Alkyl halides Nucleophilic substitution and elimination reactions E

Alkyl halides Nucleophilic substitution and elimination reactions © E. V. Blackburn, 2010
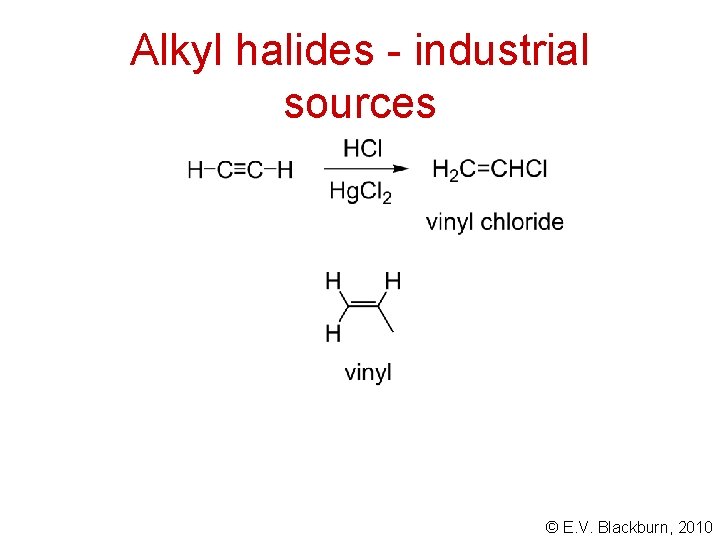
Alkyl halides - industrial sources © E. V. Blackburn, 2010
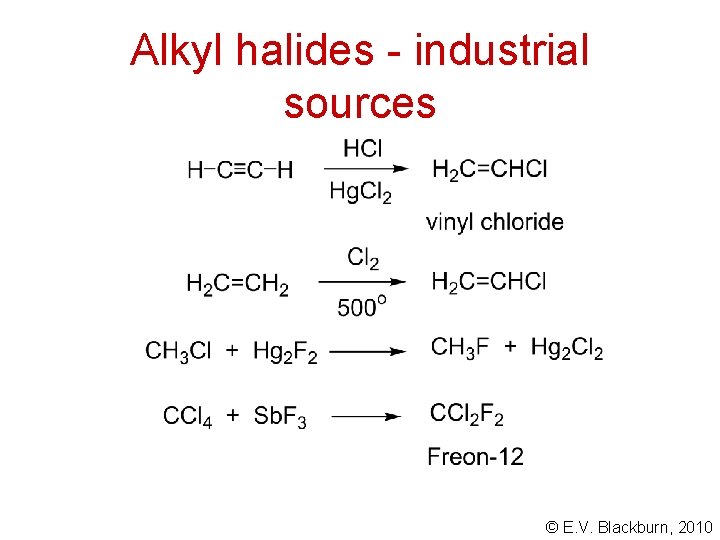
Alkyl halides - industrial sources © E. V. Blackburn, 2010
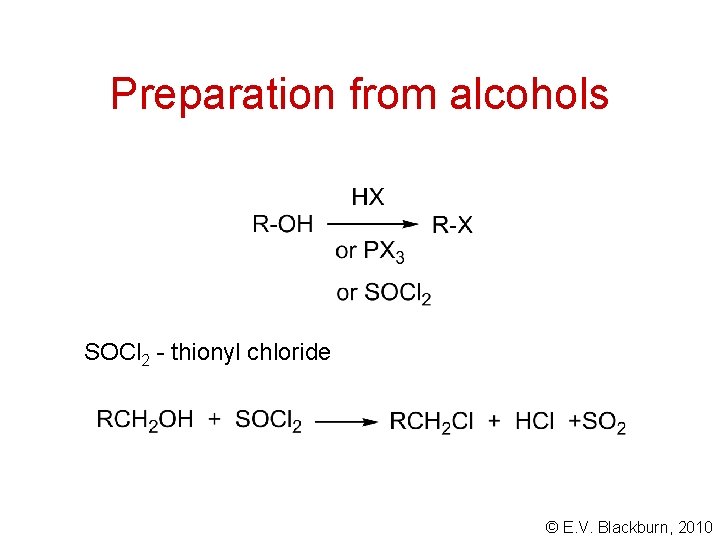
Preparation from alcohols SOCl 2 - thionyl chloride © E. V. Blackburn, 2010
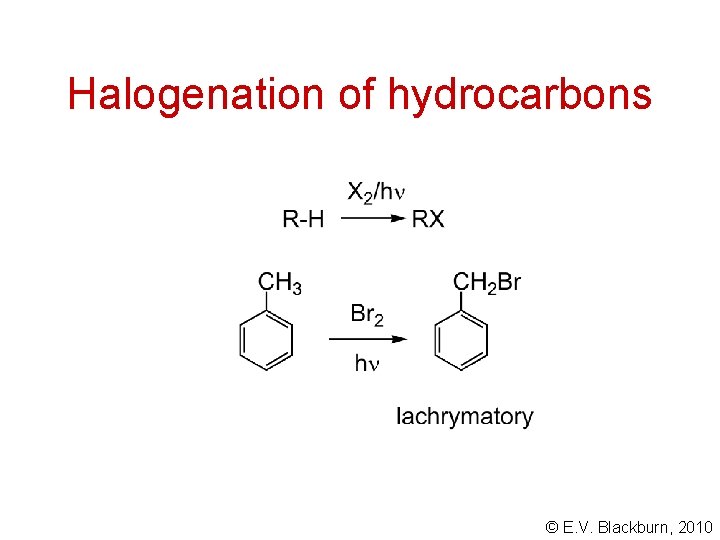
Halogenation of hydrocarbons © E. V. Blackburn, 2010
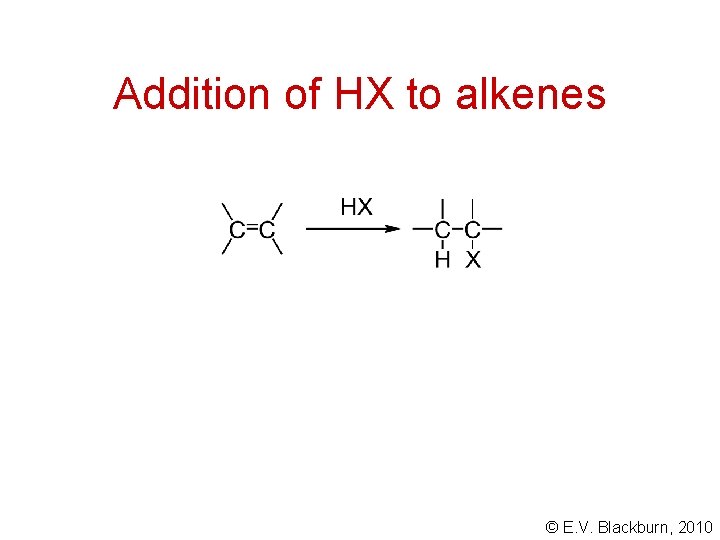
Addition of HX to alkenes © E. V. Blackburn, 2010
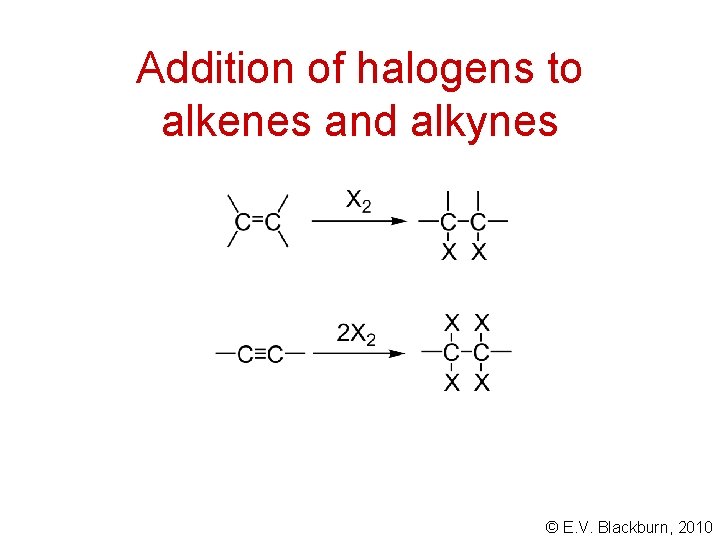
Addition of halogens to alkenes and alkynes © E. V. Blackburn, 2010

