ALK IN LUNG CANCER PAST PRESENT AND FUTURE










![ALK-Positive Timeline EML 4 -ALK chromosomal rearrangements reported in NSCLC[1] EML 4 -ALK defines ALK-Positive Timeline EML 4 -ALK chromosomal rearrangements reported in NSCLC[1] EML 4 -ALK defines](https://slidetodoc.com/presentation_image_h2/e821d06de875b5435fddb1b00c96a686/image-38.jpg)


- Slides: 40

ALK IN LUNG CANCER: PAST, PRESENT, AND FUTURE


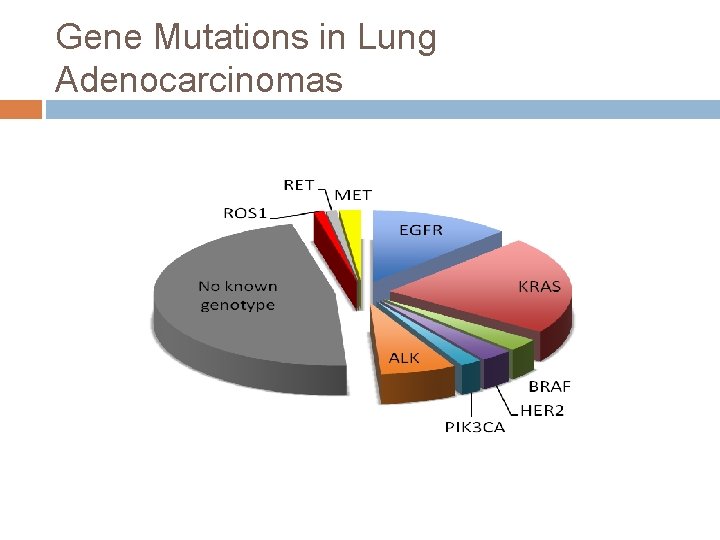
Gene Mutations in Lung Adenocarcinomas

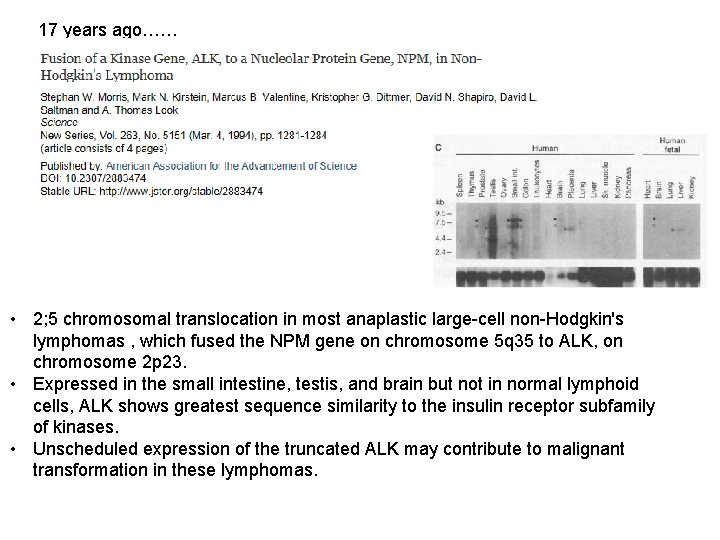
17 years ago…… • 2; 5 chromosomal translocation in most anaplastic large-cell non-Hodgkin's lymphomas , which fused the NPM gene on chromosome 5 q 35 to ALK, on chromosome 2 p 23. • Expressed in the small intestine, testis, and brain but not in normal lymphoid cells, ALK shows greatest sequence similarity to the insulin receptor subfamily of kinases. • Unscheduled expression of the truncated ALK may contribute to malignant transformation in these lymphomas.

