Alkenes Properties Nomenclature Stability Addition Reactions Ethylene Electron

Alkenes Properties Nomenclature Stability Addition Reactions
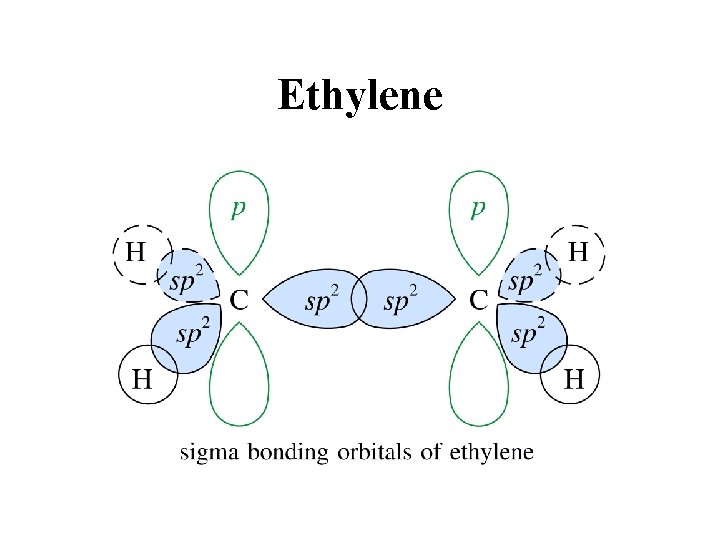
Ethylene
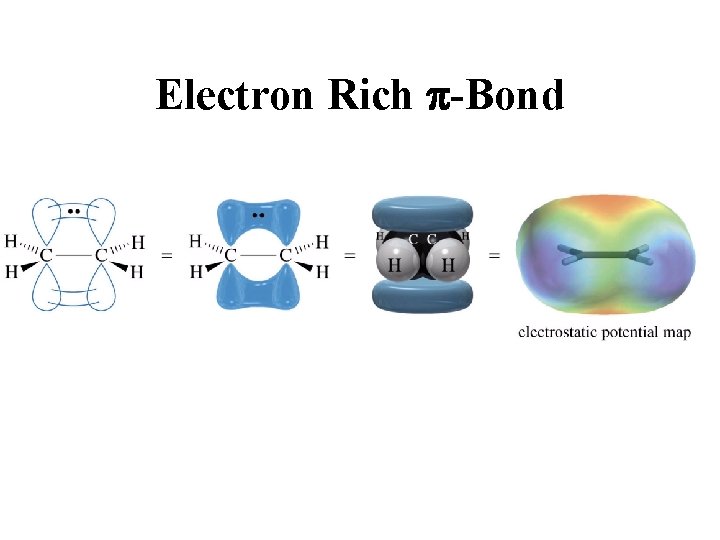
Electron Rich p-Bond
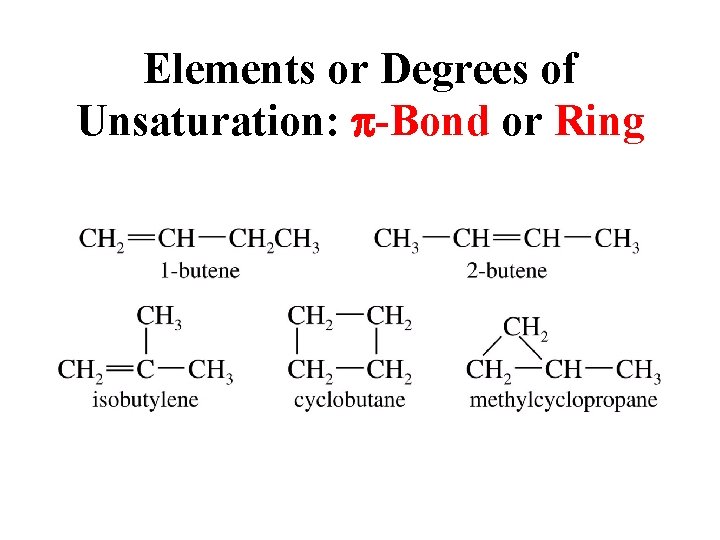
Elements or Degrees of Unsaturation: p-Bond or Ring
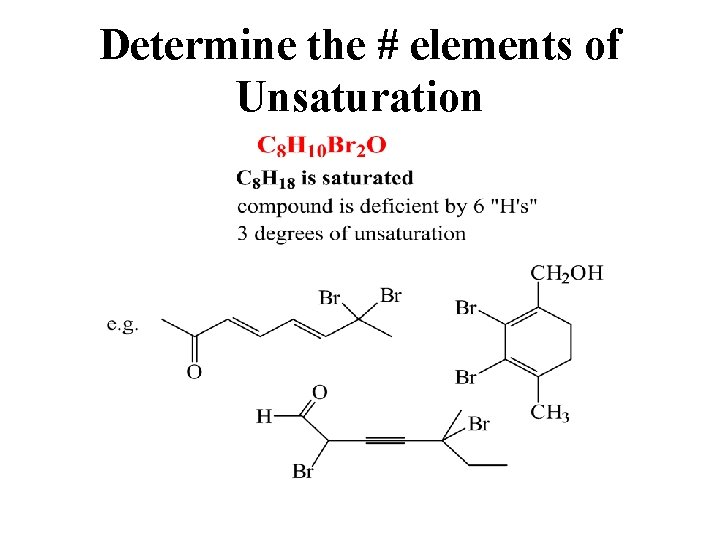
Determine the # elements of Unsaturation

