Algebraic Symmetries in Quantum Chemistry Clifford Algebra and

Algebraic Symmetries in Quantum Chemistry Clifford Algebra and Para-Fermi Algebra in Correlated Many. Electron Theories Nicholas D. K. Petraco John Jay College and the Graduate Center City University of New York

● ● Part I, The Setting – Quantum Chemistry and many-electron wave functions – Solving the Schrödinger equation including electron correlation Part II, Mathematical Tools – ● ● Outline Representation Theory Part III, Application – Spin-adaptation and some algebra – Representation theory of the unitary group U(n) – Para-Fermi algebra – The Clifford algebra unitary group U(2 n) – U(n) module in U(2 n) form – Matrix element evaluation scheme – Speculation! Acknowledgements
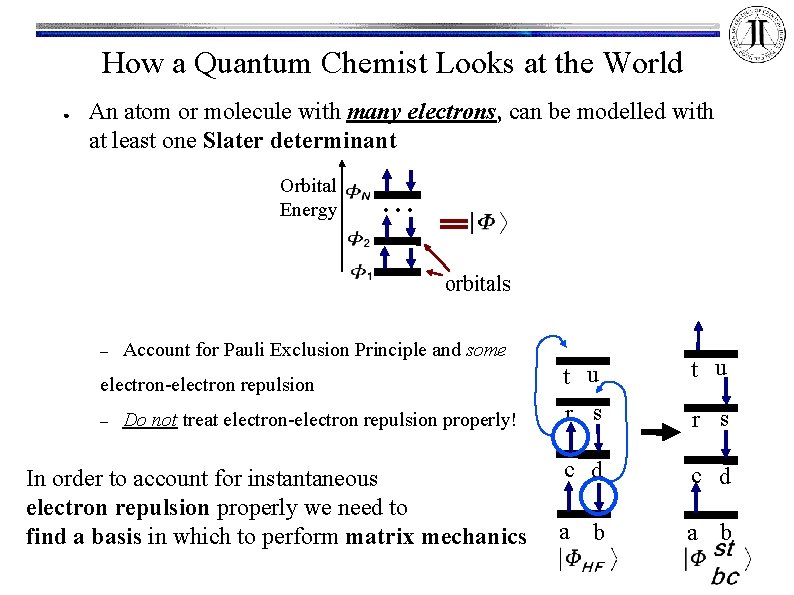
How a Quantum Chemist Looks at the World ● An atom or molecule with many electrons, can be modelled with at least one Slater determinant Orbital Energy . . . orbitals – Account for Pauli Exclusion Principle and some electron-electron repulsion – Do not treat electron-electron repulsion properly! In order to account for instantaneous electron repulsion properly we need to find a basis in which to perform matrix mechanics t u r s c d a b

How a Quantum Chemist Looks at the World • Solve the time-independent Schrödinger equation for atomic and molecular systems o Choose a finite one-electron basis set composed of 2 n spin-orbitals. o This lets us write the Hamiltonian in second quantized form as: o For an N-electron system expand exact wave function in “configurations” from the totally antisymmetric tensor product space:

Problems, Problems! This simplistic approach presents a horrendous computational problem! ● The many electron basis scales as: o ● ● Three principle approaches to solve the Schrödinger equation 1. Configuration Interaction (CI). Requires many 2. Perturbation Theory (PT). Requires many hard to evaluate 3. Coupled Cluster Theory (CC). Requires hard to evaluate Physical inconsistencies creep into the determinant representation of the many-electron basis!

