Alcohols phenols and ethers E V Blackburn 2008

Alcohols, phenols, and ethers © E. V. Blackburn, 2008
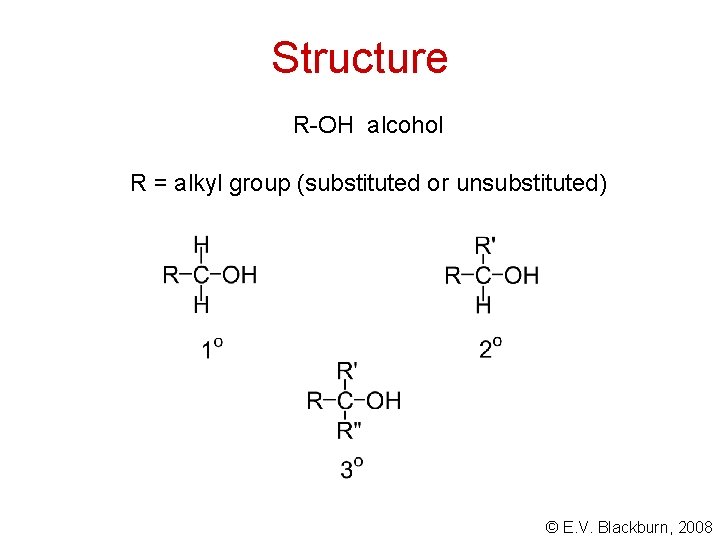
Structure R-OH alcohol R = alkyl group (substituted or unsubstituted) © E. V. Blackburn, 2008
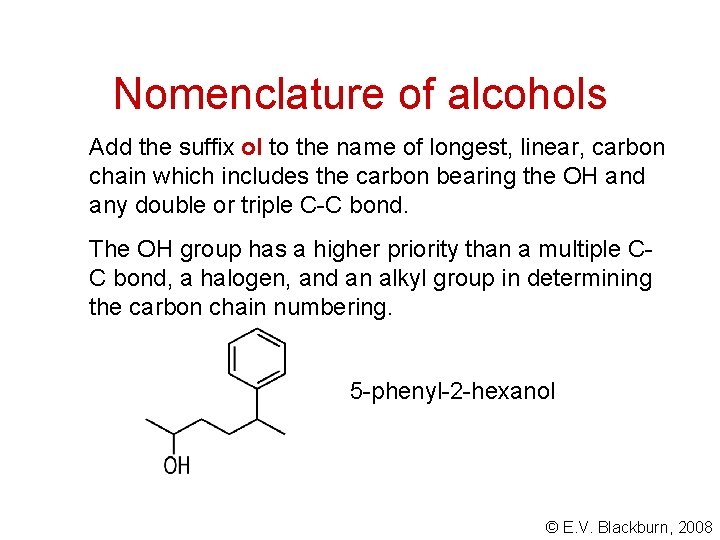
Nomenclature of alcohols Add the suffix ol to the name of longest, linear, carbon chain which includes the carbon bearing the OH and any double or triple C-C bond. The OH group has a higher priority than a multiple CC bond, a halogen, and an alkyl group in determining the carbon chain numbering. 5 -phenyl-2 -hexanol © E. V. Blackburn, 2008
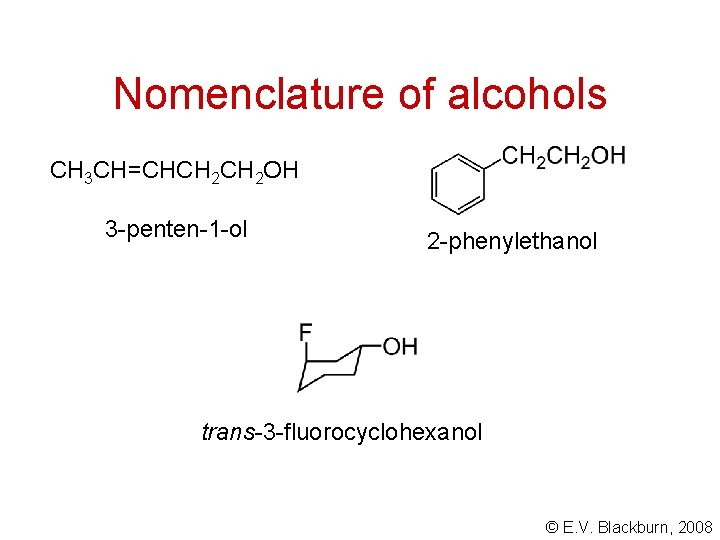
Nomenclature of alcohols CH 3 CH=CHCH 2 OH 3 -penten-1 -ol 2 -phenylethanol trans-3 -fluorocyclohexanol © E. V. Blackburn, 2008
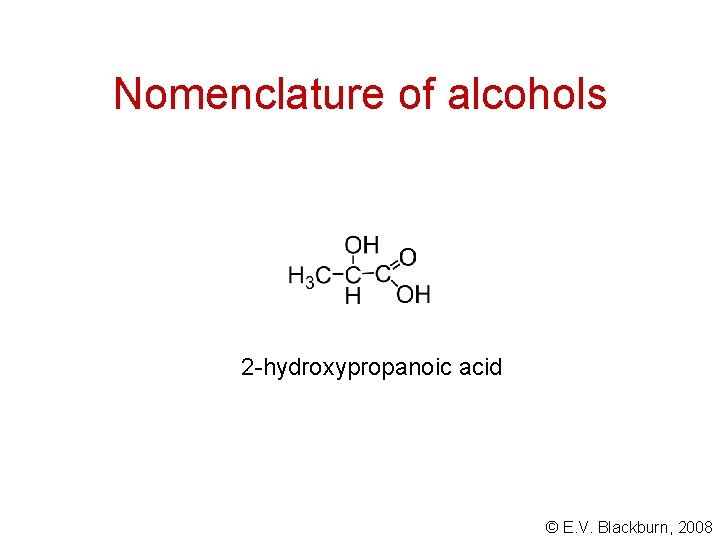
Nomenclature of alcohols 2 -hydroxypropanoic acid © E. V. Blackburn, 2008
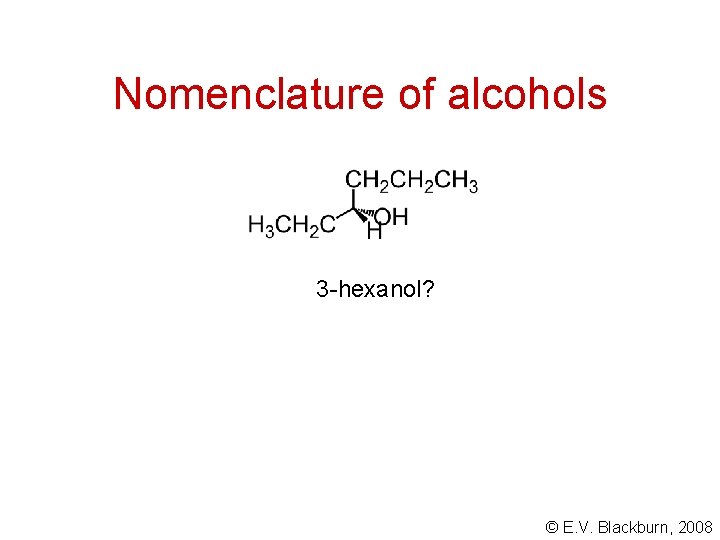
Nomenclature of alcohols 3 -hexanol? © E. V. Blackburn, 2008

Cahn - Prelog - Ingold rules Step 1: assign a priority to the 4 atoms or groups of atoms bonded to the stereogenic carbon: 1. If the 4 atoms are all different, priority is determined by atomic number. The atom of higher atomic number has the higher priority. © E. V. Blackburn, 2008