Finkelstein reaction © E. V. Blackburn, 2010

Nucleophilic substitution reactions The halide ion is the conjugate base of a strong acid. It is therefore a very weak base and little disposed to share its electrons. When bonded to a carbon, the halogen is easily displaced as a halide ion by stronger nucleophiles - it is a good leaving group. The typical reaction of alkyl halides is a nucleophilic substitution: © E. V. Blackburn, 2010
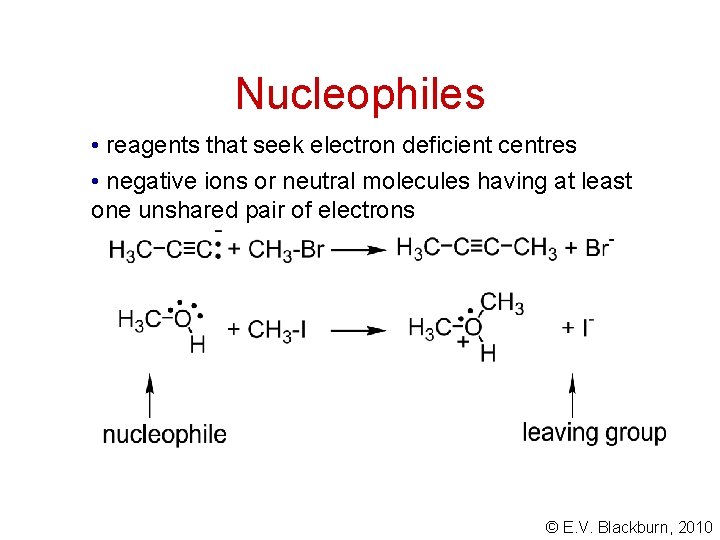
Nucleophiles • reagents that seek electron deficient centres • negative ions or neutral molecules having at least one unshared pair of electrons © E. V. Blackburn, 2010

