Aldehydes and Ketones E V Blackburn 2008 Structure

Aldehydes and Ketones © E. V. Blackburn, 2008
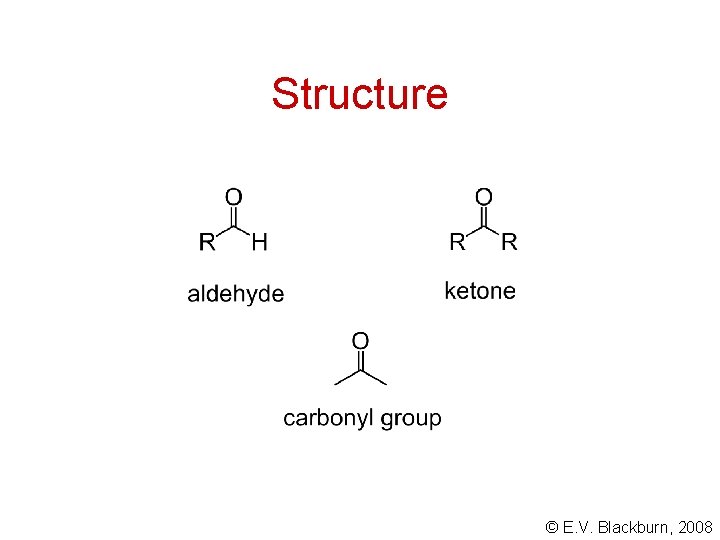
Structure © E. V. Blackburn, 2008
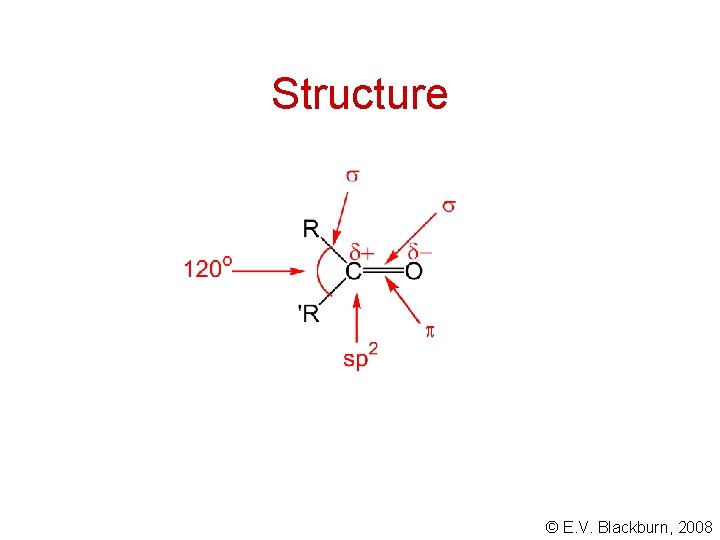
Structure © E. V. Blackburn, 2008

Nomenclature of aldehydes Aldehydes are named by adding the suffix -al to the name of the corresponding hydrocarbon. The carbonyl carbon is given the number 1. ethanal acetaldehyde © E. V. Blackburn, 2008
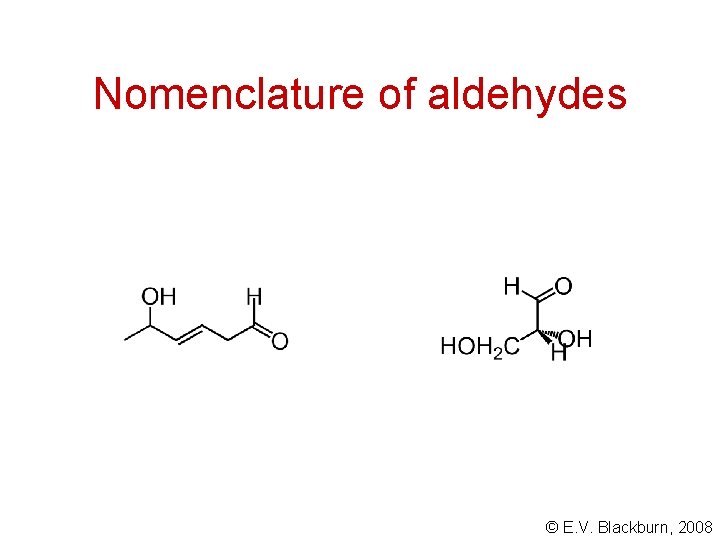
Nomenclature of aldehydes © E. V. Blackburn, 2008
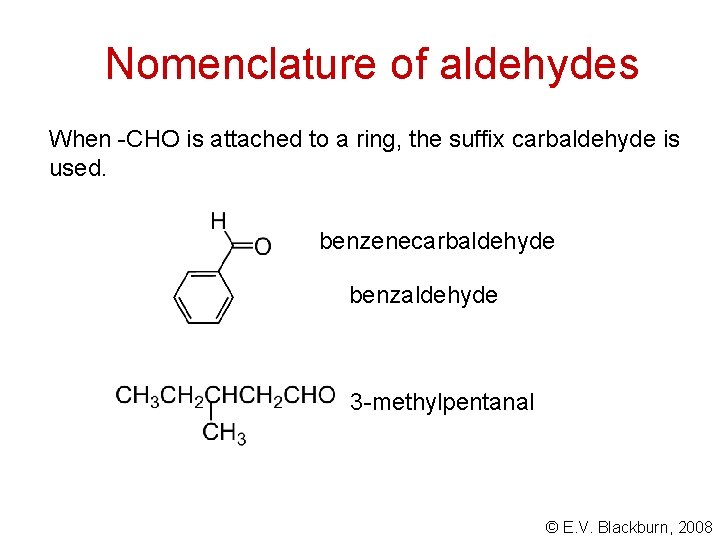
Nomenclature of aldehydes When -CHO is attached to a ring, the suffix carbaldehyde is used. benzenecarbaldehyde benzaldehyde 3 -methylpentanal © E. V. Blackburn, 2008