Determination of priority 2. If priority cannot be determined by (1), it is determined by a similar comparison of atoms working out from the stereogenic carbon. In the methyl group, the second atoms are H, H, H whereas in the ethyl group, they are C, H, H. The priority sequence is therefore Cl, C 2 H 5, CH 3, H. © E. V. Blackburn, 2008
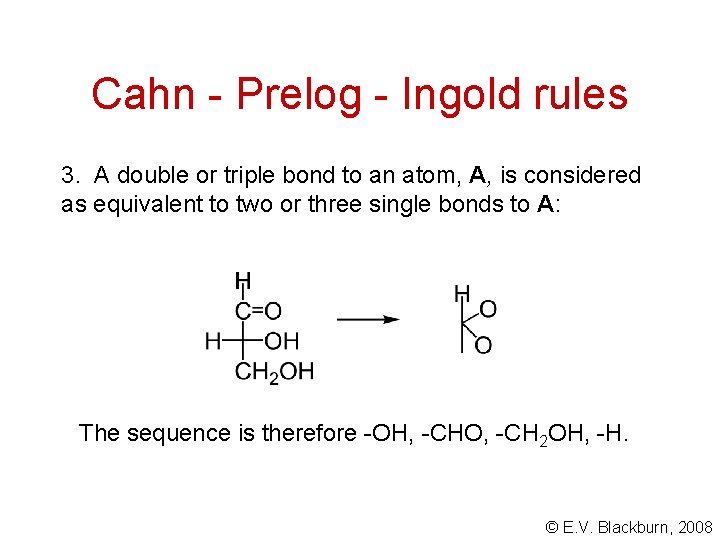
Cahn - Prelog - Ingold rules 3. A double or triple bond to an atom, A, is considered as equivalent to two or three single bonds to A: The sequence is therefore -OH, -CHO, -CH 2 OH, -H. © E. V. Blackburn, 2008
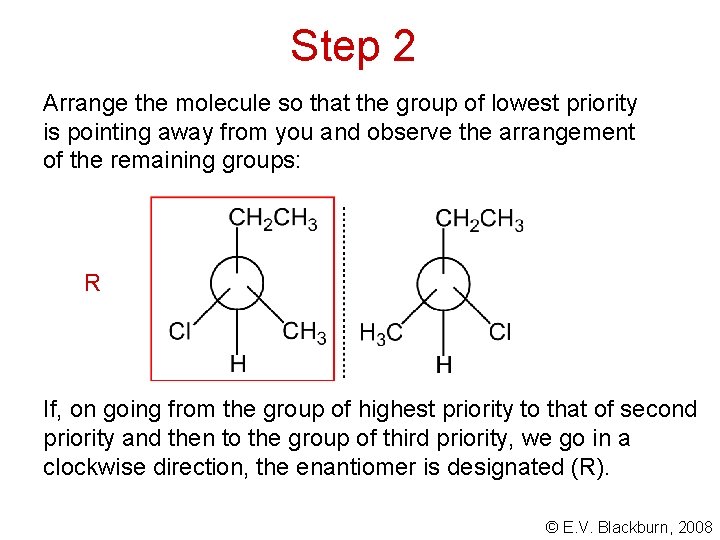
Step 2 Arrange the molecule so that the group of lowest priority is pointing away from you and observe the arrangement of the remaining groups: R If, on going from the group of highest priority to that of second priority and then to the group of third priority, we go in a clockwise direction, the enantiomer is designated (R). © E. V. Blackburn, 2008
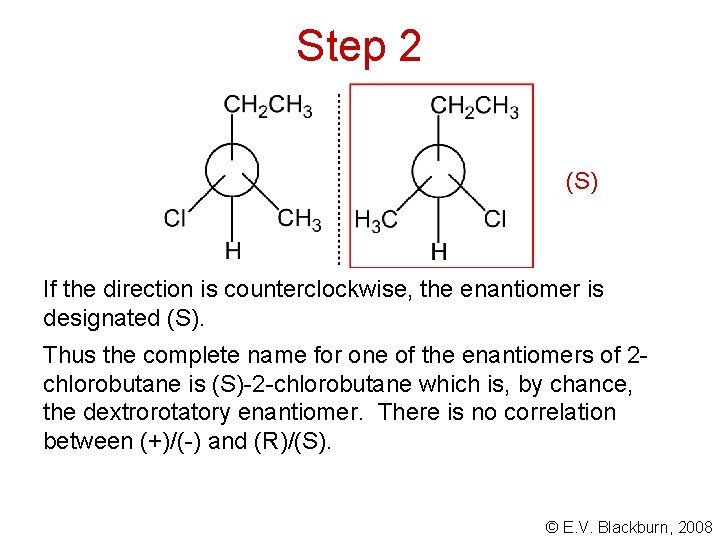
