Alkyl Halides 145 Chem 1 Alkyl Halides 145





- Slides: 15

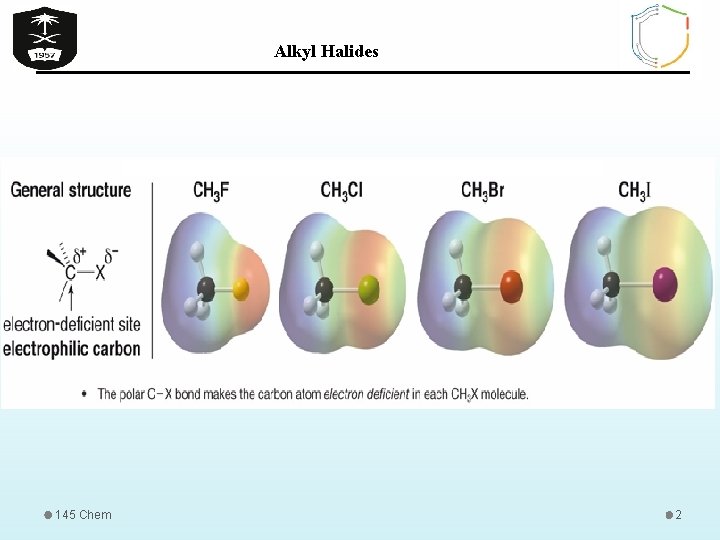
Alkyl Halides 145 Chem 1

Alkyl Halides 145 Chem 2

Alkyl Halides Classes and Names of Halogen Compounds According to hydrocarbon gp. 1 -Alkyl halides( R-X) : has a halogen atom bonded to one sp 3 hybrid C atom 2 -Allylic halides : has a halogen atom bonded to one sp 3 hybrid C atom 3 -Vinylic halides : has a halogen atom bonded to one sp 2 hybrid C atom 4 -Benzylic halides : has a halogen atom bonded to Carbone away from aromatic ring 5 -Aryl halides : has a halogen atom bonded directly to an aromatic ring 1. Alkyl halides( R-X) • CH 3 -X and R-CH 2 -X : Primary alkyl halide. • (R)2 -CH-X : Secondary alkyl halide. • (R)3 -C-X : Tertiary alkyl halide. 145 Chem 3

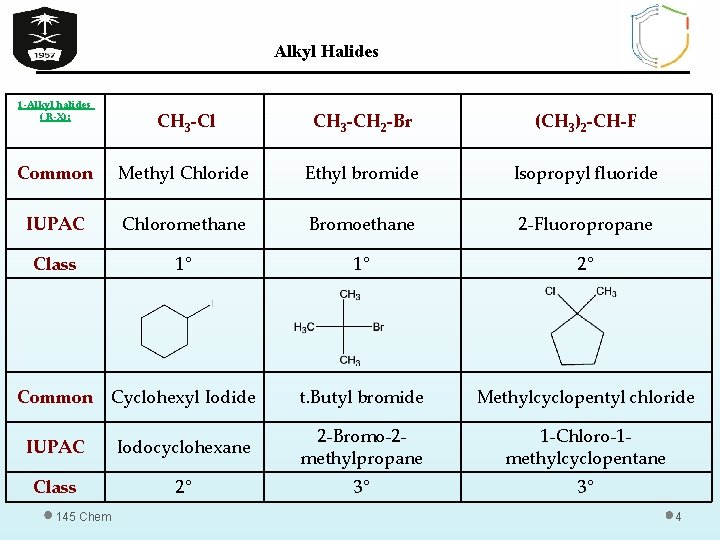
Alkyl Halides 1 -Alkyl halides ( R-X): CH 3 -Cl CH 3 -CH 2 -Br (CH 3)2 -CH-F Common Methyl Chloride Ethyl bromide Isopropyl fluoride IUPAC Chloromethane Bromoethane 2 -Fluoropropane Class 1° 1° 2° Common Cyclohexyl Iodide t. Butyl bromide Methylcyclopentyl chloride IUPAC Iodocyclohexane 2 -Bromo-2 methylpropane 1 -Chloro-1 methylcyclopentane Class 2° 3° 3° 145 Chem 4