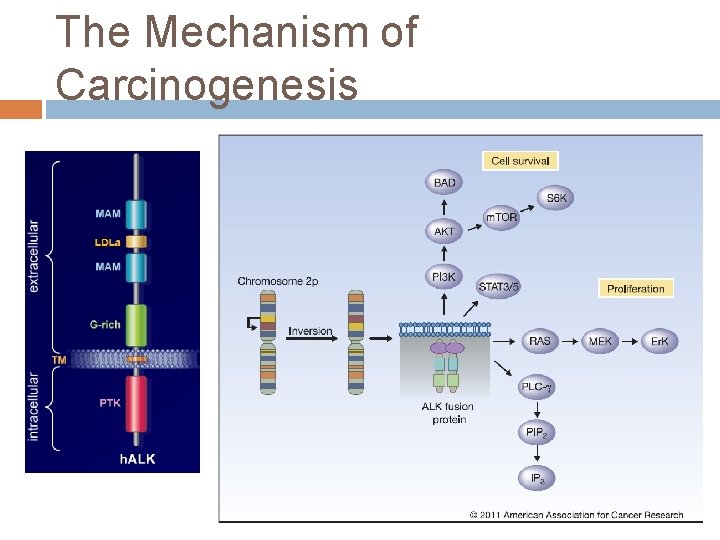
The Mechanism of Carcinogenesis


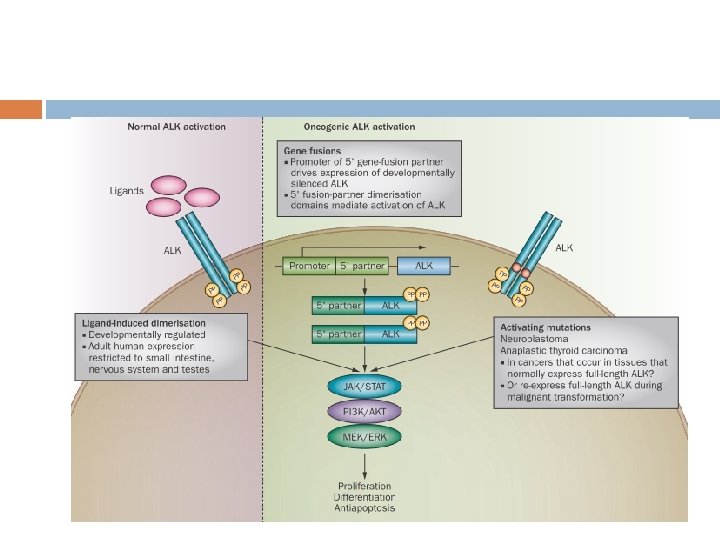
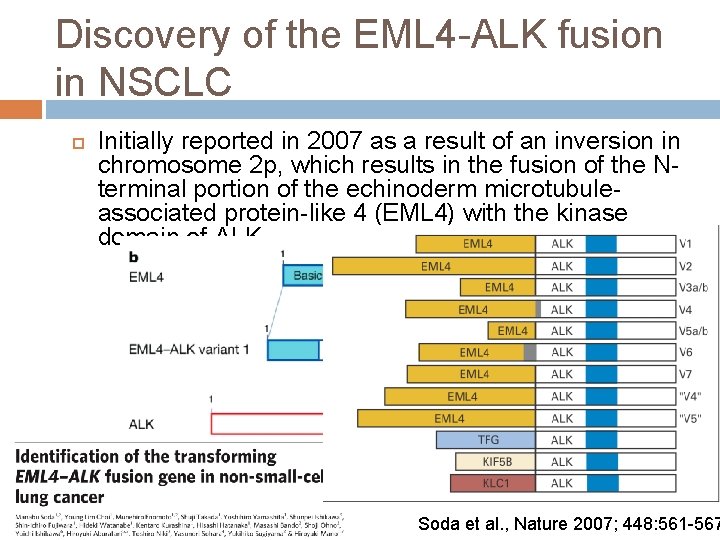
Discovery of the EML 4 -ALK fusion in NSCLC Initially reported in 2007 as a result of an inversion in chromosome 2 p, which results in the fusion of the Nterminal portion of the echinoderm microtubuleassociated protein-like 4 (EML 4) with the kinase domain of ALK. Soda et al. , Nature 2007; 448: 561 -567

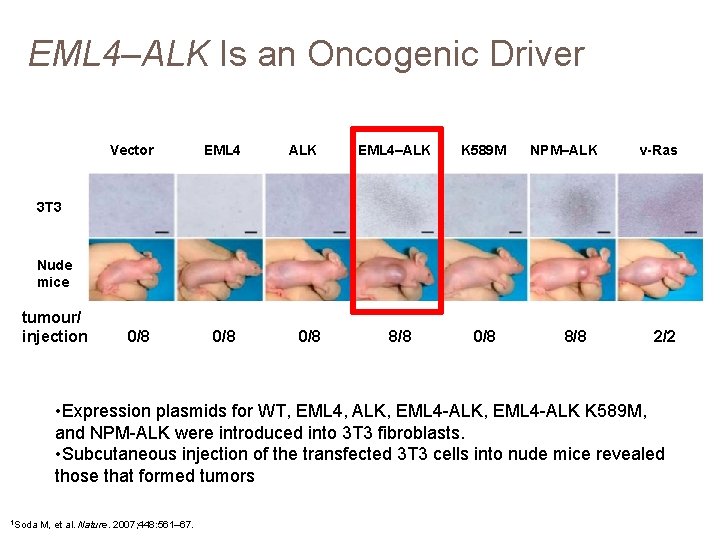
EML 4–ALK Is an Oncogenic Driver Vector EML 4 ALK EML 4–ALK K 589 M NPM–ALK v-Ras 3 T 3 Nude mice tumour/ injection 0/8 0/8 8/8 2/2 • Expression plasmids for WT, EML 4, ALK, EML 4 -ALK K 589 M, and NPM-ALK were introduced into 3 T 3 fibroblasts. • Subcutaneous injection of the transfected 3 T 3 cells into nude mice revealed those that formed tumors 1 Soda M, et al. Nature. 2007; 448: 561– 67.

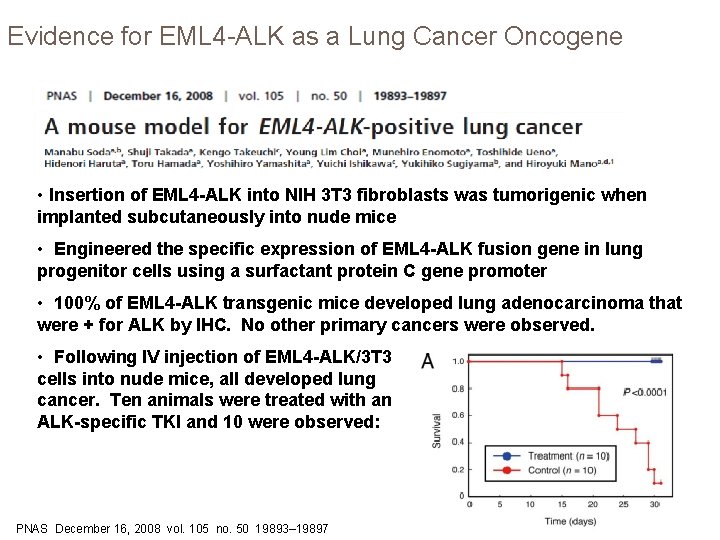
Evidence for EML 4 -ALK as a Lung Cancer Oncogene • Insertion of EML 4 -ALK into NIH 3 T 3 fibroblasts was tumorigenic when implanted subcutaneously into nude mice • Engineered the specific expression of EML 4 -ALK fusion gene in lung progenitor cells using a surfactant protein C gene promoter • 100% of EML 4 -ALK transgenic mice developed lung adenocarcinoma that were + for ALK by IHC. No other primary cancers were observed. • Following IV injection of EML 4 -ALK/3 T 3 cells into nude mice, all developed lung cancer. Ten animals were treated with an ALK-specific TKI and 10 were observed: PNAS December 16, 2008 vol. 105 no. 50 19893– 19897