Step 2 (S) If the direction is counterclockwise, the enantiomer is designated (S). Thus the complete name for one of the enantiomers of 2 chlorobutane is (S)-2 -chlorobutane which is, by chance, the dextrorotatory enantiomer. There is no correlation between (+)/(-) and (R)/(S). © E. V. Blackburn, 2008
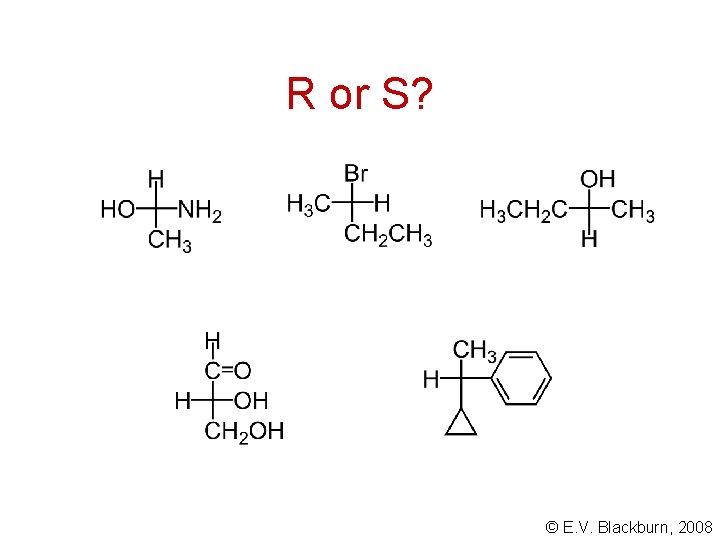
R or S? © E. V. Blackburn, 2008
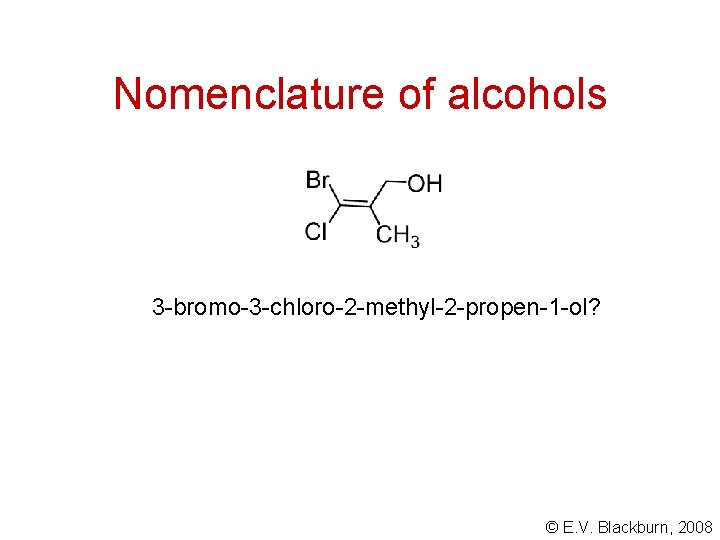
Nomenclature of alcohols 3 -bromo-3 -chloro-2 -methyl-2 -propen-1 -ol? © E. V. Blackburn, 2008

E-Z designations (Z)- 3 -bromo-3 -chloro-2 -methyl-2 -propen-1 -ol • use the Cahn-Ingold-Prelog system to assign priorities to the two groups on each carbon of the double bond. • then compare the relative positions of the groups of higher priority on these two carbons. • if the two groups are on the same side, the compound has the Z configuration (zusammen, German, together). • if the two groups are on opposite sides, the compound has the E configuration (entgegen, German, across). © E. V. Blackburn, 2008
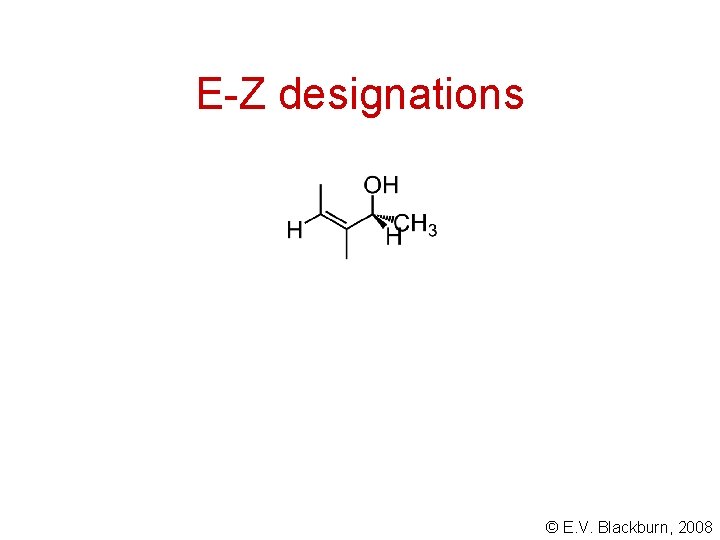
E-Z designations © E. V. Blackburn, 2008
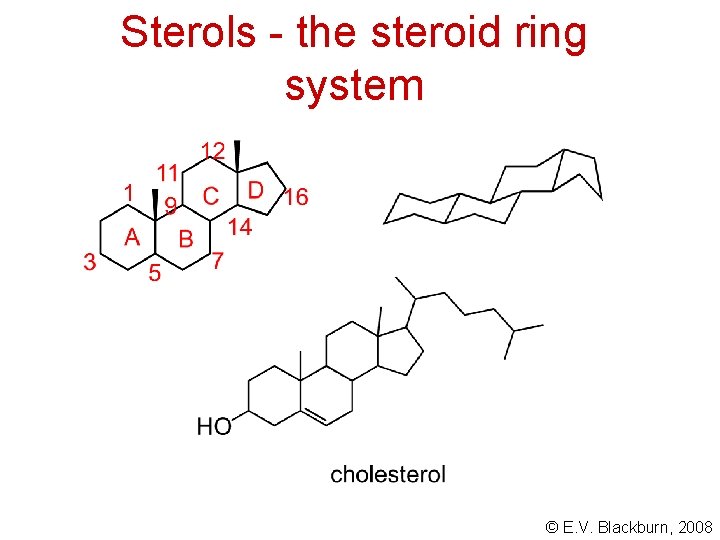
Sterols - the steroid ring system © E. V. Blackburn, 2008

