Alkenes and Alkynes Addition Reactions E V Blackburn

Alkenes and Alkynes Addition Reactions © E. V. Blackburn, 2008

Reactions of alkenes catalytic hydrogenation Heat of hydrogenation - the heat liberated during this reaction. H is ~125 k. J/mol for each double bond in the compound. © E. V. Blackburn, 2008
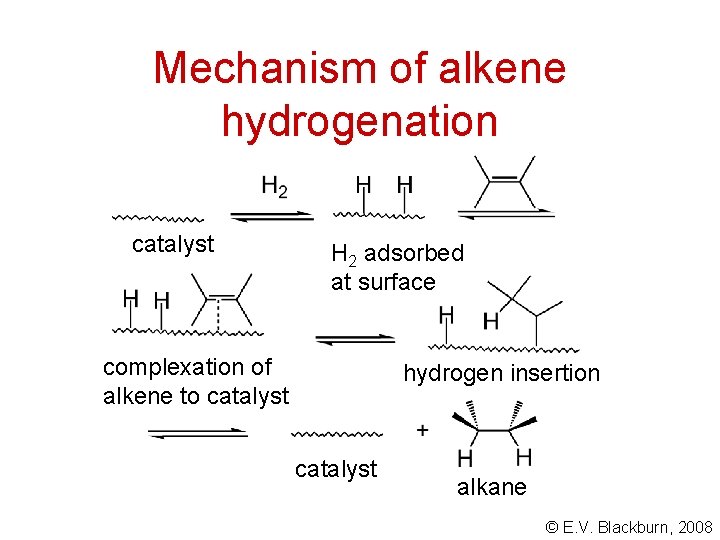
Mechanism of alkene hydrogenation catalyst H 2 adsorbed at surface complexation of alkene to catalyst hydrogen insertion catalyst alkane © E. V. Blackburn, 2008

Mechanism of alkene hydrogenation Hydrogenation is stereospecific. The two hydrogens add to the same side of the double bond - a syn addition. © E. V. Blackburn, 2008

Reactions of alkenes The electrons are less tightly held than the electrons. The double bond therefore acts as a source of electrons - a base – a nucleophile. It reacts with electron deficient compounds - acids electrophiles. © E. V. Blackburn, 2008