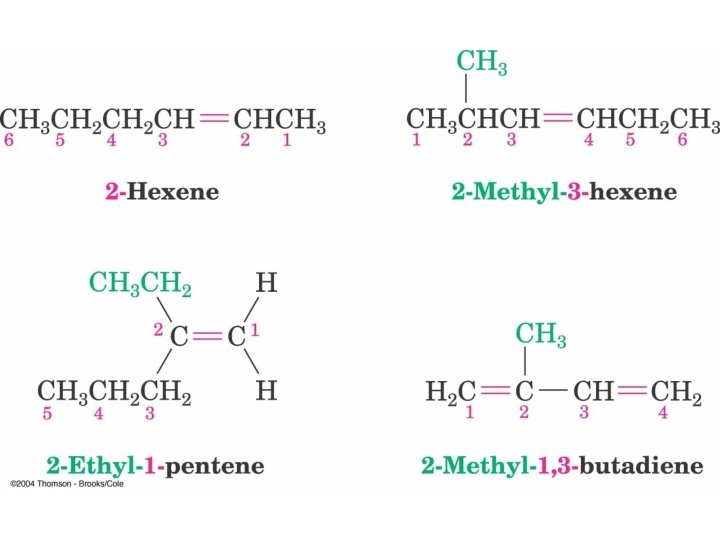
Alkene Nomenclature
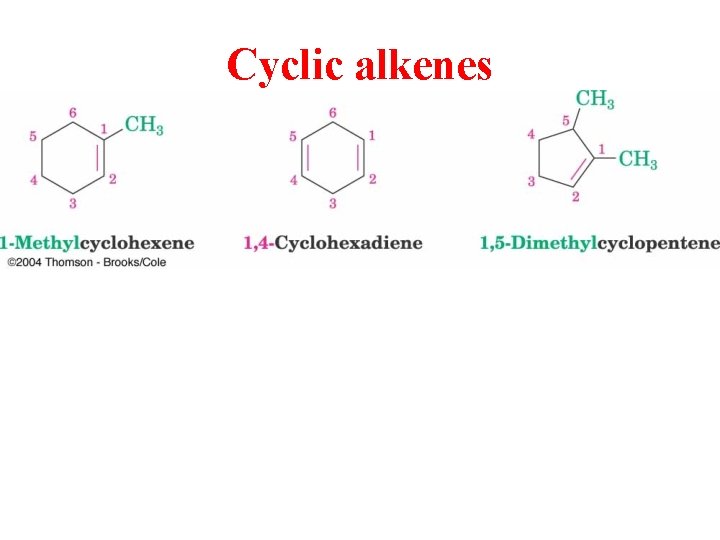
Cyclic alkenes
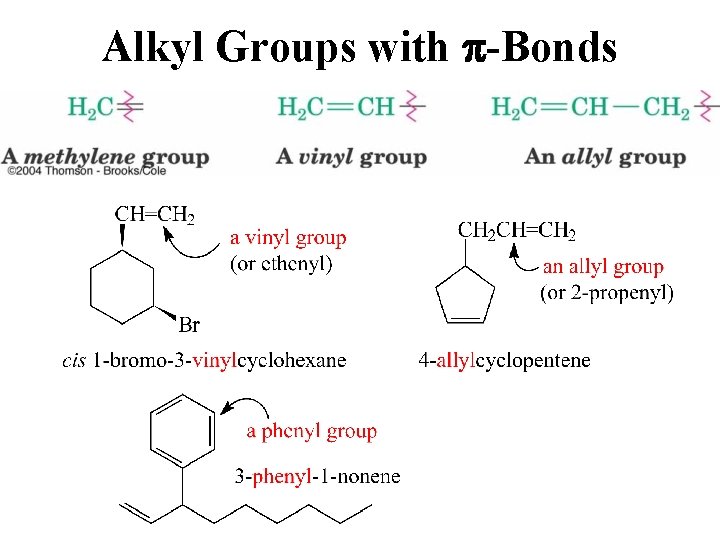
Alkyl Groups with p-Bonds
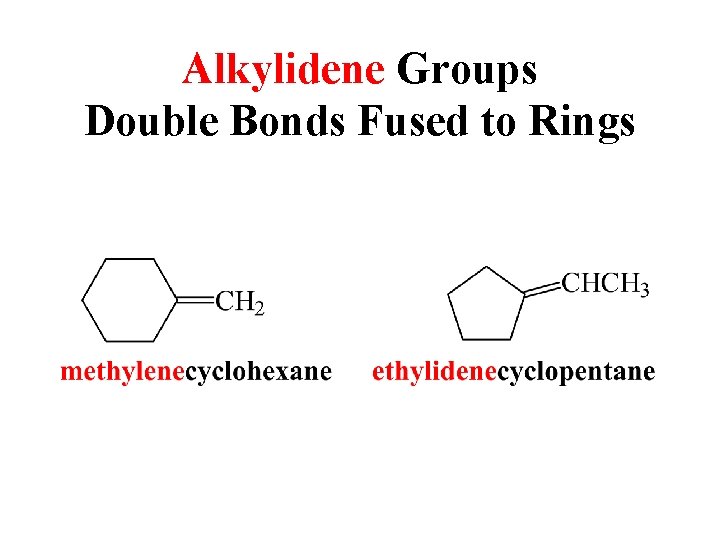
Alkylidene Groups Double Bonds Fused to Rings
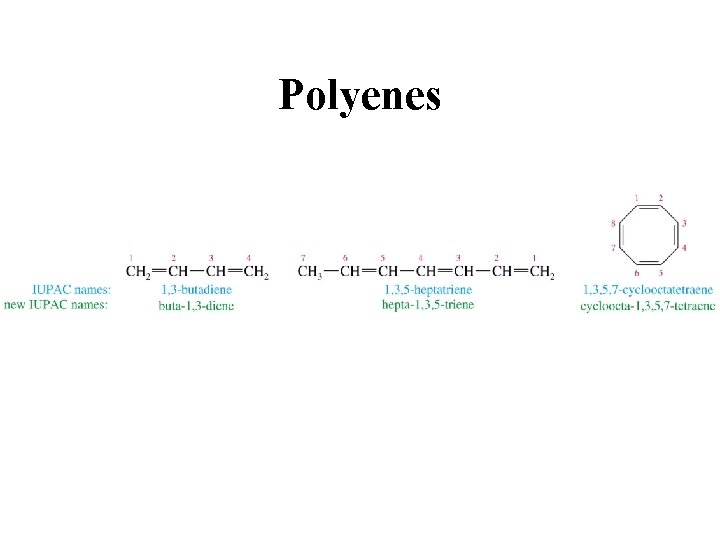
Polyenes
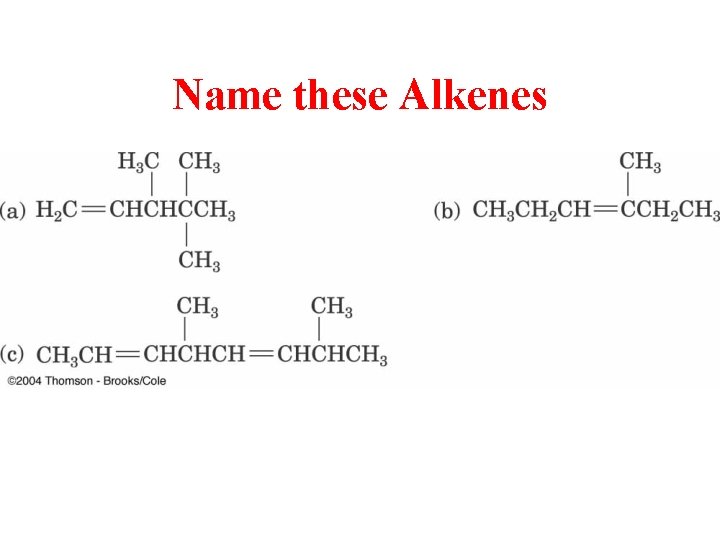
Name these Alkenes
