ALCOHOLS WORKSHEET 1 Draw name and classify as
- Slides: 7

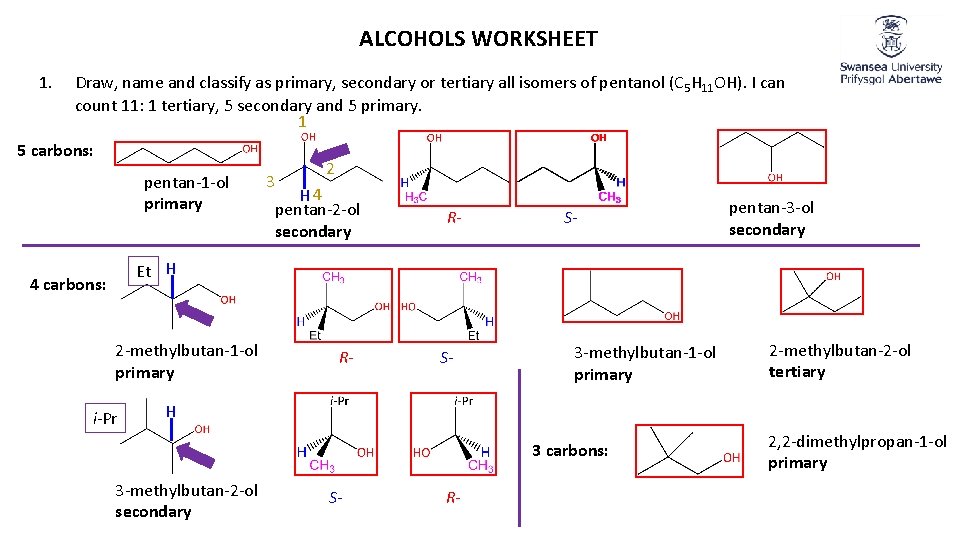
ALCOHOLS WORKSHEET 1. Draw, name and classify as primary, secondary or tertiary all isomers of pentanol (C 5 H 11 OH). I can count 11: 1 tertiary, 5 secondary and 5 primary. 1 5 carbons: pentan-1 -ol primary 3 2 H 4 pentan-2 -ol secondary R- S- pentan-3 -ol secondary Et H 4 carbons: 2 -methylbutan-1 -ol primary i-Pr R- S- 3 -methylbutan-1 -ol primary 2 -methylbutan-2 -ol tertiary H 3 carbons: 3 -methylbutan-2 -ol secondary S- R- 2, 2 -dimethylpropan-1 -ol primary

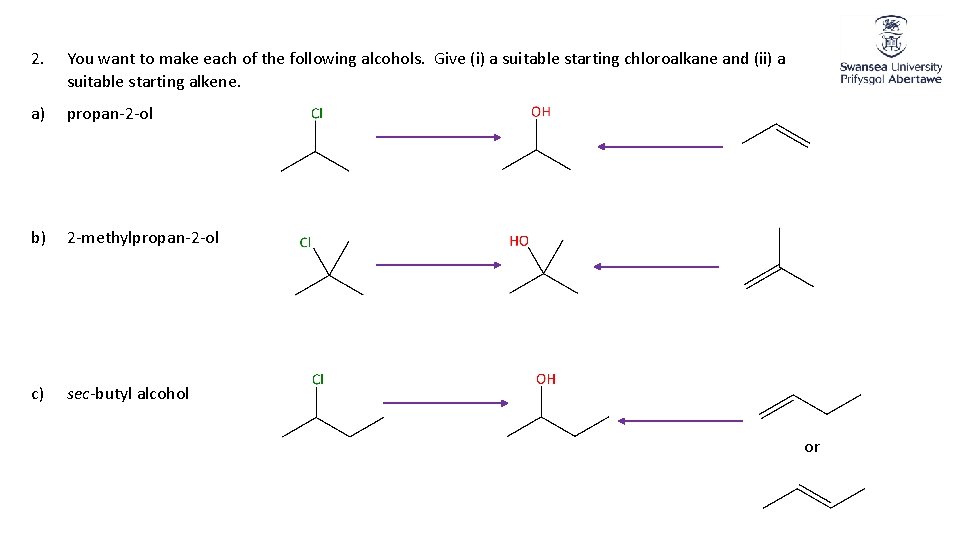
2. You want to make each of the following alcohols. Give (i) a suitable starting chloroalkane and (ii) a suitable starting alkene. a) propan-2 -ol b) 2 -methylpropan-2 -ol c) sec-butyl alcohol or