Leaving groups • a substituent that can leave as a weakly basic molecule or ion © E. V. Blackburn, 2010

Nucleophilic substitution A knowledge of how reaction rates depend on reactant concentrations provides invaluable information about reaction mechanisms. What is known about this reaction? © E. V. Blackburn, 2010
![Nucleophilic substitution [CH 3 Br]I 0. 001 M 0. 002 M [OH-]I 1. 0 Nucleophilic substitution [CH 3 Br]I 0. 001 M 0. 002 M [OH-]I 1. 0](http://slidetodoc.com/presentation_image_h2/98b10e36bc051e422a15b2154af59d53/image-13.jpg)
Nucleophilic substitution [CH 3 Br]I 0. 001 M 0. 002 M [OH-]I 1. 0 M 2. 0 M initial rate 3 x 10 -7 mol L-1 s-1 6 x 10 -7 mol L-1 s-1 1. 2 x 10 -6 mol L-1 s-1 rate a [CH 3 Br][OH-] rate = k[CH 3 Br][OH-] © E. V. Blackburn, 2010

Order - a summary The order of a reaction is equal to the sum of the exponents in the rate equation. Thus for the rate equation rate = k[A]m[B]n, the overall order is m + n. The order with respect to A is m and the order with respect to B is n. © E. V. Blackburn, 2010
![Nucleophilic substitution [(CH 3)3 CBr]I 0. 001 M 0. 002 M [OH-]I 1. 0 Nucleophilic substitution [(CH 3)3 CBr]I 0. 001 M 0. 002 M [OH-]I 1. 0](http://slidetodoc.com/presentation_image_h2/98b10e36bc051e422a15b2154af59d53/image-15.jpg)