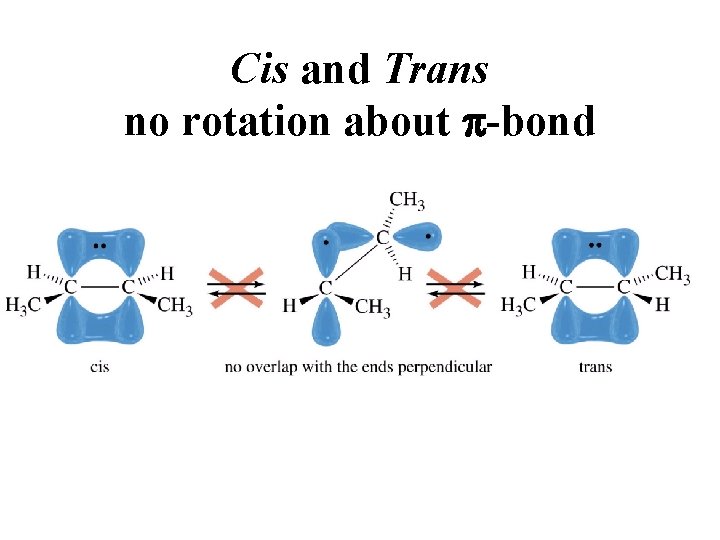
Cis and Trans no rotation about p-bond
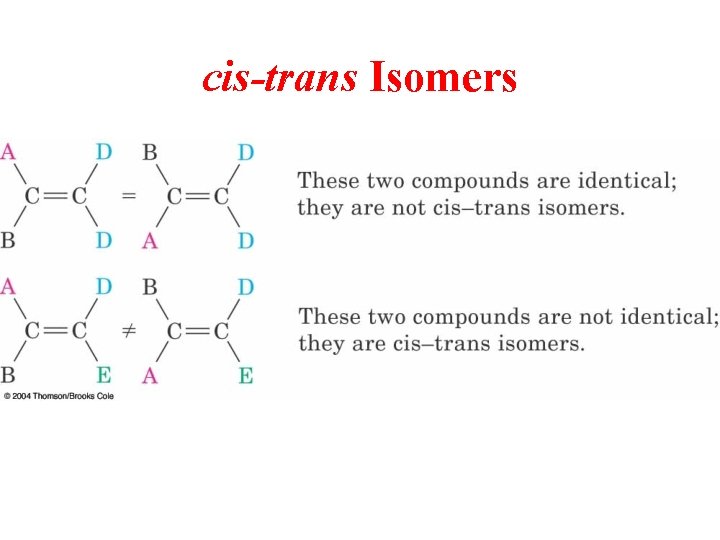
cis-trans Isomers
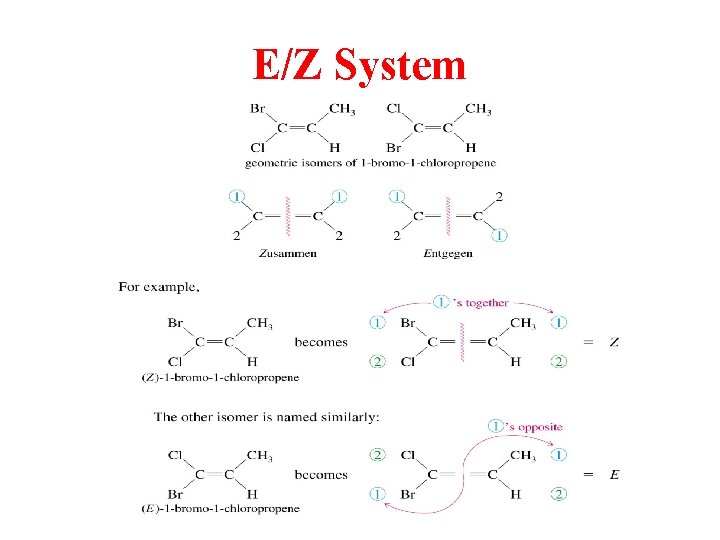
E/Z System
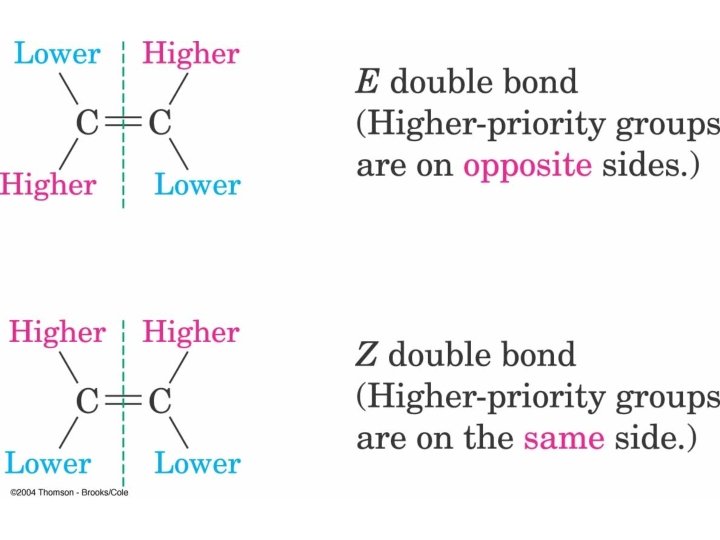
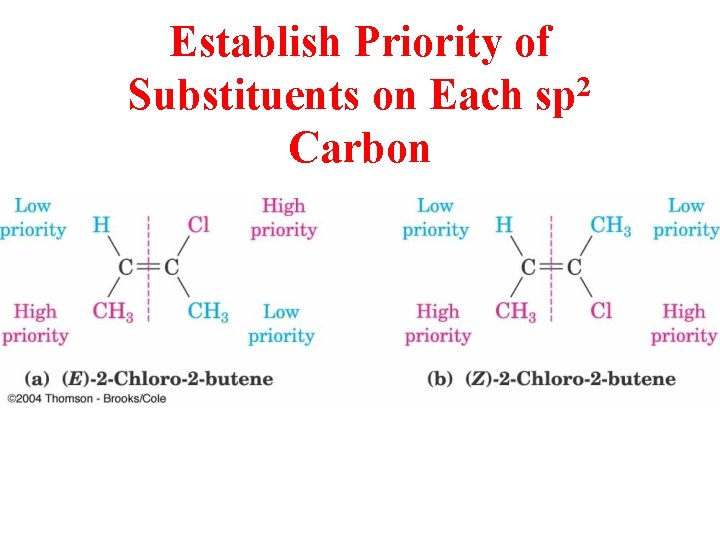
Establish Priority of 2 Substituents on Each sp Carbon
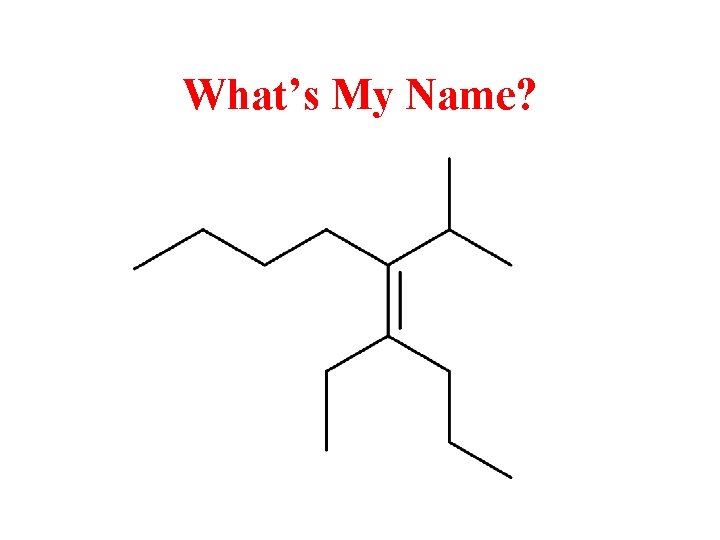
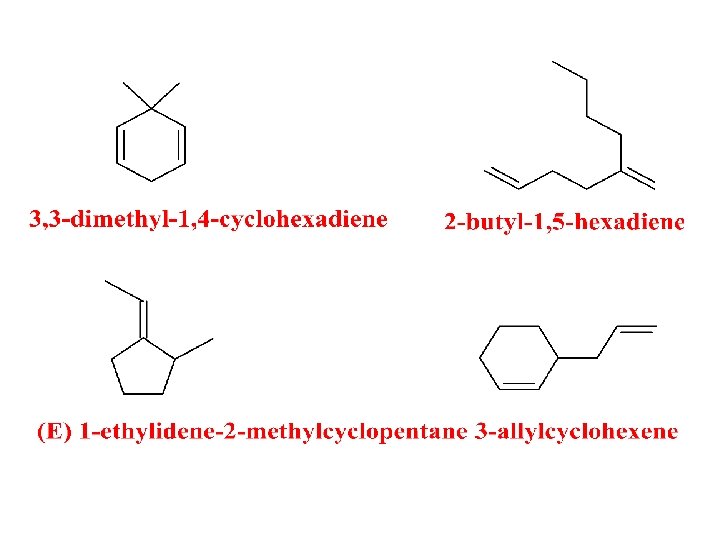
What’s My Name?