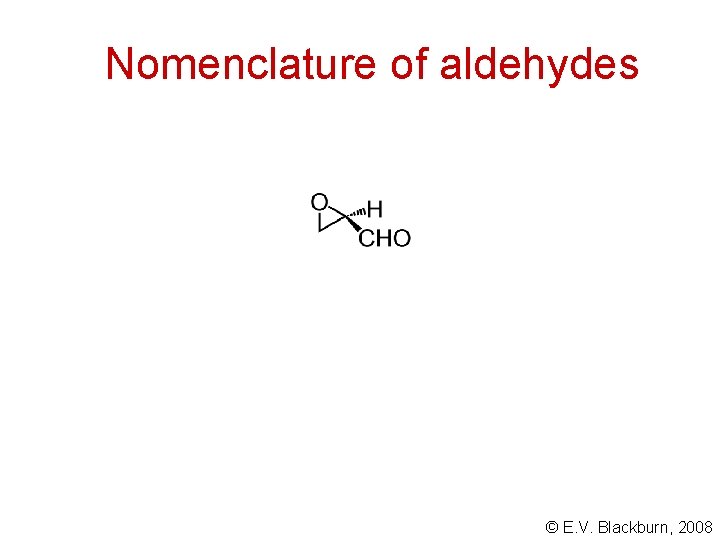
Nomenclature of aldehydes © E. V. Blackburn, 2008

Nomenclature of ketones Ketones are named using the name of the corresponding hydrocarbon followed by the suffix -one. The position of the carbonyl carbon must be indicated. propanone acetone dimethyl ketone One can also name the two radicals, R and R’, followed by the word ketone. © E. V. Blackburn, 2008
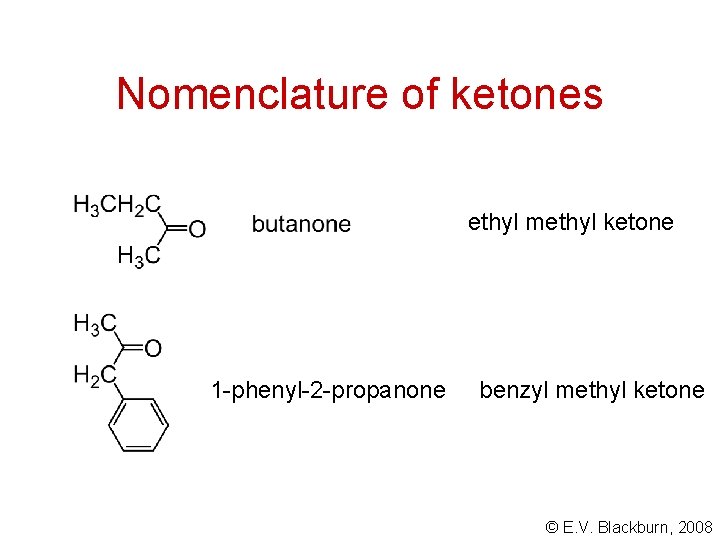
Nomenclature of ketones ethyl methyl ketone 1 -phenyl-2 -propanone benzyl methyl ketone © E. V. Blackburn, 2008
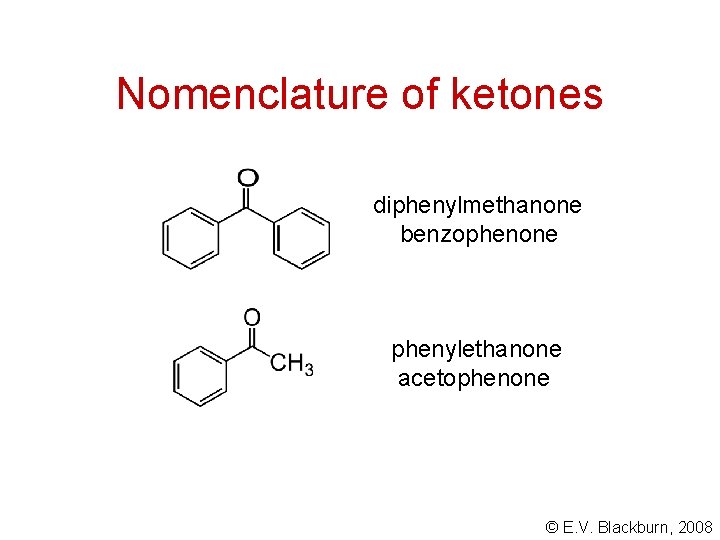
Nomenclature of ketones diphenylmethanone benzophenone phenylethanone acetophenone © E. V. Blackburn, 2008
Nomenclature of ketones © E. V. Blackburn, 2008
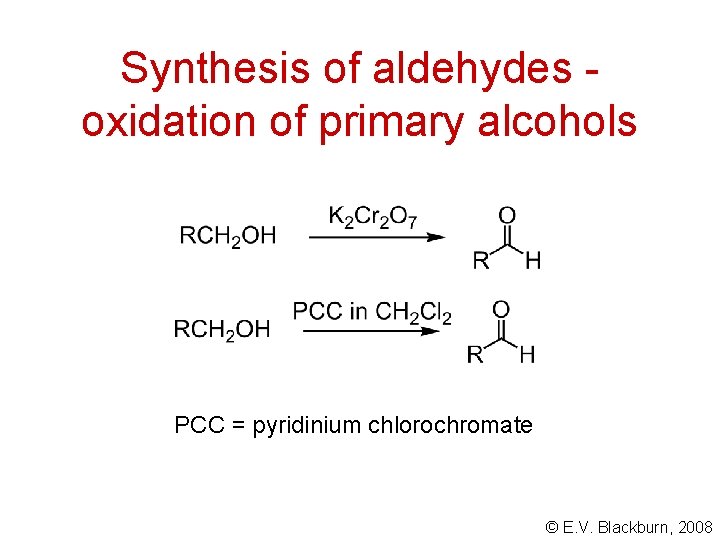
