Aldehydes and Ketones Prepared by Dr S Ignatius

Aldehydes and Ketones Prepared by Dr. S. Ignatius Arockiam Dept of chemistry

INTRODUCTION ØMany of the names of aldehydes and ketones are derived from the names of the corresponding carboxylic acids. ØThe information about the nomenclature of carboxylic acids is very useful to give the nomenclature of aldehydes and ketones

Naming of Carboxylic acids R-COOH, R-CO 2 H, Common names HCO 2 H - Formic acid CH 3 CO 2 H - Acetic acid CH 3 CH 2 CO 2 H - Propionic acid CH 3 CH 2 CO 2 H - Butyric acid CH 3 CH 2 CH 2 CO 2 H - Valeric acid
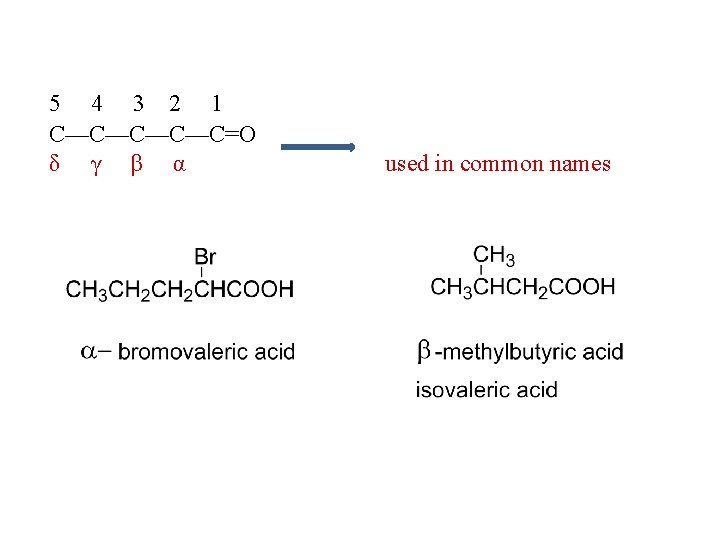
5 4 3 2 1 C—C—C=O δ γ β α used in common names

NAMING OF AROMATIC COMPOUNDS
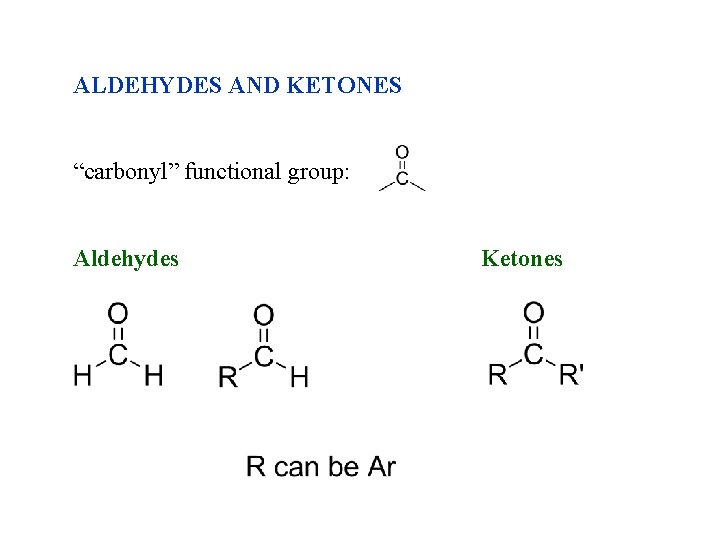
ALDEHYDES AND KETONES “carbonyl” functional group: Aldehydes Ketones

NAMING OF ALDEHYDES FROM THEIR COMMON NAMES (i) To name the aldehydes remove –ic acid from the carboxylic acid (ii) add the suffix aldehyde in the end of the compound. CH 3 CH 2 CH=O butyraldehyde CH 3 CHCH=O isobutyraldehyde (α-methylpropionaldehyde)