Electrophilic addition Electron seeking reagents are called electrophilic reagents. The typical reaction of alkenes is one of electrophilic addition - an acid - base reaction. Don’t forget that free radicals are electron deficient. They undergo addition reactions with alkenes. © E. V. Blackburn, 2008
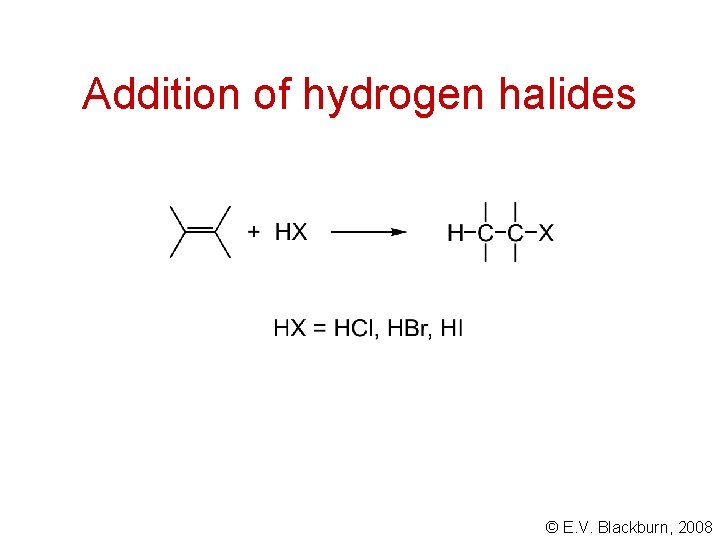
Addition of hydrogen halides © E. V. Blackburn, 2008
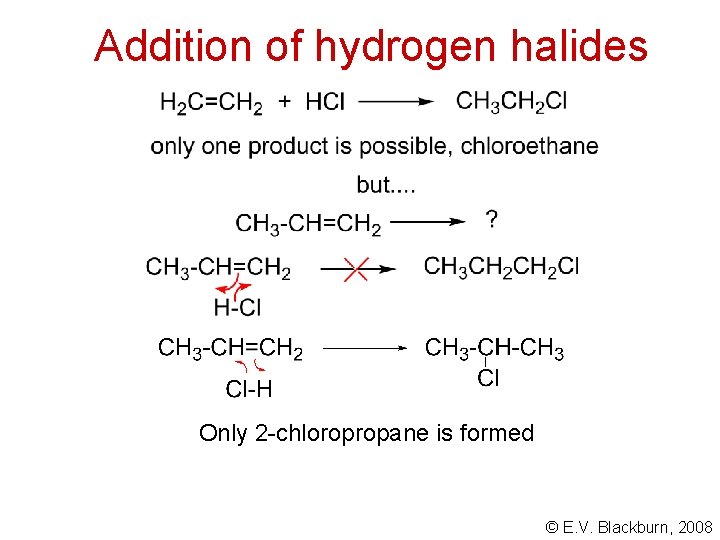
Addition of hydrogen halides Only 2 -chloropropane is formed © E. V. Blackburn, 2008

Markovnikov’s rule In 1869, Markovnikov proposed that in the addition of an acid to an alkene, the hydrogen of the acid bonds to the carbon which is already bonded to the greater number of hydrogens. © E. V. Blackburn, 2008
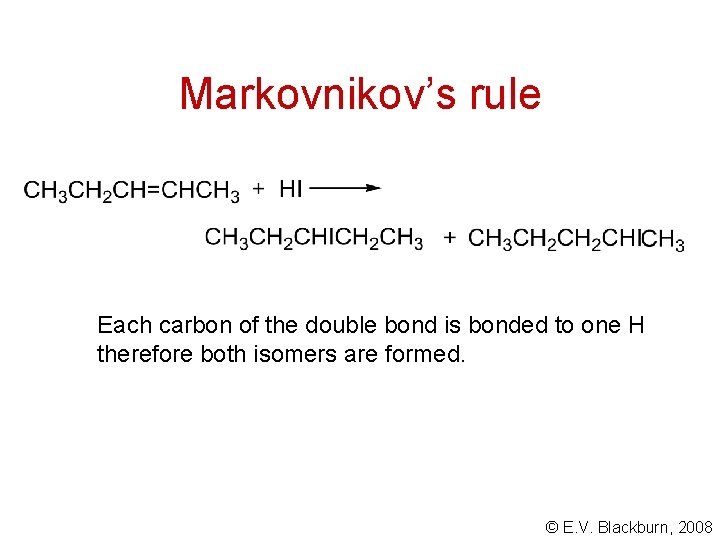
Markovnikov’s rule Each carbon of the double bond is bonded to one H therefore both isomers are formed. © E. V. Blackburn, 2008

Markovnikov addition - a regioselective reaction These reactions are said to be regioselective because only one of the two possible directions of addition occurs. Regioselectivity - the preferential formation of one isomer in those situations where a choice is possible. © E. V. Blackburn, 2008