Nucleophilic substitution [(CH 3)3 CBr]I 0. 001 M 0. 002 M [OH-]I 1. 0 M 2. 0 M initial rate 4 x 10 -7 mol L-1 s-1 8 x 10 -7 mol L-1 s-1 rate a [(CH 3)3 CBr][OH-]0 rate = k[(CH 3)3 CBr] © E. V. Blackburn, 2010
![The SN 2 mechanism rate = k[CH 3 Br][OH-] References of interest: E. D. The SN 2 mechanism rate = k[CH 3 Br][OH-] References of interest: E. D.](http://slidetodoc.com/presentation_image_h2/98b10e36bc051e422a15b2154af59d53/image-16.jpg)
The SN 2 mechanism rate = k[CH 3 Br][OH-] References of interest: E. D. Hughes, C. K. Ingold, and C. S. Patel, J. Chem. Soc. , 526 (1933) J. L. Gleave, E. D. Hughes and C. K. Ingold, J. Chem. Soc. , 236 (1935) © E. V. Blackburn, 2010
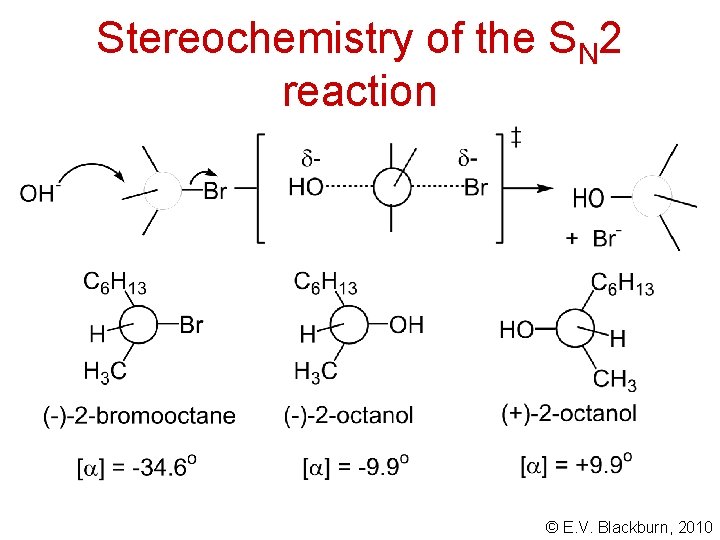
Stereochemistry of the SN 2 reaction © E. V. Blackburn, 2010
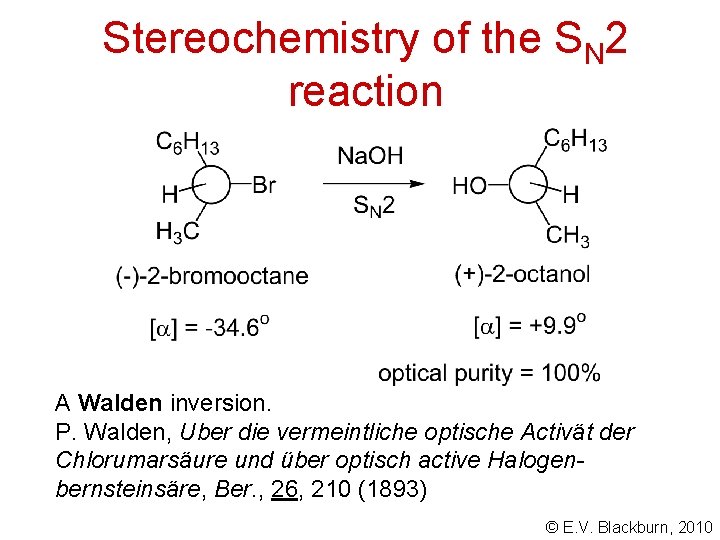
Stereochemistry of the SN 2 reaction A Walden inversion. P. Walden, Uber die vermeintliche optische Activät der Chlorumarsäure und über optisch active Halogenbernsteinsäre, Ber. , 26, 210 (1893) © E. V. Blackburn, 2010
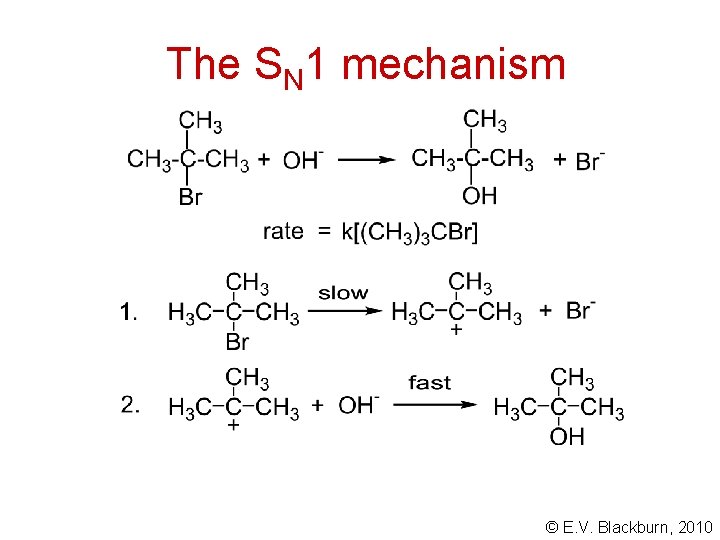
The SN 1 mechanism © E. V. Blackburn, 2010
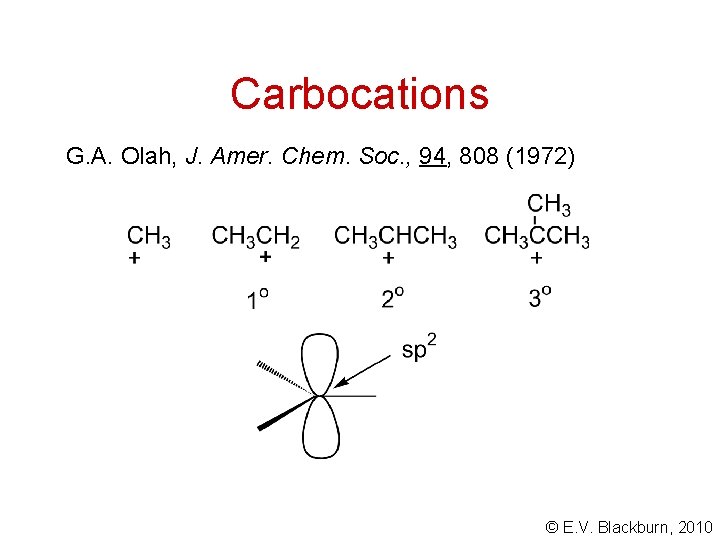
Carbocations G. A. Olah, J. Amer. Chem. Soc. , 94, 808 (1972) © E. V. Blackburn, 2010