Physical properties of alcohols Alcohols are noticeably less volatile; their melting points are greater and they are more water soluble than the corresponding hydrocarbons having similar molecular weights. These differences are due to the OH group which renders a certain polarity to the molecule. The result is an important intermolecular attraction: © E. V. Blackburn, 2008
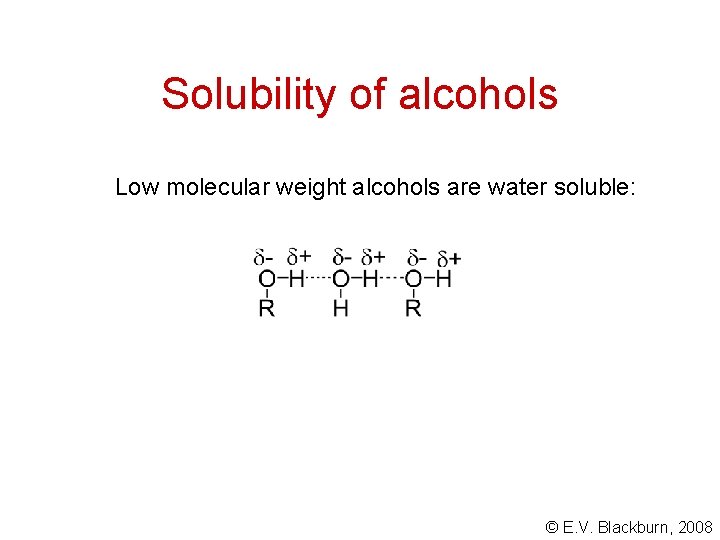
Solubility of alcohols Low molecular weight alcohols are water soluble: © E. V. Blackburn, 2008

Spectroscopic properties IR: Unassociated alcohols show a fairly sharp absorption near 3600 cm-1 due to O-H stretching. Associated alcohols (hydrogen bonded) show a broad absorption in the 3300 3400 cm-1 range. 1 H NMR: Absorption occurs in the range = 3. 5 to 4. 5. Coupling is not oberved due to rapid H - H exchange. © E. V. Blackburn, 2008

Fermentation of sugar by yeast gives C 2 H 5 OH. Methanol is added to denature it. © E. V. Blackburn, 2008

Azeotropic mixtures The bp of ethanol is 78. 