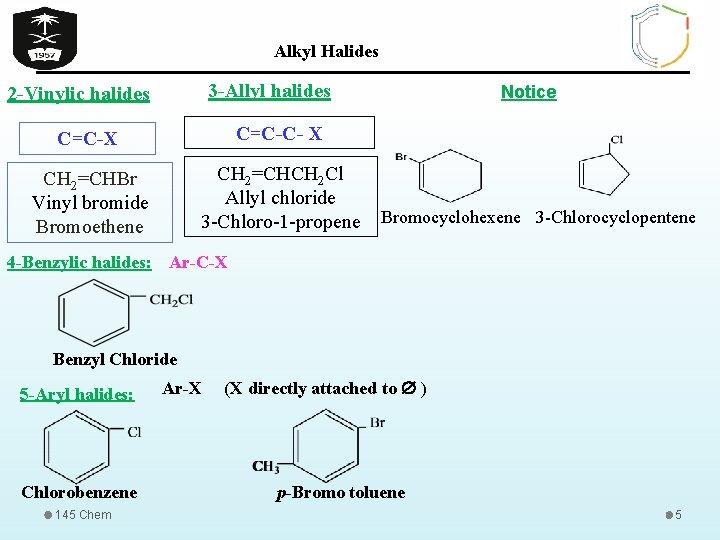
Alkyl Halides 3 -Allyl halides 2 -Vinylic halides C=C-X C=C-C- X CH 2=CHBr Vinyl bromide Bromoethene CH 2=CHCH 2 Cl Allyl chloride 3 -Chloro-1 -propene Notice Bromocyclohexene 3 -Chlorocyclopentene 4 -Benzylic halides: Ar-C-X Benzyl Chloride 5 -Aryl halides: Chlorobenzene 145 Chem Ar-X (X directly attached to ) p-Bromo toluene 5

Alkyl Halides PHYSICAL PROPERTIES OF ORGANIC HALIDES • All organic halides are insoluble in water and soluble in common organic solvents. • In an alkyl halide, the halogen is more electronegative than carbon • The boiling points increases with increasing in molecular weights. • Alkyl halides have higher melting point than the corresponding alkanes, alkenes, and alkynes because: 1. Polarity 2. Molecular weight (In series of halides BP. F <Cl <Br <I) CH 3 CH 2 F bp= 470 C 145 Chem CH 3 CH 2 Br bp= 710 C CH 3 CH 2 I bp= 1020 C 6

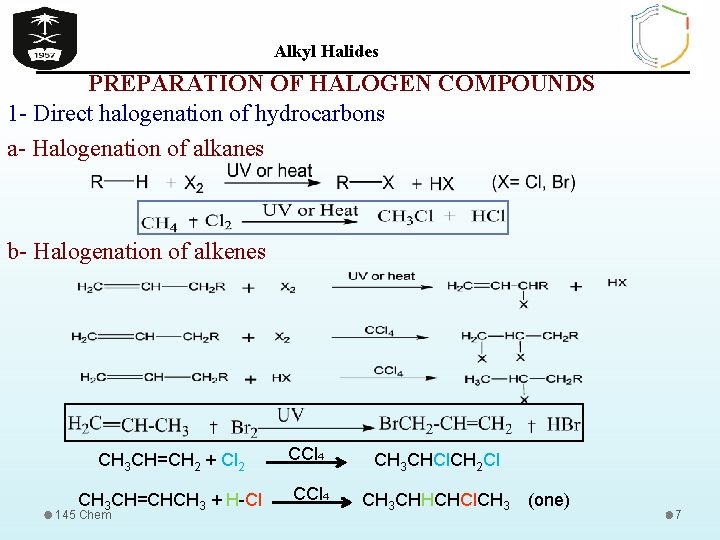
Alkyl Halides PREPARATION OF HALOGEN COMPOUNDS 1 - Direct halogenation of hydrocarbons a- Halogenation of alkanes b- Halogenation of alkenes CH 3 CH=CH 2 + Cl 2 CH 3 CH=CHCH 3 + H-Cl 145 Chem CCl 4 CH 3 CHCl. CH 2 Cl CH 3 CHHCHCl. CH 3 (one) 7

