Alignment of Flexible Protein Structures Calmodulin Motivation Proteins
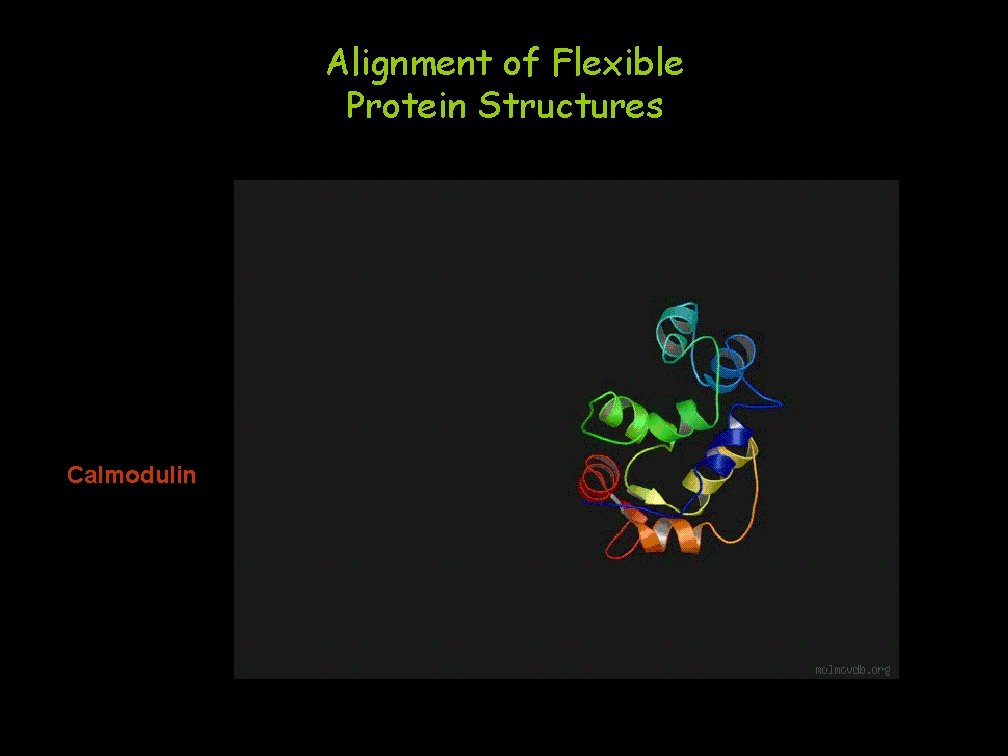
Alignment of Flexible Protein Structures Calmodulin

Motivation • Proteins are flexible. One would like to align proteins modulo the flexibility. • Hinge and shear protein domain motions (Gerstein, Lesk , Chotia).
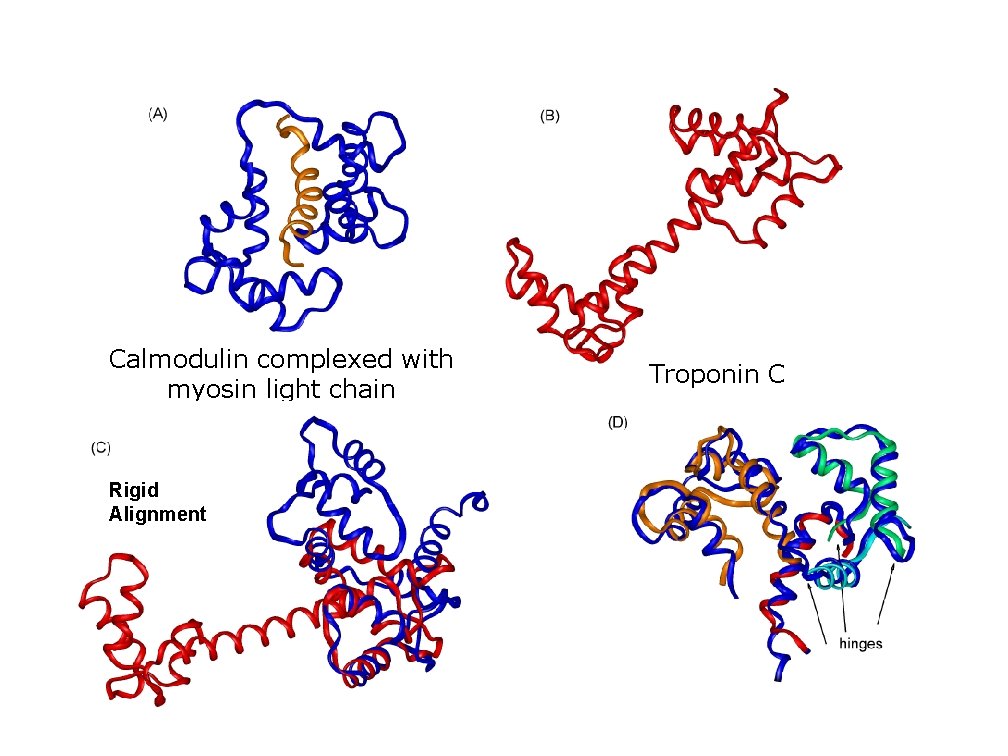
Calmodulin complexed with myosin light chain Rigid Alignment Troponin C

Proteins are flexible. One would like to align proteins modulo the flexibility. Flex. Prot algorithm: – Flexible structural alignment of hinge-bent proteins without prior knowledge of the flexible hinge regions. – Simultaneously detects hinge regions and aligns rigid subparts. – It is insensitive to insertions/deletions.