Synthesis of aldehydes oxidation of primary alcohols PCC = pyridinium chlorochromate © E. V. Blackburn, 2008

Rosemund reduction RCHO or Ar. CHO A special catalyst is used: palladium on barium sulfate which has been deactivated (poisoned) with an amine such as quinoline. © E. V. Blackburn, 2008
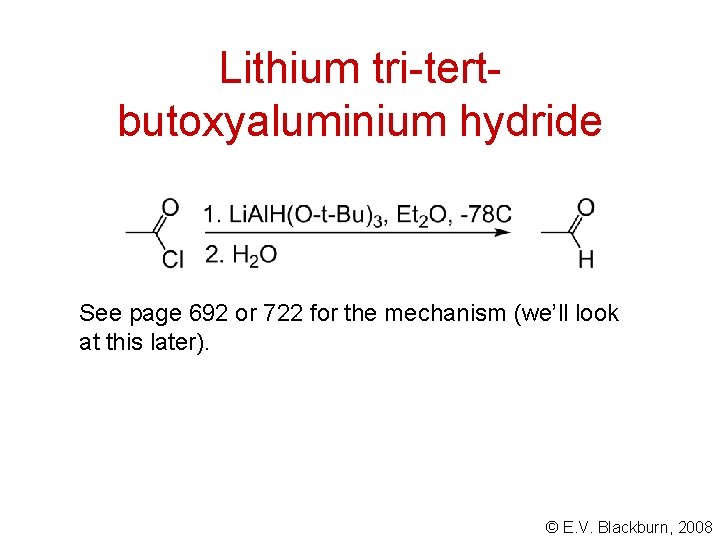
Lithium tri-tertbutoxyaluminium hydride See page 692 or 722 for the mechanism (we’ll look at this later). © E. V. Blackburn, 2008
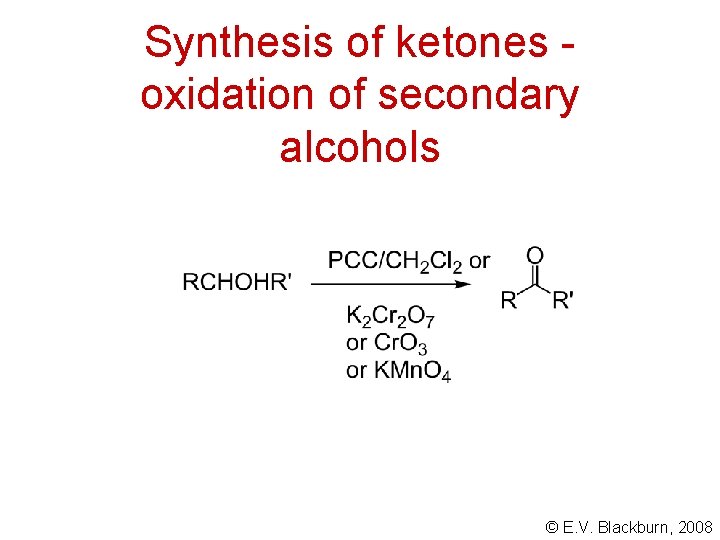
Synthesis of ketones oxidation of secondary alcohols © E. V. Blackburn, 2008
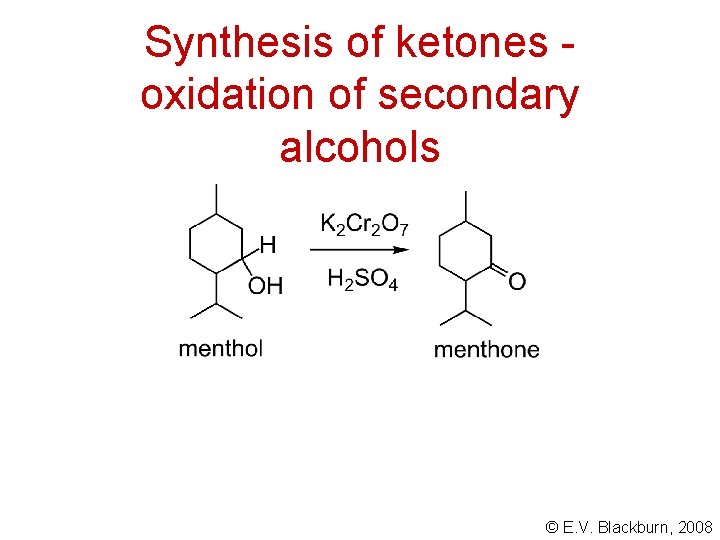
Synthesis of ketones oxidation of secondary alcohols © E. V. Blackburn, 2008
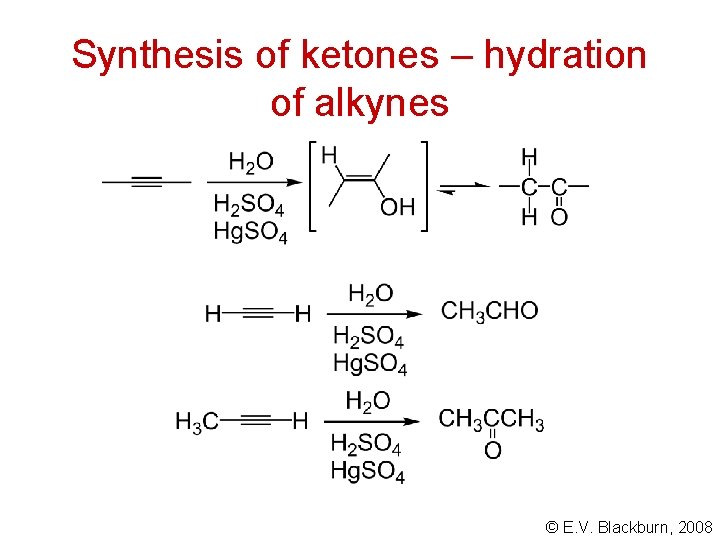
