different synthesis for each compound: 1 -bromobutane neopentyl chloride n-propyl iodide tert-butyl bromide
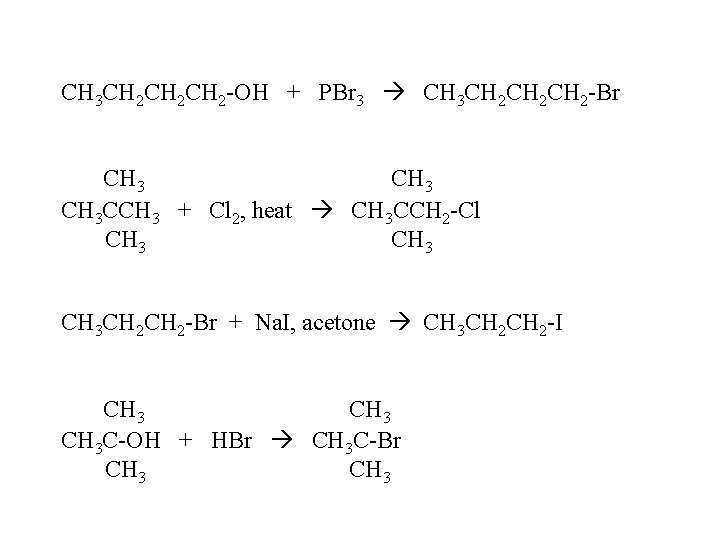
CH 3 CH 2 CH 2 -OH + PBr 3 CH 3 CH 2 CH 2 -Br CH 3 CCH 3 + Cl 2, heat CH 3 CCH 2 -Cl CH 3 CH 2 -Br + Na. I, acetone CH 3 CH 2 -I CH 3 C-OH + HBr CH 3 C-Br CH 3

R-H Acids Bases Active Metals Oxidants Reductants Halogens NR NR NR R-X NR NR

Reactions of alkyl halides: 1. Nucleophilic substitution Best with 1 o or CH 3!!!!!! 2. R-X + : Z- R-Z + : X- 3. 2. (later) 3. Preparation of Grignard Reagent 4. R-X + Mg RMg. X 4. Reduction 5. R-X + Mg 6. R-X + Sn, HCl R-H + H 2 O R-H

nucleophilic substitution R-W substrate + : Znucleophile R-Z substitution product good nucleophile strong base good leaving group weak base + : Wleaving group

R-X + : OH- ROH + : X- alcohol R-X + H 2 O ROH + HX alcohol R-X + : OR´- R-O-R´ + R-X + -: C CR´ R-C CR´ + : X- alkyne R-X + : I- R-I iodide R-X + : CN- R-C N + : X- nitrile R-X + : NH 3 R-NH 2 + HX primary amine R-X + : NH 2 R´ R-X + : SH- R-SH + : X- thiol R-X + : SR´ R-SR´ + : X- thioether + : X- R-NHR´ Etc. Best when R-X is CH 3 or 1 o! : X- + HX ether secondary amine
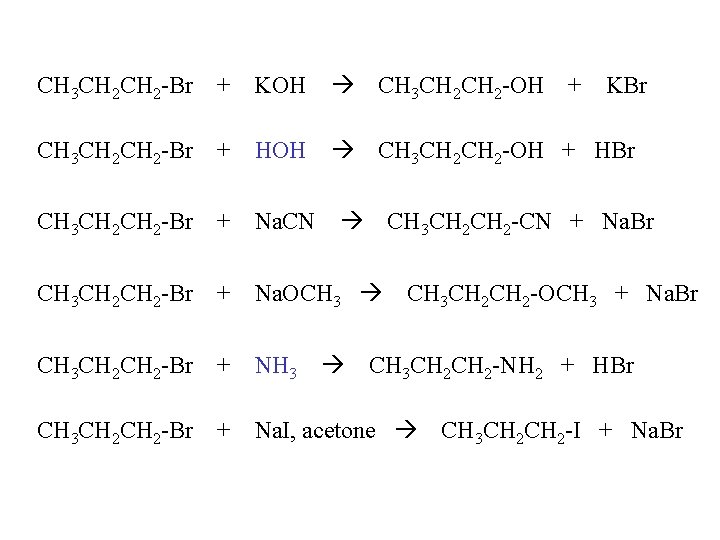
CH 3 CH 2 -Br + KOH CH 3 CH 2 CH 2 -Br + HOH CH 3 CH 2 -OH + HBr CH 3 CH 2 -Br + Na. CN CH 3 CH 2 -Br + Na. OCH 3 CH 3 CH 2 -Br + NH 3 CH 3 CH 2 -Br + Na. I, acetone + KBr CH 3 CH 2 -CN + Na. Br CH 3 CH 2 -OCH 3 + Na. Br CH 3 CH 2 -NH 2 + HBr CH 3 CH 2 -I + Na. Br