Carbocation stability Hyperconjugation stabilizes the positive charge. © E. V. Blackburn, 2010
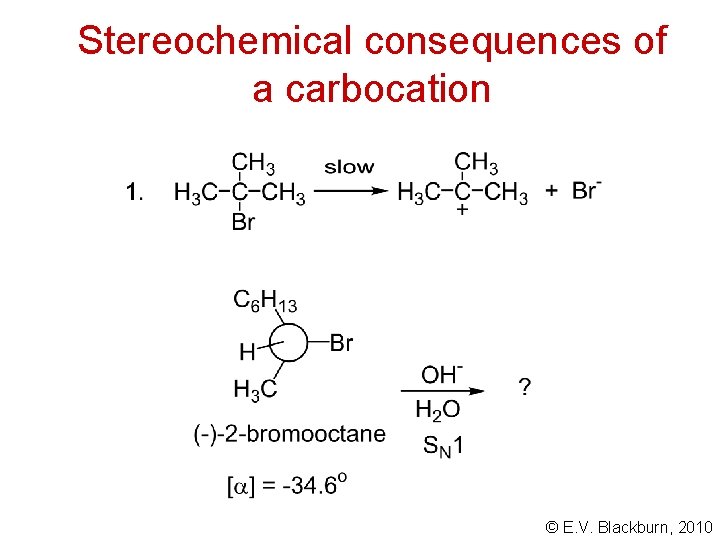
Stereochemical consequences of a carbocation © E. V. Blackburn, 2010
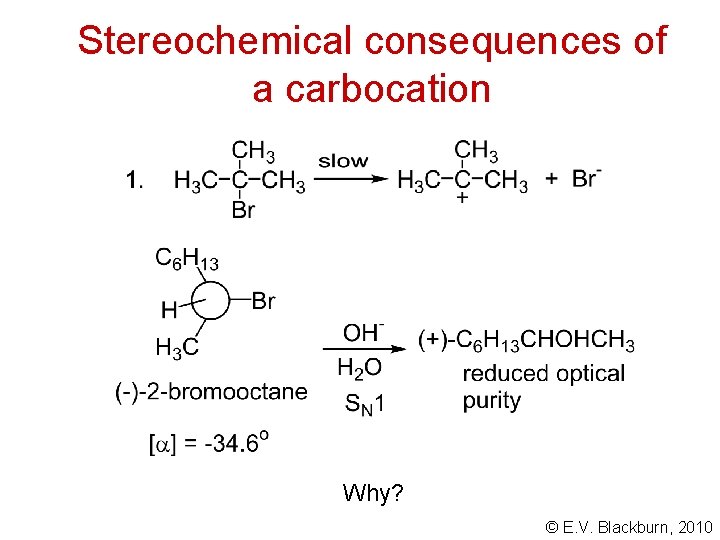
Stereochemical consequences of a carbocation Why? © E. V. Blackburn, 2010
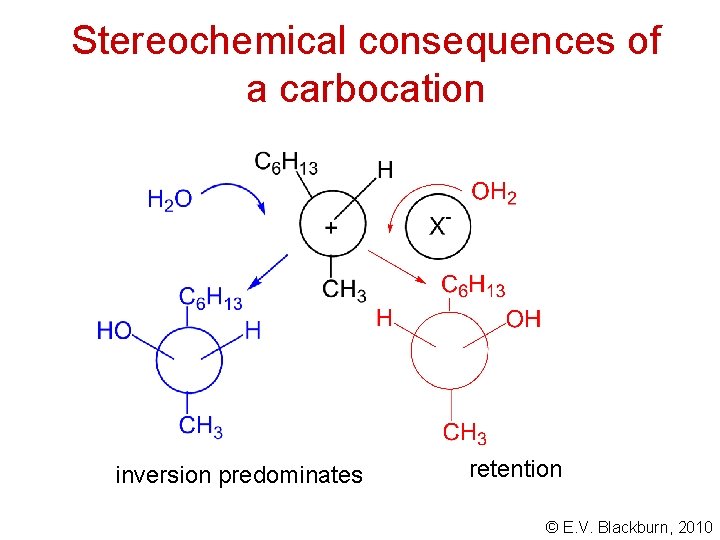
Stereochemical consequences of a carbocation inversion predominates retention © E. V. Blackburn, 2010
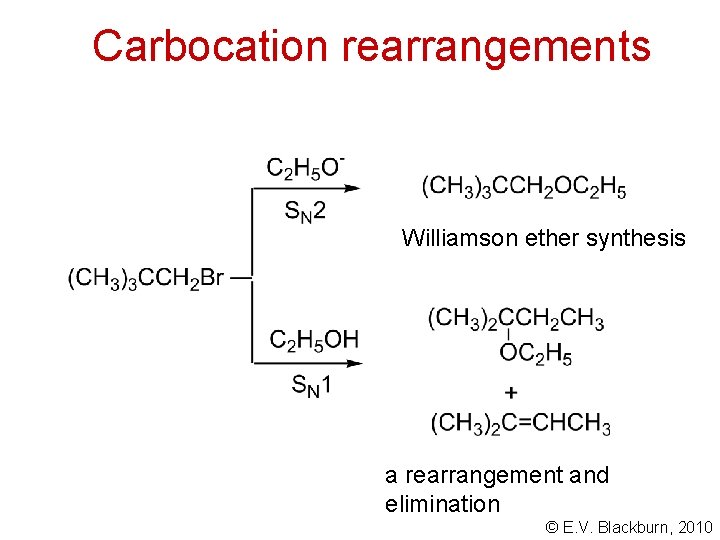