Synthesis of ketones – hydration of alkynes © E. V. Blackburn, 2008
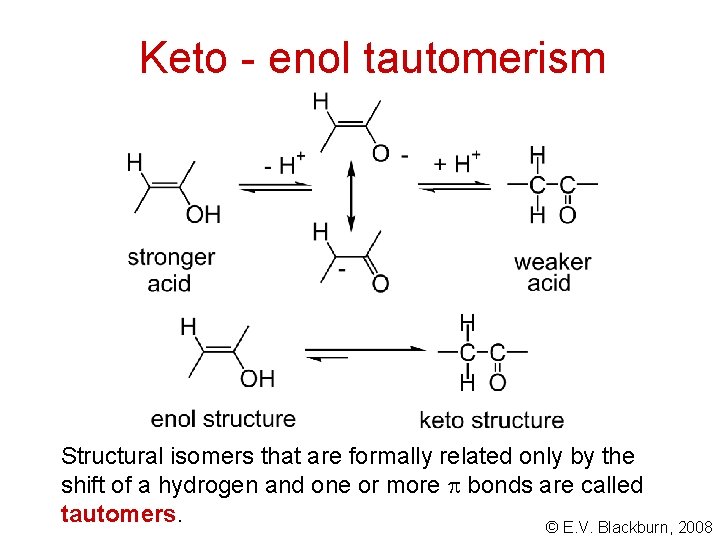
Keto - enol tautomerism Structural isomers that are formally related only by the shift of a hydrogen and one or more bonds are called tautomers. © E. V. Blackburn, 2008
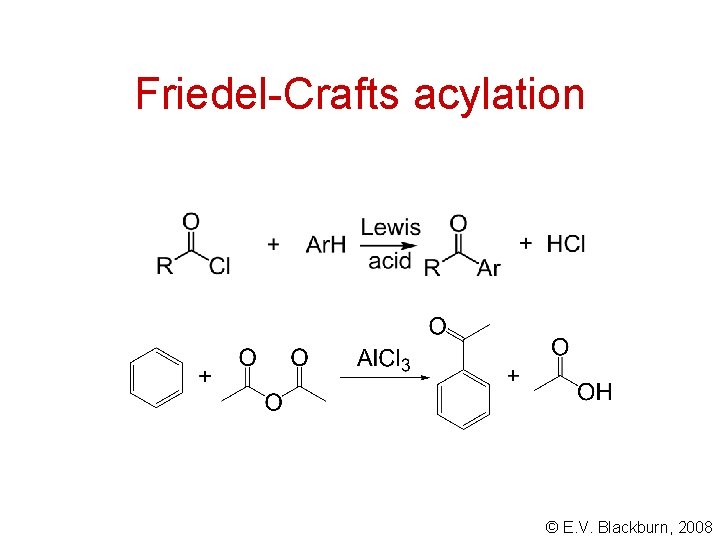
Friedel-Crafts acylation © E. V. Blackburn, 2008
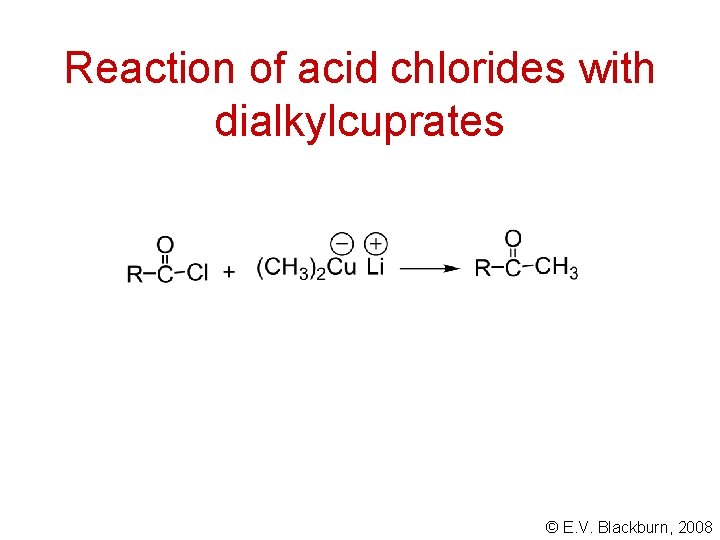
Reaction of acid chlorides with dialkylcuprates © E. V. Blackburn, 2008
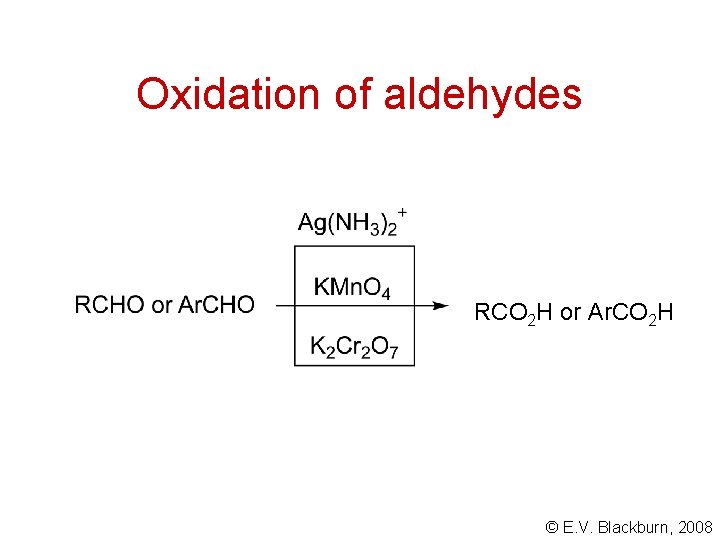
Oxidation of aldehydes RCO 2 H or Ar. CO 2 H © E. V. Blackburn, 2008
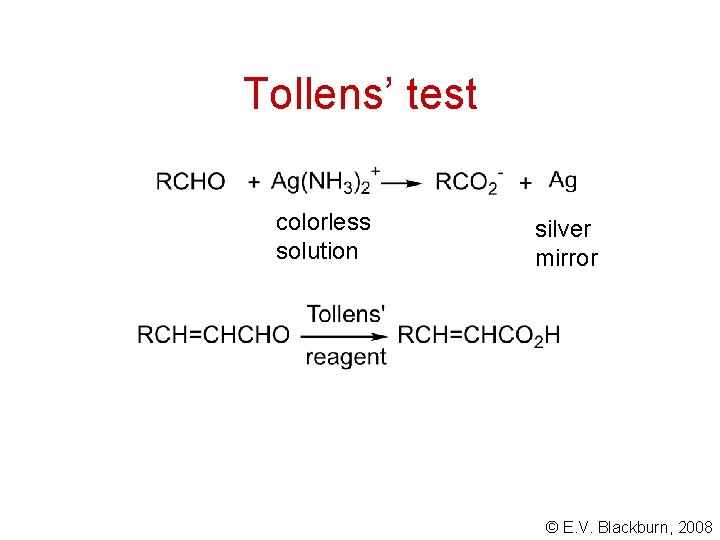
Tollens’ test colorless solution silver mirror © E. V. Blackburn, 2008
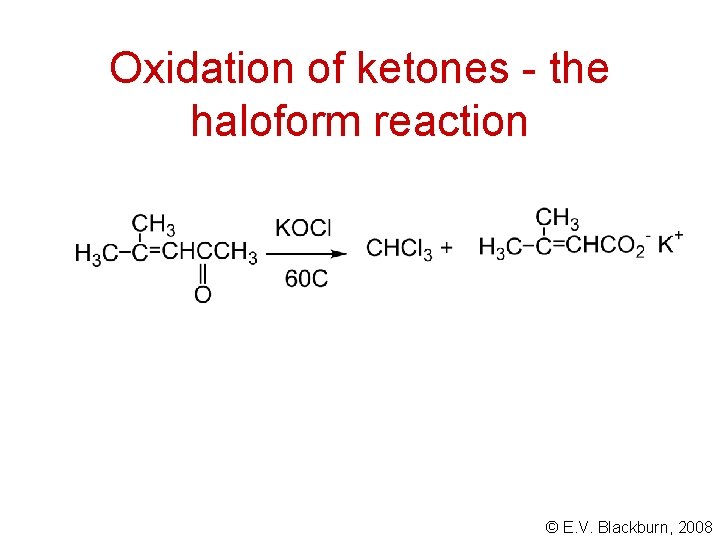
Oxidation of ketones - the haloform reaction © E. V. Blackburn, 2008