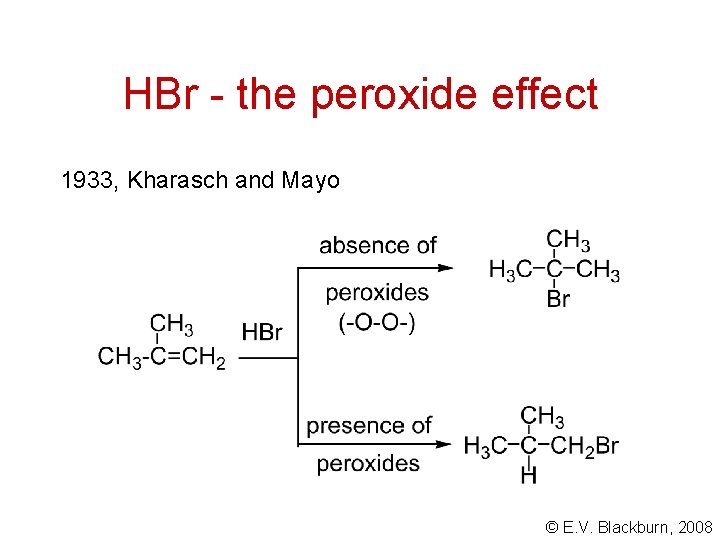
HBr - the peroxide effect 1933, Kharasch and Mayo © E. V. Blackburn, 2008
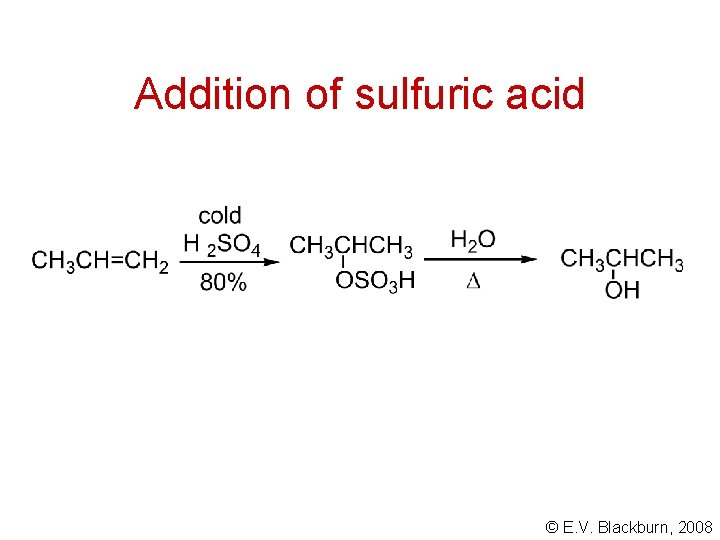
Addition of sulfuric acid © E. V. Blackburn, 2008
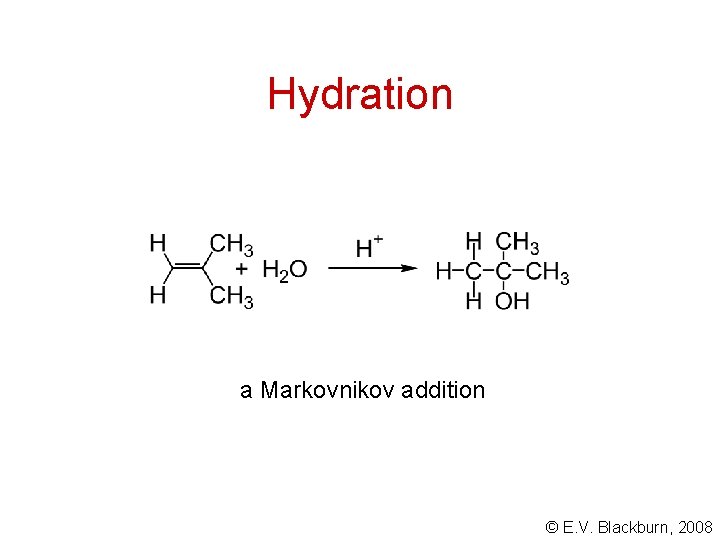
Hydration a Markovnikov addition © E. V. Blackburn, 2008
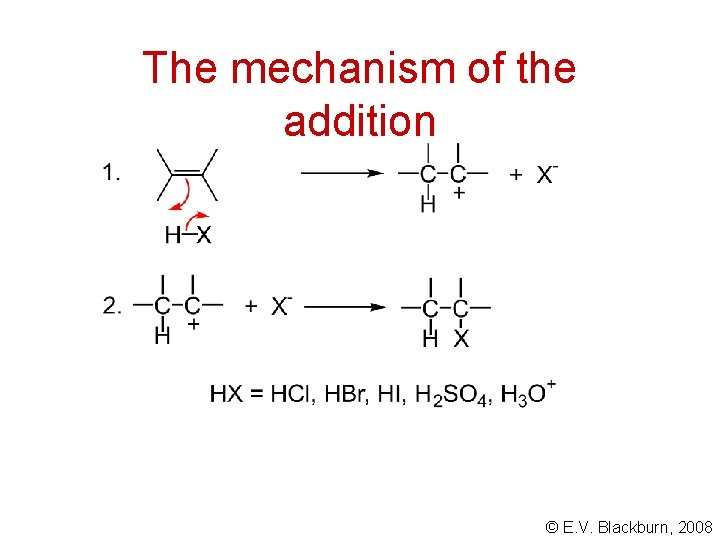
The mechanism of the addition © E. V. Blackburn, 2008
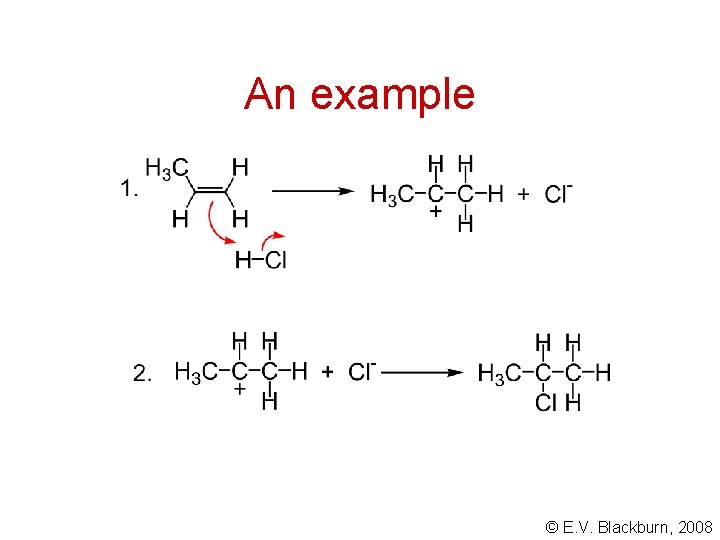
An example © E. V. Blackburn, 2008
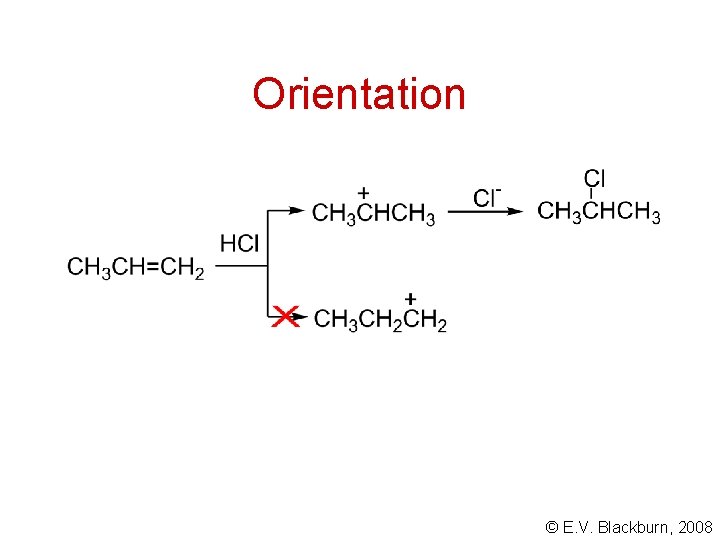
Orientation © E. V. Blackburn, 2008
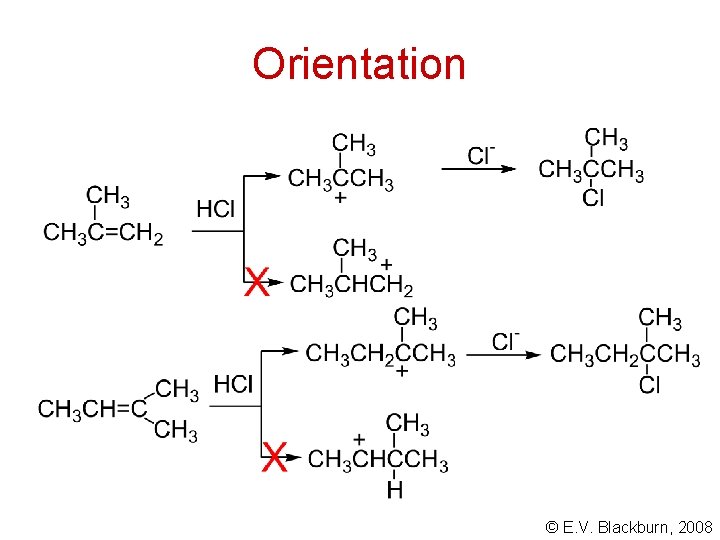
Orientation © E. V. Blackburn, 2008
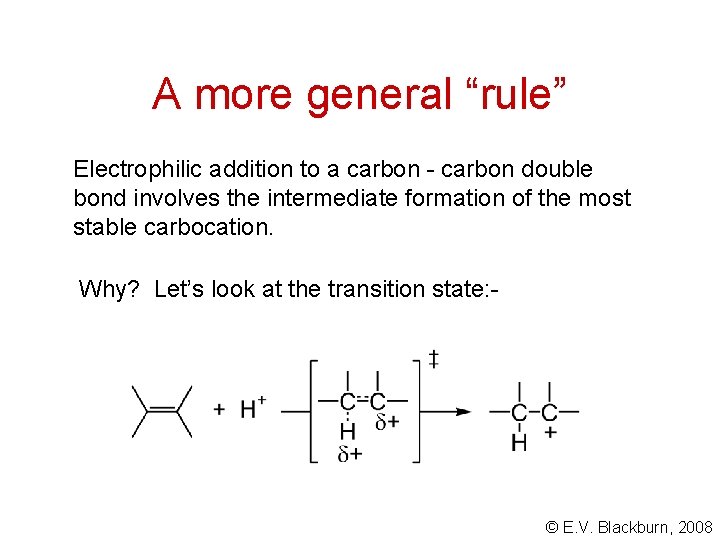
A more general “rule” Electrophilic addition to a carbon - carbon double bond involves the intermediate formation of the most stable carbocation. Why? Let’s look at the transition state: - © E. V. Blackburn, 2008
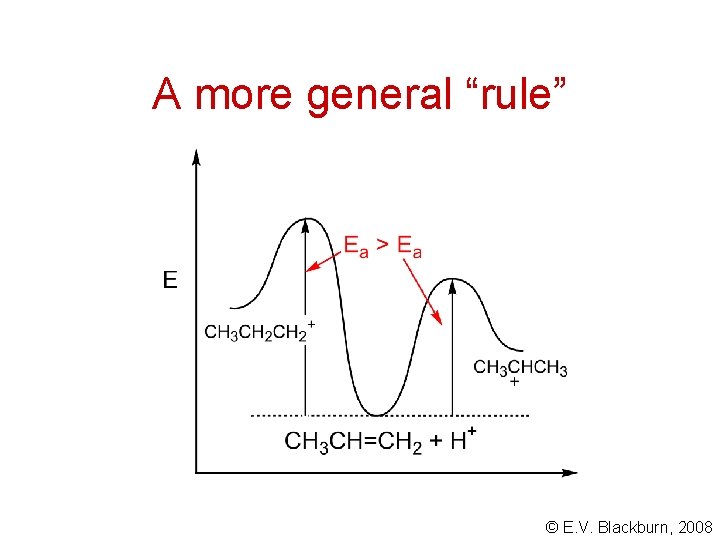
A more general “rule” © E. V. Blackburn, 2008
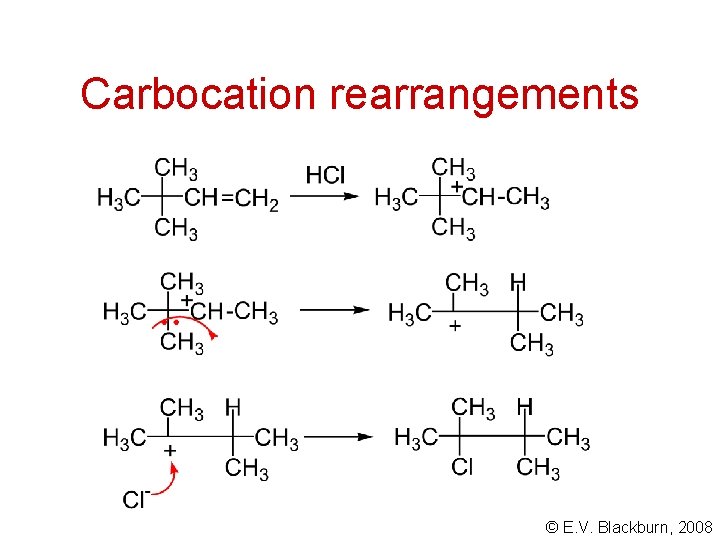
Carbocation rearrangements © E. V. Blackburn, 2008

