alcohols ROH Methanol Ehanol Aliphatic Glycerol Benzyl alcohol

alcohols R-OH Methanol Ehanol Aliphatic Glycerol Benzyl alcohol Aromatic
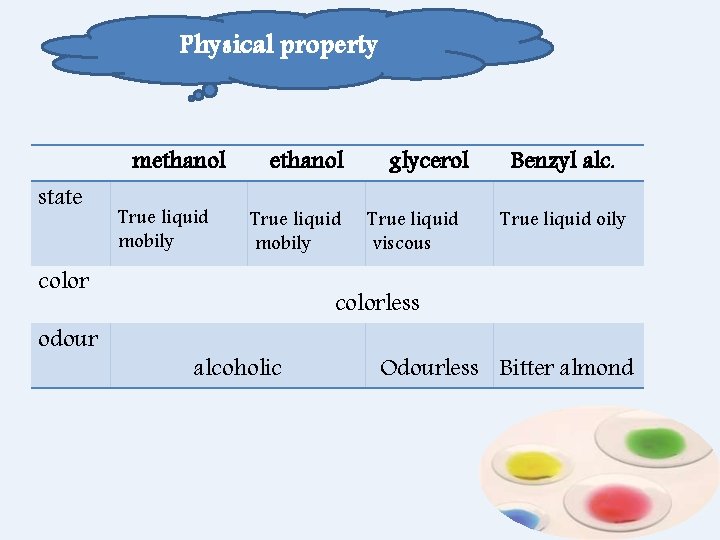
Physical property state methanol True liquid mobily color odour glycerol True liquid viscous Benzyl alc. True liquid oily colorless alcoholic Odourless Bitter almond
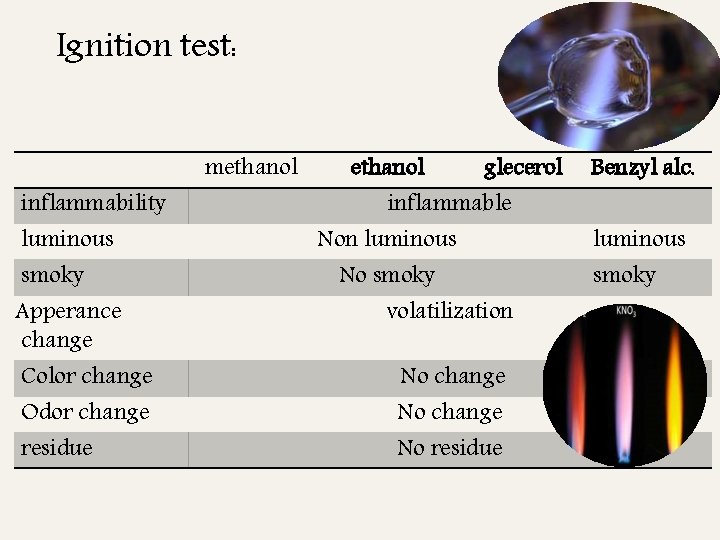
Ignition test: inflammability luminous smoky Apperance change Color change Odor change residue methanol glecerol inflammable Non luminous No smoky volatilization No change No residue Benzyl alc. luminous smoky
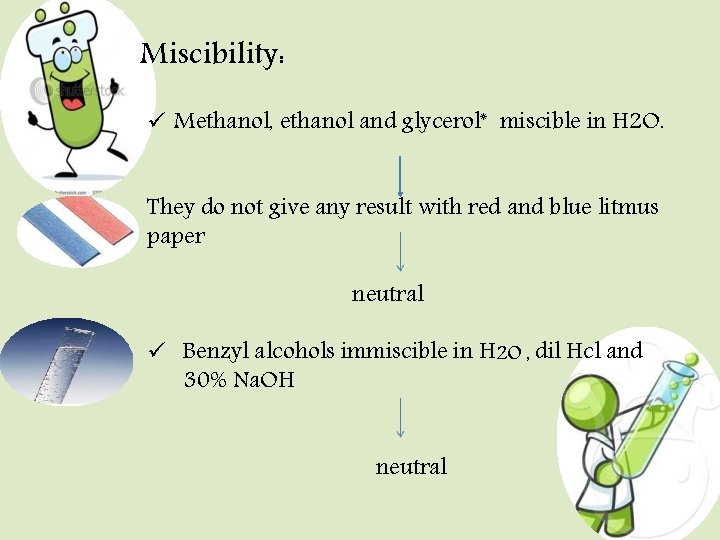
Miscibility: ü Methanol, ethanol and glycerol* miscible in H 2 O. They do not give any result with red and blue litmus paper neutral ü Benzyl alcohols immiscible in H 2 O , dil Hcl and 30% Na. OH neutral