3 C whereas that of water is 100 C (at least on Vancouver’s waterfront!). Can we separate a mixture by distillation? No! An azeotropic mixture forms! An azeotropic mixture is one whose liquid and vapor forms have identical compositions. The mixture cannot be separated by distillation. eg C 2 H 5 OH (95%) and H 2 O (5%) - bp 78. 13 C H 2 O (7. 5%), C 2 H 5 OH (18. 5%) and C 6 H 6 (74%) - bp 64. 9 C © E. V. Blackburn, 2008
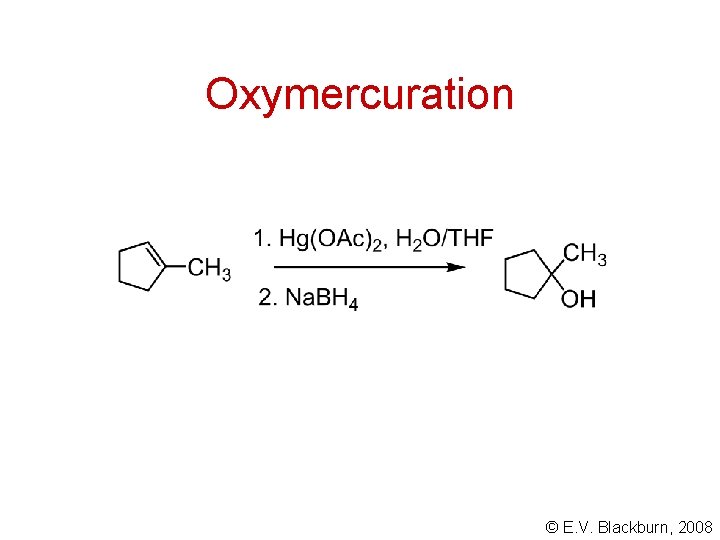
Oxymercuration © E. V. Blackburn, 2008
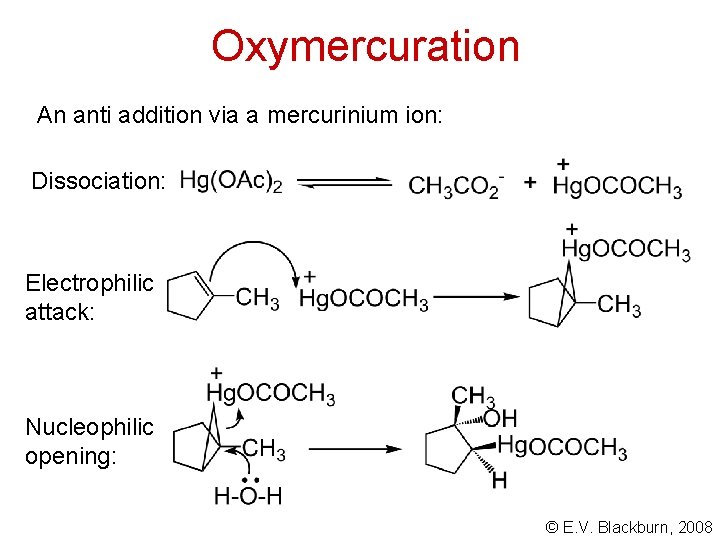
Oxymercuration An anti addition via a mercurinium ion: Dissociation: Electrophilic attack: Nucleophilic opening: © E. V. Blackburn, 2008
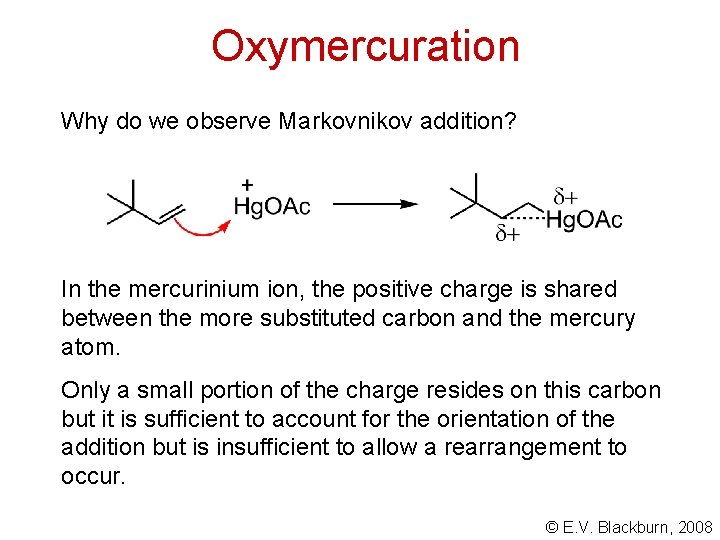
Oxymercuration Why do we observe Markovnikov addition? In the mercurinium ion, the positive charge is shared between the more substituted carbon and the mercury atom. Only a small portion of the charge resides on this carbon but it is sufficient to account for the orientation of the addition but is insufficient to allow a rearrangement to occur. © E. V. Blackburn, 2008
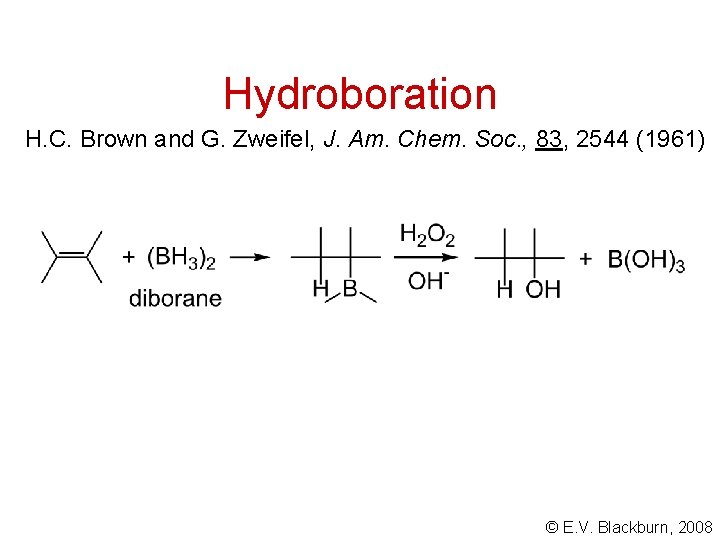
Hydroboration H. C. Brown and G. Zweifel, J. Am. Chem. Soc. , 83, 2544 (1961) © E. V. Blackburn, 2008
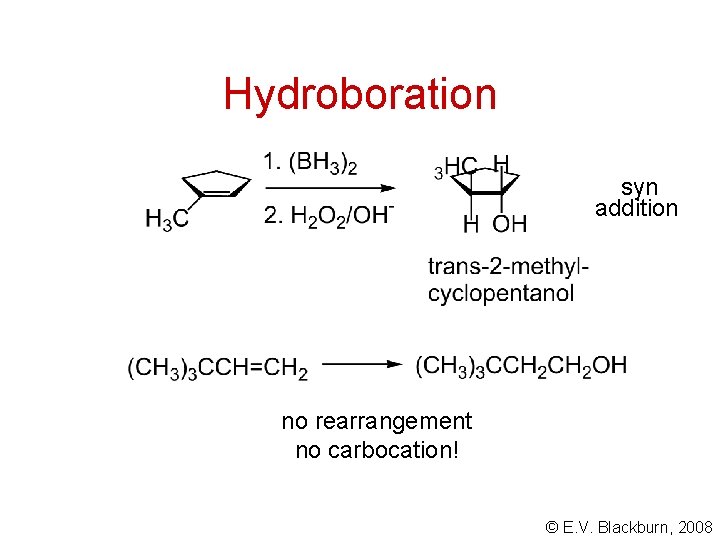