Problems Try problems 8. 3 – 8. 8 on pages 336 – 340 of Solomons and Fryhle. © E. V. Blackburn, 2008
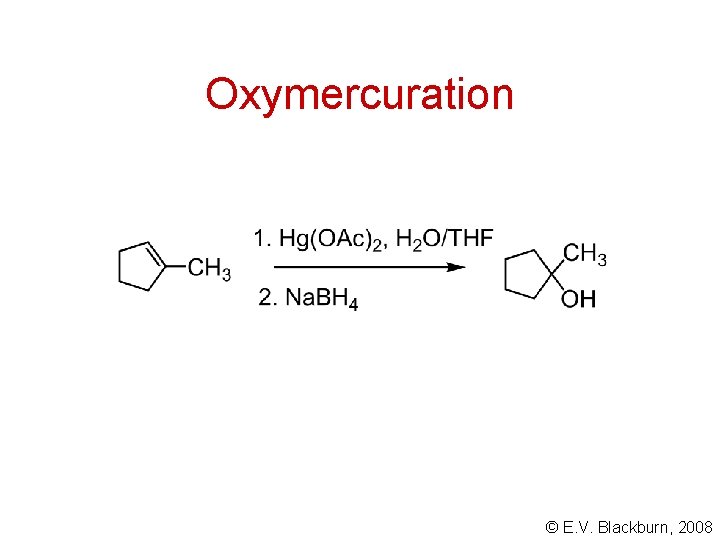
Oxymercuration © E. V. Blackburn, 2008
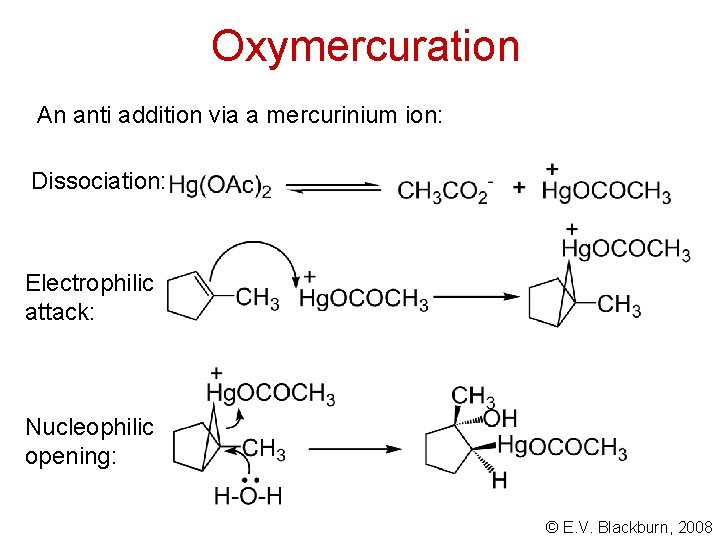
Oxymercuration An anti addition via a mercurinium ion: Dissociation: Electrophilic attack: Nucleophilic opening: © E. V. Blackburn, 2008
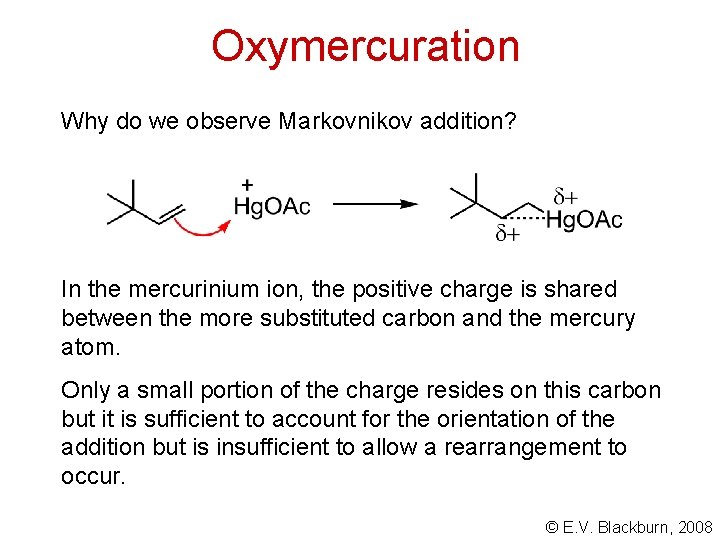
Oxymercuration Why do we observe Markovnikov addition? In the mercurinium ion, the positive charge is shared between the more substituted carbon and the mercury atom. Only a small portion of the charge resides on this carbon but it is sufficient to account for the orientation of the addition but is insufficient to allow a rearrangement to occur. © E. V. Blackburn, 2008
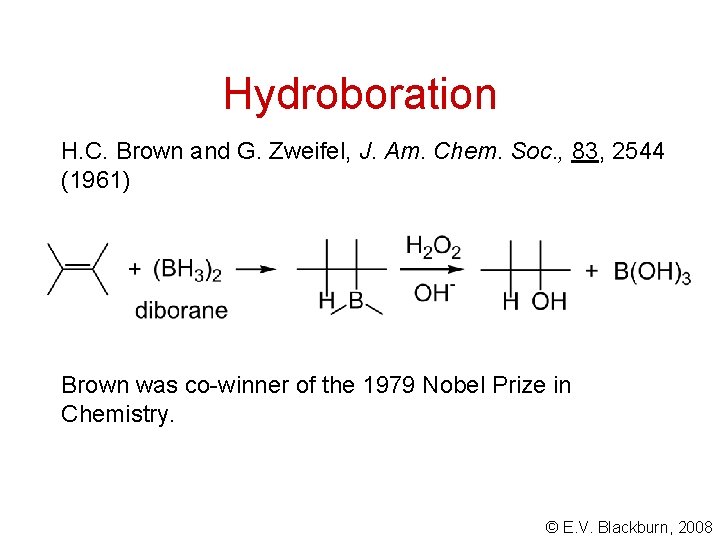
Hydroboration H. C. Brown and G. Zweifel, J. Am. Chem. Soc. , 83, 2544 (1961) Brown was co-winner of the 1979 Nobel Prize in Chemistry. © E. V. Blackburn, 2008
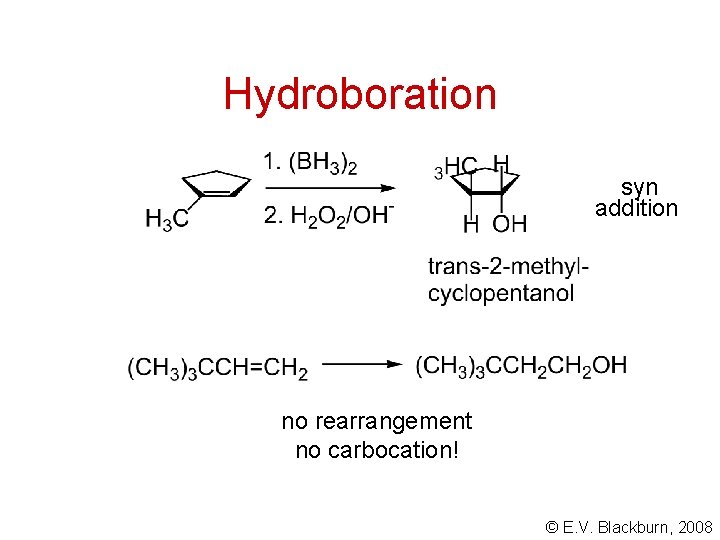
Hydroboration syn addition no rearrangement no carbocation! © E. V. Blackburn, 2008