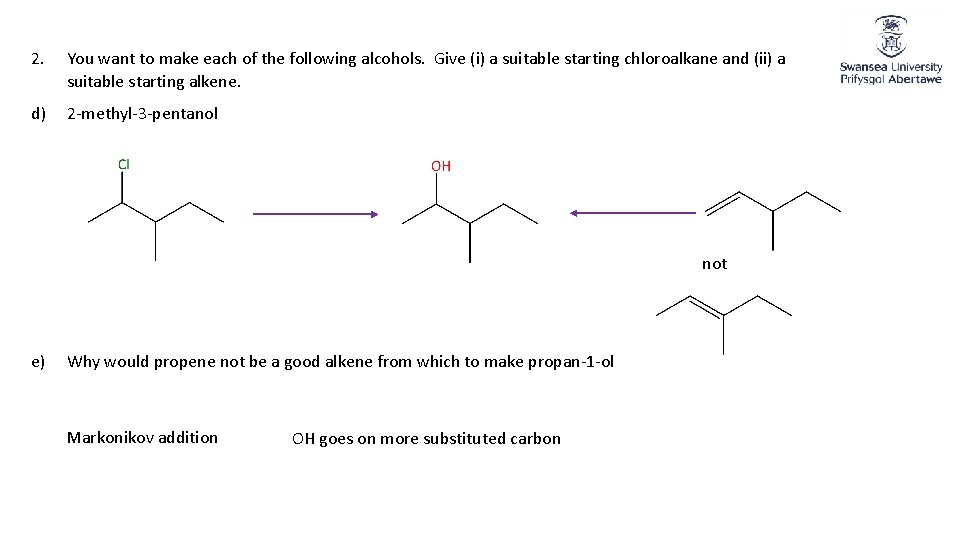
2. You want to make each of the following alcohols. Give (i) a suitable starting chloroalkane and (ii) a suitable starting alkene. d) 2 -methyl-3 -pentanol not e) Why would propene not be a good alkene from which to make propan-1 -ol Markonikov addition OH goes on more substituted carbon

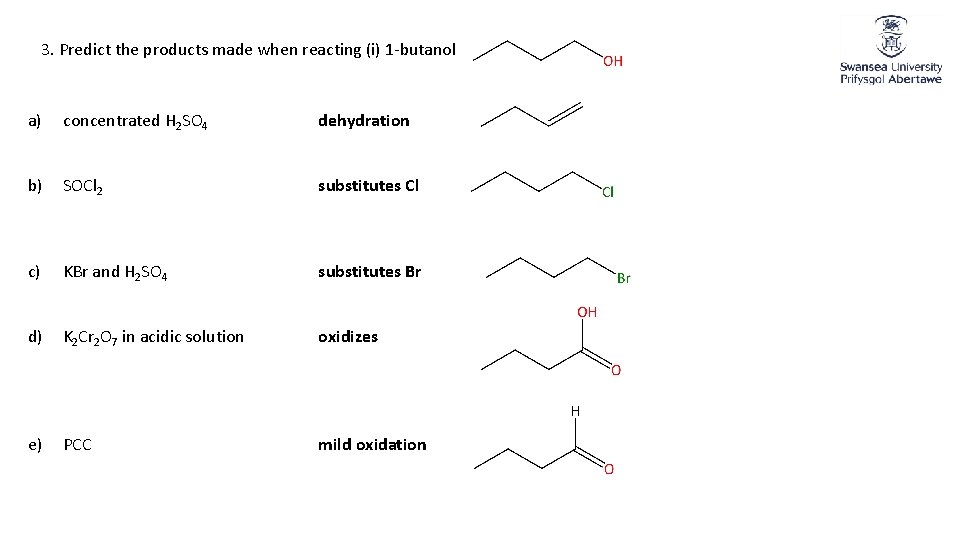
3. Predict the products made when reacting (i) 1 -butanol a) concentrated H 2 SO 4 dehydration b) SOCl 2 substitutes Cl c) KBr and H 2 SO 4 substitutes Br d) K 2 Cr 2 O 7 in acidic solution oxidizes e) PCC mild oxidation

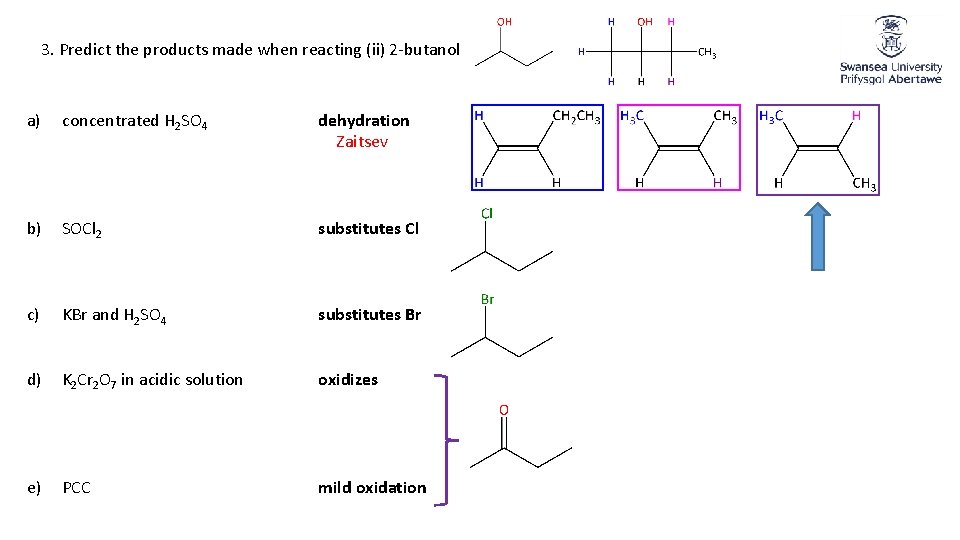
3. Predict the products made when reacting (ii) 2 -butanol a) concentrated H 2 SO 4 dehydration Zaitsev b) SOCl 2 substitutes Cl c) KBr and H 2 SO 4 substitutes Br d) K 2 Cr 2 O 7 in acidic solution oxidizes e) PCC mild oxidation