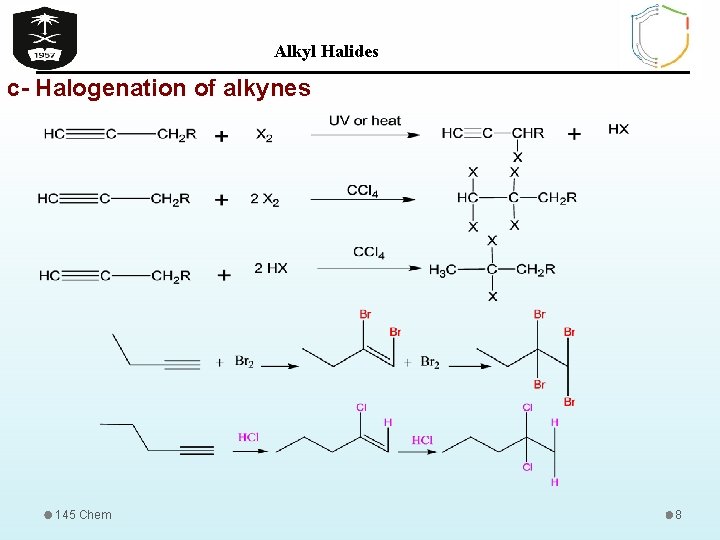
Alkyl Halides c- Halogenation of alkynes 145 Chem 8

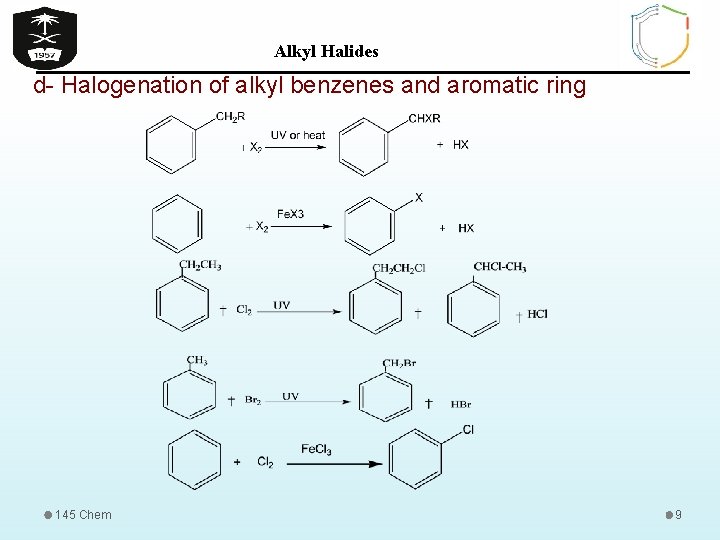
Alkyl Halides d- Halogenation of alkyl benzenes and aromatic ring 145 Chem 9

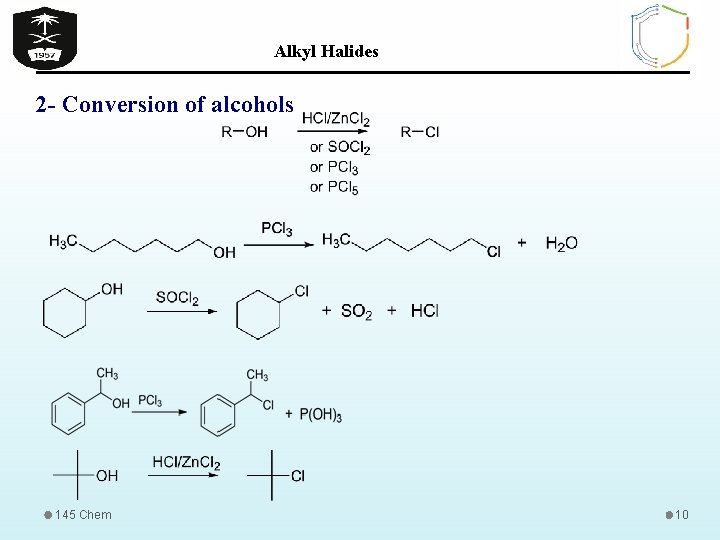
Alkyl Halides 2 - Conversion of alcohols 145 Chem 10