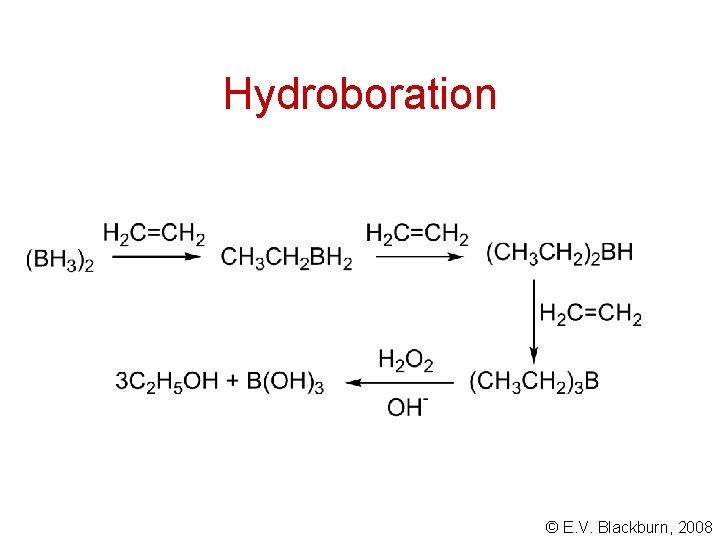
Hydroboration © E. V. Blackburn, 2008
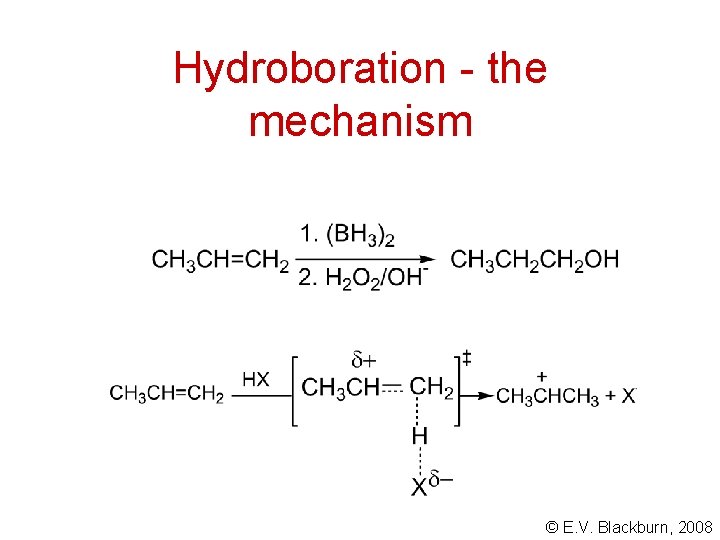
Hydroboration - the mechanism © E. V. Blackburn, 2008
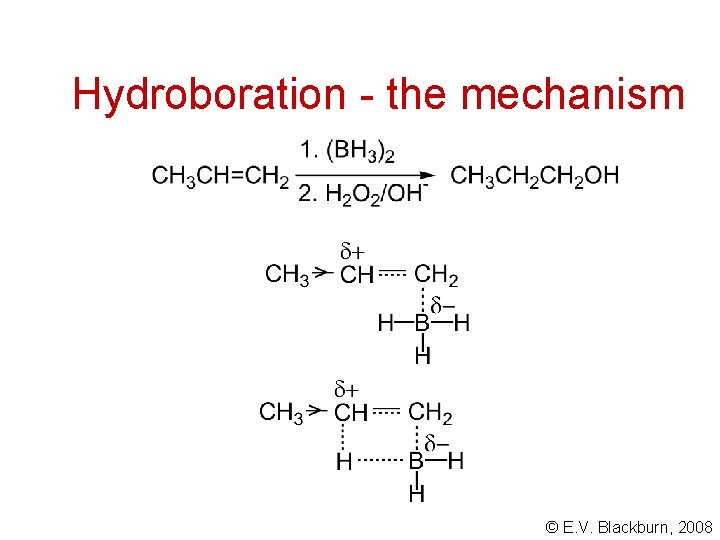
Hydroboration - the mechanism © E. V. Blackburn, 2008
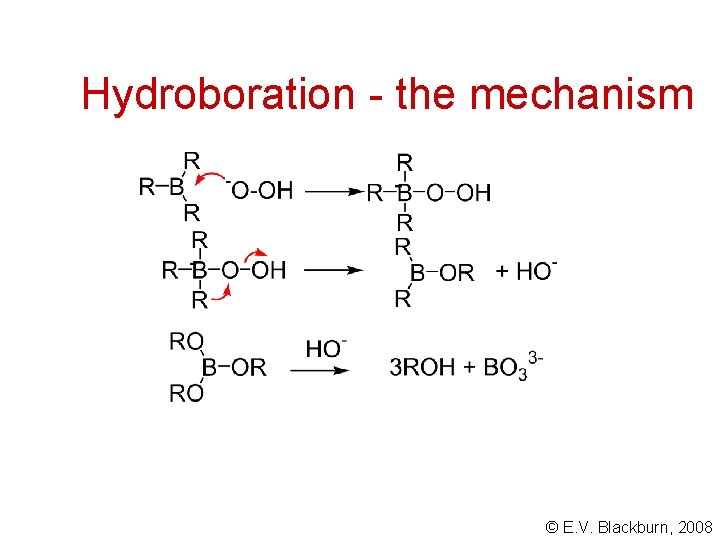
Hydroboration - the mechanism © E. V. Blackburn, 2008
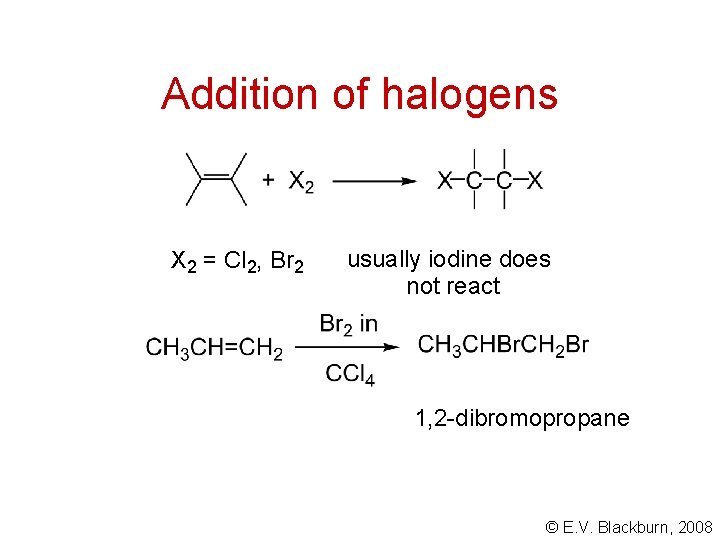
Addition of halogens X 2 = Cl 2, Br 2 usually iodine does not react 1, 2 -dibromopropane © E. V. Blackburn, 2008
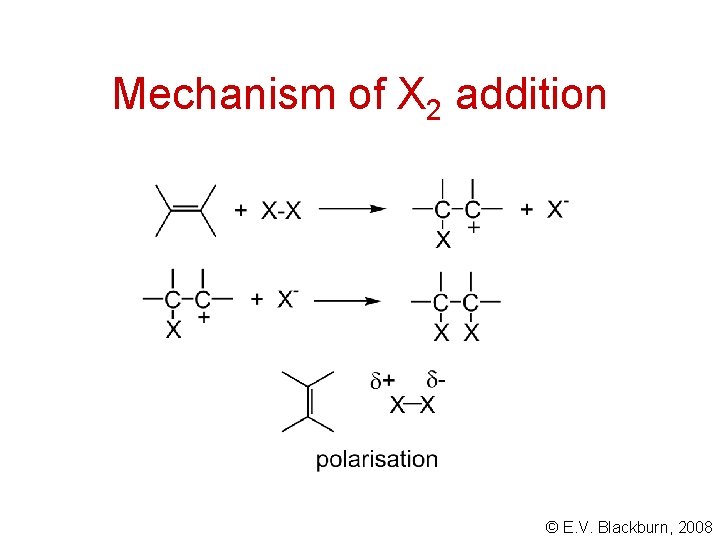
Mechanism of X 2 addition © E. V. Blackburn, 2008
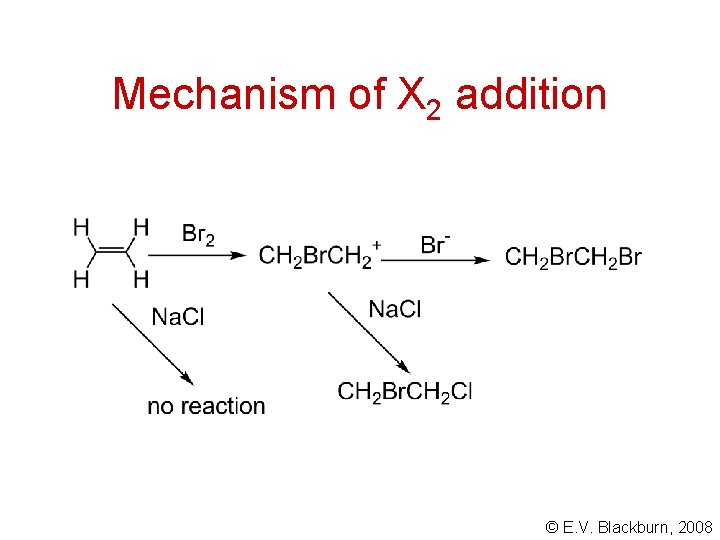
Mechanism of X 2 addition © E. V. Blackburn, 2008

Stereospecific reactions (Z)-2 -butene gives racemic 2, 3 -dibromobutane and no meso compound is formed. (E)-2 -butene gives only meso-2, 3 -dibromobutane. A reaction is stereospecific if a particular stereoisomer of the reactant produces a specific stereoisomer of the product. © E. V. Blackburn, 2008
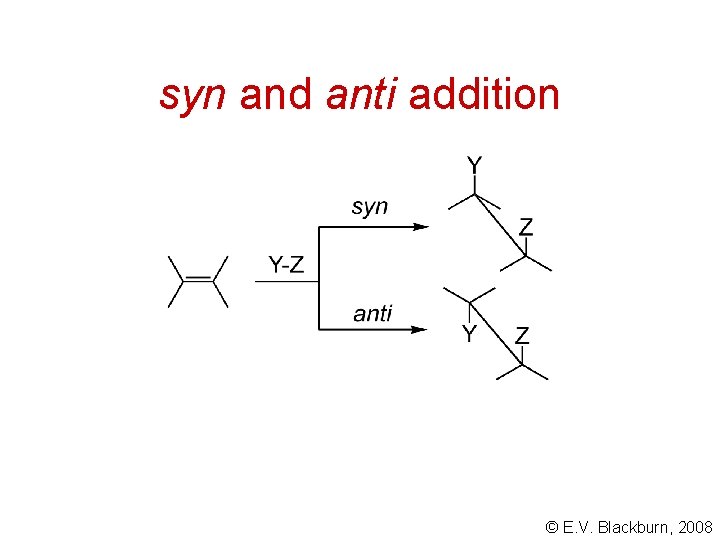