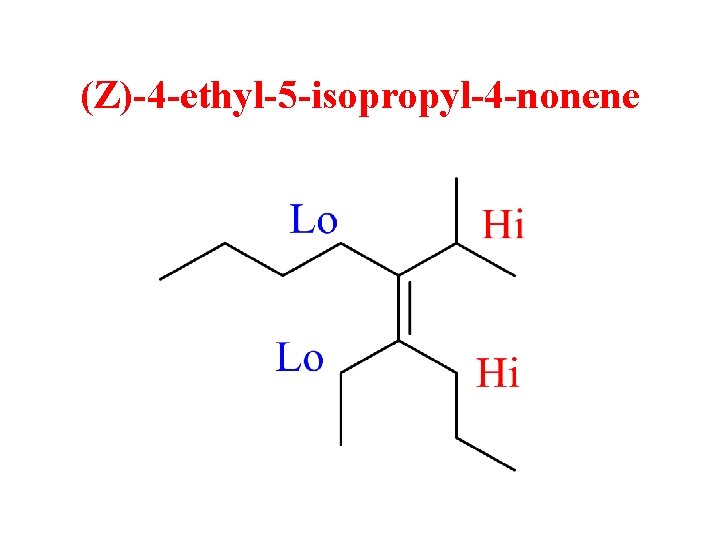
(Z)-4 -ethyl-5 -isopropyl-4 -nonene
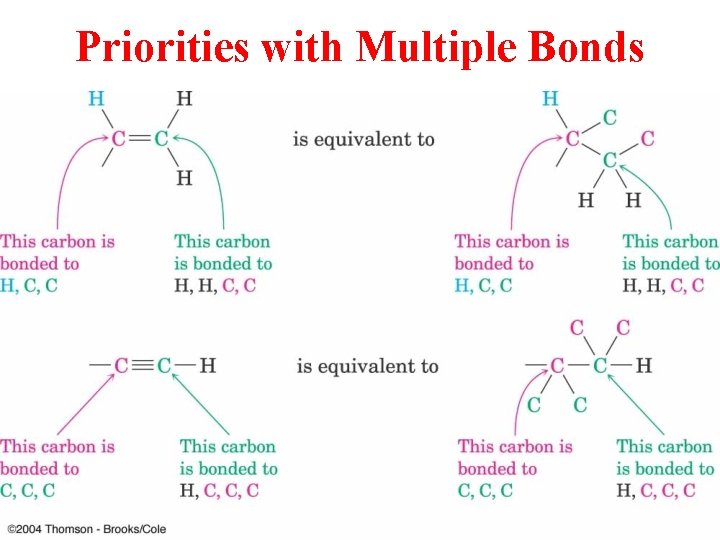
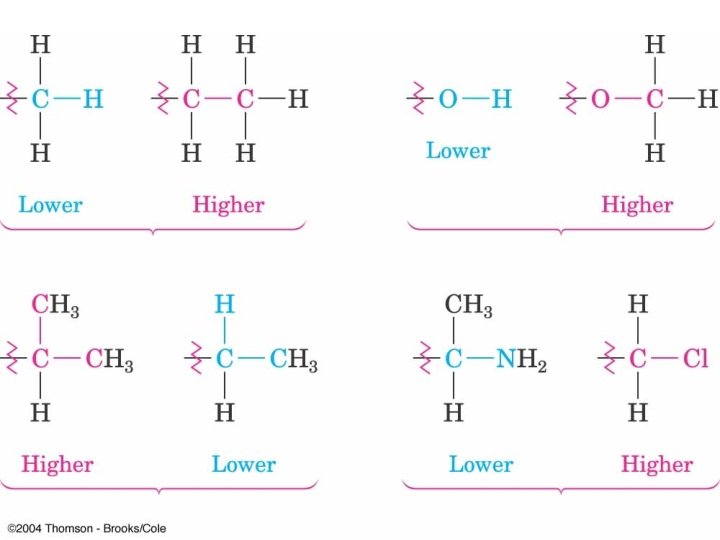
Priorities with Multiple Bonds
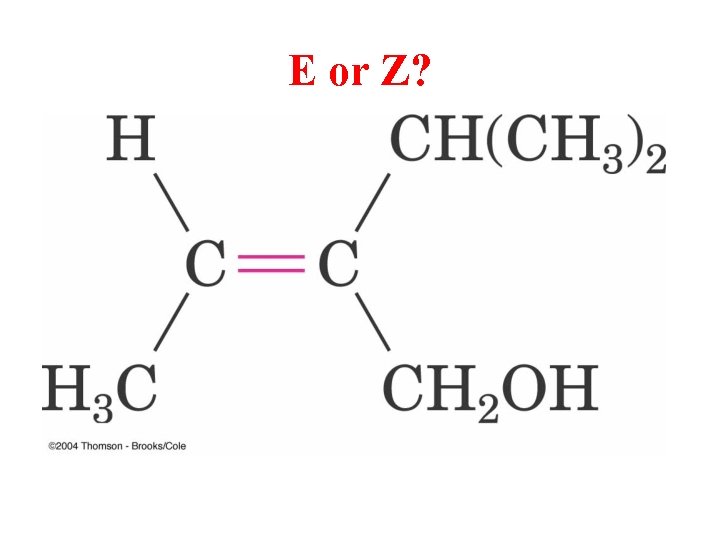
E or Z?
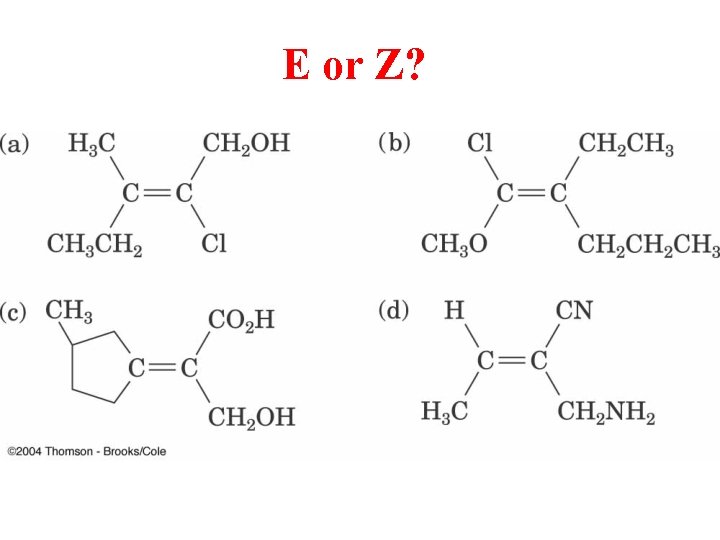
E or Z?