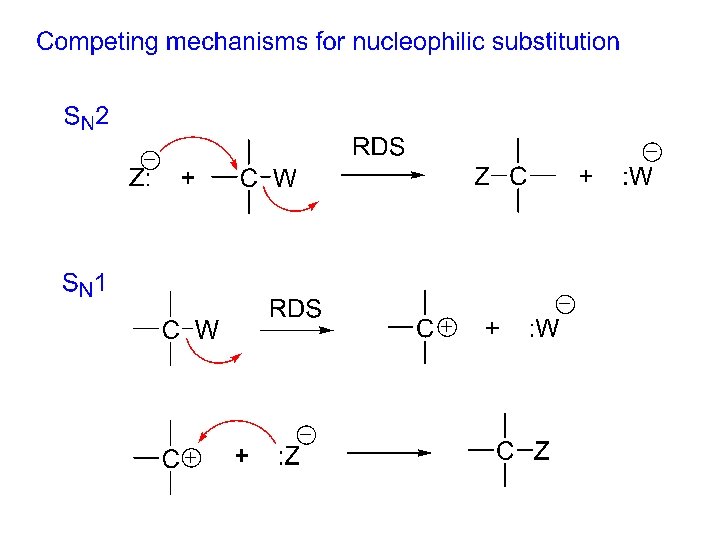
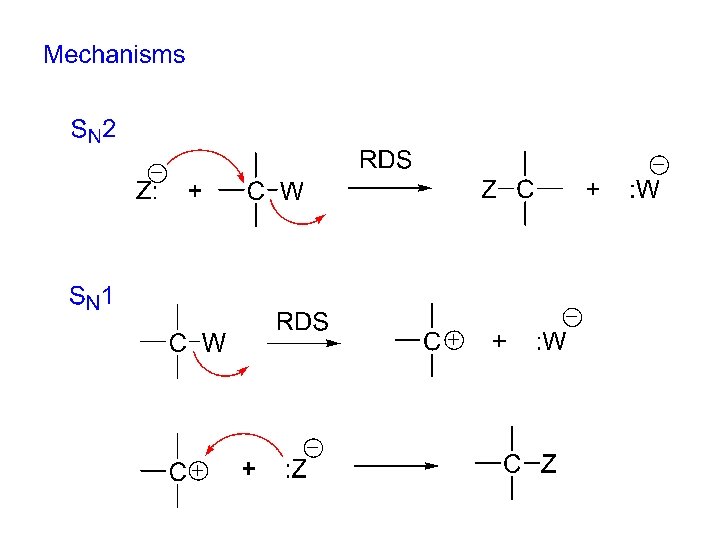
Mechanism for nucleophilic substitution: “substitution, nucleophilic, bimolecular” “curved arrow formalism” uses arrows to show the movement of pairs of electrons in a mechanism.

Kinetics – study of the effect of changes in concentration on rates of reactions. CH 3—Br + Na. OH CH 3—OH + Na. Br rate = k [ CH 3 -Br ] [ OH- ] Tells us that both CH 3 -Br and OH- are involved in the rate determining step of the mechanism. “bimolecular”