syn and anti addition © E. V. Blackburn, 2008
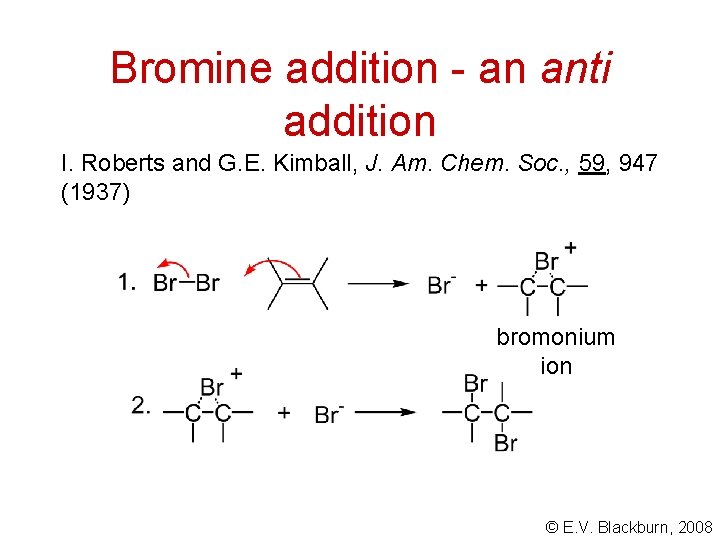
Bromine addition - an anti addition I. Roberts and G. E. Kimball, J. Am. Chem. Soc. , 59, 947 (1937) bromonium ion © E. V. Blackburn, 2008
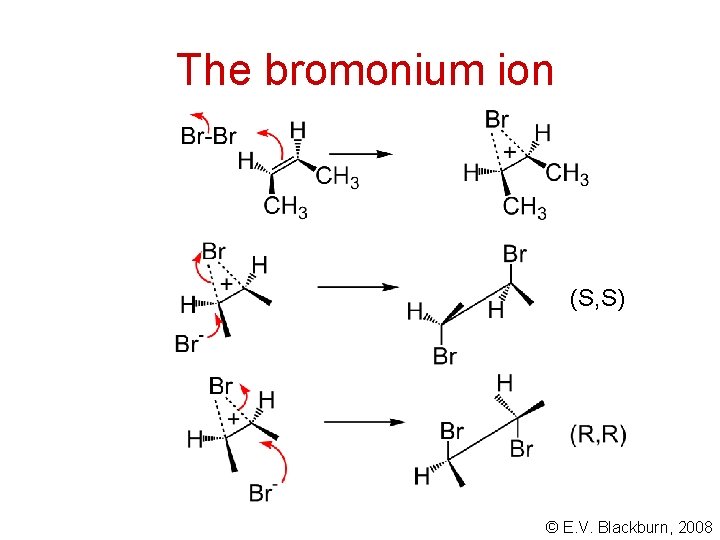
The bromonium ion (S, S) © E. V. Blackburn, 2008
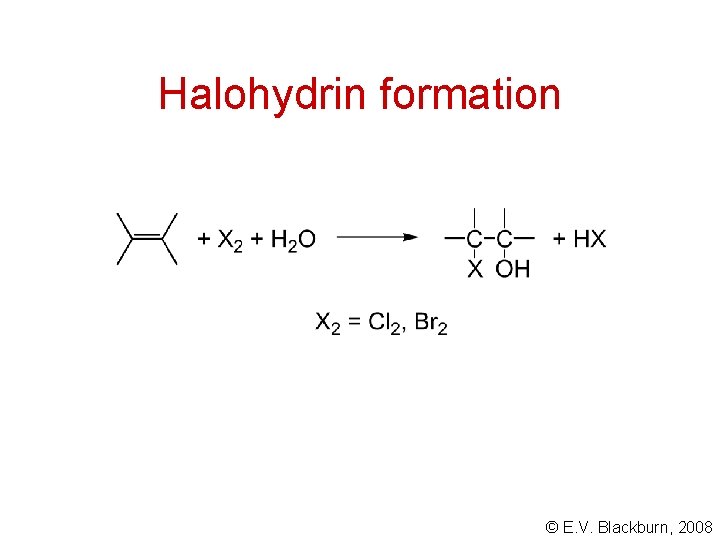
Halohydrin formation © E. V. Blackburn, 2008
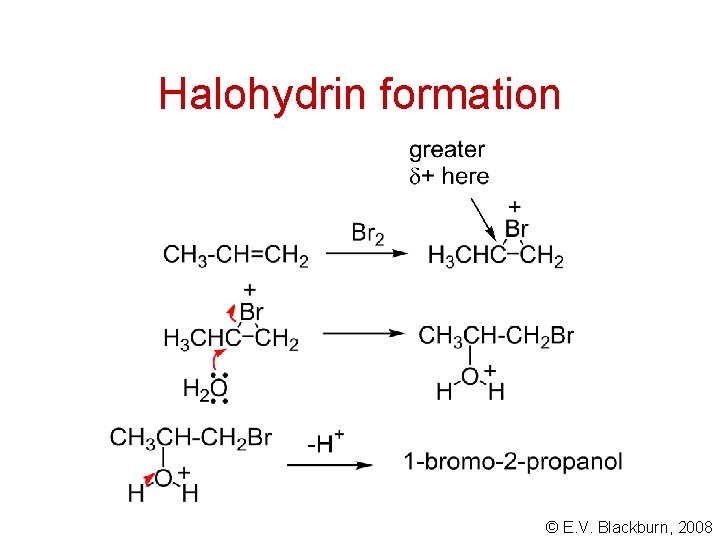
Halohydrin formation © E. V. Blackburn, 2008
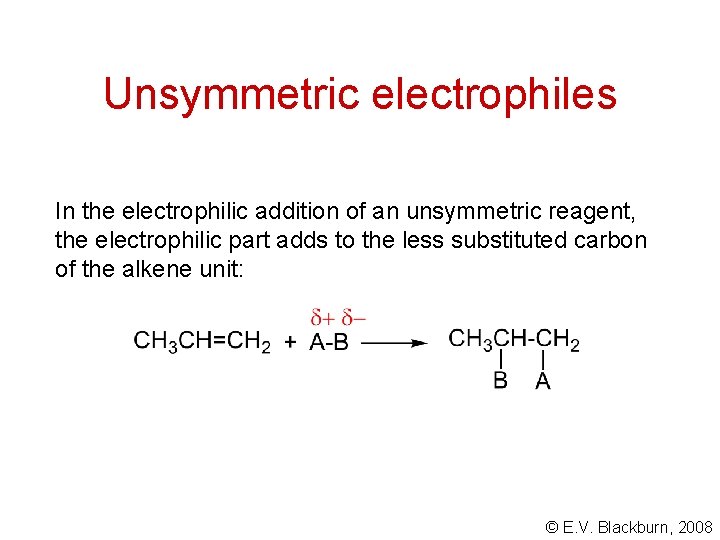
Unsymmetric electrophiles In the electrophilic addition of an unsymmetric reagent, the electrophilic part adds to the less substituted carbon of the alkene unit: © E. V. Blackburn, 2008
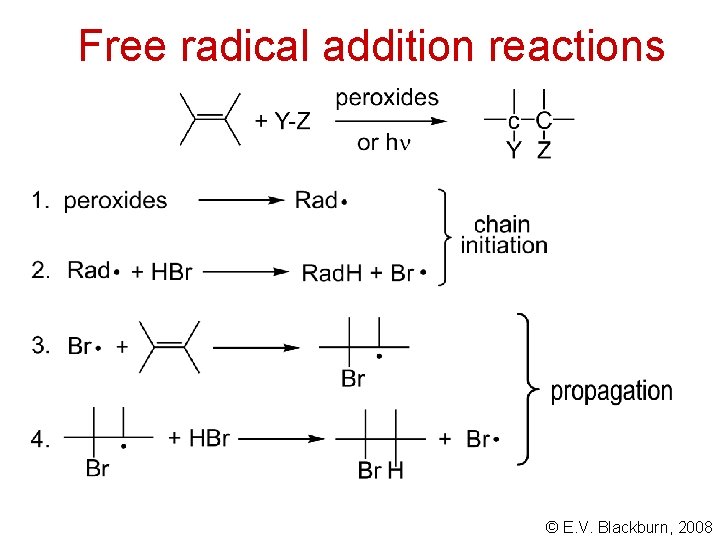
Free radical addition reactions © E. V. Blackburn, 2008
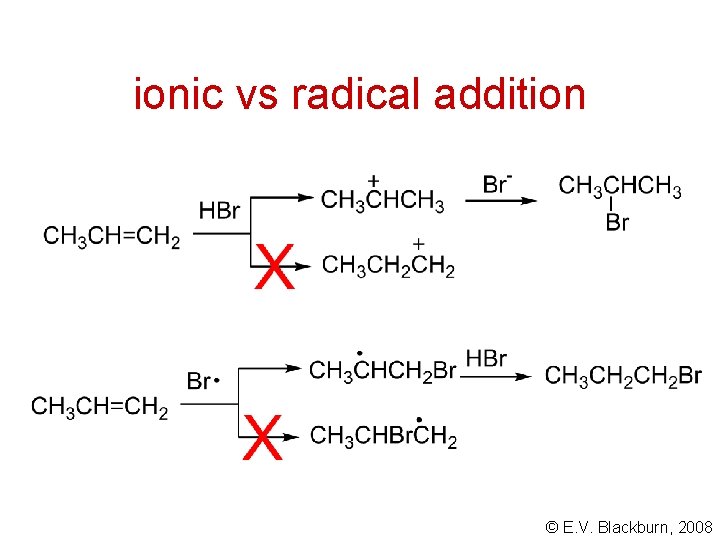
ionic vs radical addition © E. V. Blackburn, 2008

Polymerization A polymer is a long chain molecule made up of structural units (monomers) joined together. © E. V. Blackburn, 2008

Free radical polymerization of alkenes Initiation © E. V. Blackburn, 2008