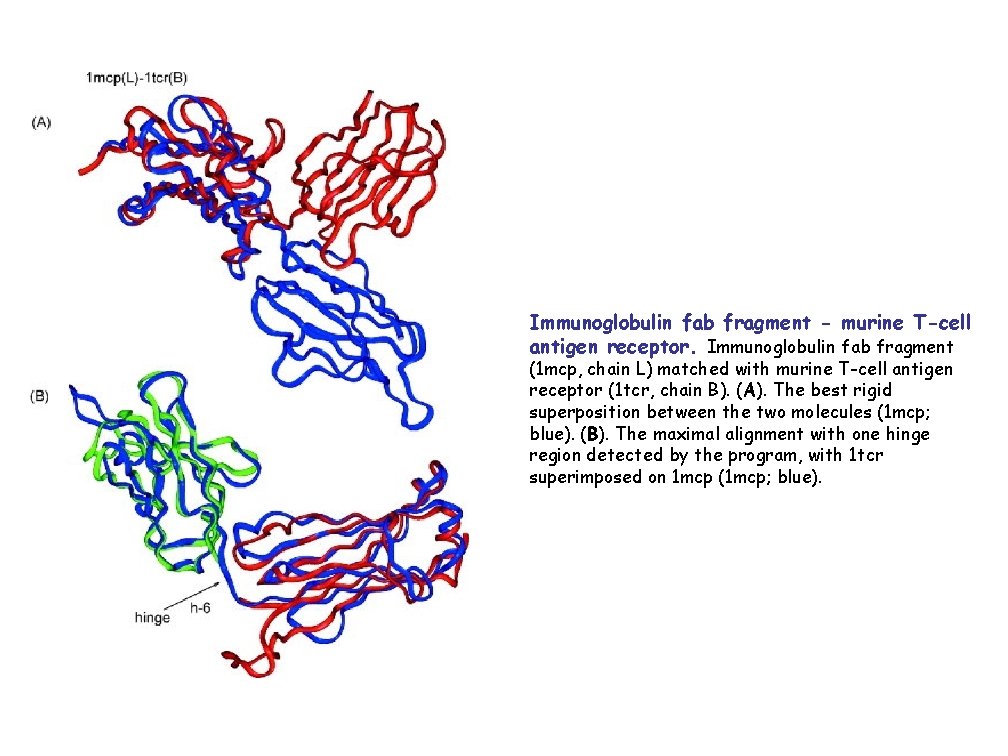
Immunoglobulin fab fragment - murine T-cell antigen receptor. Immunoglobulin fab fragment (1 mcp, chain L) matched with murine T-cell antigen receptor (1 tcr, chain B). (A). The best rigid superposition between the two molecules (1 mcp; blue). (B). The maximal alignment with one hinge region detected by the program, with 1 tcr superimposed on 1 mcp (1 mcp; blue).