Relative rates of R—X R-I > R-Br > R-Cl “element effect” C—X bond is broken in the rate determining step of the mechanism.
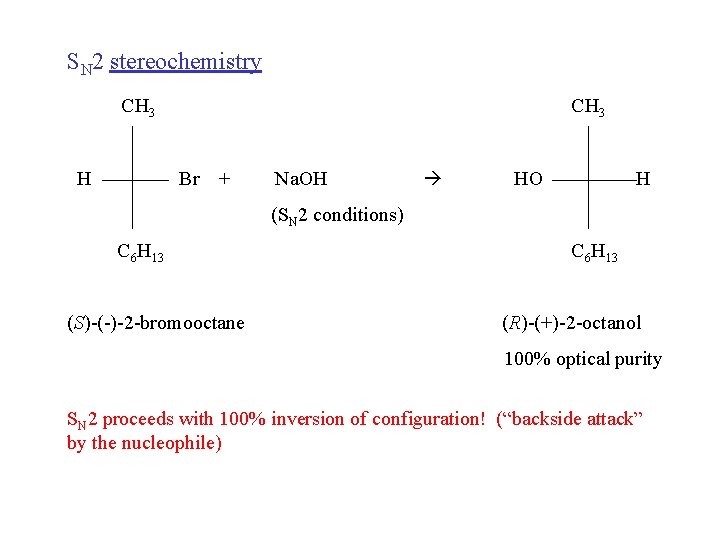
SN 2 stereochemistry CH 3 H CH 3 Br + Na. OH HO H (SN 2 conditions) C 6 H 13 (S)-(-)-2 -bromooctane C 6 H 13 (R)-(+)-2 -octanol 100% optical purity SN 2 proceeds with 100% inversion of configuration! (“backside attack” by the nucleophile)

SN 2 100% backside attack by the nucleophile Evidence: stereochemistry = 100% inversion of configuration Reasonable? 1) incoming nucleophile and negatively charged leaving group are as far apart as they can get. 2) there is more room on the backside of the carbon for the incoming nucleophile to begin to bond to the carbon.

Relative rates for alkyl halides in SN 2: CH 3 -X > 1 o > 2 o > 3 o 37 : 1. 0 : 0. 2 : 0. 0008 The transition state has five groups crowded around the carbon. If the substrate is CH 3 X then three of the five groups are Hydrogens. If the alkyl halide is 3 o then there are three bulky alkyl groups crowded around the carbon in the transition state. “Steric factors” explain the relative reactivity of alkyl halides in the SN 2 mechanism.

CH 3 CCH 3 + Br OH- CH 3 CCH 3 OH + Br- + alkene rate = k [ tert-butyl bromide ] The rate of this reaction depends on only the concentration of the alkyl halide. Therefore the nucleophile is not involved in the RDS here, cannot be SN 2 mechanism!? “unimolecular”
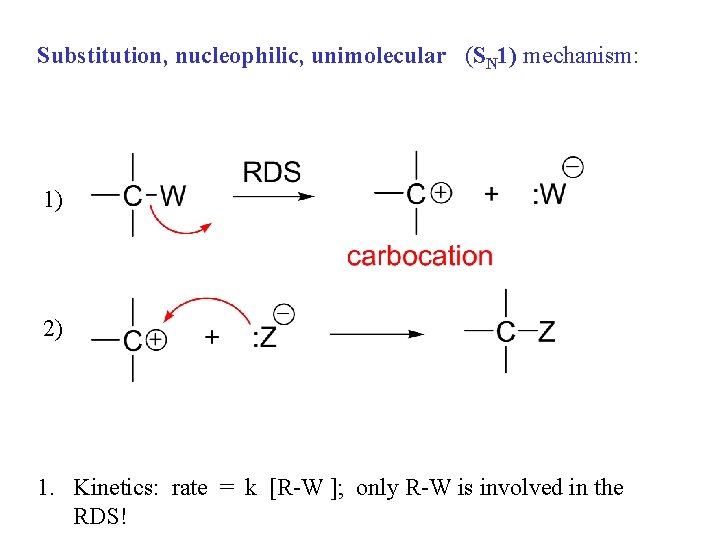
Substitution, nucleophilic, unimolecular (SN 1) mechanism: 1) 2) 1. Kinetics: rate = k [R-W ]; only R-W is involved in the RDS!
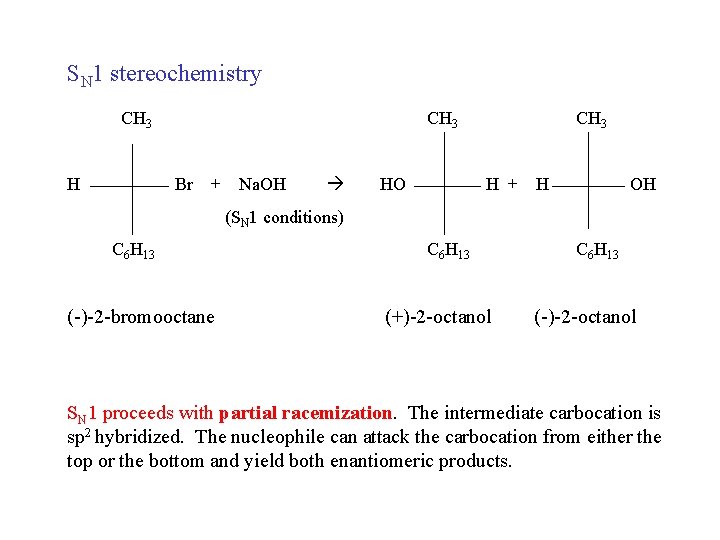
SN 1 stereochemistry CH 3 H CH 3 Br + Na. OH HO CH 3 H + H OH (SN 1 conditions) C 6 H 13 (-)-2 -bromooctane C 6 H 13 (+)-2 -octanol C 6 H 13 (-)-2 -octanol SN 1 proceeds with partial racemization. The intermediate carbocation is sp 2 hybridized. The nucleophile can attack the carbocation from either the top or the bottom and yield both enantiomeric products.