Carbocation rearrangements Williamson ether synthesis a rearrangement and elimination © E. V. Blackburn, 2010
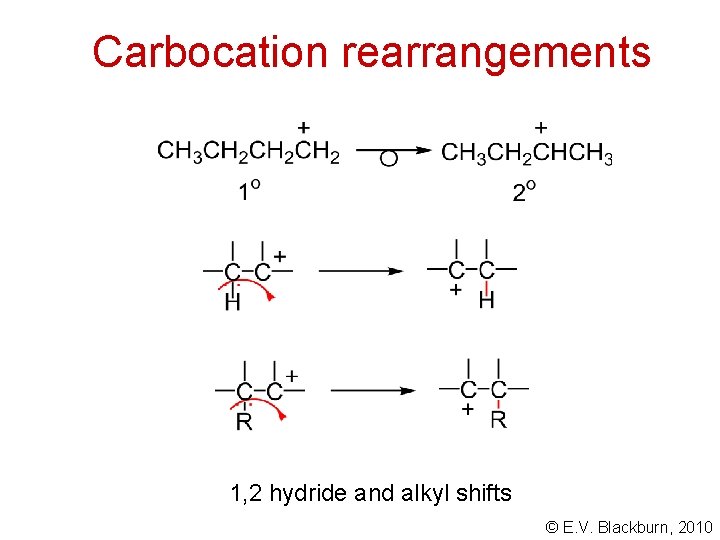
Carbocation rearrangements 1, 2 hydride and alkyl shifts © E. V. Blackburn, 2010
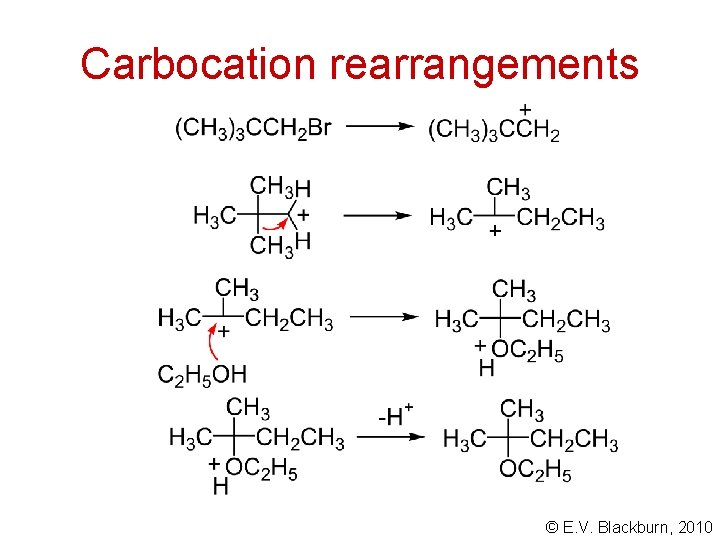
Carbocation rearrangements © E. V. Blackburn, 2010
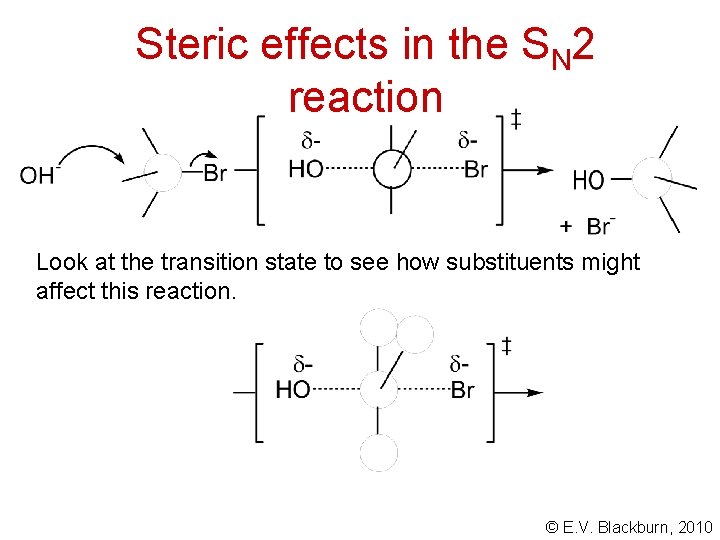
Steric effects in the SN 2 reaction Look at the transition state to see how substituents might affect this reaction. © E. V. Blackburn, 2010