“New Tools” A Crash Course in Representation Theory ● ● ● A “group” is a special collection of “operators” which transform a given set of “vectors” V, among themselves Really we only need the “algebra” associated with the group Our vectors will be many-electron wave functions The operators that make up our algebra are electron excitation operators and electron number operators A “representation” G is a recipe (homomorphism) for writing each operator in the algebra A as a “matrix” Given a “vector space”, V and an algebra A then subspace” (“invariant module”) if is an “invariant

An “irreducible module” (“irreducible representation space” or “irrep”) is an invariant subspace that cannot be written as a sum of smaller invariant subspaces ● Every irrep can be labelled by a list of “weights”, G[m] ● o Our lists of weights correspond to pictures called “Weyl frames” o Weights are eigenvalues of “invariance operators” algebra o Invariance operators are in general elements in the “universal enveloping algebra” of A, o Every vector in the irrep is assigned a unique pattern of weights stemming from the algebra A and any if its subalgebras ● , made from elements in the The weight patterns we will use are called “Weyl tableaux”

A Closer Look At Spin ● ● ● Since to good approximation, and the Hamiltonian for most chemical systems is spin independent: Also and are invariants in , so their eigenvalues label irreps of the su(2)-module of many electron wave functions Slater determinants are a common and convenient basis used for many-electron problems (i. e. basis for ). Slater dets. are always eigenfunctions of but not always of ! This basis yields “spin-contaminated” solutions to the Schrödinger eq. We loose the advantage of partial diagonalization of adapted basis. in a non-spin-

Unitary Transformation of Orbitals ● V 2 n is invariant to unitary transformations: Thus: where Therefore V 2 n carries the fundamental irrep, ● of U(2 n)! Through the same analysis: Vn carries the fundamental irrep of U(n) S 2 carries the fundamental irrep of U(2)

Now For Some Algebra ● Let Generators of: U(2 n) and with: U(n) Lie product of u(n): U(2)

Method to construct eigenstates of Tensor Irreps of U(n) Gel’fand Tsetlin canonical orthonormal basis. ● o Gel’fand-Tsetlin basis adapted to the subgroup chain: o Irreps of U(k) characterized by highest weight vectors mk o for N-electron wave functions carries the totally antisymmetric irrep of U(2 n), ● Gel’fand-Tsetlin (GT) basis of U(2 n) is not an eigenbasis of ● We consider the subgroup chain instead: – ● Only two column irreps of U(n) need to be considered The GT basis of U(n) is an eigenbasis of !

A Detour Through the Strange Land of Para-Fermi Algebra ● Operators forming a para-fermi algebra satisfy ● The o ● The and can be built from the “Green ansatz” are “weird” kinds of Fermions satisfying However, unitary group generators can be built as

Para-Fermi Algebra in Quantum Chemistry ● Define the operators ● Then the “weird” fermions are given as ● And the para-fermi operators are defined through o Two terms in the sum, therefore electronic orbitals are para-fermions of order 2

Clifford Algebra Unitary Group U(2 n) Consider the multispinor space spanned by nth-rank tensors of (single particle Fermionic) spin eigenvectors ● carries the fundamental reps of SO(m), m = 2 n or 2 n+1 and the unitary group U(2 n) ● carries tensor irreps of U(2 n) o ● Using para-Fermi algebra, one can show only contains the p-column irrep of U(n) at least once. of U(2 n) For the many-electron problem p = 2 and thus All G[2 a 1 b 0 c] of U(n) are contained in G[2] of U(2 n), the dynamical group of Quantum Chemistry!

Where the Clifford Algebra Part Comes in ● The monomials are a basis for the Clifford algebra Cn: primitive “Clifford numbers” ● The monomials can be used to construct generators of U(2 n).

● Since m is a vector of 0’s and 1’s then using maps: we can go between the binary and base 10 numbers with m = m 2 ● Elements of a 2 -column U(n)-module, are linear combinations of two-box (Weyl) tableaux

Action of U(n) Generators on ● ● Action of U(2 n) generators on in Form is trivial to evaluate: Since any two-column tableau can be expressed as a linear combination of two-box tableaux, expand U(n) generators in terms of U(2 n) generators: weights of the ith component in the pth monomial hard to get sign for specific E copious!!!