SN 1 reactivity: R—Br 3 o > 2 o > 1 o > CH 3 R+ + Br- CH 3—Br ΔH = 219 Kcal/mole CH 3+ CH 3 CH 2—Br ΔH = 184 Kcal/mole 1 o CH 3 CH—Br CH 3 ΔH = 164 Kcal/mole 2 o ΔH = 149 Kcal/mole 3 o CH 3 C—Br CH 3

SN 1 order of reactivity = 3 o > 2 o > 1 o > CH 3 Stability of carbocations = 3 o > 2 o > 1 o > CH 3+ RDS in SN 1: R—W R+ R—X [ R-----X ] δ+ δ- + : WR+ + X-
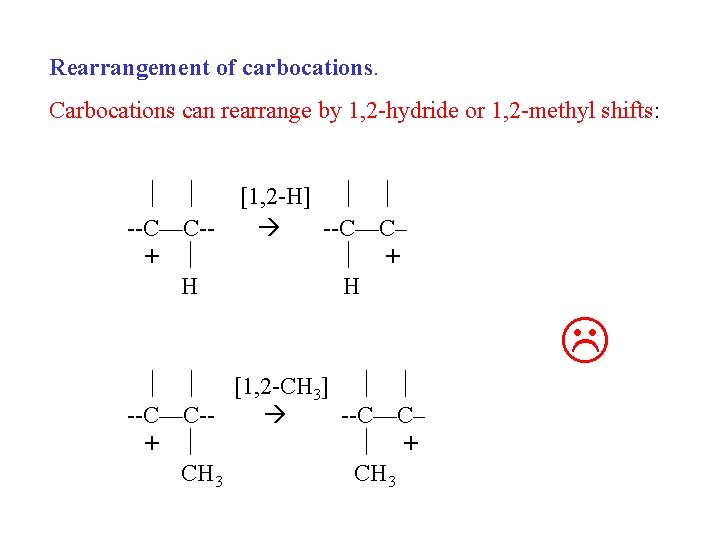
Rearrangement of carbocations. Carbocations can rearrange by 1, 2 -hydride or 1, 2 -methyl shifts: --C—C-+ H [1, 2 -H] --C—C– + H [1, 2 -CH 3] --C—C- --C—C– + + CH 3