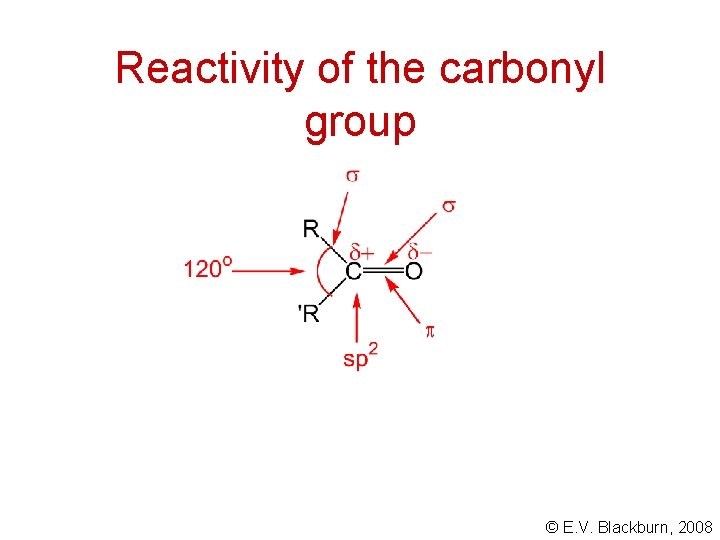
Reactivity of the carbonyl group © E. V. Blackburn, 2008
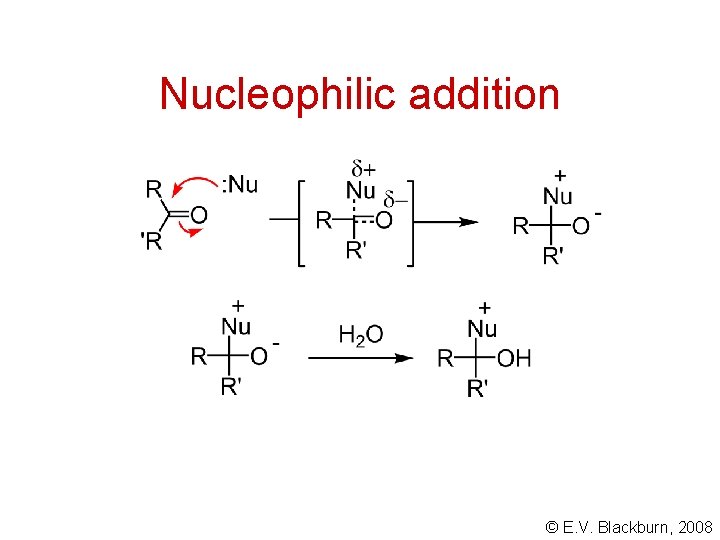
Nucleophilic addition © E. V. Blackburn, 2008
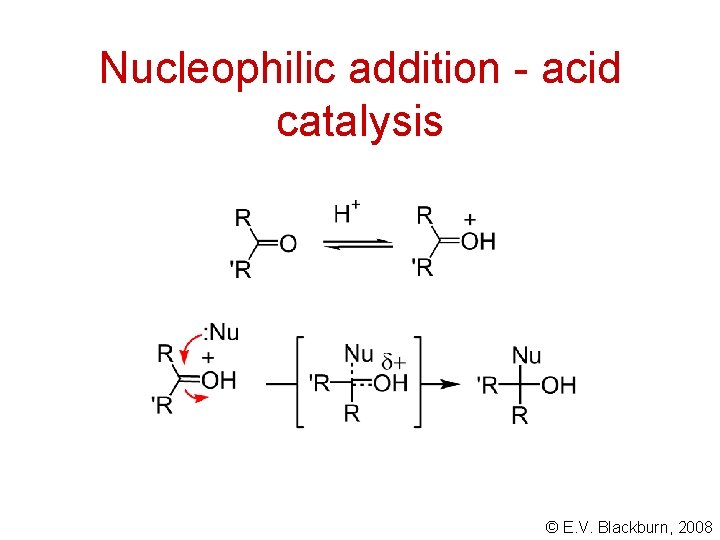
Nucleophilic addition - acid catalysis © E. V. Blackburn, 2008
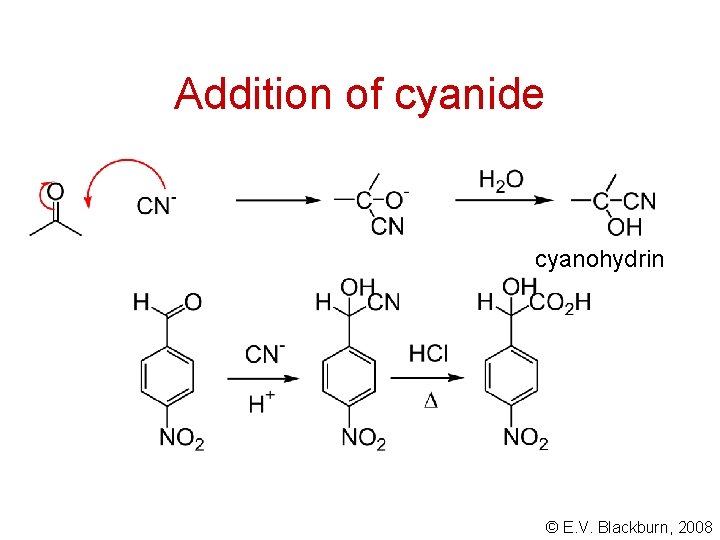
Addition of cyanide cyanohydrin © E. V. Blackburn, 2008
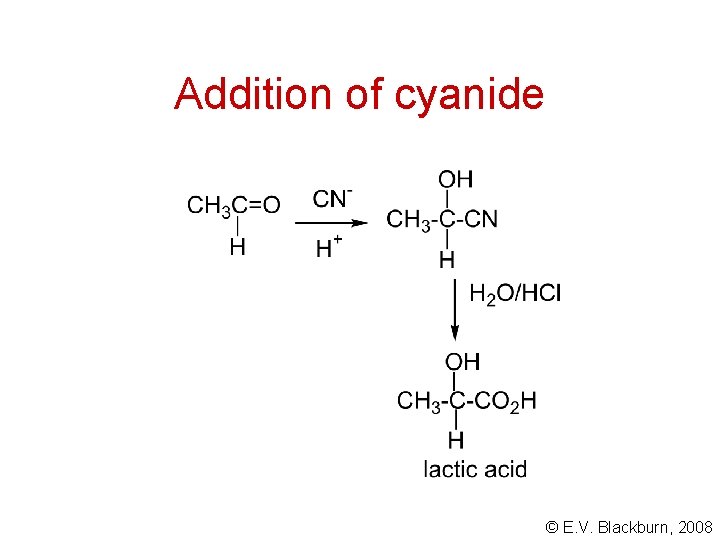
Addition of cyanide © E. V. Blackburn, 2008

Addition of Grignard reagents © E. V. Blackburn, 2008
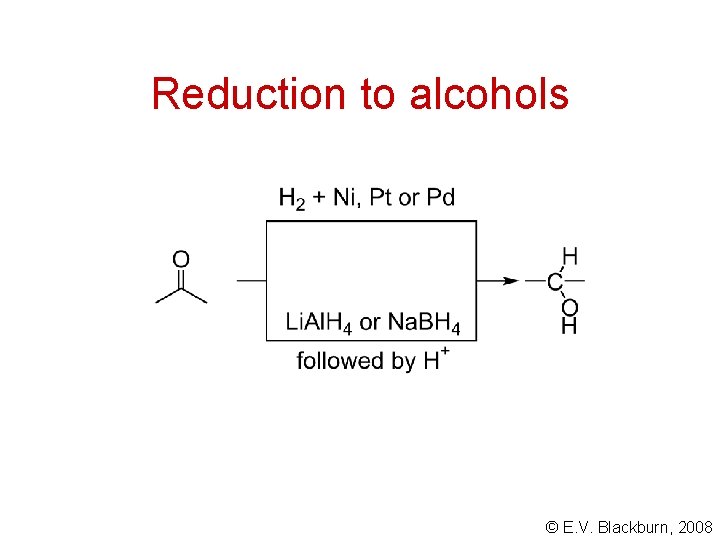
Reduction to alcohols © E. V. Blackburn, 2008
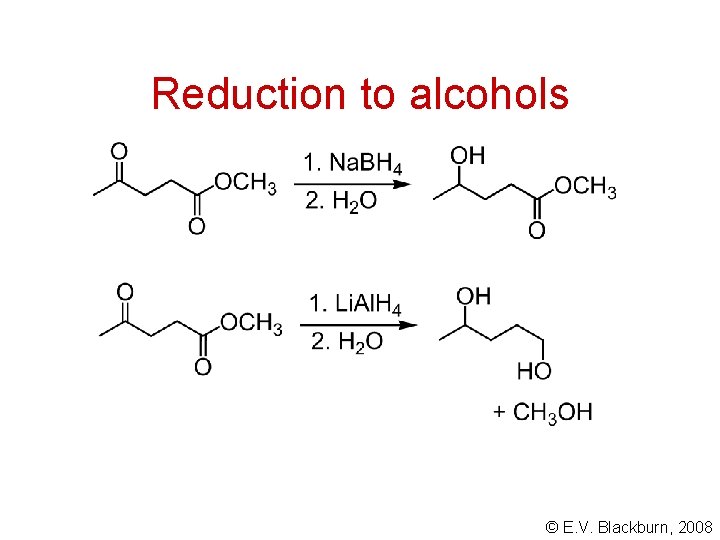
Reduction to alcohols © E. V. Blackburn, 2008
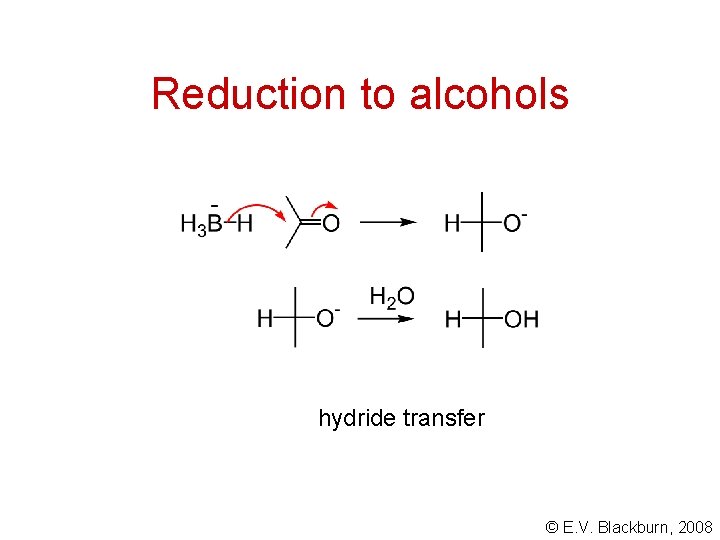