Hydroboration syn addition no rearrangement no carbocation! © E. V. Blackburn, 2008
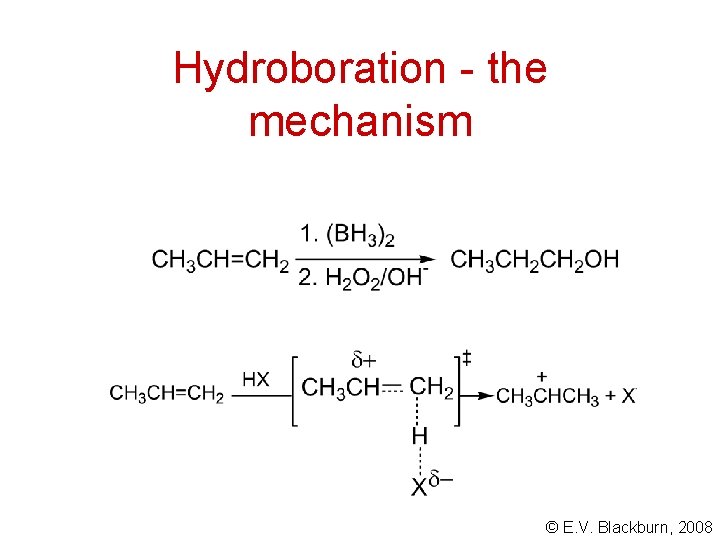
Hydroboration - the mechanism © E. V. Blackburn, 2008
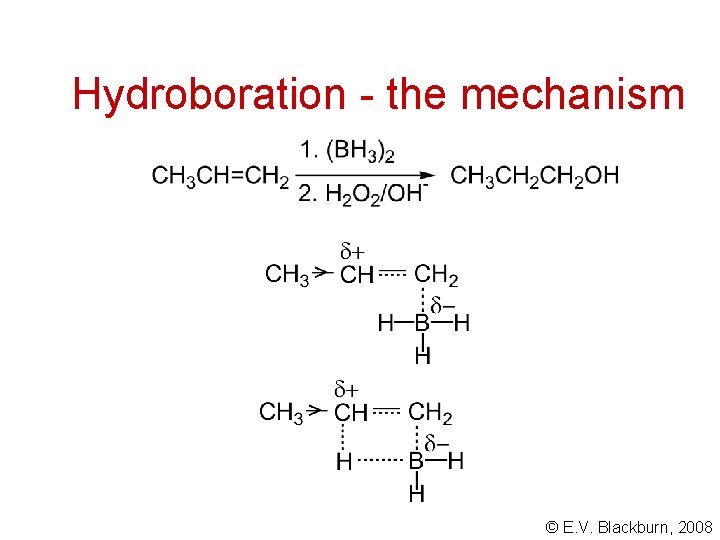
Hydroboration - the mechanism © E. V. Blackburn, 2008
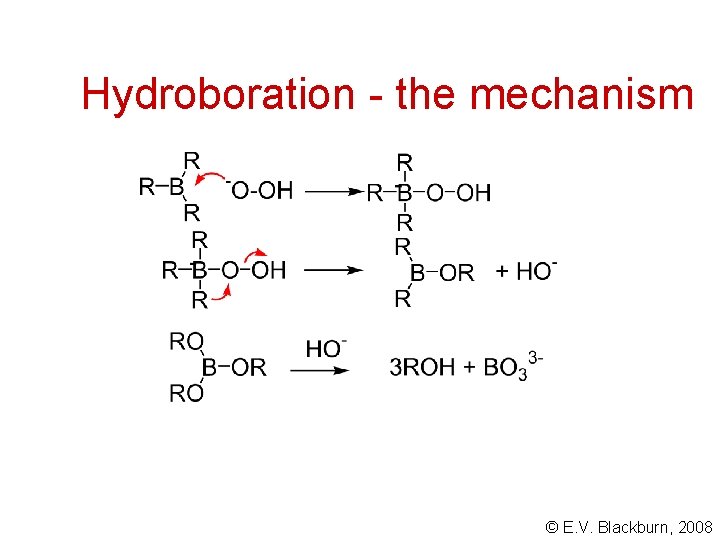
Hydroboration - the mechanism © E. V. Blackburn, 2008
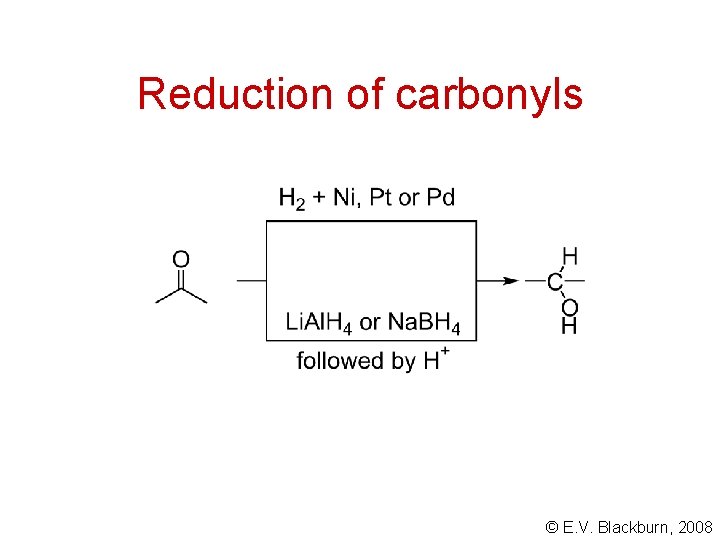
Reduction of carbonyls © E. V. Blackburn, 2008
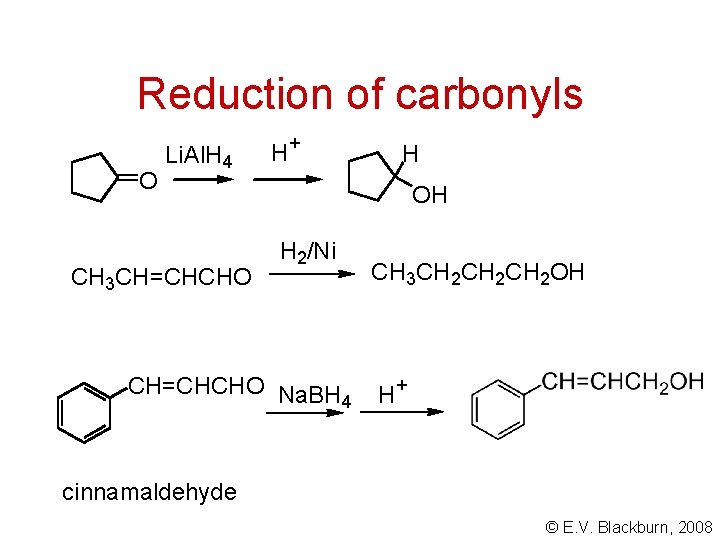
Reduction of carbonyls O Li. Al. H 4 CH 3 CH=CHCHO H + H OH H 2/Ni CH=CHCHO Na. BH 4 CH 3 CH 2 CH 2 OH H + cinnamaldehyde © E. V. Blackburn, 2008
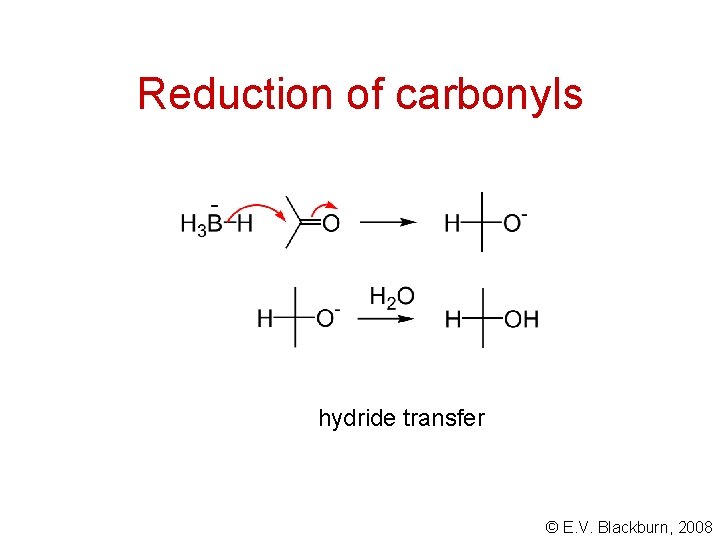
Reduction of carbonyls hydride transfer © E. V. Blackburn, 2008
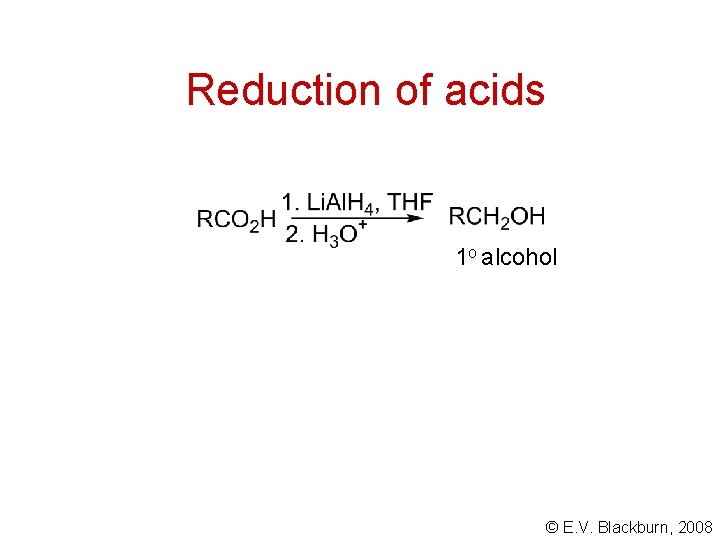
Reduction of acids 1 o alcohol © E. V. Blackburn, 2008
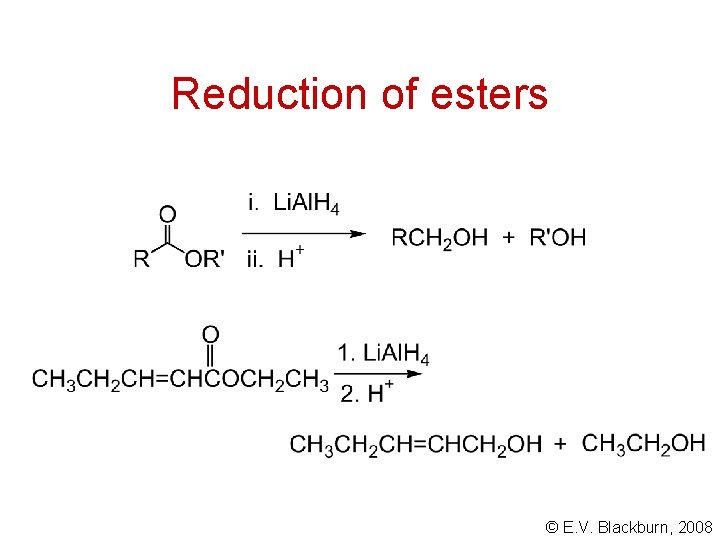
Reduction of esters © E. V. Blackburn, 2008
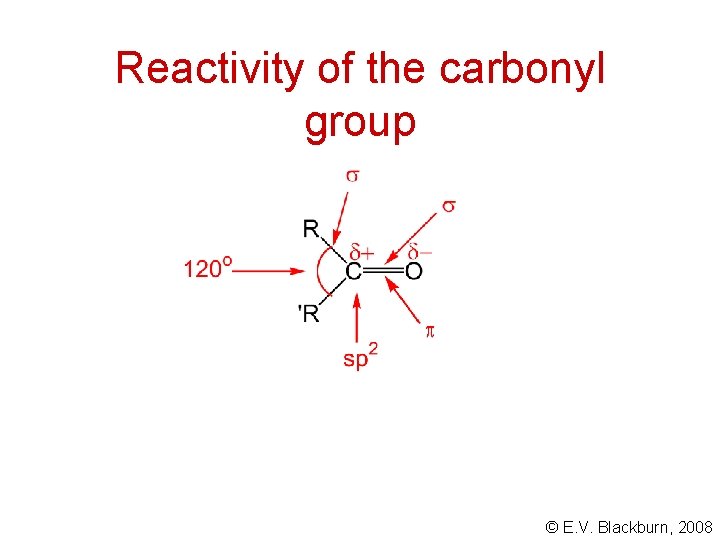
Reactivity of the carbonyl group © E. V. Blackburn, 2008
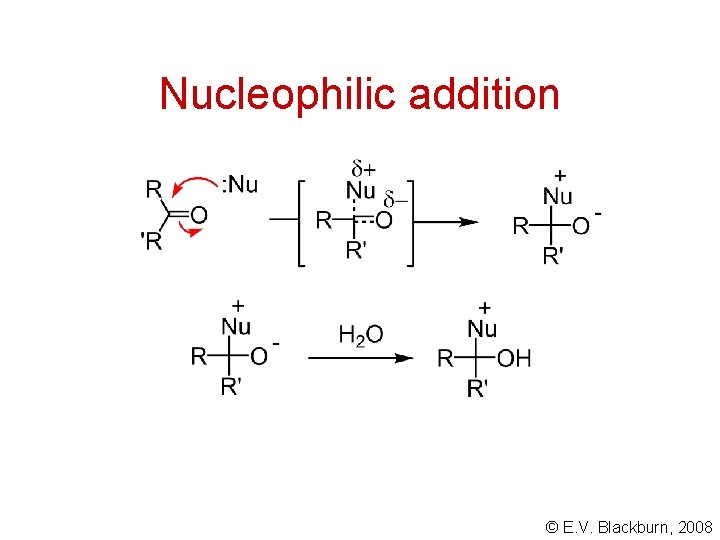
Nucleophilic addition © E. V. Blackburn, 2008
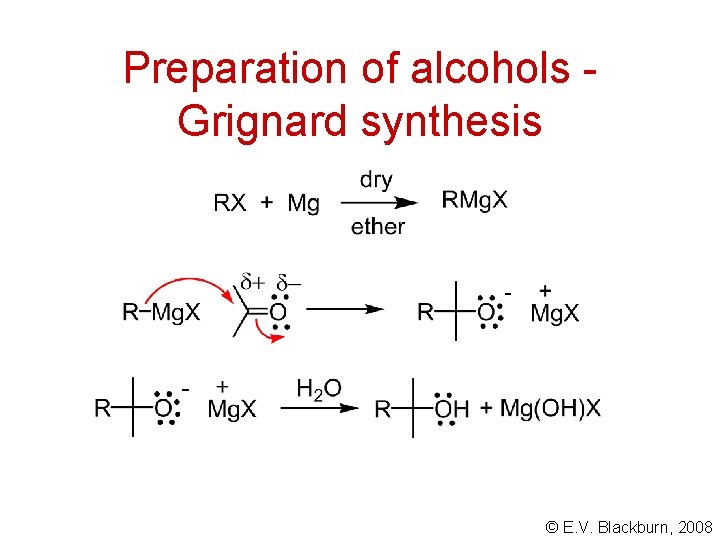