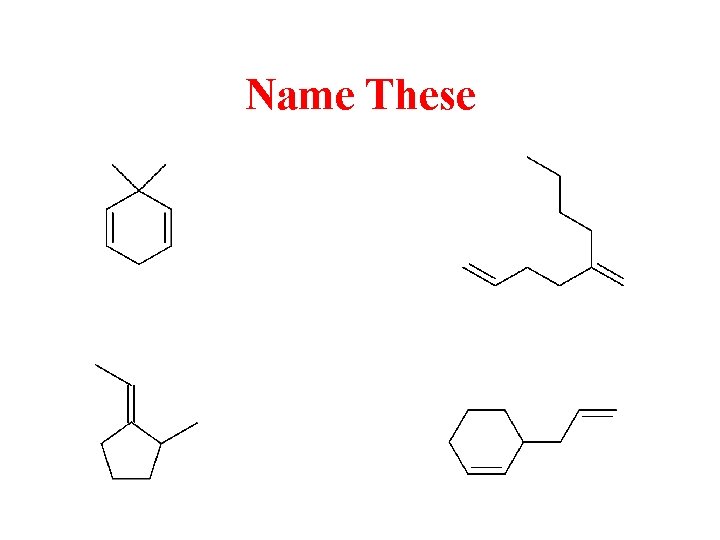
Name These

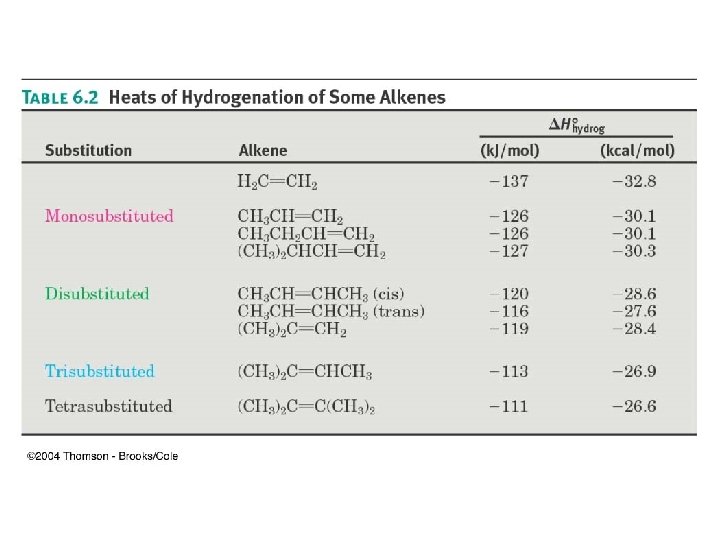
Hydrogenation Data Helps to Determine Stability DHhydrogenation of Alkenes
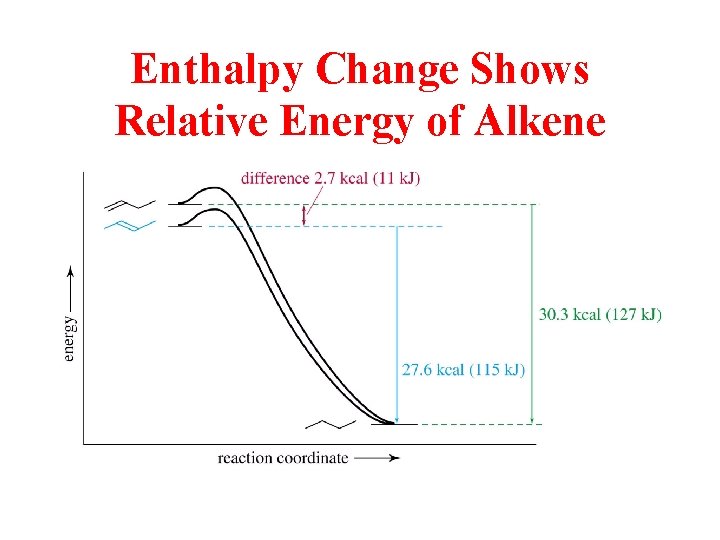
Enthalpy Change Shows Relative Energy of Alkene
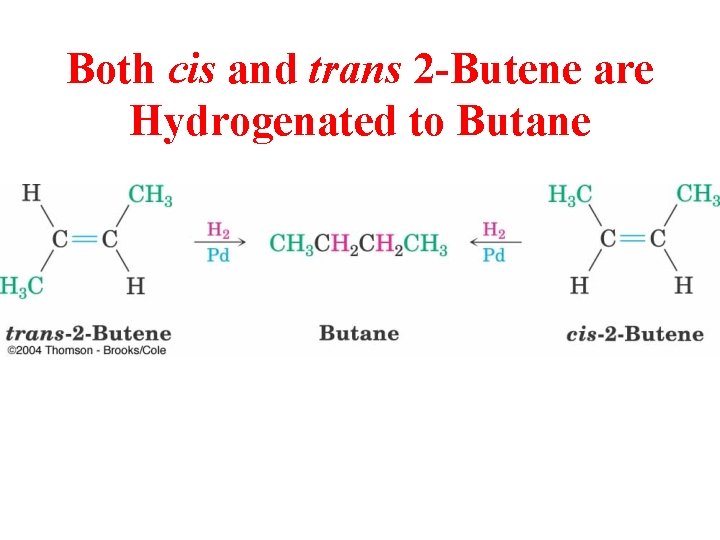
Both cis and trans 2 -Butene are Hydrogenated to Butane
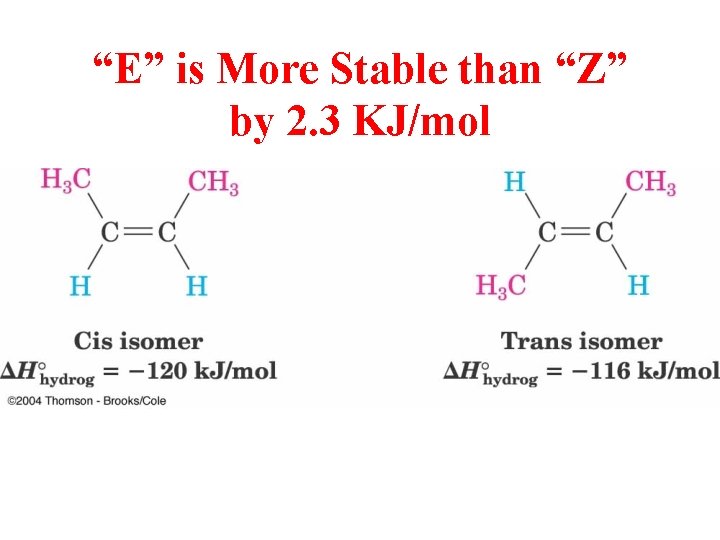
“E” is More Stable than “Z” by 2. 3 KJ/mol
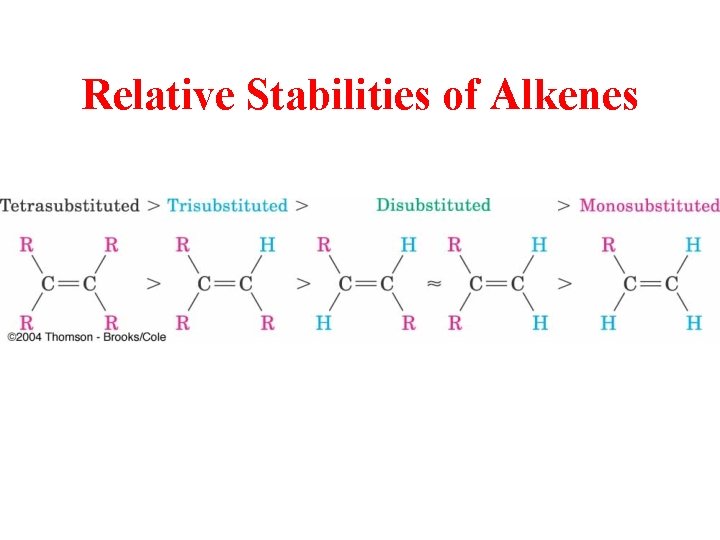
Relative Stabilities of Alkenes