Reduction to alcohols hydride transfer © E. V. Blackburn, 2008
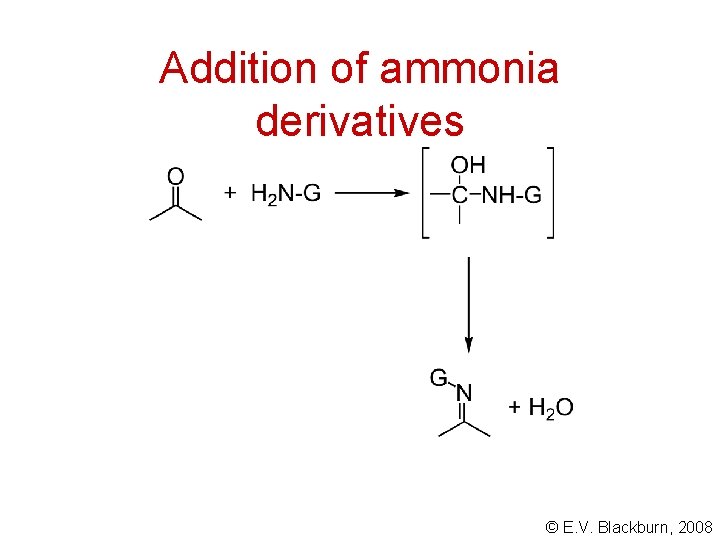
Addition of ammonia derivatives © E. V. Blackburn, 2008
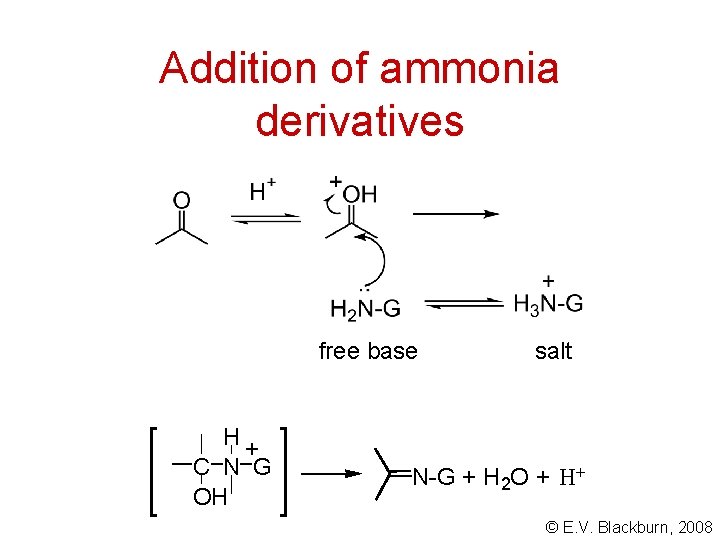
Addition of ammonia derivatives free base H+ C N G OH salt N-G + H 2 O + H+ © E. V. Blackburn, 2008
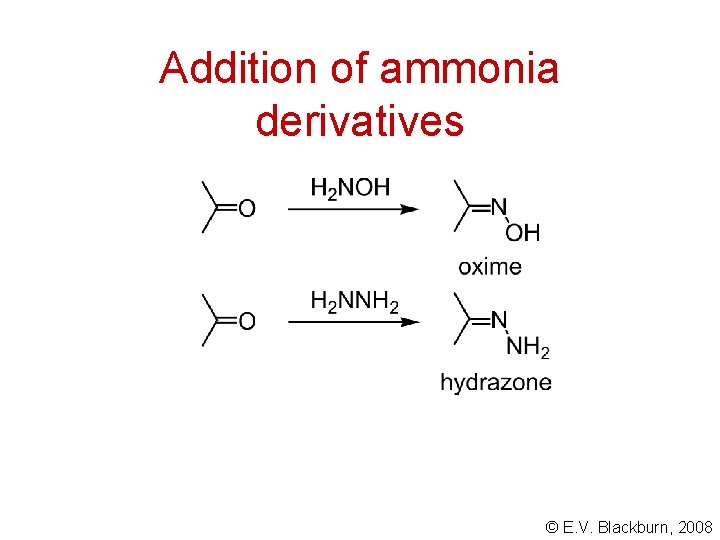
Addition of ammonia derivatives © E. V. Blackburn, 2008
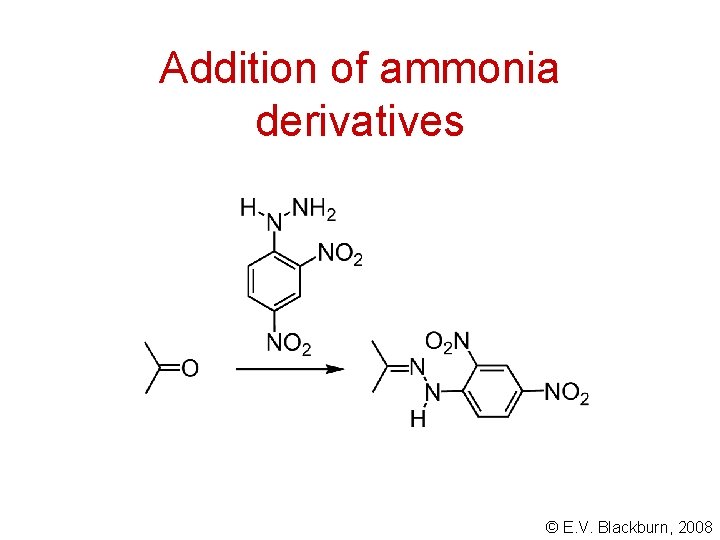
Addition of ammonia derivatives © E. V. Blackburn, 2008
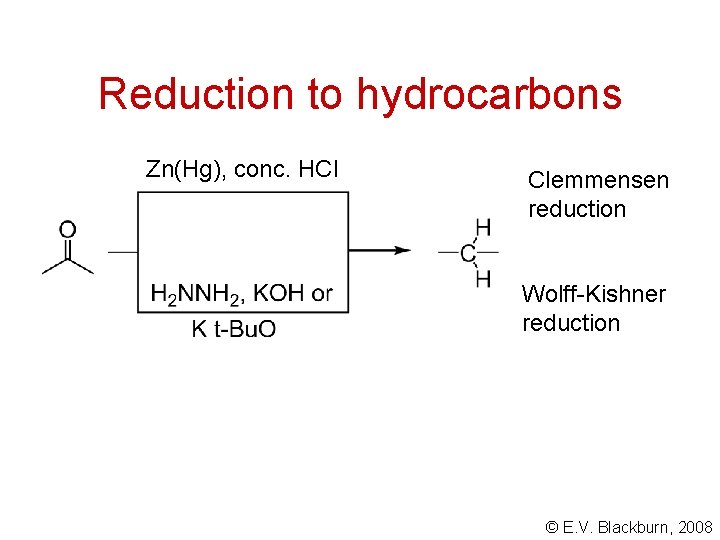
Reduction to hydrocarbons Zn(Hg), conc. HCl Clemmensen reduction Wolff-Kishner reduction © E. V. Blackburn, 2008
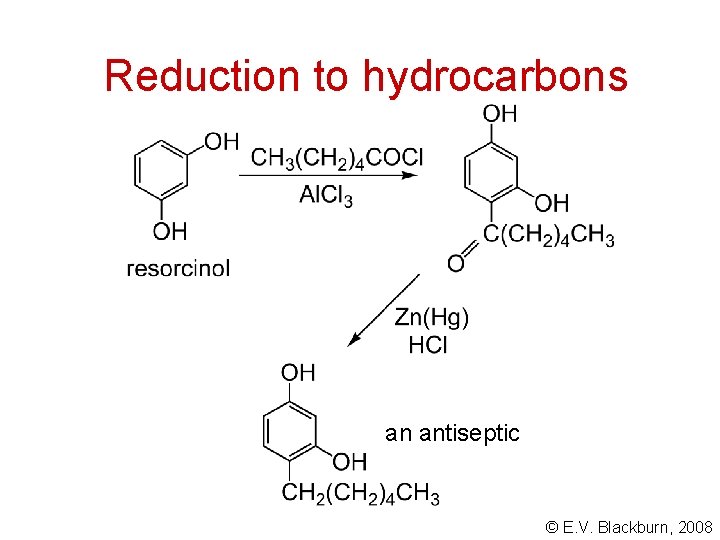
Reduction to hydrocarbons an antiseptic © E. V. Blackburn, 2008
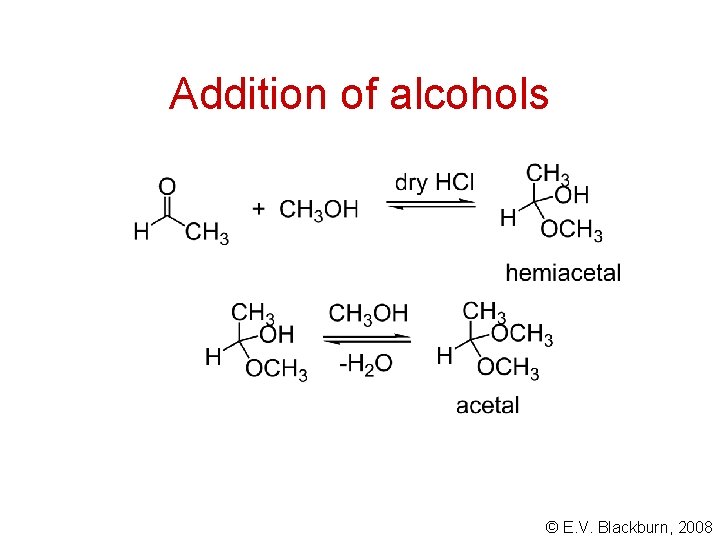