Preliminary tests: ü soda lime : not done (why)? . ü 30%Na. OH : no reaction. ü Fecl 3 : no reaction. ü Conc. H 2 SO 4* : no reaction with all except benzyl alc. Benzyl alc+ 4 dps Conc. H 2 SO 4 (on wall) gelatinous ppt. dirty white
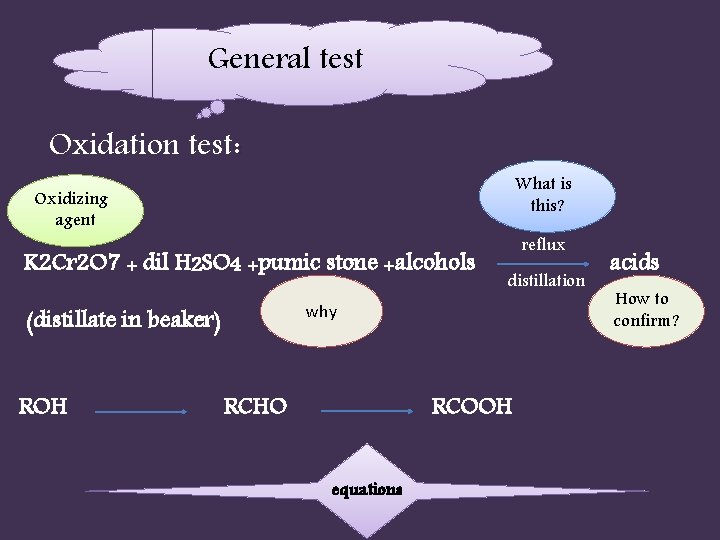
General test Oxidation test: What is this? Oxidizing agent K 2 Cr 2 O 7 + dil H 2 SO 4 +pumic stone +alcohols distillation why (distillate in beaker) ROH reflux RCHO RCOOH equations acids How to confirm?

Specific tests mehanol ethanol Iodoform test: meth. Or eth. +10% Na. OH+ xss I 2 No yellow ppt. Yellow ppt Wait and warm Esterfication test: (meth. + salic. Or eth. + acetic. ) +Conc. H 2 SO 4→pour it after heat into beaker contain water Oil of winter green (vicks odor) with white ppt. (methyl salicylate) Bad apple odor (ribe apple) ethyl acetate) Heat until fussion

Borax test of glycerol Pink color Borax powder dissolve in water +phenol phthaline (ph. ph) +glycerol→ disappear pink color Conc. H 2 SO 4 of benzyl alcohol Dirty white gelatinous ppt.
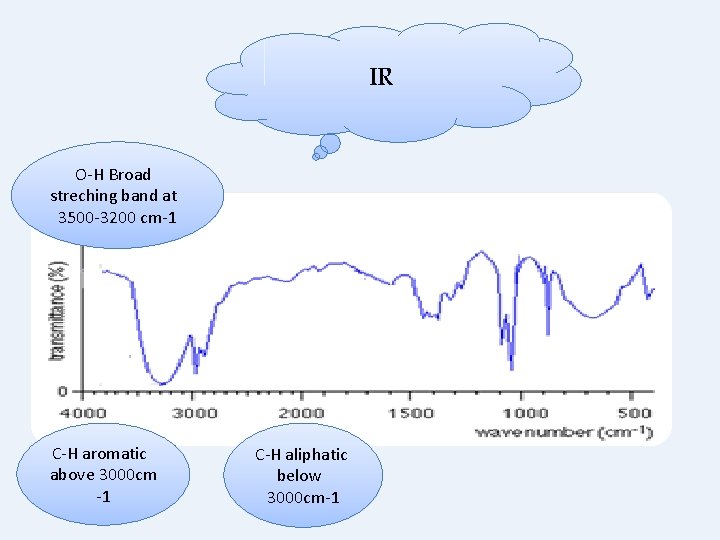
IR O-H Broad streching band at 3500 -3200 cm-1 C-H aromatic above 3000 cm -1 C-H aliphatic below 3000 cm-1
- Slides: 11