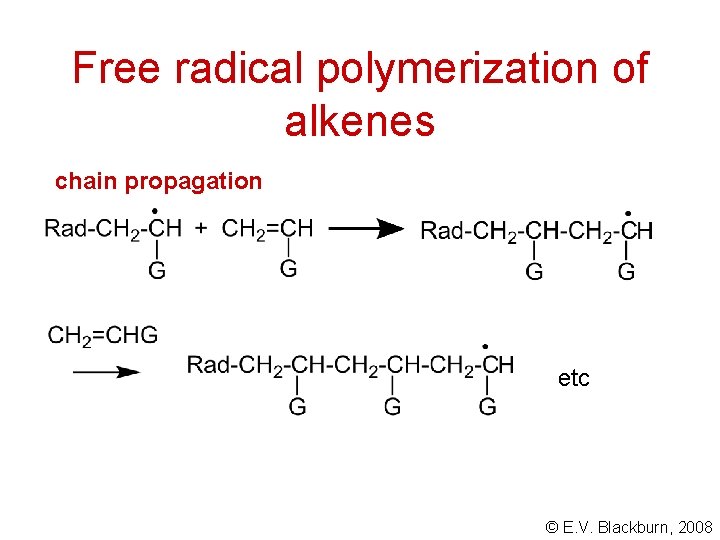
Free radical polymerization of alkenes chain propagation etc © E. V. Blackburn, 2008
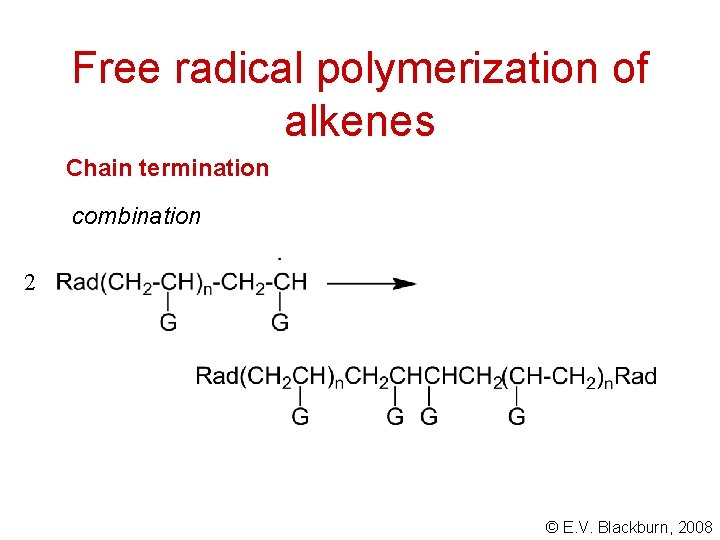
Free radical polymerization of alkenes Chain termination combination 2 © E. V. Blackburn, 2008
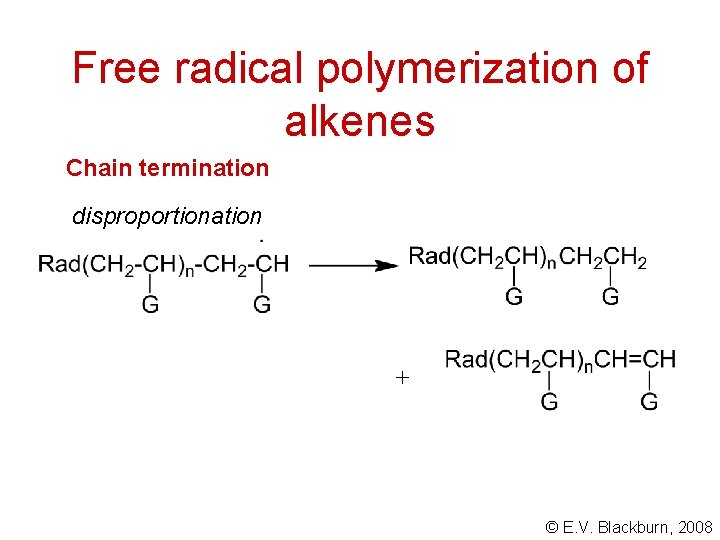
Free radical polymerization of alkenes Chain termination disproportionation + © E. V. Blackburn, 2008

Examples G Cl CN monomer polymer CH 2=CHCl -CH 2 CHCl-CH 2 - vinyl chloride polyvinyl chloride, PVC CH 2=CHCN -CH 2 CHCN- acrylonitrile polyacrylonitrile Orlon, Acrilon © E. V. Blackburn, 2008
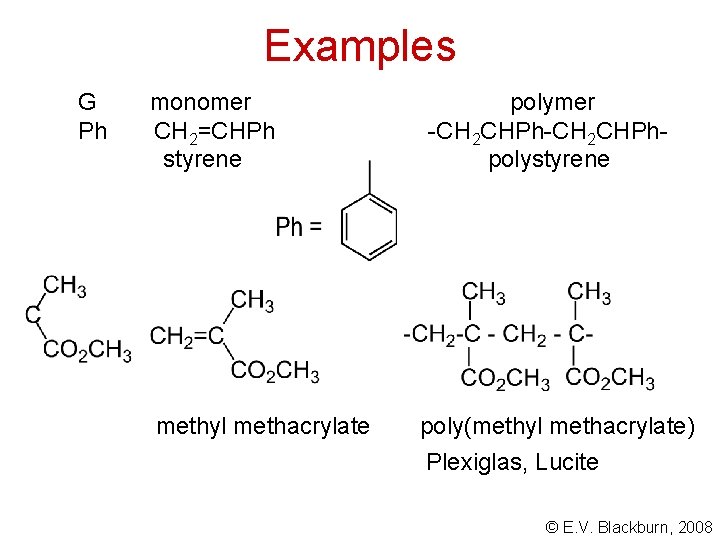
Examples G Ph monomer CH 2=CHPh styrene methyl methacrylate polymer -CH 2 CHPhpolystyrene poly(methyl methacrylate) Plexiglas, Lucite © E. V. Blackburn, 2008
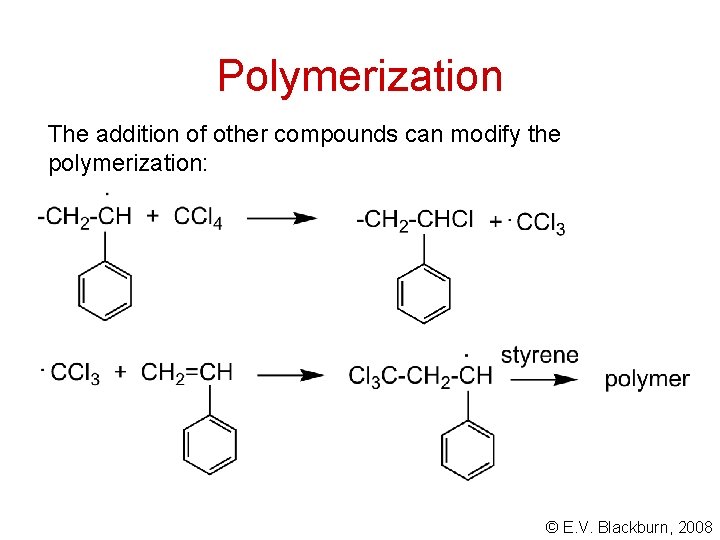
Polymerization The addition of other compounds can modify the polymerization: © E. V. Blackburn, 2008
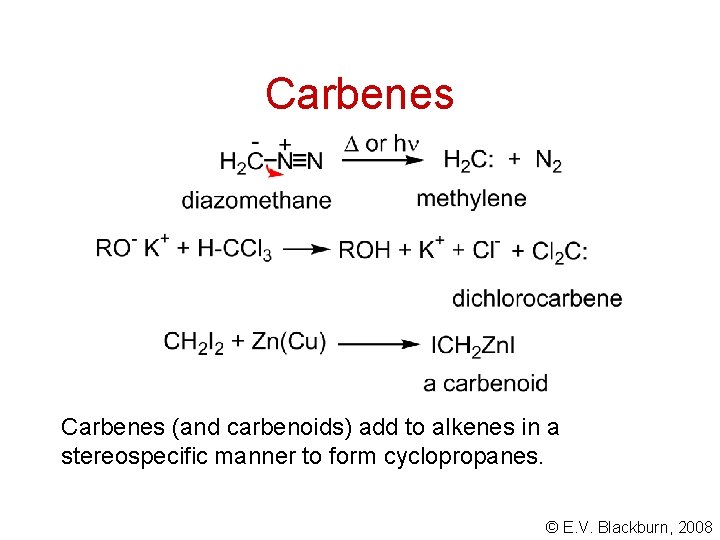