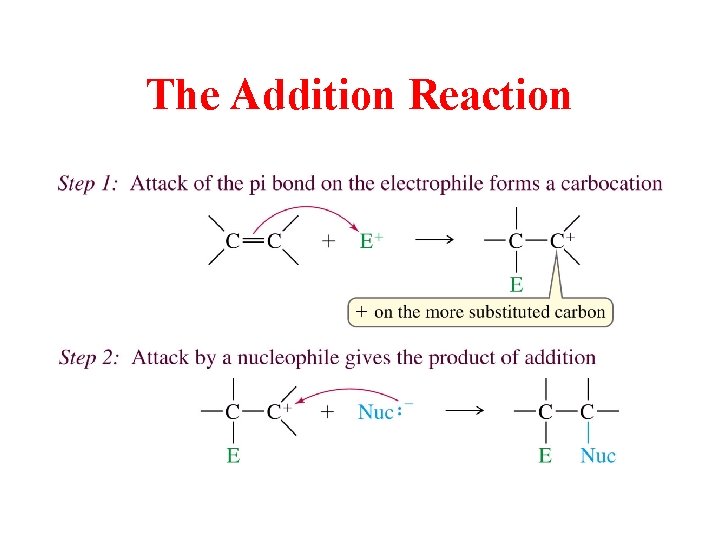
The Addition Reaction

HBr Addition
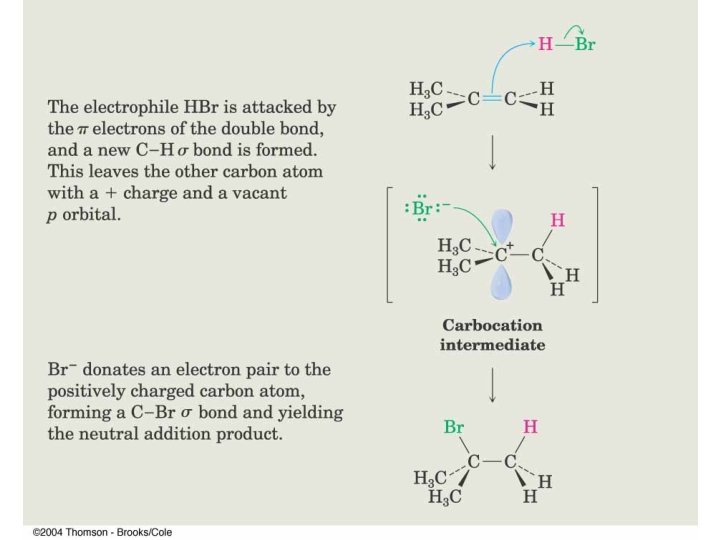
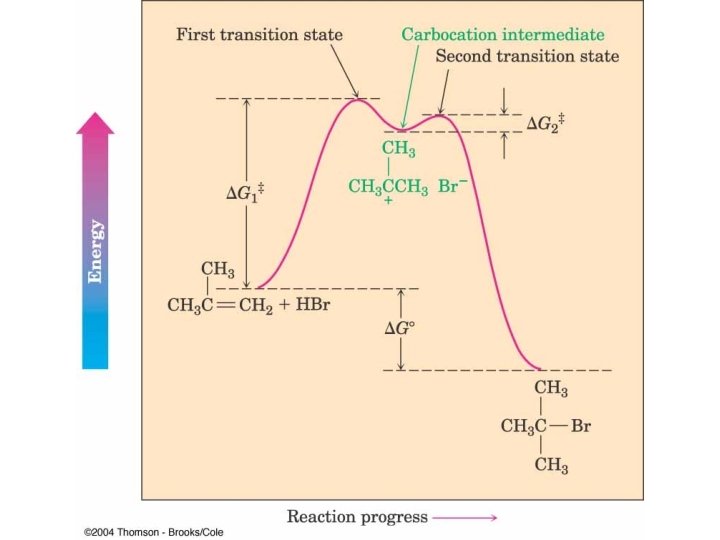

Markovnikov’s Rule The addition of H-X across a double bond results in the more highly substituted alkyl halide as the major product.
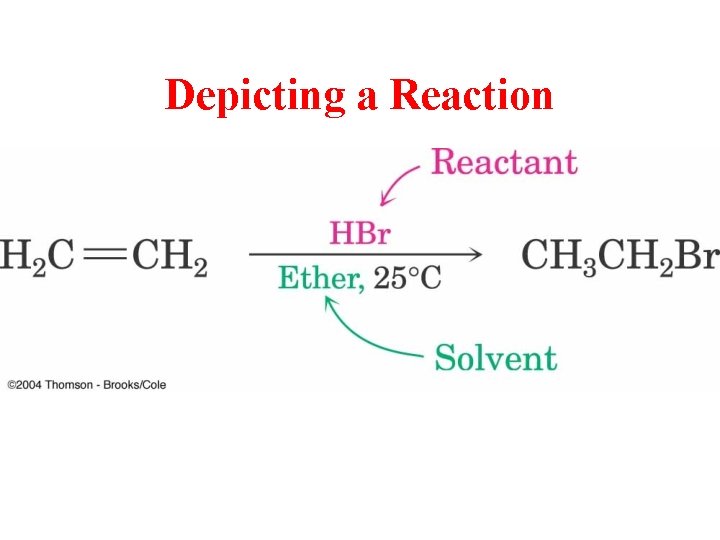
Depicting a Reaction
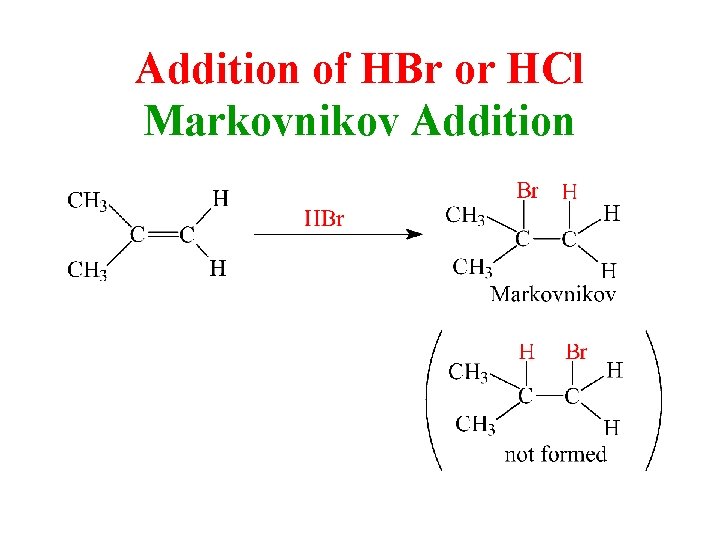
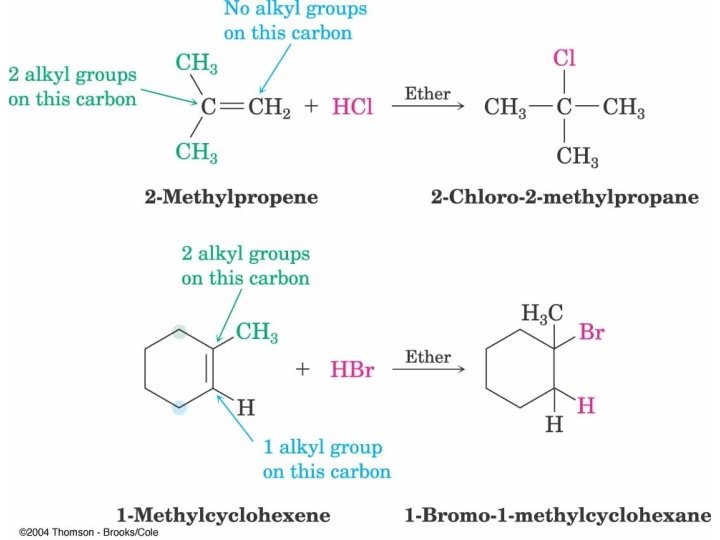
Addition of HBr or HCl Markovnikov Addition
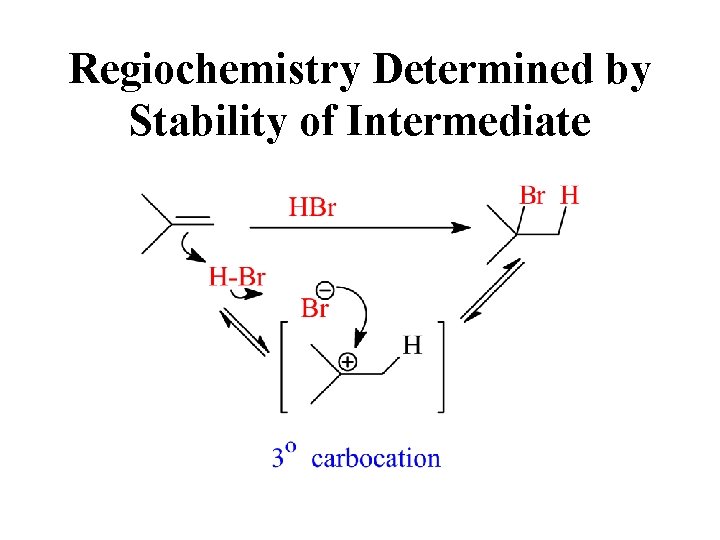