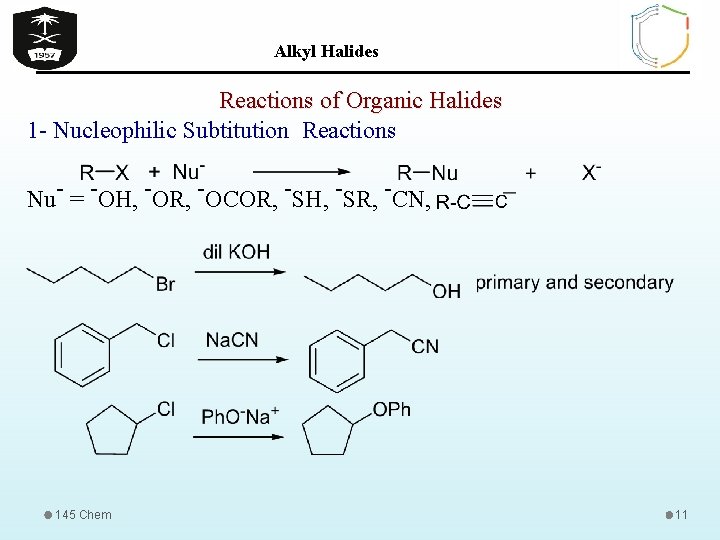
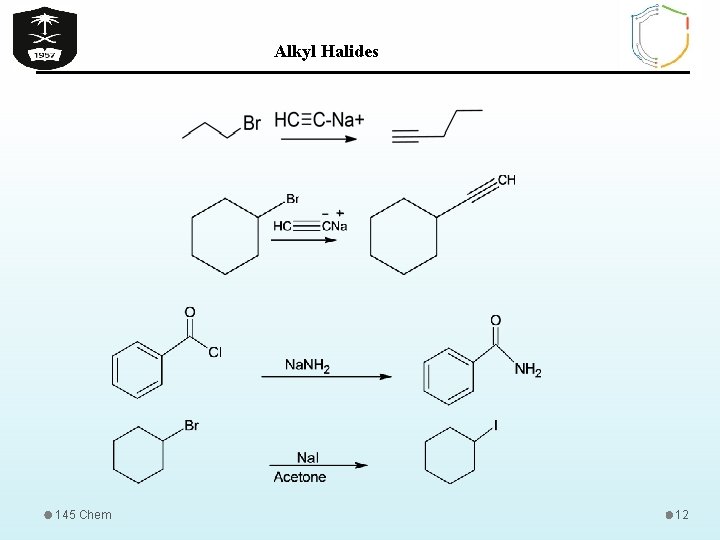
Alkyl Halides Reactions of Organic Halides 1 - Nucleophilic Subtitution Reactions Nu- = -OH, -OR, -OCOR, -SH, -SR, -CN, 145 Chem 11

Alkyl Halides 145 Chem 12

Alkyl Halides 2 - Elimination Reactions 145 Chem 13

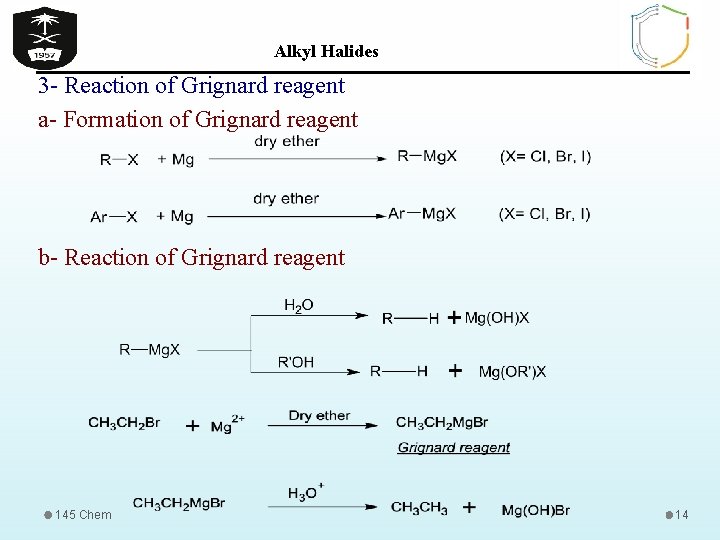
Alkyl Halides 3 - Reaction of Grignard reagent a- Formation of Grignard reagent b- Reaction of Grignard reagent 145 Chem 14

Alkyl Halides Thank You for your kind attention ! Questions? Comments 15