Preparation of alcohols Grignard synthesis © E. V. Blackburn, 2008
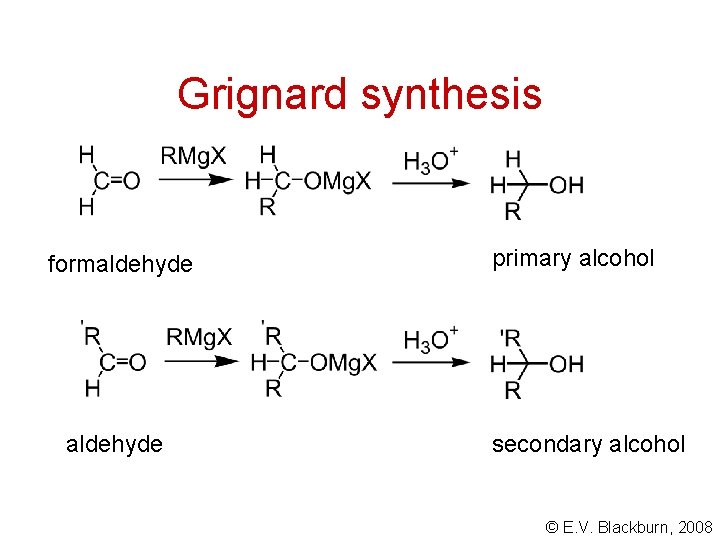
Grignard synthesis formaldehyde primary alcohol secondary alcohol © E. V. Blackburn, 2008
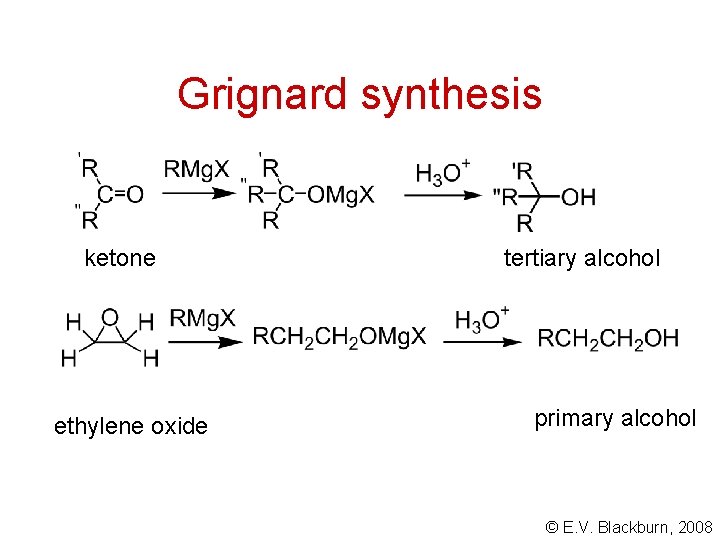
Grignard synthesis ketone ethylene oxide tertiary alcohol primary alcohol © E. V. Blackburn, 2008

Planning a Grignard synthesis © E. V. Blackburn, 2008

Limitations • Any hydrogen bonded to an electronegative element (including an acetylenic hydrogen) is sufficiently acidic to react with a Grignard reagent. CH 3 Mg. I + H 2 O CH 4 + Mg(OH)I • Grignard reagents react with O 2, CO 2 and with almost all organic compounds which contain multiply bonded C-O or C-N units. © E. V. Blackburn, 2008

Reactions of alcohols The reactions of alcohols involve one of two processes: • breaking of the O-H bond • breaking of the C-O bond © E. V. Blackburn, 2008
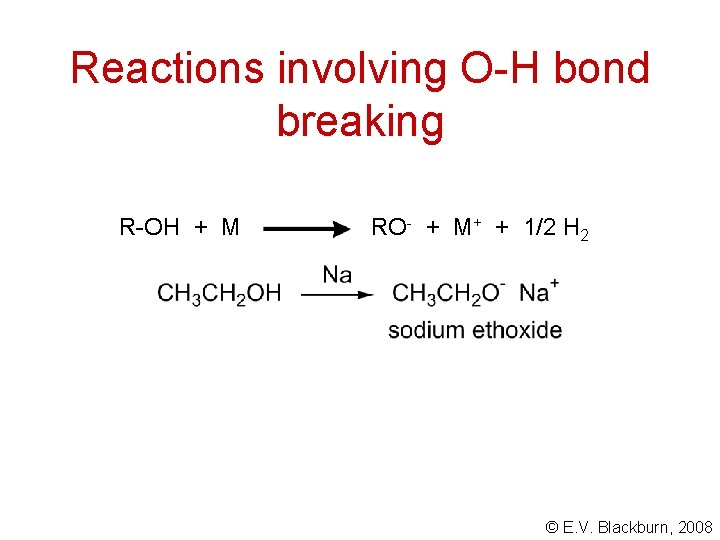
Reactions involving O-H bond breaking R-OH + M RO- + M+ + 1/2 H 2 © E. V. Blackburn, 2008
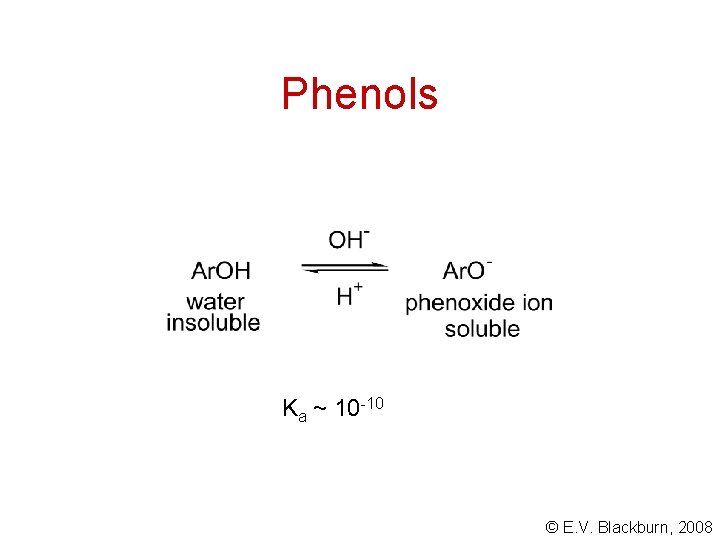
Phenols Ka ~ 10 -10 © E. V. Blackburn, 2008
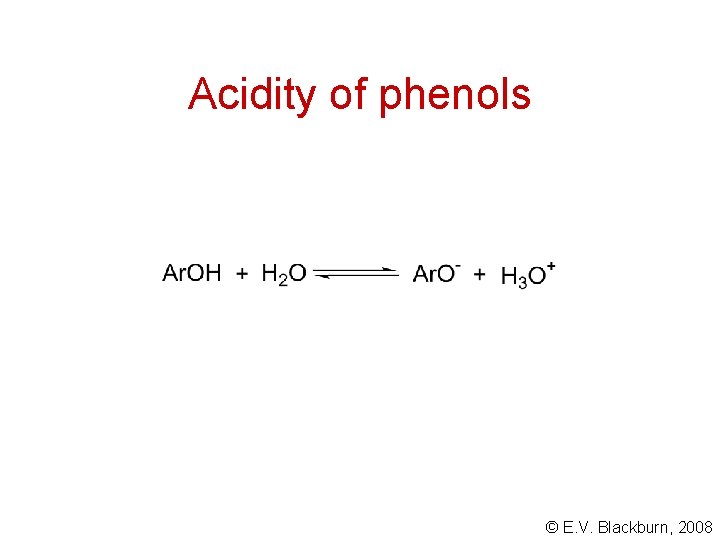
Acidity of phenols © E. V. Blackburn, 2008