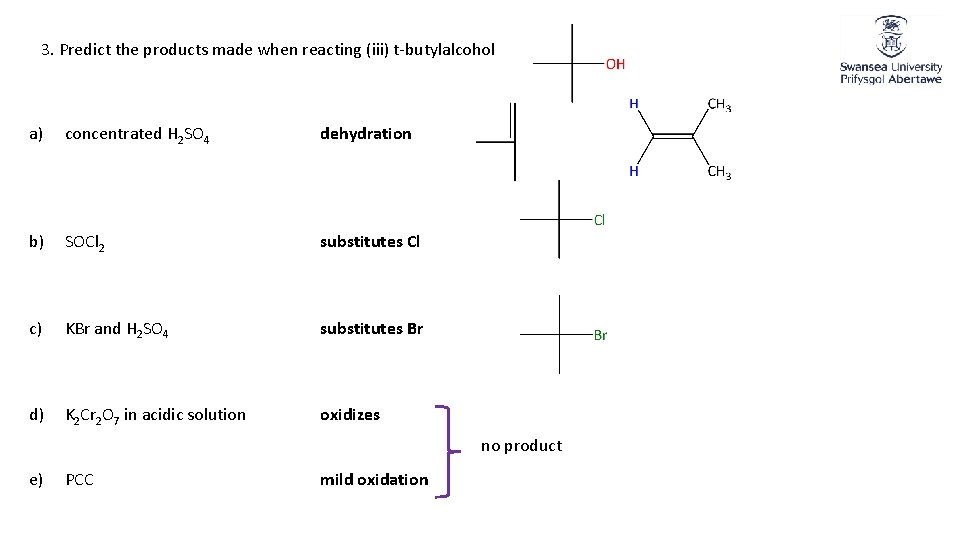
3. Predict the products made when reacting (iii) t-butylalcohol a) concentrated H 2 SO 4 dehydration b) SOCl 2 substitutes Cl c) KBr and H 2 SO 4 substitutes Br d) K 2 Cr 2 O 7 in acidic solution oxidizes no product e) PCC mild oxidation

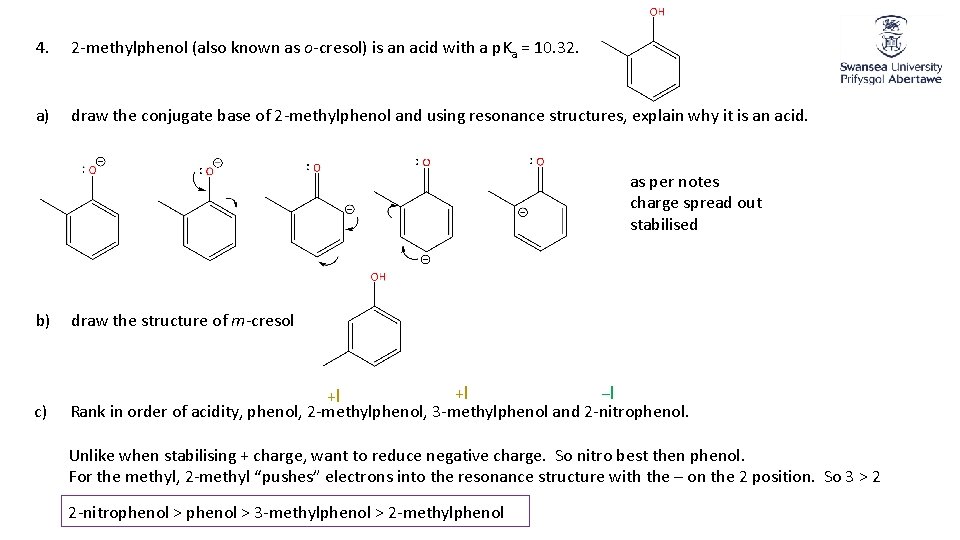
4. 2 -methylphenol (also known as o-cresol) is an acid with a p. Ka = 10. 32. a) draw the conjugate base of 2 -methylphenol and using resonance structures, explain why it is an acid. as per notes charge spread out stabilised b) draw the structure of m-cresol c) +I −I +I Rank in order of acidity, phenol, 2 -methylphenol, 3 -methylphenol and 2 -nitrophenol. Unlike when stabilising + charge, want to reduce negative charge. So nitro best then phenol. For the methyl, 2 -methyl “pushes” electrons into the resonance structure with the – on the 2 position. So 3 > 2 2 -nitrophenol > 3 -methylphenol > 2 -methylphenol