Action of U(n) Generators on in Form Given a G[2 a 1 b 0 c] the highest weight state in two-box form ● can be lowered to generate the rest of the module. Get around long expansion by “selecting out” a non-zero result on the to the right. ● – Consider ● with Examine if e. g. If ● contains and (lowering generator) and/or then Generate r from i and j with p and/or q e. g. If contains then: that yield contains .
![Basis Selection and Generation ● Given a G[2 a 1 b 0 c] lower Basis Selection and Generation ● Given a G[2 a 1 b 0 c] lower](http://slidetodoc.com/presentation_image_h2/27af6c95f6d6810e9c42c495baec8efc/image-19.jpg)
Basis Selection and Generation ● Given a G[2 a 1 b 0 c] lower from highest weight state according to a number of schemes Clifford-Weyl Basis Generate by simple lowering action and thus spin-adapted Equivalent to Rumer-Weyl “Valence Bond” basis Can be stored in distinct row table and thus has directed graph representation NOT ORTHAGONAL Gel’fand-Tsetlin Basis Generate by Schmidt orthagonalizing CW basis or lowering with Nagel. Moshinsky lowering operators Can be stored in DRT Orthagonal Lacks certain unitary invariance properties required by open shell coupled cluster theory

Basis Selection and Generation Jezorski-Paldus-Jankowski Basis Use U(n) tensor “excitation” operators adapted to the chain: Resulting operators have a nice “hole-particle” interpretation No need to generate basis explicitly Orthagonal and spin-adapted Has proper invariance properties required for open-shell Coupled Cluster Symmetry adaptation accomplished with Wigner operators from SN group algebra Operators in general contain “spectator” indices which lengthen computations and result in even more “unnatural scaling” Determinant Basis Just use the two-box tableau Easy to generate Symmetric Tensor Product between two determinants Orthagonal NOT SPIN-ADAPTED

Formulation of Common Correlated Quantum Chemical Methods ● Equations of all these methods can be formulated in terms of coefficients (known or unknown) multiplied by a matrix elements sandwiching elements of : ● Configure Interaction ● Coupled Cluster Theory ● Rayleigh-Schrödinger Perturbation Theory

Formulation of Common Correlated Quantum Chemical Methods One can use induction on the indices of each orbital subspace ● – core – active – virtual to show that the multi-generator matrix elements are invariant to the addition or subtraction of orbitals within each subspace – This invariance allows one to use numerical indices on these matrix elements and generate closed formulas

Speculation A composite particle composed of two electrons occupy “geminals” ● o There are various schemes to form geminals from pairs of orbitals Most forms of geminals behave as bosons form a secondquantized point of view: ● o The geminals that are formed from “bosonized” orbitals are really only approximate bosons, form a purists point of view o Also, there are lots of ways to bosonize orbitals An openshell molecule is composed of paired and unpaired electrons in orbitals ● o SPECULATION! Can we treat general openshell atoms and molecules as interacting systems of (approximate) bosons and fermions?

Supersymmetry in Quantum Chemistry? Composite systems of interacting bosons and fermions with the added symmetry that boson and fermions can transform into one another can be treated be Lie superalgebras ● In nuclear physics there is the “supersymmetric interacting boson-fermion models” of Iachello et al. ● ● o Pairs of nucleons are treated approximately as bosons o Any unpaired nucleons are treated as fermions o “Excitation” of a boson to a fermion changes the net particle number and interrelates nuclei of different masses by classifying them as transforming as the same superirrep (the totally supersymmetric one) o Model works ok when compared to experiment Can we interrelate molecules in different sectors of Fock space and include proper spin adaptation by studying U(m|n) modules?

Acknowledgments ● John Jay College and CUNY ● My collaborators and colleagues: o Prof. Josef Paldus o Prof. Marcel Nooijen o Prof. Debashis Mukherjee o Sunita Ramsarran o Chris Barden o Prof. Jon Riensrta-Kiracofe
- Slides: 25