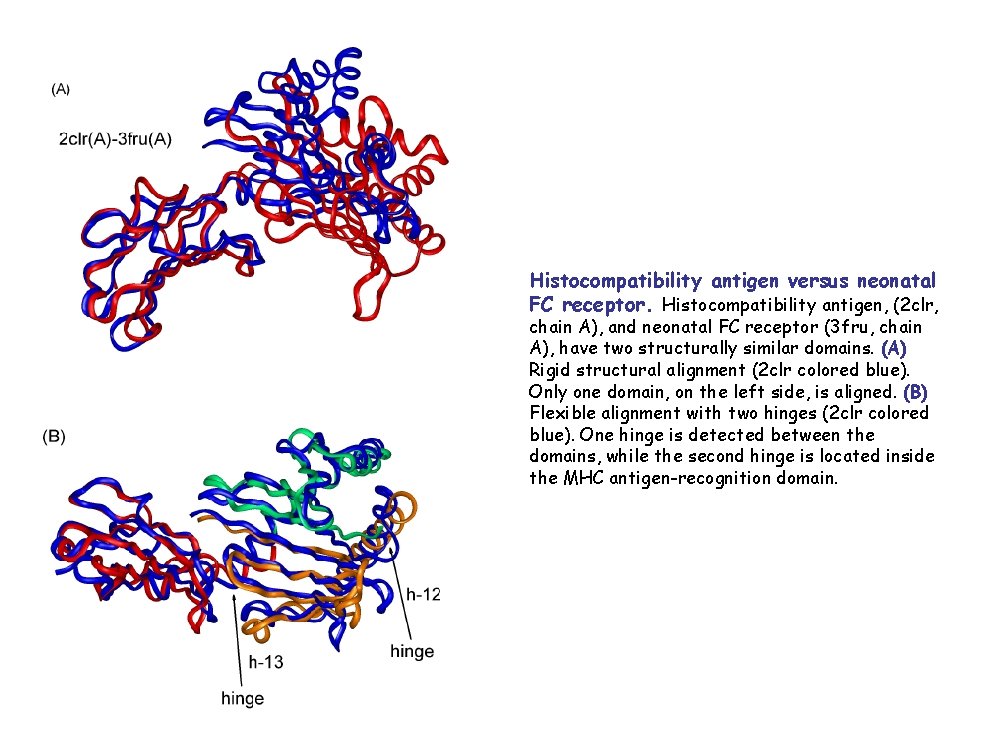
Histocompatibility antigen versus neonatal FC receptor. Histocompatibility antigen, (2 clr, chain A), and neonatal FC receptor (3 fru, chain A), have two structurally similar domains. (A) Rigid structural alignment (2 clr colored blue). Only one domain, on the left side, is aligned. (B) Flexible alignment with two hinges (2 clr colored blue). One hinge is detected between the domains, while the second hinge is located inside the MHC antigen-recognition domain.
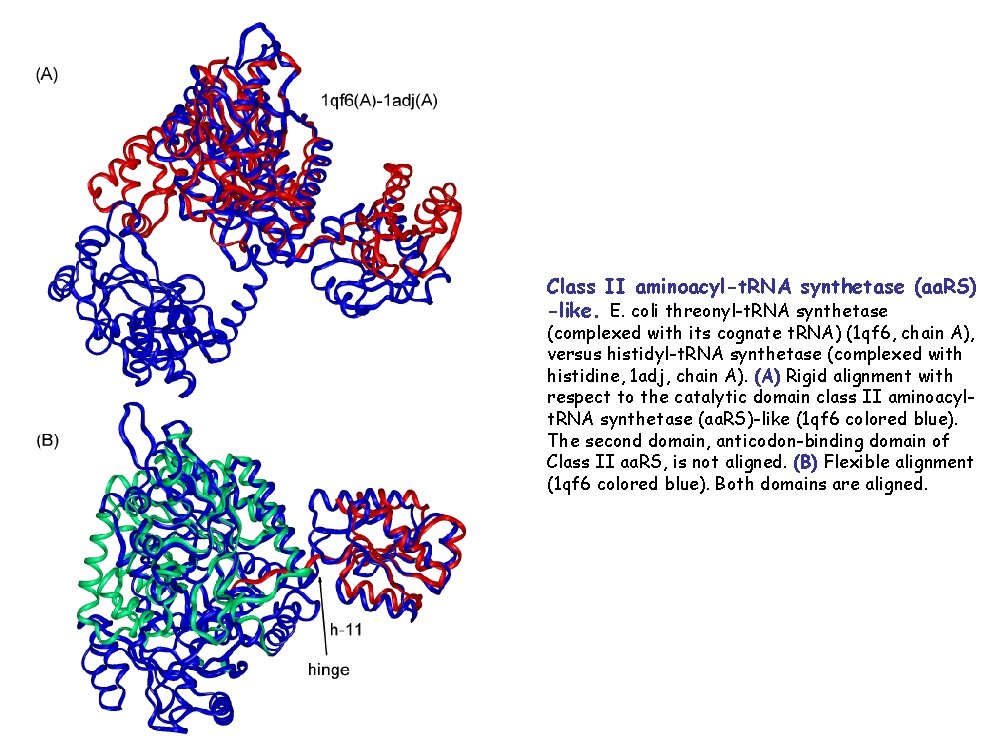
Class II aminoacyl-t. RNA synthetase (aa. RS) -like. E. coli threonyl-t. RNA synthetase (complexed with its cognate t. RNA) (1 qf 6, chain A), versus histidyl-t. RNA synthetase (complexed with histidine, 1 adj, chain A). (A) Rigid alignment with respect to the catalytic domain class II aminoacylt. RNA synthetase (aa. RS)-like (1 qf 6 colored blue). The second domain, anticodon-binding domain of Class II aa. RS, is not aligned. (B) Flexible alignment (1 qf 6 colored blue). Both domains are aligned.
Flex. Prot online http: //bioinfo 3 d. cs. tau. ac. il/Flex. Prot/
- Slides: 9