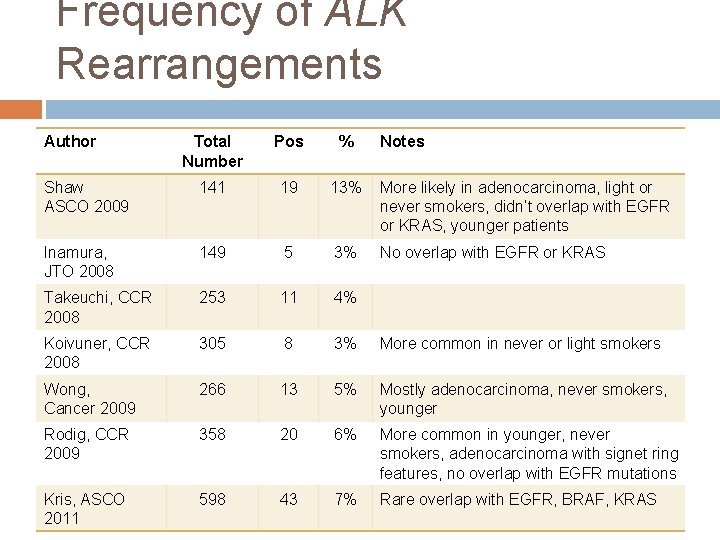
Frequency of ALK Rearrangements Author Total Number Pos % Notes Shaw ASCO 2009 141 19 13% More likely in adenocarcinoma, light or never smokers, didn’t overlap with EGFR or KRAS, younger patients Inamura, JTO 2008 149 5 3% No overlap with EGFR or KRAS Takeuchi, CCR 2008 253 11 4% Koivuner, CCR 2008 305 8 3% More common in never or light smokers Wong, Cancer 2009 266 13 5% Mostly adenocarcinoma, never smokers, younger Rodig, CCR 2009 358 20 6% More common in younger, never smokers, adenocarcinoma with signet ring features, no overlap with EGFR mutations Kris, ASCO 2011 598 43 7% Rare overlap with EGFR, BRAF, KRAS

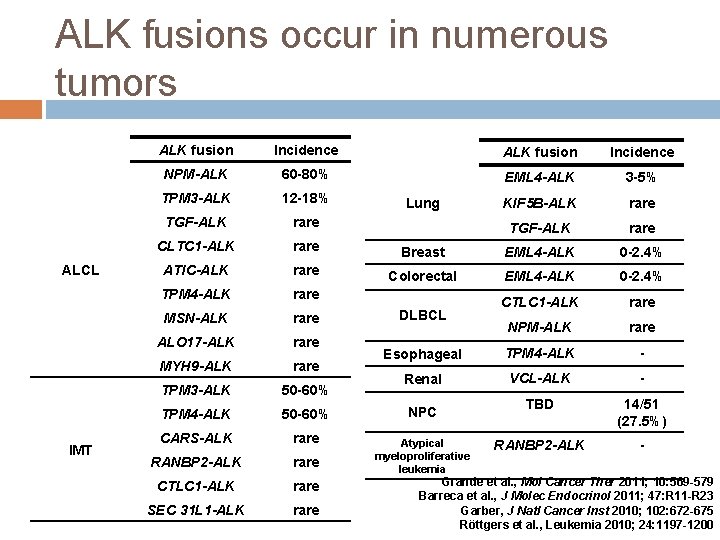
ALK fusions occur in numerous tumors ALCL IMT ALK fusion Incidence NPM-ALK 60 -80% EML 4 -ALK 3 -5% TPM 3 -ALK 12 -18% KIF 5 B-ALK rare TGF-ALK rare CLTC 1 -ALK rare Breast EML 4 -ALK 0 -2. 4% ATIC-ALK rare Colorectal EML 4 -ALK 0 -2. 4% TPM 4 -ALK rare CTLC 1 -ALK rare MSN-ALK rare ALO 17 -ALK rare NPM-ALK rare MYH 9 -ALK rare Esophageal TPM 4 -ALK - TPM 3 -ALK 50 -60% Renal VCL-ALK - TPM 4 -ALK 50 -60% TBD 14/51 (27. 5%) CARS-ALK rare RANBP 2 -ALK - RANBP 2 -ALK rare CTLC 1 -ALK rare SEC 31 L 1 -ALK rare Lung DLBCL NPC Atypical myeloproliferative leukemia Grande et al. , Mol Cancer Ther 2011; 10: 569 -579 Barreca et al. , J Molec Endocrinol 2011; 47: R 11 -R 23 Garber, J Natl Cancer Inst 2010; 102: 672 -675 Röttgers et al. , Leukemia 2010; 24: 1197 -1200

Other ALK alterations (mutations, gene amplification) ALK alteration Incidence Thyroid Mutations (L 1198 F, G 1201 E) 11% 6 -8% Neuroblastoma Mutations (F 1174 L, R 1275 Q) Amplification 4% ALK protein expression Glioblastoma Growth factor PTN protein, m. RNA expression - Murugan et al. , Cancer Res 2011; 71: 4403– 4411 Grande et al. , Mol Cancer Ther 2011; 10: 569 -579 Powers et al. , J Biol Chem 2002; 277: 14153 -14158 Lu et al. , J Biol Chem 2005; 280: 26953 -26964

How do we test for ALK rearrangments? Histology/IHC FISH/Cytogenetics PCR Sequencing

What can we do in ALK+ NSCLC patient?