NOMENCLATURE OF ORGANIC COMPOUNDS v. Select the longest one from the parent chain containing the carbonyl group. v Remove the last letter “e” and then add “al” in the name of the aldehyde. HCHO - Methanal CH 3 CHO - Ethanal CH 3 CH 2 CHO - butanal CH 3 CH(CH 3)CHO - 2 -methylpropanal
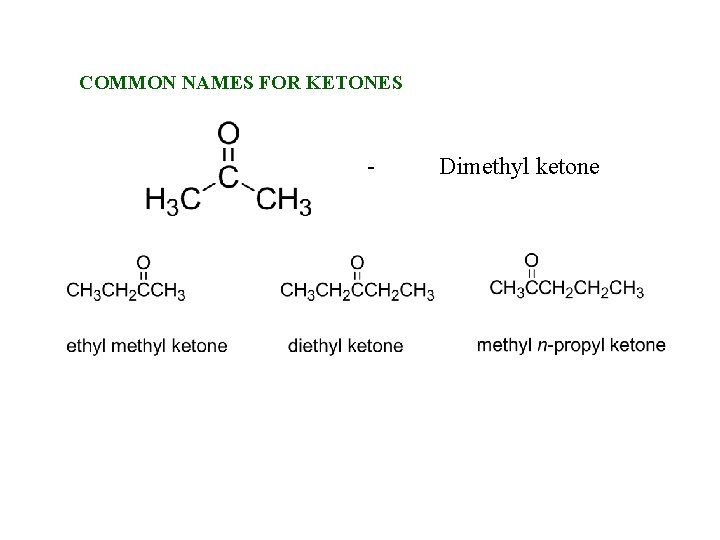
COMMON NAMES FOR KETONES - Dimethyl ketone
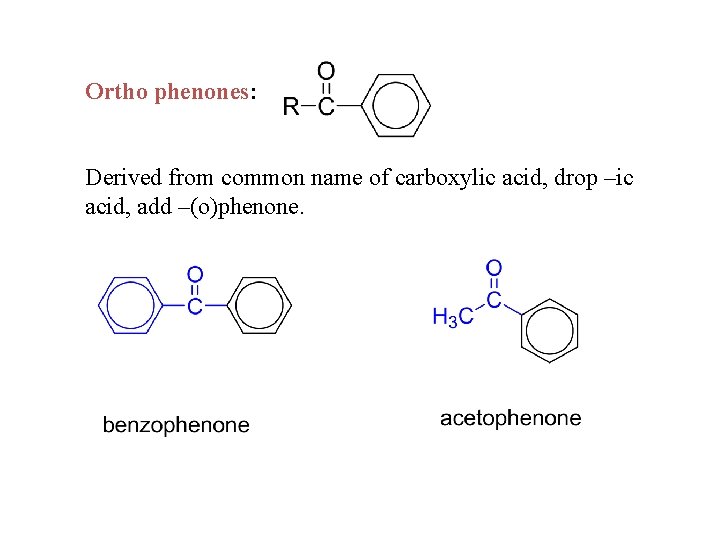
Ortho phenones: Derived from common name of carboxylic acid, drop –ic acid, add –(o)phenone.

Ketones: IUPAC nomenclature (i) Parent chain is the longest continuous carbon chain containing the carbonyl group. (ii) drop –e, add –one. Prefix a locant for the position of the carbonyl using the principle of lower number.

Physical properties: (i)Carbonyl compounds are polar in nature and there is no hydrogen bonding between the atoms (ii)Melting and boiling points are relatively moderate for covalent substances (iii) Compounds are water insoluble. Except the compounds having four carbons or less)

Spectroscopic analysis: IR gives the spectroscopy gives the following strectching frequencies Compound Strectching frequency (cm-1) RCHO 1725 Ar. CHO 1700 R 2 CO 1710

Oxidation/Reduction: oxidation numbers oxidation number of the compounds increases from alkane to carboxylic acid. oxidation -4 CH 4 -2 CH 3 OH 0 H 2 C=O +2 HCO 2 H alkane alcohol aldehyde carboxylic acid reduction

Thank you
- Slides: 16