Alkane Nomenclature in alkanes all carbons are saturated

Alkane Nomenclature

• in alkanes, all carbons are “saturated” (have four single bonds) • Alkanes have the general formula Cn. H 2 n+2 • This is the basis of all organic nomenclature

• 1. Find the longest continuous chain of carbons and name. # of carbons name 1 methane 2 ethane 3 propane 4 butane 5 pentane 6 hexane 7 heptane

• • 8 9 10 11 12 13 14 octane nonane decane undecane dodecane tridecane tetradecane 15 16 17 18 19 20 pentadecane hexadecane heptadecane octadecane nonadecane eicosane

• 2. Number the chain so that the branches have the lowest possible number. • 3. Name and number each branching group (name with –yl ending)
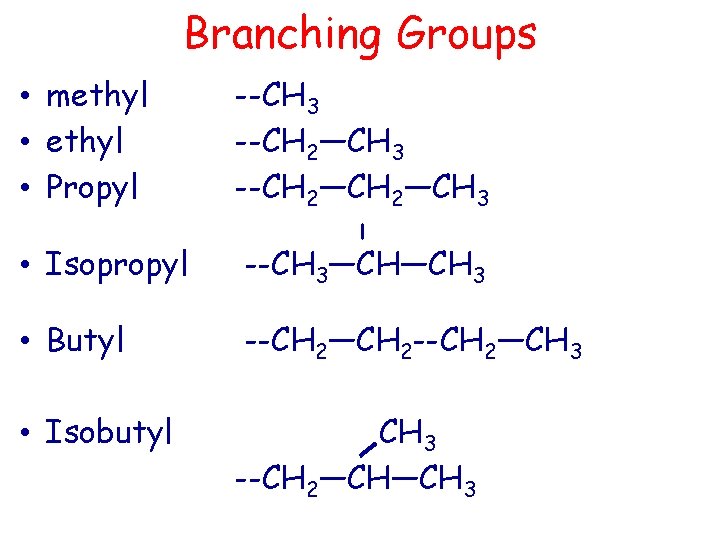
Branching Groups • methyl • Propyl --CH 3 --CH 2—CH 3 l • Isopropyl --CH 3—CH—CH 3 • Butyl --CH 2—CH 2 --CH 2—CH 3 • Isobutyl CH 3 --CH 2—CH—CH 3
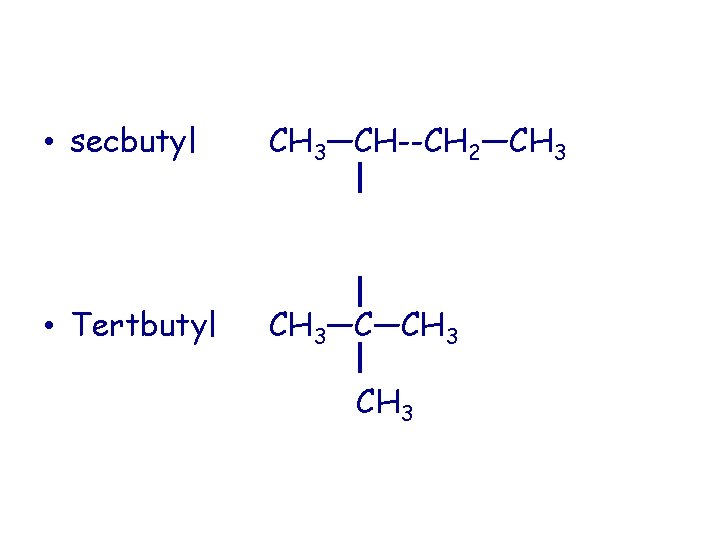
• secbutyl CH 3—CH--CH 2—CH 3 • Tertbutyl CH 3—C—CH 3

• 4. Alphabetize the groups. • 5 If there is more than one group with the same name, use the appropriate prefix with the group name. 2 groups – di 5 groups – penta 3 groups – tri 6 groups – hexa 4 groups – tetra- 7 groups – hepta-

• 6. Punctuate a. Numbers are separated by commas b. Numbers are separated from words by hyphens • 7. If a branching group chain consists of more than 4 carbons, the group is named as if it were a compound using the same rules (1 -6), with the following exceptions: a. the chain must start at the branching carbon b. the name ends in –yl, not in –ane c. The group is enclosed in parentheses with its position # outside the parentheses d. Alphabetize the group by the 1 st letter inside the parentheses.

• 8. If there are two chains of equal length, the chain with the greatest number of side chains is chosen as the main chain.
- Slides: 10