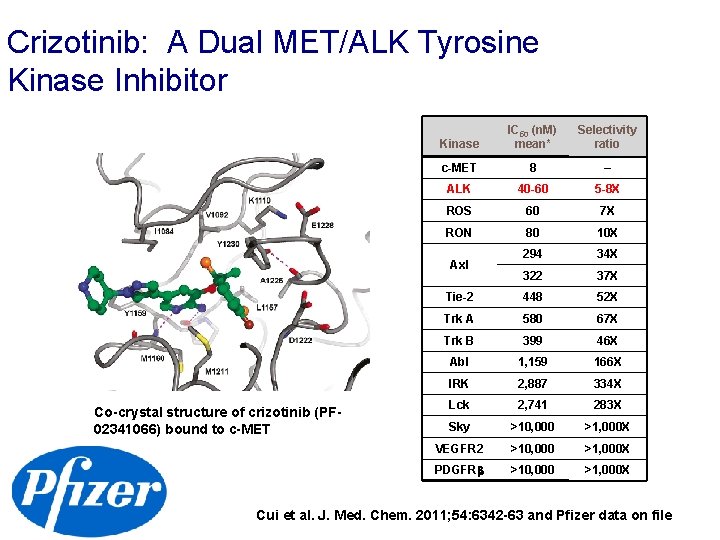
Crizotinib: A Dual MET/ALK Tyrosine Kinase Inhibitor Kinase IC 50 (n. M) mean* Selectivity ratio c-MET 8 – ALK 40 -60 5 -8 X ROS 60 7 X RON 80 10 X 294 34 X 322 37 X Tie-2 448 52 X Trk A 580 67 X Trk B 399 46 X Abl 1, 159 166 X IRK 2, 887 334 X Lck 2, 741 283 X Sky >10, 000 >1, 000 X VEGFR 2 >10, 000 >1, 000 X PDGFR >10, 000 >1, 000 X Axl Co-crystal structure of crizotinib (PF 02341066) bound to c-MET Cui et al. J. Med. Chem. 2011; 54: 6342 -63 and Pfizer data on file

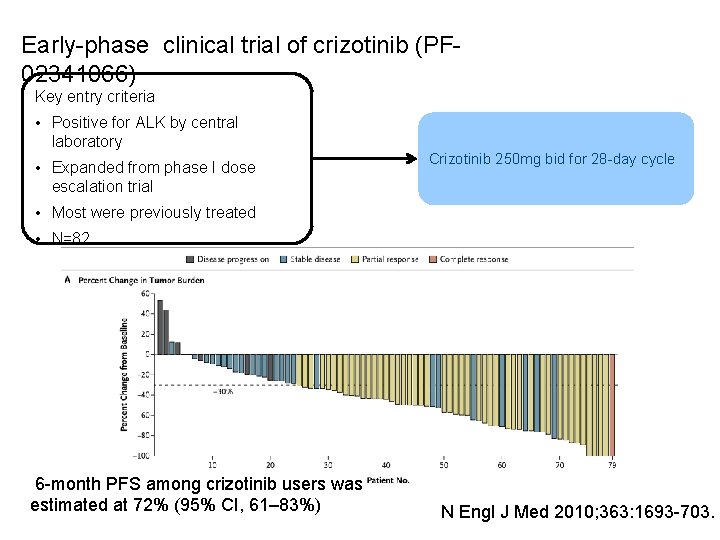
Early-phase clinical trial of crizotinib (PF 02341066) Key entry criteria • Positive for ALK by central laboratory • Expanded from phase I dose escalation trial Crizotinib 250 mg bid for 28 -day cycle • Most were previously treated • N=82 6 -month PFS among crizotinib users was estimated at 72% (95% CI, 61– 83%) N Engl J Med 2010; 363: 1693 -703.

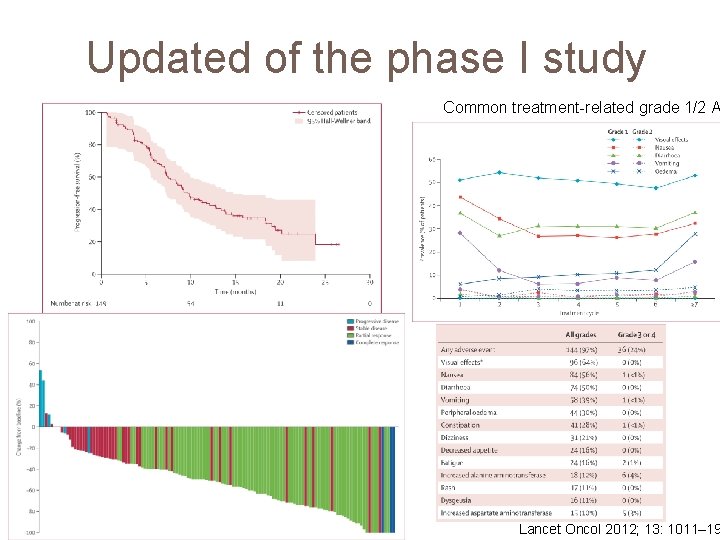
Updated of the phase I study Common treatment-related grade 1/2 A Lancet Oncol 2012; 13: 1011– 19

Overview of ongoing trials ● ● ALK inhibition, NSCLC – PROFILE 1007 – Ph III 2 nd line (NCT 00932893) – PROFILE 1014 – Ph III frontline (NCT 01154140) – PROFILE 1005 – Ph II pretreated (NCT 00932451) – PROFILE 1001 – Ph II expansion cohort (NCT 00585195) ALK inhibition, other tumor types – ● PROFILE 1013 – Ph I in non-NSCLC (NCT 01121588) Met inhibition – Study 1002 – Ph I/II with erlotinib (NCT 00965731) – Study 1006 – Ph I with PF-0299804 (dacomitinib), NSCLC (NCT 01121575)