Steric effects in the SN 2 reaction The order of reactivity of RX in these SN 2 reactions is CH 3 X > 1 o > 2 o > 3 o © E. V. Blackburn, 2010

Steric effects in the SN 2 reaction © E. V. Blackburn, 2010

Structural effects in SN 1 reactions 3 o > 2 o > 1 o > CH 3 X © E. V. Blackburn, 2010

Nucleophilicity Rates of SN 2 reactions depend on concentration and nucleophilicity of the nucleophile. A base is more nucleophilic than its conjugate acid: CH 3 Cl + H 2 O CH 3 OH 2+ slow CH 3 Cl + HO- CH 3 OH fast The nucleophilicity of nucleophiles having the same nucleophilic atom parallels basicity: RO- > HO- >> RCO 2 - > ROH >H 2 O © E. V. Blackburn, 2010

Nucleophilicity When the nucleophilic atoms are different, their relative strengths do not always parallel their basicity. In protic solvents, the larger the nucleophilic atom, the better: I- > Br- > Cl- > FIn protic solvents, the smaller the anion, the greater its solvation due to hydrogen bonding. This shell of solvent molecules reduces its ability to attack. © E. V. Blackburn, 2010

Nucleophilicity Aprotic solvents tend to solvate cations rather than anions. Thus the unsolvated anion has a greater nucleophilicity in an aprotic solvent. © E. V. Blackburn, 2010
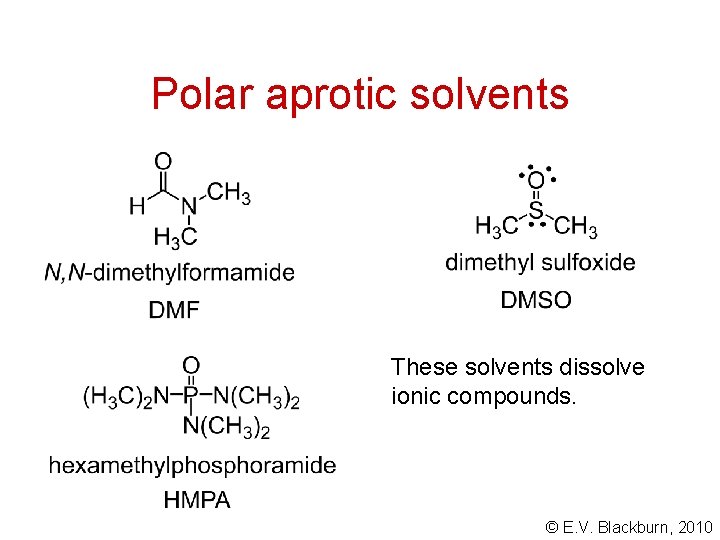
Polar aprotic solvents These solvents dissolve ionic compounds. © E. V. Blackburn, 2010
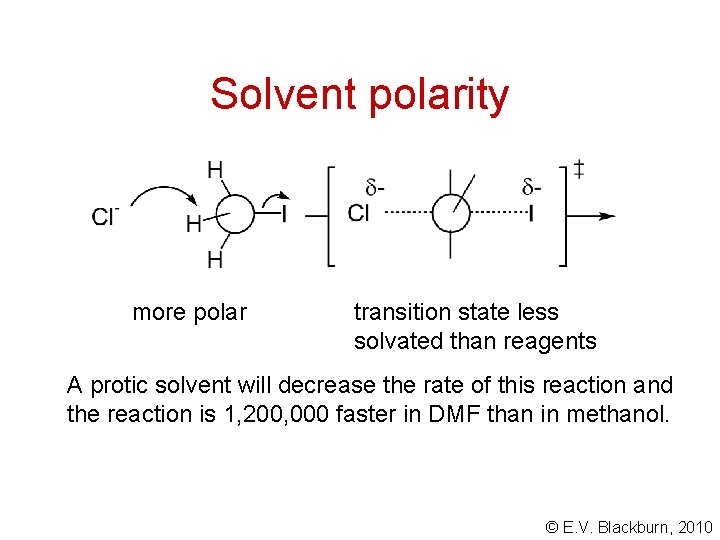
Solvent polarity more polar transition state less solvated than reagents A protic solvent will decrease the rate of this reaction and the reaction is 1, 200, 000 faster in DMF than in methanol. © E. V. Blackburn, 2010

Solvent polarity less polar more polar greater stabilization by polar solvent The transition state is more polarized. Therefore the rate of this reaction increases with increase in solvent polarity. A protic solvent is particularly effective as it stabilizes the transition state by forming hydrogen bonds with the leaving group. © E. V. Blackburn, 2010

Solvent polarity Explain the solvent effects for each of the following second order reactions: a) 131 I- + CH 3 I CH 3131 I + IRelative rates: in water, 1; in methanol, 16; in ethanol, 44 b) (n-C 3 H 7)3 N + CH 3 I (n-C 3 H 7)3 N+CH 3 IRelative rates: in n-hexane, 1; in chloroform, 13 000 © E. V. Blackburn, 2010

Leaving group ability Weak bases are good leaving groups. They are better able to accommodate a negative charge and therefore stabilize the transition state. Thus I- is a better leaving group than Br-. I- > Br- > Cl- > H 2 O > F- > OH- © E. V. Blackburn, 2010

S N 1 v S N 2 kinetics: S N 1 S N 2 1 st order second order reactivity: 3 o > 2 o > 1 o > CH 3 X rearrangements partial inversion CH 3 X > 1 o > 2 o > 3 o no rearrangements inversion of configuration eliminations possible © E. V. Blackburn, 2010