Carbocations can rearrange by 1, 2 -hydride or 1, 2 -methyl shifts but only do so when the resultant carbocation is more stable. 1 o carbocation will rearrange to 2 o 1 o carbocation will rearrange to 3 o 2 o carbocation will rearrange to 3 o (only goes “down hill”)
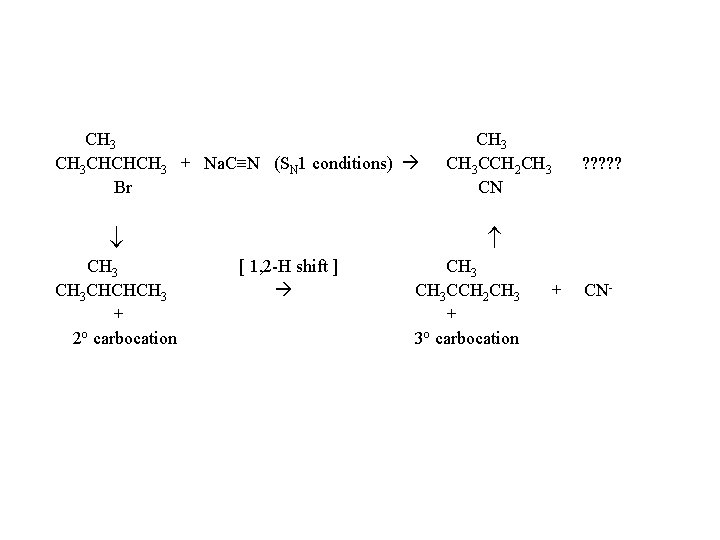
CH 3 CHCHCH 3 + Na. C N (SN 1 conditions) Br CH 3 CHCHCH 3 + 2 o carbocation CH 3 CCH 2 CH 3 CN ? ? ? [ 1, 2 -H shift ] CH 3 CCH 2 CH 3 + 3 o carbocation + CN-
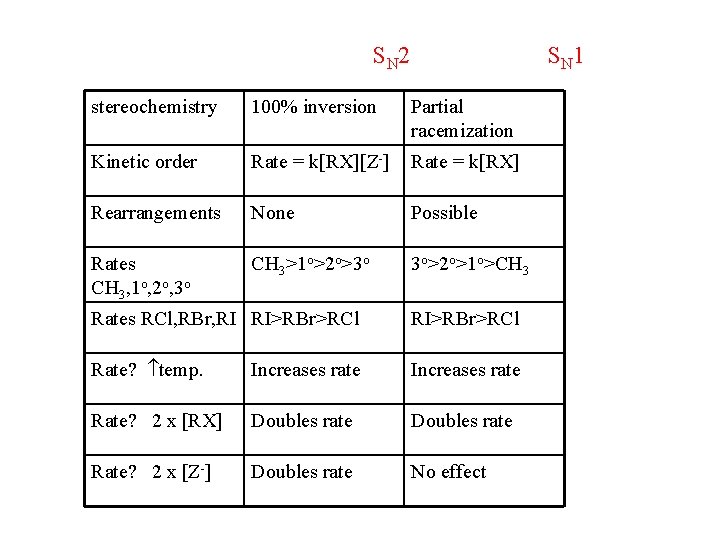
S N 2 S N 1 stereochemistry 100% inversion Partial racemization Kinetic order Rate = k[RX][Z-] Rate = k[RX] Rearrangements None Possible Rates CH 3, 1 o, 2 o, 3 o CH 3>1 o>2 o>3 o 3 o>2 o>1 o>CH 3 Rates RCl, RBr, RI RI>RBr>RCl Rate? temp. Increases rate Rate? 2 x [RX] Doubles rate Rate? 2 x [Z-] Doubles rate No effect

R-X + Z- R-Z + X- which mechanism? S N 2 - CH 3 1 o 2 o 3 o - S N 1 SN 2 “steric factors” CH 3 > 1 o > 2 o > 3 o SN 1 carbocation stability 3 o > 2 o > 1 o > CH 3

Effect of solvent polarity on SN 1/SN 2: water = polar ethanol = less polar Solvent: mixture of ethanol/water Add more water = more polar; add more ethanol = less polar. SN 1: R-W R+ + Wionization favored by polar solvents SN 2: Z: - + R-W Z-R + : Xsolvent polarity does not affect rate

Alkyl halide + base ? ? SN 2: best with CH 3 or 1 o RX, concentrated, strong base (SN 1: 2 o or 3 o, dilute, weak base, polar solvent; rearrangements are possible , alkene by-products )

Synthesis of alkyl halides: 1. From alcohols a) HX b) PX 3 2. Halogenation of certain hydrocarbons 3. 3. (later) 4. 5. 4. (later) 6. 5. Halide exchange for iodide

Reactions of alkyl halides: 1. Nucleophilic substitution Best with 1 o or CH 3!!!!!! 2. R-X + : Z- R-Z + : X- 3. 2. (later) 3. Preparation of Grignard Reagent 4. R-X + Mg RMg. X 4. Reduction 5. R-X + Mg 6. R-X + Sn, HCl R-H + H 2 O R-H
- Slides: 40