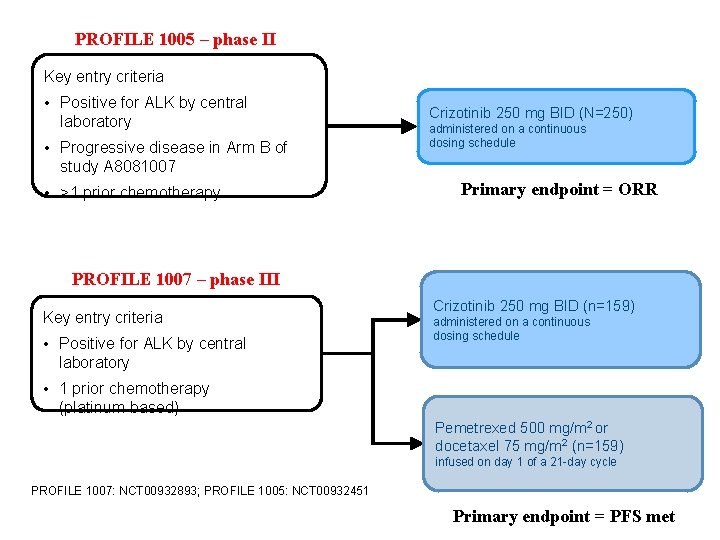
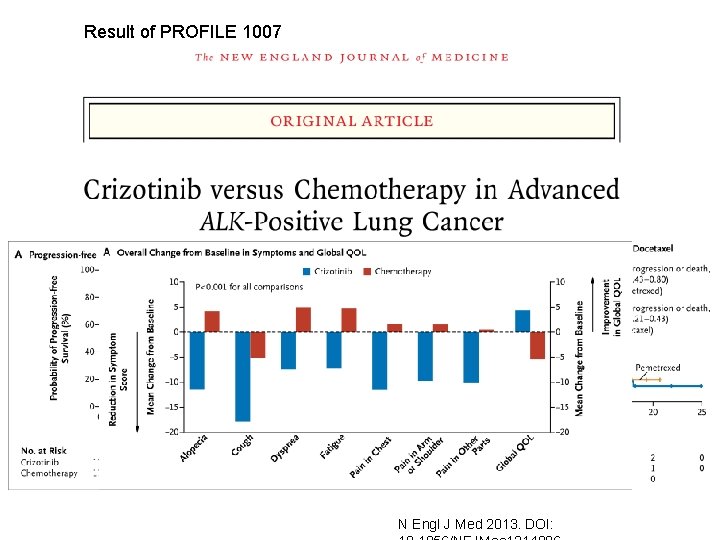
PROFILE 1005 – phase II Key entry criteria • Positive for ALK by central laboratory • Progressive disease in Arm B of study A 8081007 • >1 prior chemotherapy Crizotinib 250 mg BID (N=250) administered on a continuous dosing schedule Primary endpoint = ORR PROFILE 1007 – phase III Key entry criteria • Positive for ALK by central laboratory Crizotinib 250 mg BID (n=159) administered on a continuous dosing schedule • 1 prior chemotherapy (platinum-based) Pemetrexed 500 mg/m 2 or docetaxel 75 mg/m 2 (n=159) infused on day 1 of a 21 -day cycle PROFILE 1007: NCT 00932893; PROFILE 1005: NCT 00932451 Primary endpoint = PFS met

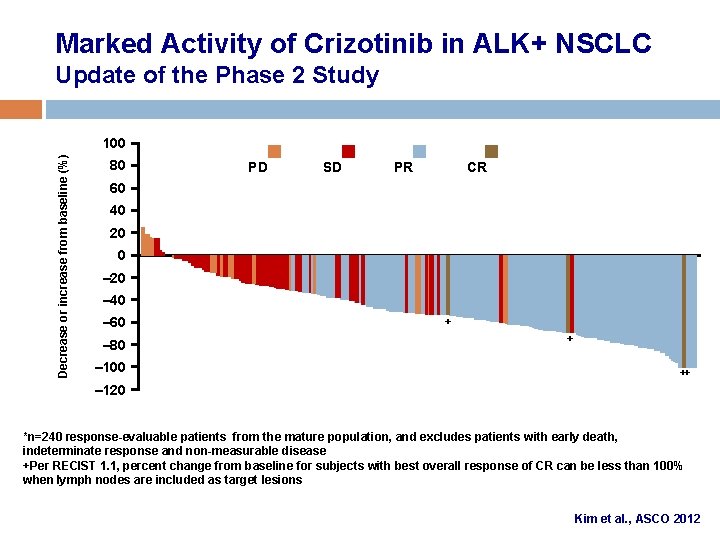
Marked Activity of Crizotinib in ALK+ NSCLC Update of the Phase 2 Study Decrease or increase from baseline (%) 100 80 PD SD PR CR 60 40 20 0 – 20 – 40 – 60 – 80 – 100 + + ++ – 120 *n=240 response-evaluable patients from the mature population, and excludes patients with early death, indeterminate response and non-measurable disease +Per RECIST 1. 1, percent change from baseline for subjects with best overall response of CR can be less than 100% when lymph nodes are included as target lesions Kim et al. , ASCO 2012

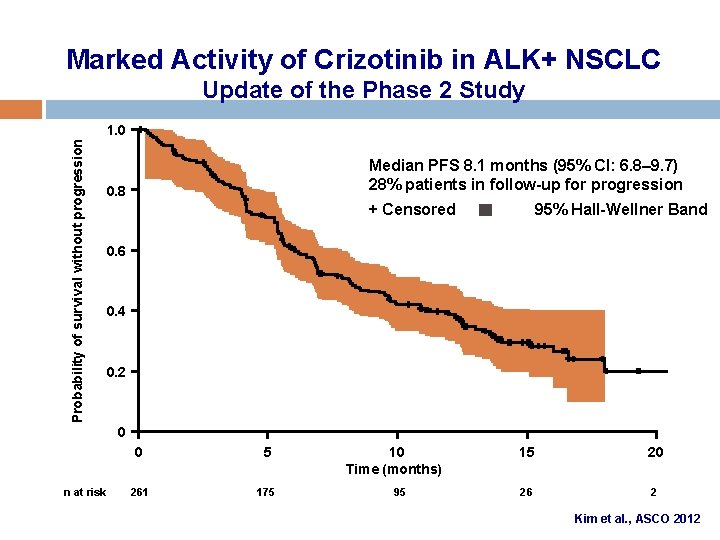
Marked Activity of Crizotinib in ALK+ NSCLC Update of the Phase 2 Study Probability of survival without progression 1. 0 Median PFS 8. 1 months (95% CI: 6. 8– 9. 7) 28% patients in follow-up for progression 0. 8 + Censored 95% Hall-Wellner Band 0. 6 0. 4 0. 2 0 n at risk 0 5 261 175 10 Time (months) 95 15 20 26 2 Kim et al. , ASCO 2012