Regiochemistry Determined by Stability of Intermediate
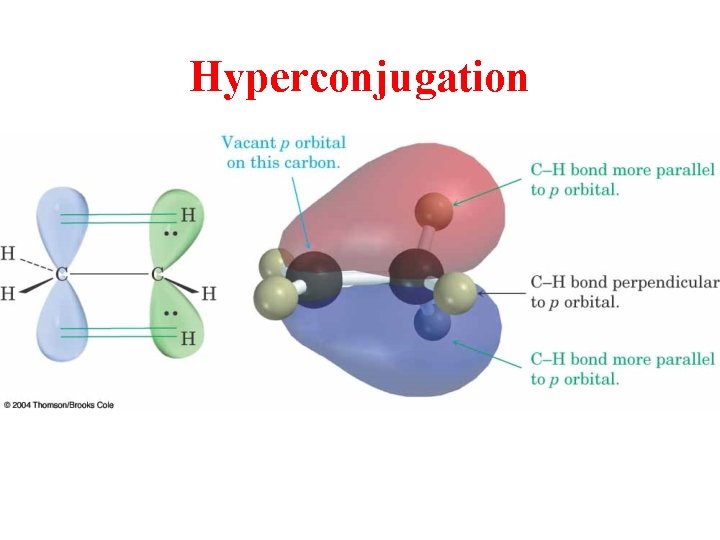
Hyperconjugation
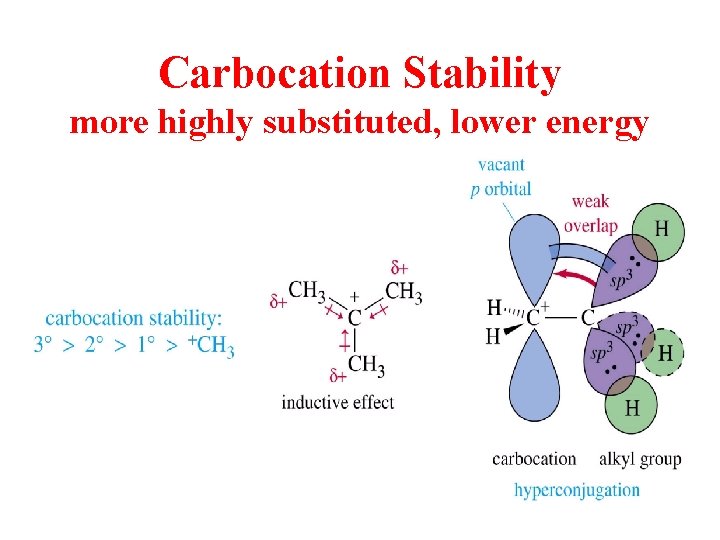
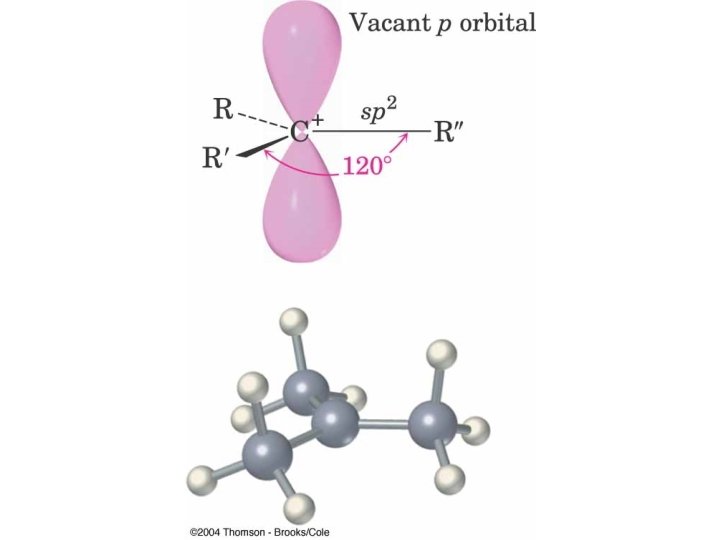
Carbocation Stability more highly substituted, lower energy
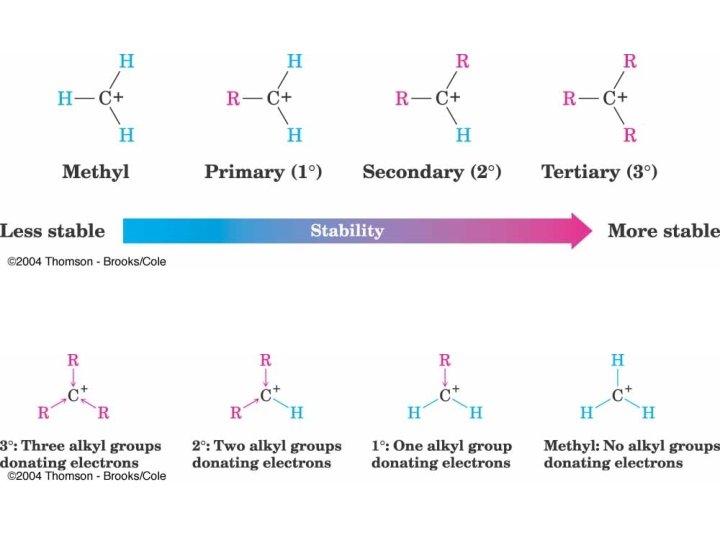

3 o Carbocation forms Preferentially
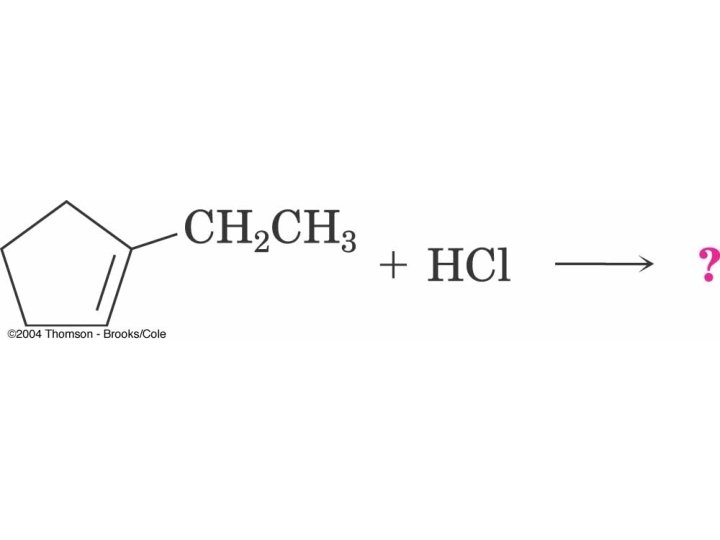
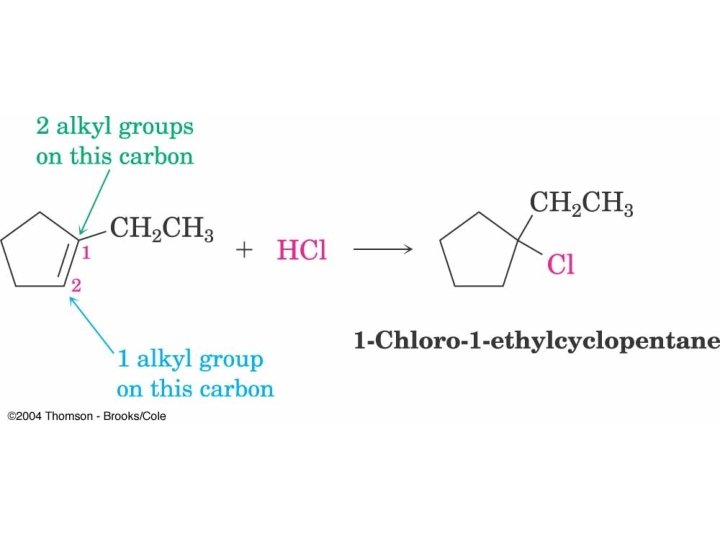
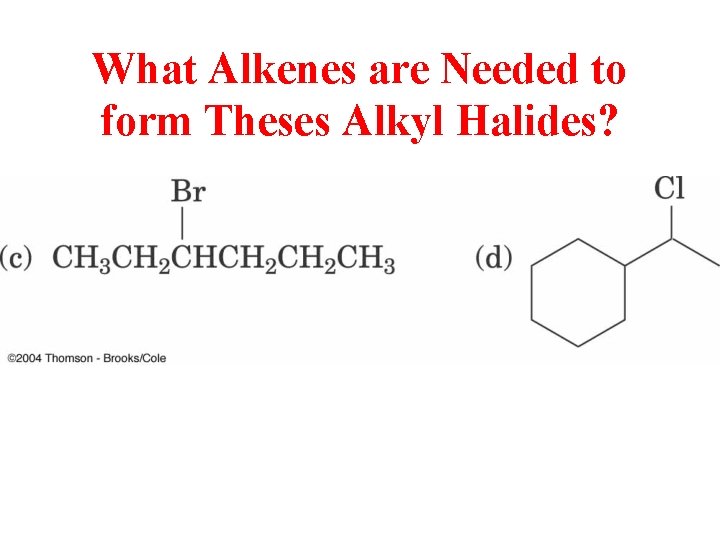
What Alkenes are Needed to form Theses Alkyl Halides?