Carbenes (and carbenoids) add to alkenes in a stereospecific manner to form cyclopropanes. © E. V. Blackburn, 2008
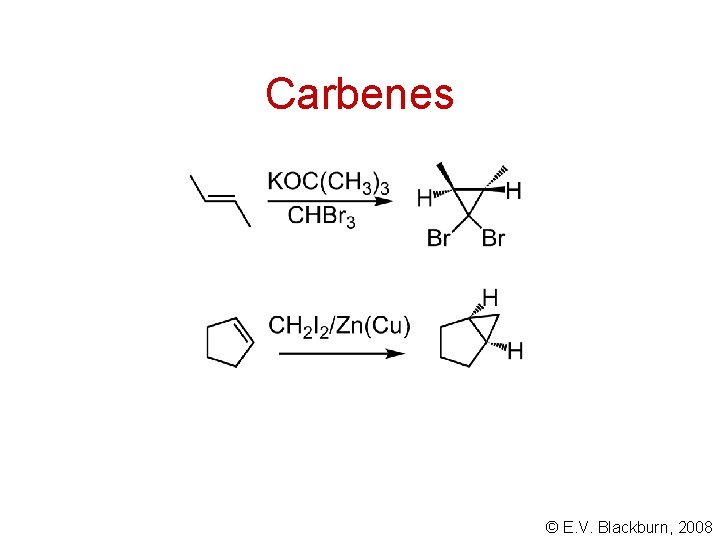
Carbenes © E. V. Blackburn, 2008
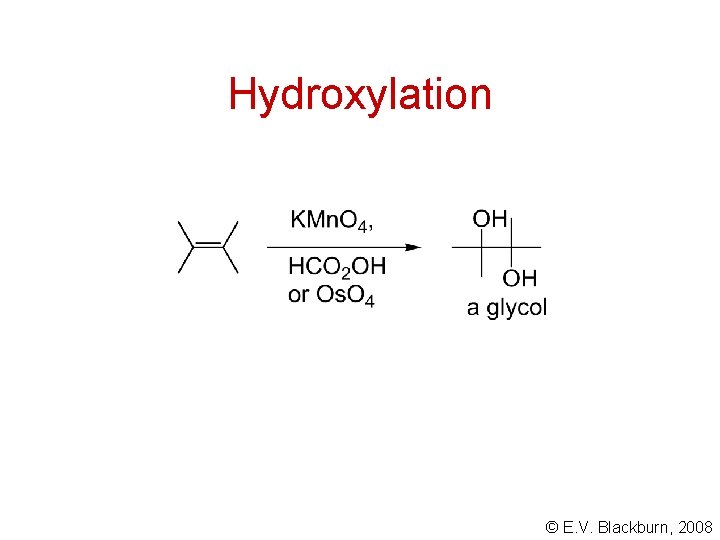
Hydroxylation © E. V. Blackburn, 2008
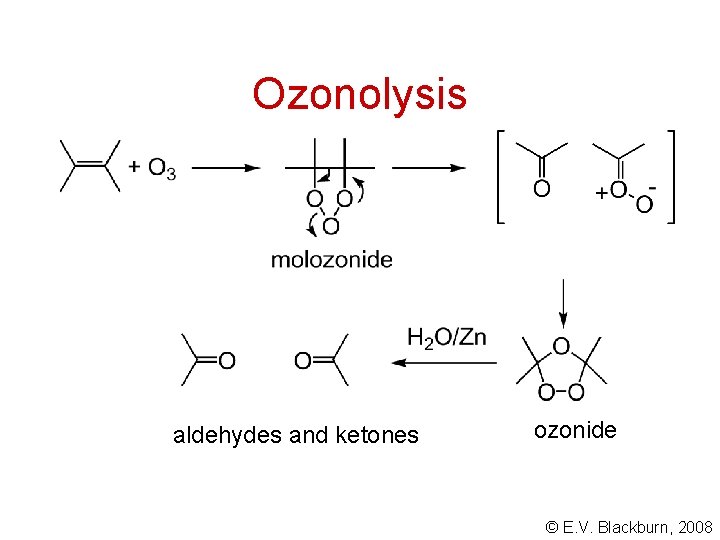
Ozonolysis aldehydes and ketones ozonide © E. V. Blackburn, 2008
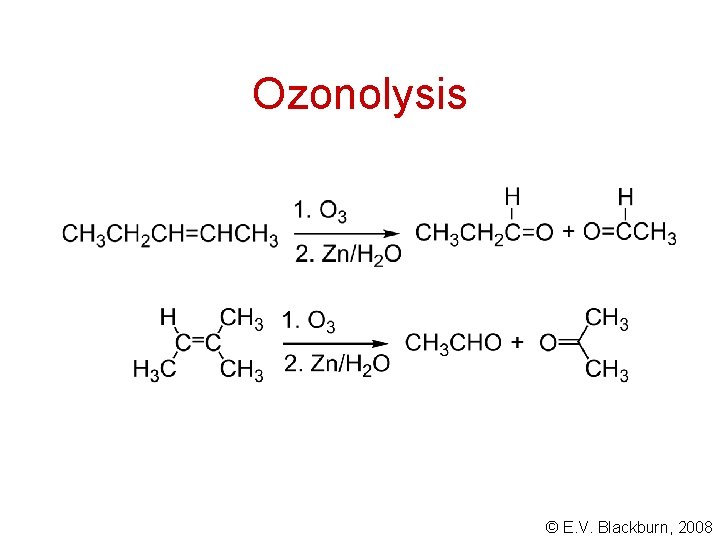
Ozonolysis © E. V. Blackburn, 2008
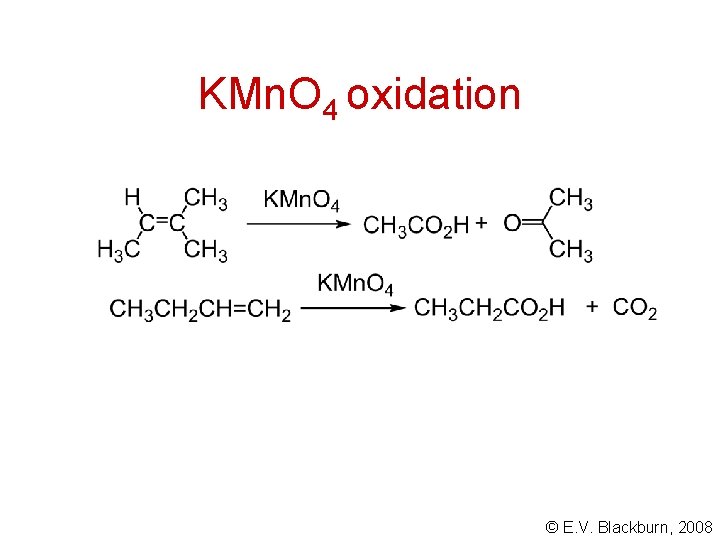
KMn. O 4 oxidation © E. V. Blackburn, 2008
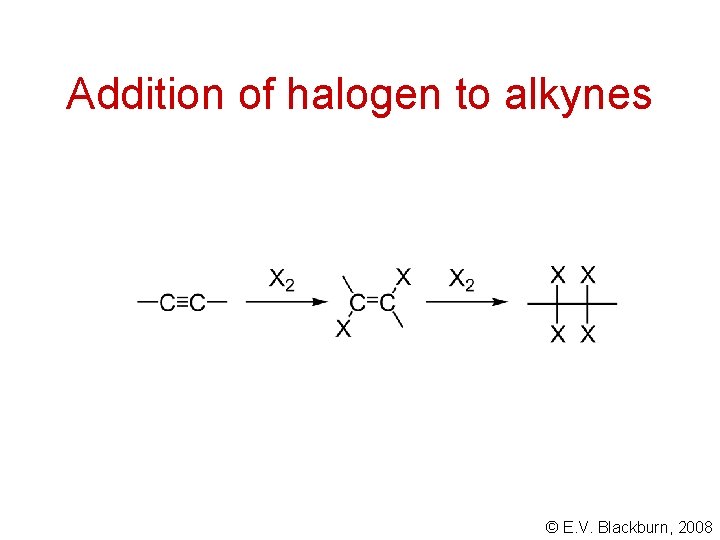
Addition of halogen to alkynes © E. V. Blackburn, 2008
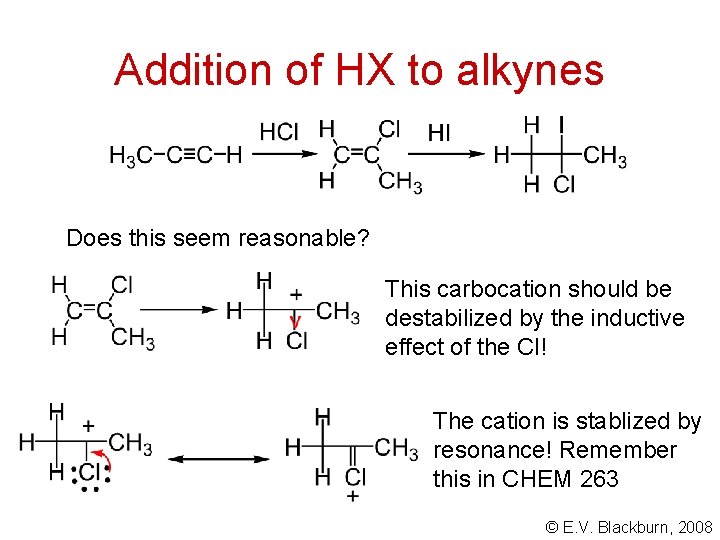
Addition of HX to alkynes Does this seem reasonable? This carbocation should be destabilized by the inductive effect of the Cl! The cation is stablized by resonance! Remember this in CHEM 263 © E. V. Blackburn, 2008
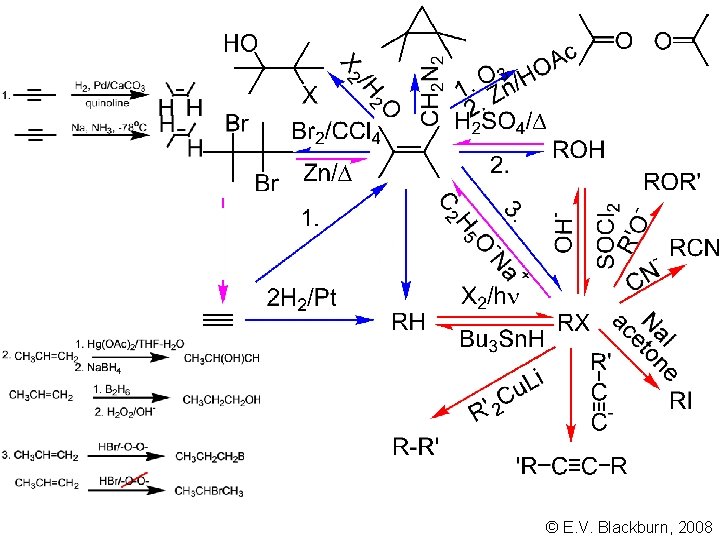
© E. V. Blackburn, 2008

Problems Try problems 8. 30, 8. 32, 8. 34, 8. 38, 8. 40, 8. 42, 8. 45, 8. 46, 8. 51, and 8. 61 on pages 376 -380 of Solomons and Fryhle. © E. V. Blackburn, 2008
- Slides: 61