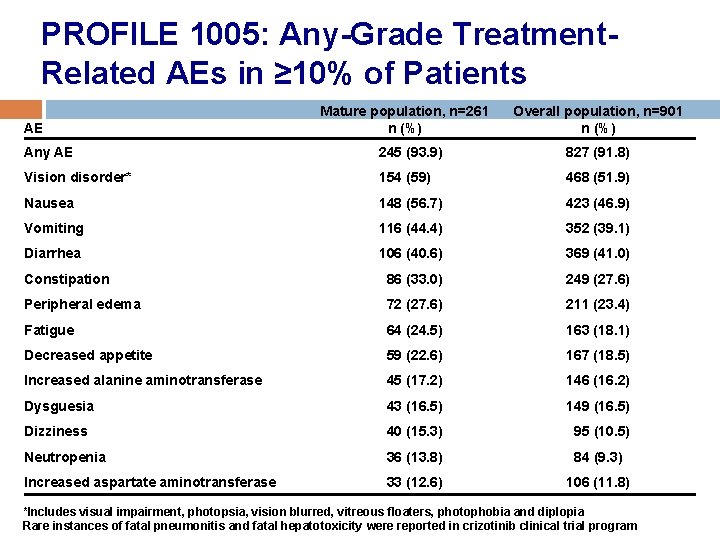
PROFILE 1005: Any-Grade Treatment. Related AEs in ≥ 10% of Patients AE Mature population, n=261 n (%) Overall population, n=901 n (%) Any AE 245 (93. 9) 827 (91. 8) Vision disorder* 154 (59) 468 (51. 9) Nausea 148 (56. 7) 423 (46. 9) Vomiting 116 (44. 4) 352 (39. 1) Diarrhea 106 (40. 6) 369 (41. 0) Constipation 86 (33. 0) 249 (27. 6) Peripheral edema 72 (27. 6) 211 (23. 4) Fatigue 64 (24. 5) 163 (18. 1) Decreased appetite 59 (22. 6) 167 (18. 5) Increased alanine aminotransferase 45 (17. 2) 146 (16. 2) Dysguesia 43 (16. 5) 149 (16. 5) Dizziness 40 (15. 3) 95 (10. 5) Neutropenia 36 (13. 8) 84 (9. 3) Increased aspartate aminotransferase 33 (12. 6) 106 (11. 8) *Includes visual impairment, photopsia, vision blurred, vitreous floaters, photophobia and diplopia Rare instances of fatal pneumonitis and fatal hepatotoxicity were reported in crizotinib clinical trial program

Result of PROFILE 1007 N Engl J Med 2013. DOI:

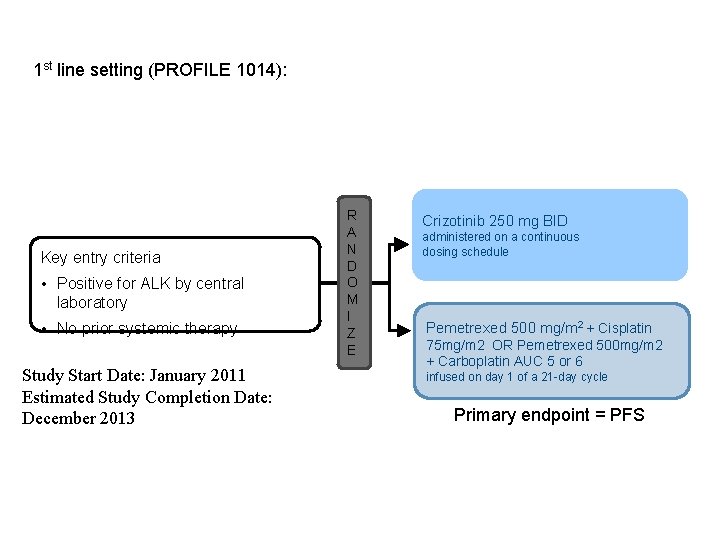
1 st line setting (PROFILE 1014): Key entry criteria • Positive for ALK by central laboratory • No prior systemic therapy Study Start Date: January 2011 Estimated Study Completion Date: December 2013 R A N D O M I Z E Crizotinib 250 mg BID administered on a continuous dosing schedule Pemetrexed 500 mg/m 2 + Cisplatin 75 mg/m 2 OR Pemetrexed 500 mg/m 2 + Carboplatin AUC 5 or 6 infused on day 1 of a 21 -day cycle Primary endpoint = PFS

The Future: Overcoming Crizotinib Resistance develops on average within the first year or two of TKI therapy…

