Alkenes Formation by elimination reactions Mc M 11

Alkenes: Formation by elimination reactions (Mc. M 11. 12, 11. 13) Substitution reactions: Cleavage of ethers (Mc. M 18. 5) Organometallic coupling react. (Mc. M 10. 9, lab ex 10) Dienes: MO description, conjugated diene (Mc. M 14. 2) Aromatic compounds: Aromaticity (repetition; Mc. M 15. 3 -15. 8) Electrophilic aromatic subst and substituent effects (Mc. M 16. 5 -16. 7, 16. 12) Nucleophilic aromatic subst (Mc. M 16. 8 -16. ) Other react on aromatic compounds (Mc. M 16. 10 -16. 11)

Dienes (Mc. M 14. 2)

Molecular orbitals of H 2 (cf fig 1. 20) - s-bond Molecular orbitals of p-bond (cf fig. 1. 21) MO 1, 3 -butadiene cf fig 14. 2 LUMO HOMO hn 1 e- Energy absorb UV spectra 4 p electron in 2 bonding orbitals (ground state)

MO orbitals benzene - aromaticity (Mc. M 15. 4 - 15. 5) Benzene 1, 3, 5 -hexatriene only bonding MO shown
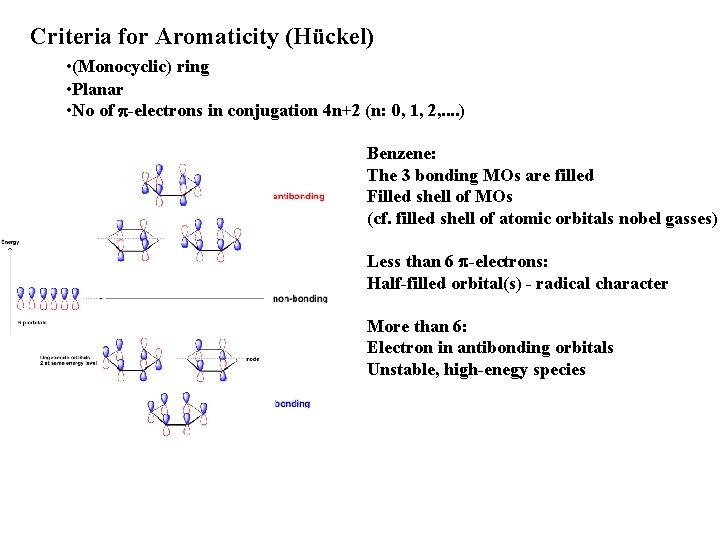
Criteria for Aromaticity (Hückel) • (Monocyclic) ring • Planar • No of p-electrons in conjugation 4 n+2 (n: 0, 1, 2, . . ) Benzene: The 3 bonding MOs are filled Filled shell of MOs (cf. filled shell of atomic orbitals nobel gasses) Less than 6 p-electrons: Half-filled orbital(s) - radical character More than 6: Electron in antibonding orbitals Unstable, high-enegy species

Ex. of an anti-aromatic compound - Cyclobutadiene

Electrophilic Aromatic Substitution and Substituent Effects (Mc. M. 16. 5 - 16. 6) • Halogenation (bromination) • Nitration • Sulfonation • Alkylation (Friedel Craft) • Acylation (Friedel Craft)� R influence: • Reactivity • Regiochemistry

R: Electron donating By resonance ex. RO-, HO-, R 2 N-, RHN, H 2 N-. RCOHN- The ring is activated for E-fil ar subst More reactive than benzene

Reaction in o or p pos. Activating, o/p directing

R: Electron donating The ring is activated for E-fil ar subst More reactive than benzene By inductive effect ex. alkyls Activating, o/p directing

R: Electron withdrawing By resonance effect: -CN, -COR, COH, CO 2 R, -NO 2, -SO 2 R etc Especially electron poor in o/p pos The ring is deactivated for E-fil ar subst Less reactive than benzene

Deactivating, m directing
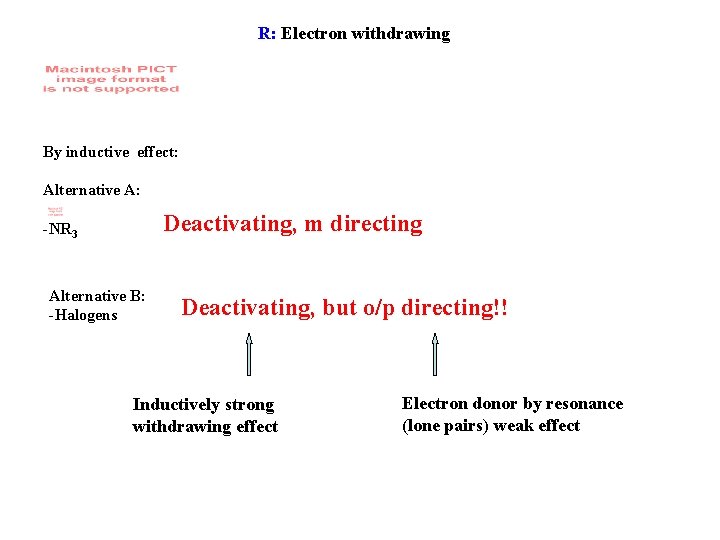
R: Electron withdrawing By inductive effect: Alternative A: Deactivating, m directing -NR 3 Alternative B: -Halogens Deactivating, but o/p directing!! Inductively strong withdrawing effect Electron donor by resonance (lone pairs) weak effect

Regiochemistry in E-fil aromatic subst of disubst. benzene derivs. (Mc. M 16. 7) Synth of trisubst. benzene deriv. - Planning of a good reaction sequence (Mc. M 16. 12)

Nucleophilic Aromatic Substitution (Mc. M 16. 8 - 16. 9)

Mechanisms: • SNAr • SN 1: Via diazonium salts (Mc. M 24. 8) • Benzyne • (SRN 1: Involves radicals) • (VNS: Vicarious nucl. Subst. )

• SNAr Only on electron defficient arenes (EWG o/p to X, Anion stabilizing effect) (Aromatic heterocycles, Mc. M 28. 6) 1 st step rate limiting (Aromaticity broken) X=F>Cl>Br>I

• Benzyne “Triple bond” between sp 2 C p-p overlap sp 2 -sp 2 overlap - weak bond Benzynes unstable / reactive intermed. Reactivity of benzynes: • Adds nucleophiles • Dienophile in Diels Alder react.

Reactivity of aromatic side chains • Ox. of alkyl side chains • Halogenation benzylic position (c. f. Lab ex. 1) • No ox. cleavage of “double bonds” in benzene • Reduction of benzene (hydrogenation) difficult
- Slides: 19