Addition of alcohols © E. V. Blackburn, 2008
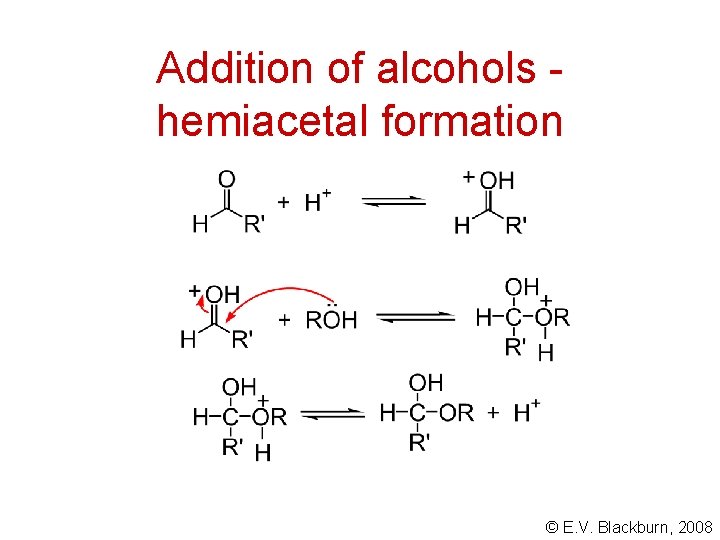
Addition of alcohols hemiacetal formation © E. V. Blackburn, 2008
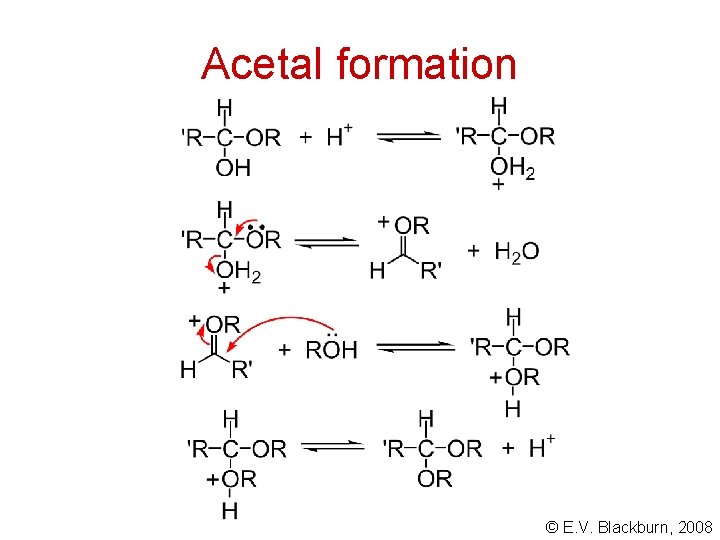
Acetal formation © E. V. Blackburn, 2008

Reactions of acetals H 'R C OR + H 2 O OR H + 2 ROH + R'-C=O H © E. V. Blackburn, 2008
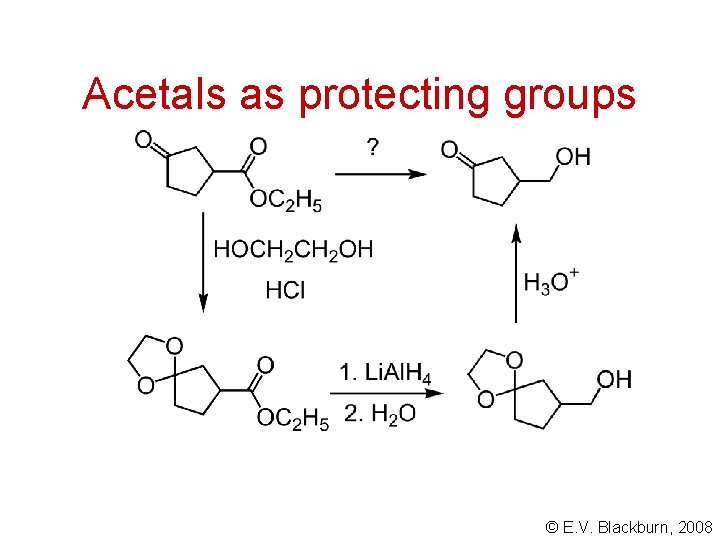
Acetals as protecting groups © E. V. Blackburn, 2008
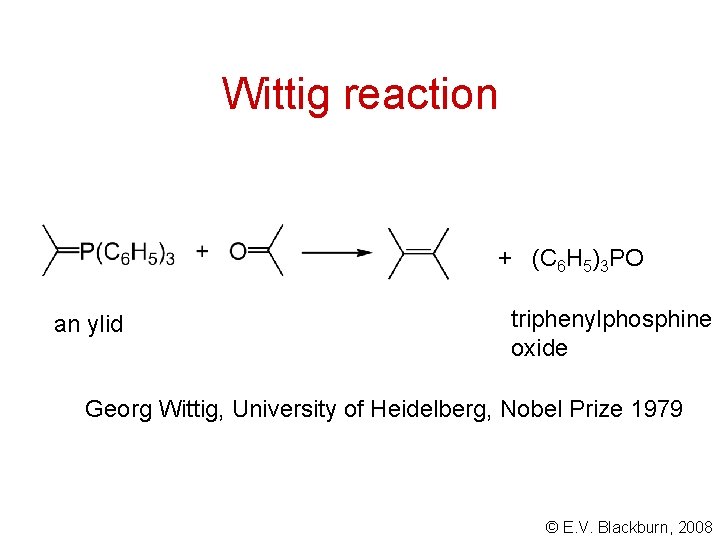
Wittig reaction + (C 6 H 5)3 PO an ylid triphenylphosphine oxide Georg Wittig, University of Heidelberg, Nobel Prize 1979 © E. V. Blackburn, 2008

Wittig reaction - ylid formation triphenylphosphine alkyltriphenylphosphonium halide an ylid © E. V. Blackburn, 2008
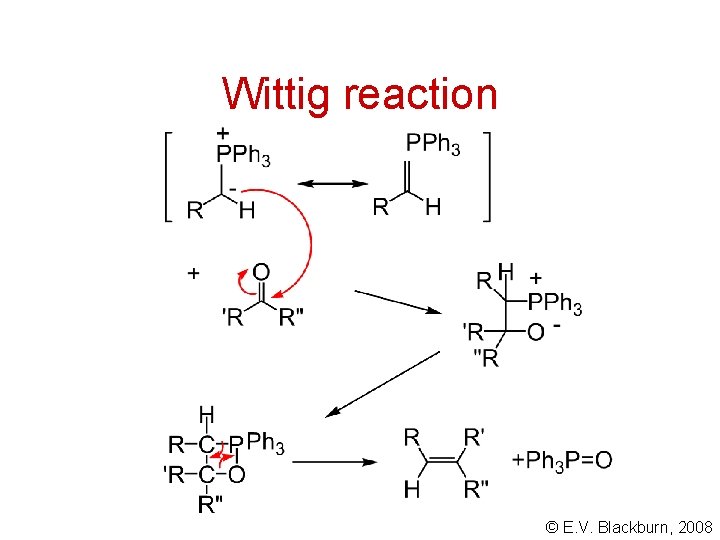