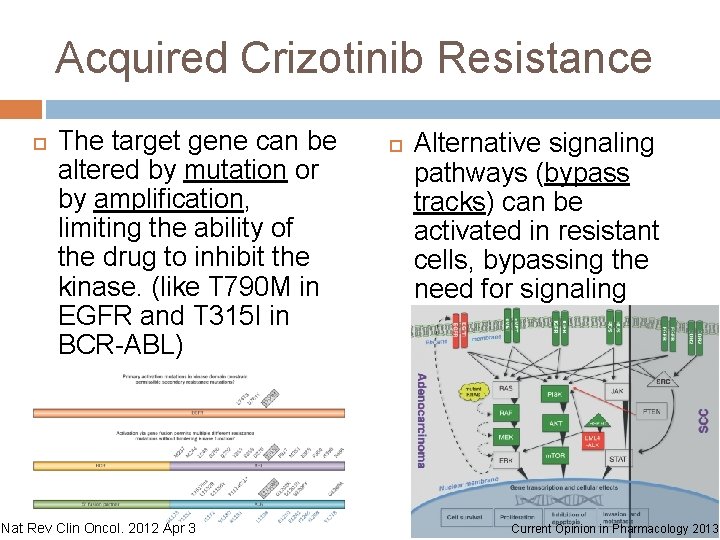
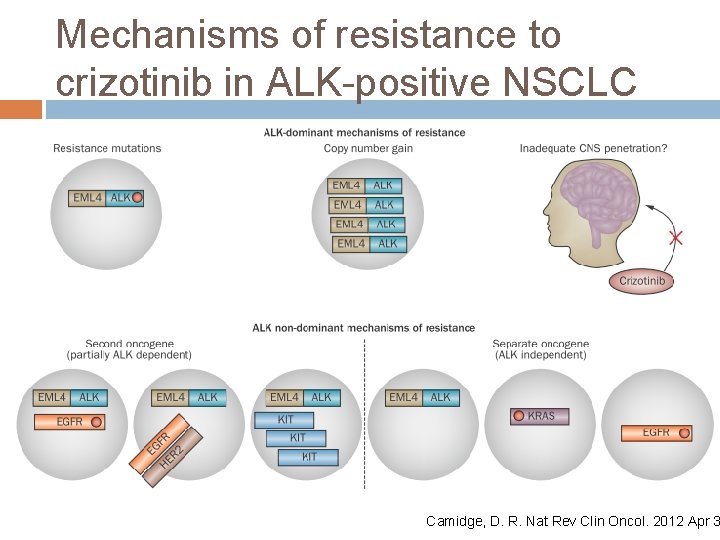
Acquired Crizotinib Resistance The target gene can be altered by mutation or by amplification, limiting the ability of the drug to inhibit the kinase. (like T 790 M in EGFR and T 315 I in BCR-ABL) Nat Rev Clin Oncol. 2012 Apr 3 Alternative signaling pathways (bypass tracks) can be activated in resistant cells, bypassing the need for signaling from the target. Current Opinion in Pharmacology 2013

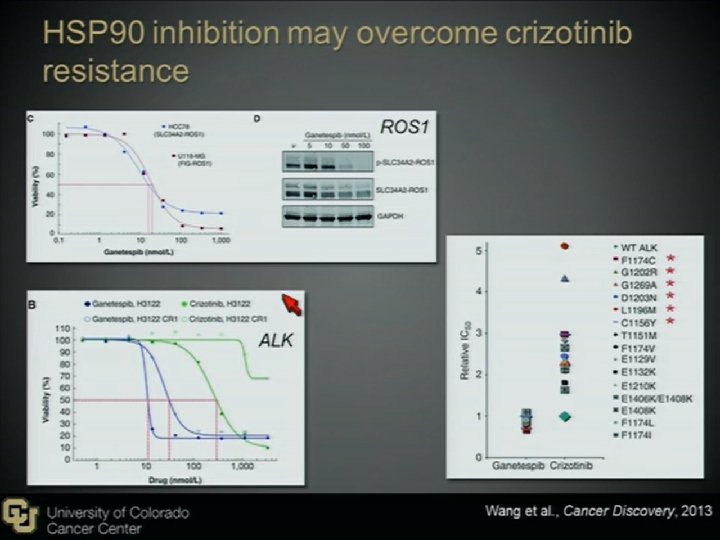
Acquired Crizotinib Resistance Mutation Up to 1/3 of relapsing patients, crizotinib resistance is mediated by secondary resistance mutations located in the ALK TK domain. Mutation Mechanism L 1196 M gatekeeper mutation, hinder TKI binding through steric hindrance G 1269 A lies directly in the ATPbinding Pocket G 1202 R and S 1206 Y Locate in solvent-exposed region of the kinase domain, decrease binding affinity of Crizotinib Most common

Acquired Crizotinib Resistance Amplification Amplification of the ALK fusion gene has also been reported in a small number of crizotinibresistant tumors

Acquired Crizotinib Resistance Alternative Pathway In crizotinib-resistant tumors, several distinct bypass tracks mediating resistance have been Sci Transl Med 2012 reported. � EGFR Cancer 2011 ½ cases with crizotinib resistance Sci Transl Med 2012 showed increased EGFR activity � c-KIT Confirmed by IHC, FISH, and c-Kit ligand Stem cell factor (SCF) Can be overcome by Imatinib combine with crizotinib � There may be more than 1 bypass

Mechanisms of resistance to crizotinib in ALK-positive NSCLC Camidge, D. R. Nat Rev Clin Oncol. 2012 Apr 3

What Can We Do?