Definitions • Regioisomers – two constitutional isomers that could result from an addition reaction. • Regiospecific – only one regiosisomer forms at the expense of the other. • Regioselective – both regioisomers are formed, but one is formed in preference.
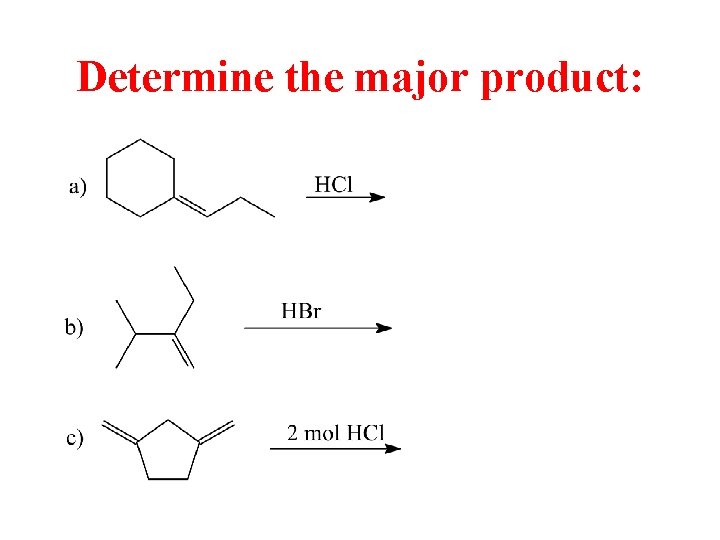
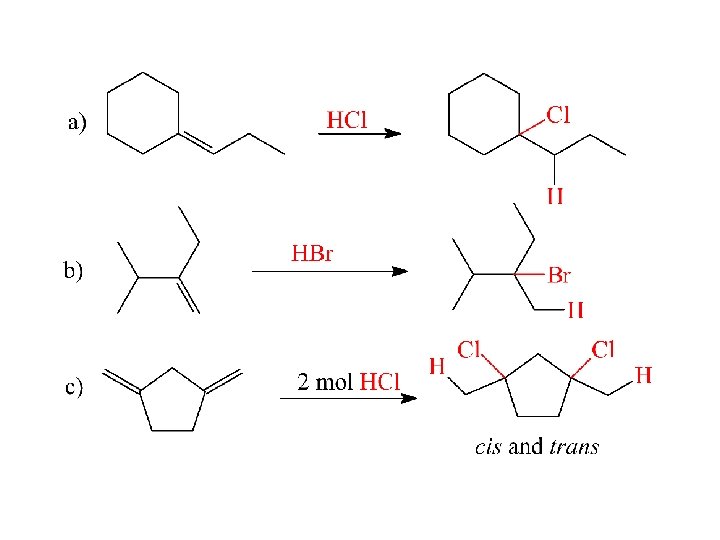
Determine the major product:

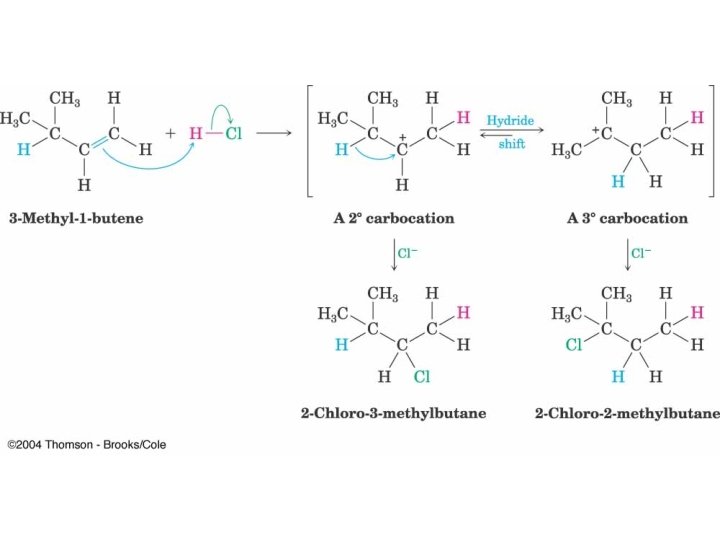
Rearrangements
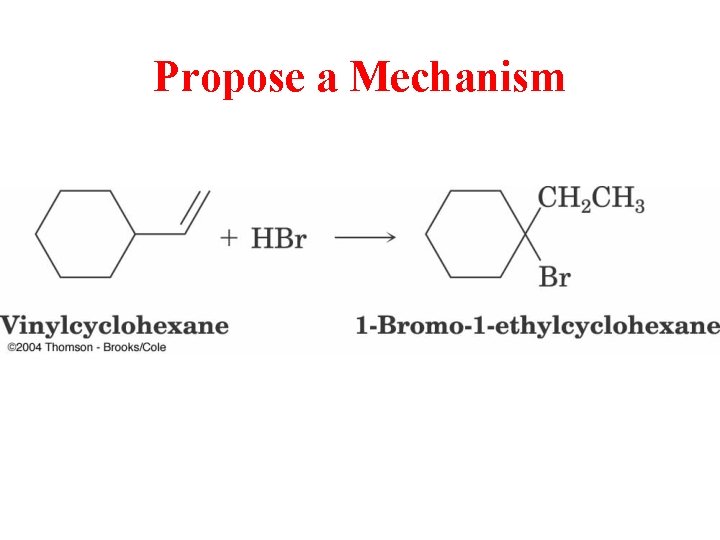
Propose a Mechanism
- Slides: 54