Problems Try problems 6. 6 – 6. 11 and 6. 14 – 6. 16 in chapter 6 of Solomons and Fryhle. © E. V. Blackburn, 2010
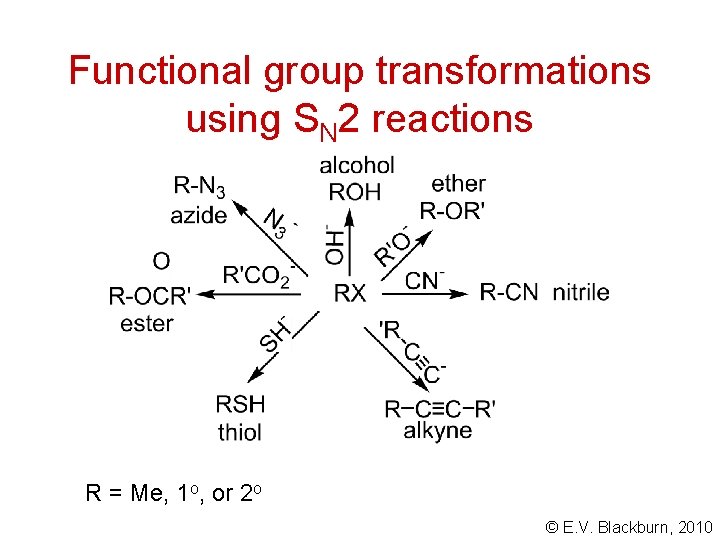
Functional group transformations using SN 2 reactions R = Me, 1 o, or 2 o © E. V. Blackburn, 2010

Problems Try problems 6. 12 and 6. 17 in chapter 6 of Solomons and Fryhle. © E. V. Blackburn, 2010

ROH + HX - an SN reaction © E. V. Blackburn, 2010
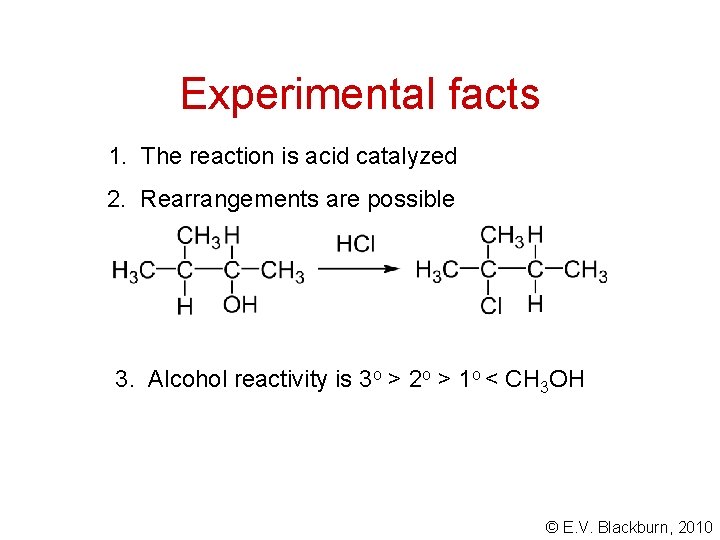
Experimental facts 1. The reaction is acid catalyzed 2. Rearrangements are possible 3. Alcohol reactivity is 3 o > 2 o > 1 o < CH 3 OH © E. V. Blackburn, 2010

The mechanism © E. V. Blackburn, 2010

Reaction of primary alcohols with HX S N 2 © E. V. Blackburn, 2010
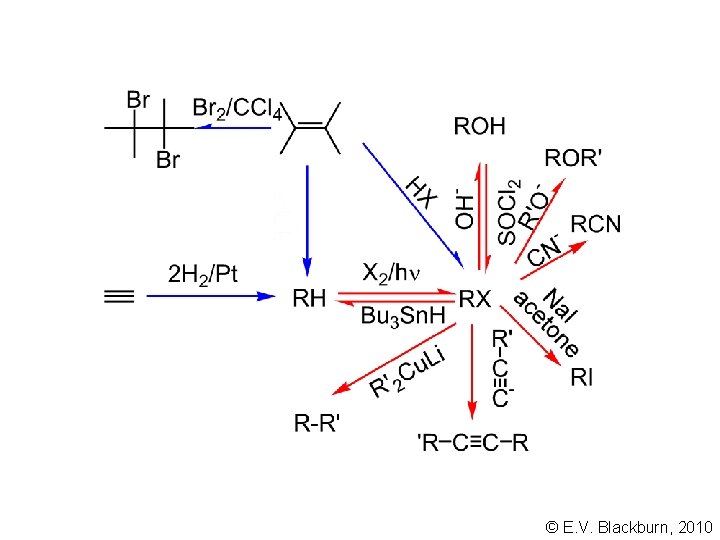
© E. V. Blackburn, 2010
- Slides: 48