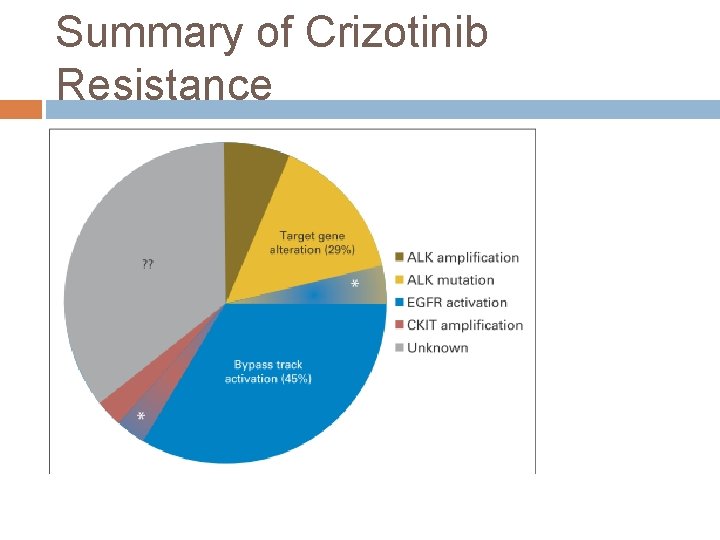
Summary of Crizotinib Resistance

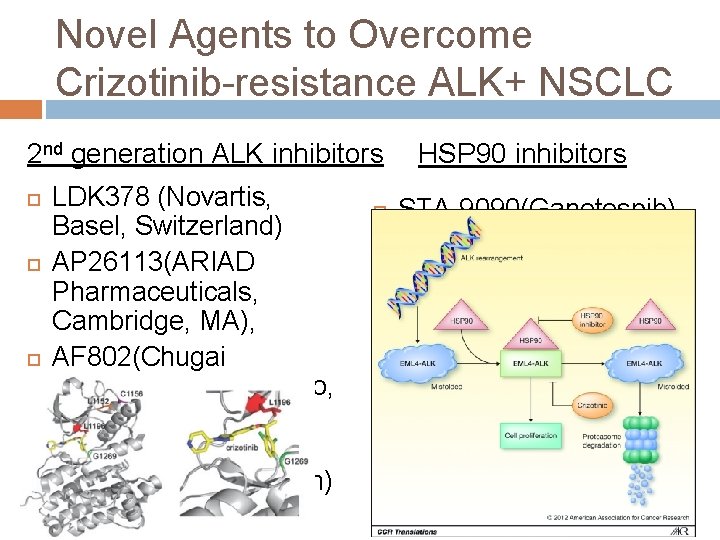
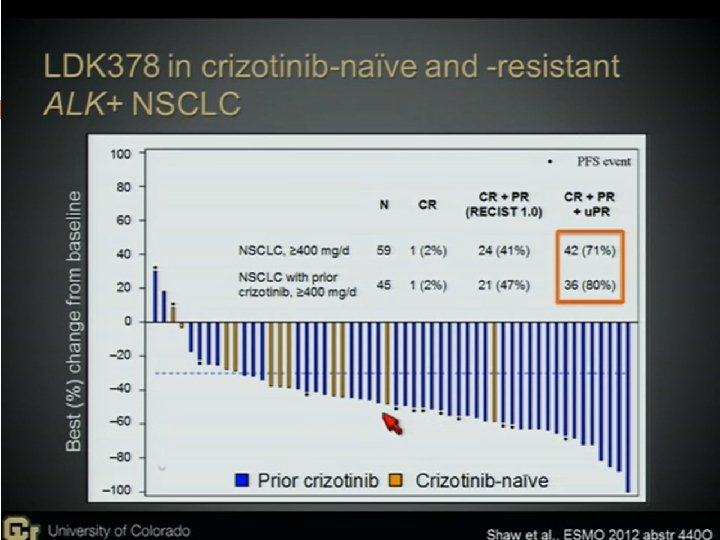
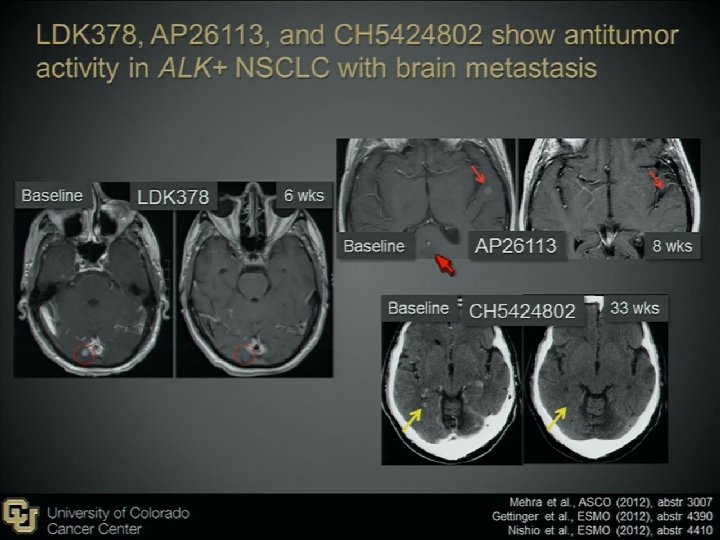
Novel Agents to Overcome Crizotinib-resistance ALK+ NSCLC 2 nd generation ALK inhibitors LDK 378 (Novartis, Basel, Switzerland) AP 26113(ARIAD Pharmaceuticals, Cambridge, MA), AF 802(Chugai Pharmaceutical, Tokyo, Japan) ASP 3026 (Astellas Pharma, Tokyo, Japan) HSP 90 inhibitors STA-9090(Ganetespib) AUY 922 IPI-504 AT 13387 DS-2248




The Possible Difficulties There may be more than 1 bypass mechanisms in one patient, therefore combination therapy may be needed. The mutation may be different in different tumor sites in the same patient. Biopsy in different site may be indicated There may be other unknown bypass pathway Frontline 2 nd ALK inhibitor, sequential use, or combine with other agent/CT (cocktail use)
![ALKPositive Timeline EML 4 ALK chromosomal rearrangements reported in NSCLC1 EML 4 ALK defines ALK-Positive Timeline EML 4 -ALK chromosomal rearrangements reported in NSCLC[1] EML 4 -ALK defines](https://slidetodoc.com/presentation_image_h2/e821d06de875b5435fddb1b00c96a686/image-38.jpg)
ALK-Positive Timeline EML 4 -ALK chromosomal rearrangements reported in NSCLC[1] EML 4 -ALK defines a molecular subset of NSCLC with distinct clinical characteristics[4] 2011 2009 2007 2008 Preclinical studies document antitumor activity of ALK inhibitors in lung cancer cell lines and xenografts[2, 3] FDA approves crizotinib for treatment of ALK+ NSCLC[6] 2010 Crizotinib produces a response in 47/82 ALK+ patients and a 6 -month PFS of 72%[5] 1. Soda M, et al. Nature 2007; 448: 561 -566. 2. Mc. Dermott U, et al. Cancer Res 2008; 68: 3389 -3395. 3. Koivunen JP, et al. Clin Cancer Res 2008; 14: 4275 -4283. 4. Shaw AT, et al. JCO 2009; 27: 4247 -4253. 5. Kwak EL, et al. N Engl J Med. 2010; 363: 1693 -1703. 6. US Food and Drug Administration. ? 2012 ? 2 nd generation ALK inhibitor TKIs and hsp inhibitors

Take Home Message EML 4 -ALK defines a new molecular subset of NSCLC Patients are more likely to be young, never/light smokers with adenocarcinoma Crizotinib results in a 6 -month PFS of 72% and overall response rate of 57% at 6. 4 months 2 nd generation ALK TKIs and HSP 90 inhibitors offer promise in patients with crizotinib resistance

Thanks for Your Attention!!