Wittig reaction © E. V. Blackburn, 2008
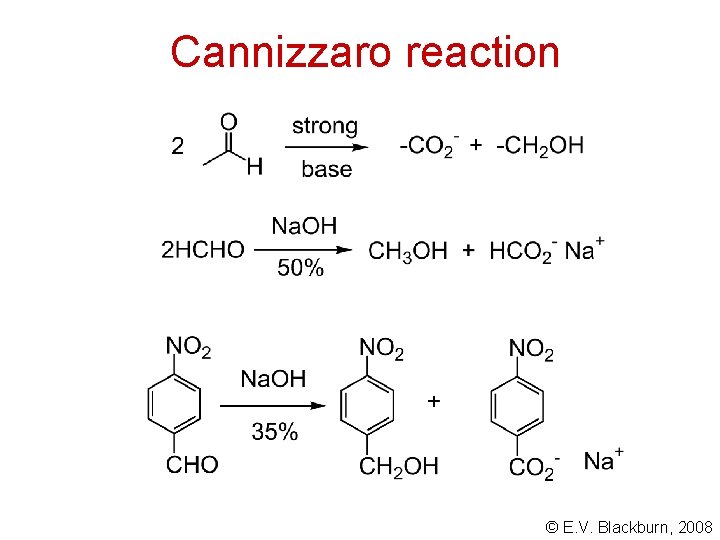
Cannizzaro reaction © E. V. Blackburn, 2008
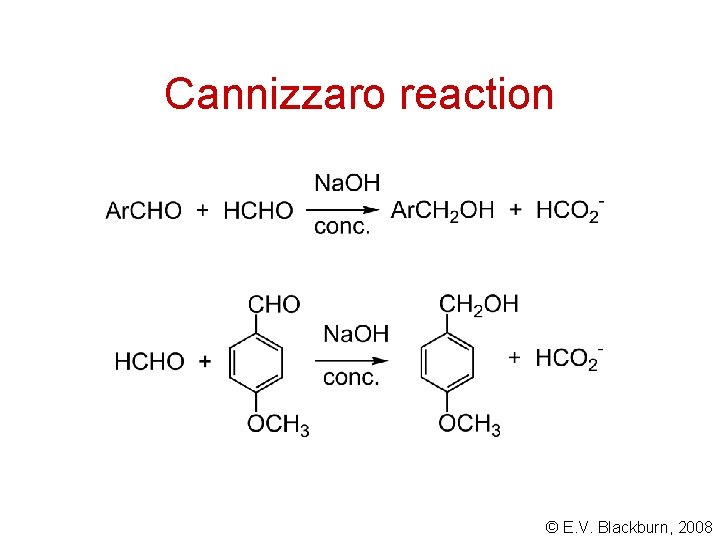
Cannizzaro reaction © E. V. Blackburn, 2008
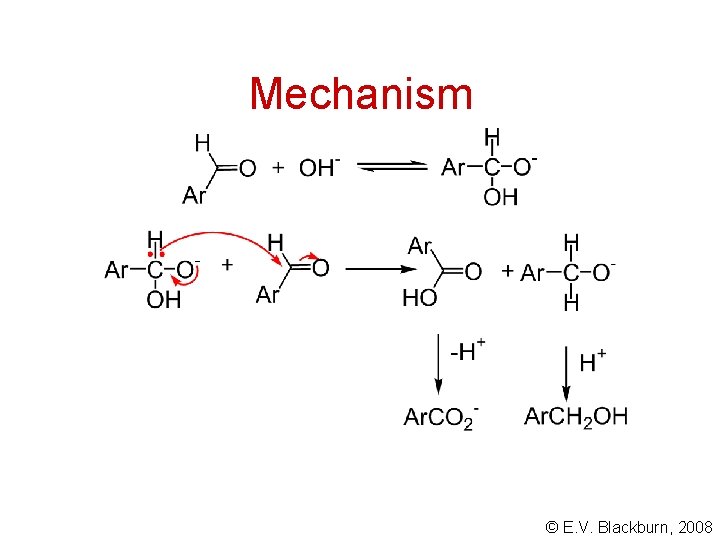
Mechanism © E. V. Blackburn, 2008

Analysis of aldehydes and ketones • They react with 2, 4 -dinitrophenylhydrazine. • Aldehydes give a positive Tollens’ test. • Methyl ketones give a positive iodoform test. © E. V. Blackburn, 2008

Spectroscopic properties IR 1705 - 1740 cm-1 - C=O stretching ~2720 cm-1 - C-H stretching in aldehydes NMR chemical shift of aldehyde proton: = 9 - 10 © E. V. Blackburn, 2008

Problems Try problems 16. 23, 16. 24, 16. 26, 16. 28, 16. 31, 16. 35, 16. 37, and 16. 42. © E. V. Blackburn, 2008
- Slides: 52