Acidity © E. V. Blackburn, 2008
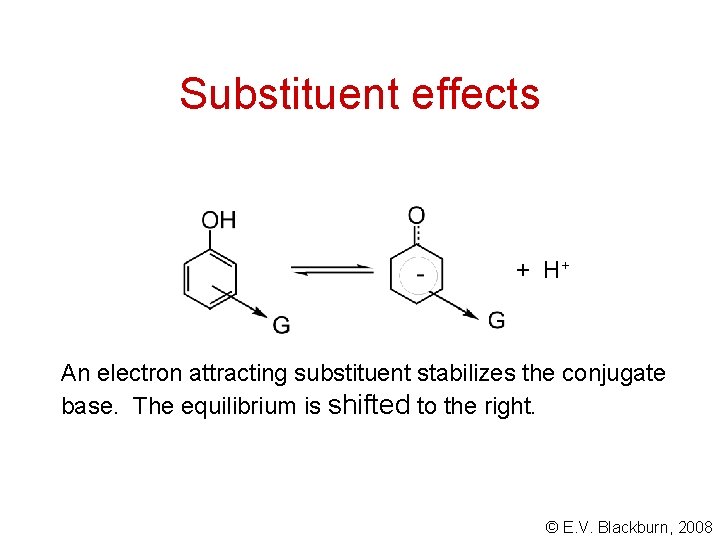
Substituent effects + H+ An electron attracting substituent stabilizes the conjugate base. The equilibrium is shifted to the right. © E. V. Blackburn, 2008
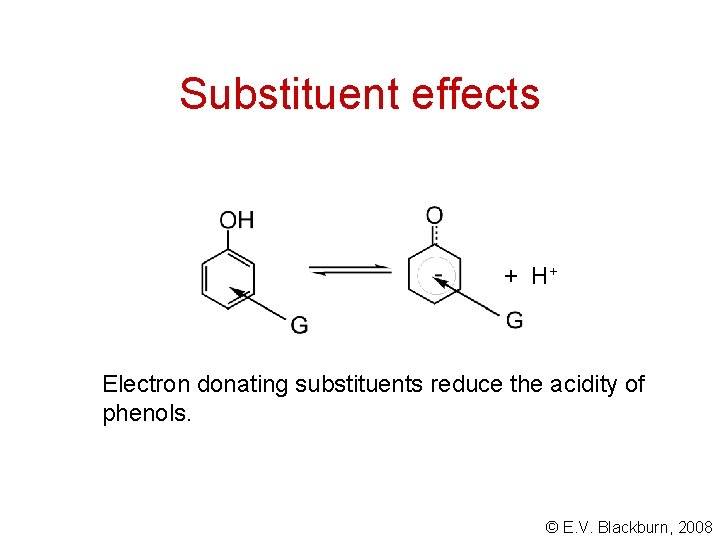
Substituent effects + H+ Electron donating substituents reduce the acidity of phenols. © E. V. Blackburn, 2008
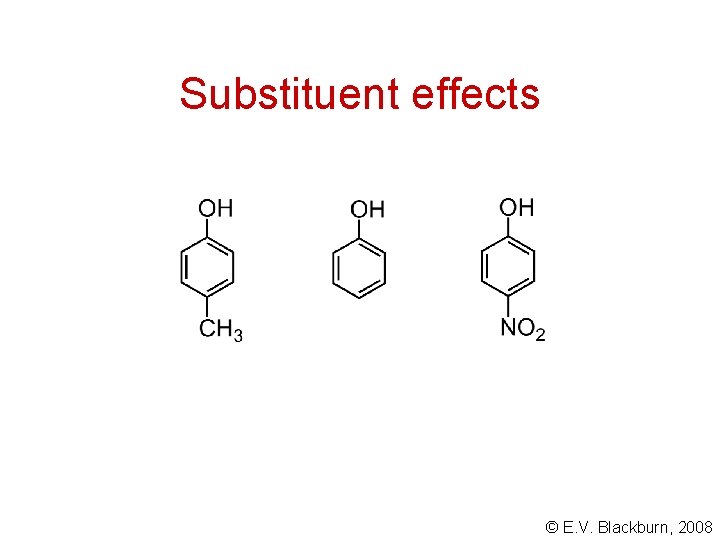
Substituent effects © E. V. Blackburn, 2008
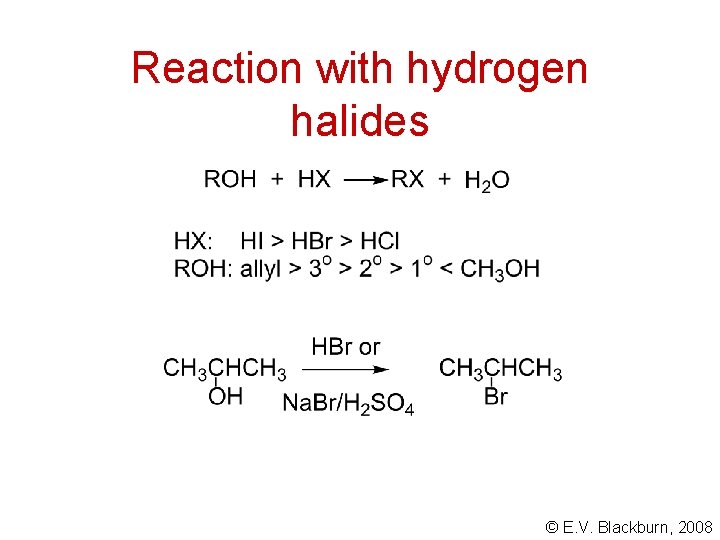
Reaction with hydrogen halides © E. V. Blackburn, 2008
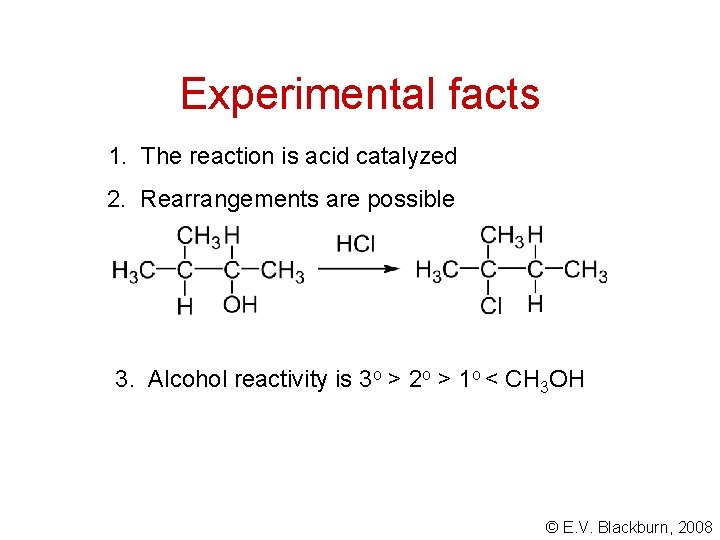
Experimental facts 1. The reaction is acid catalyzed 2. Rearrangements are possible 3. Alcohol reactivity is 3 o > 2 o > 1 o < CH 3 OH © E. V. Blackburn, 2008
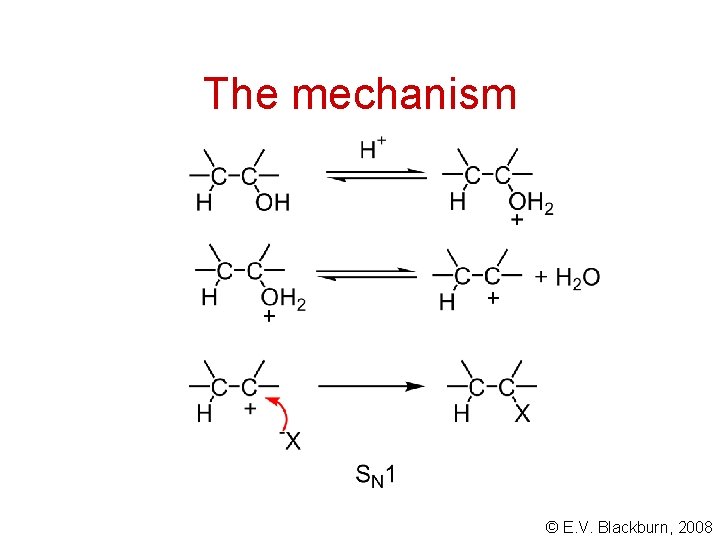
The mechanism © E. V. Blackburn, 2008

Reaction of primary alcohols with HX S N 2 This reflects nucleophile strength in a protic solvent. © E. V. Blackburn, 2008
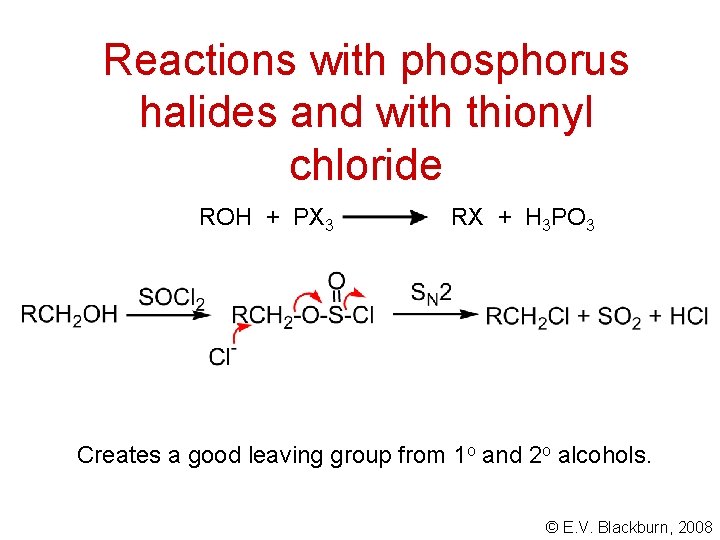
Reactions with phosphorus halides and with thionyl chloride ROH + PX 3 RX + H 3 PO 3 Creates a good leaving group from 1 o and 2 o alcohols. © E. V. Blackburn, 2008
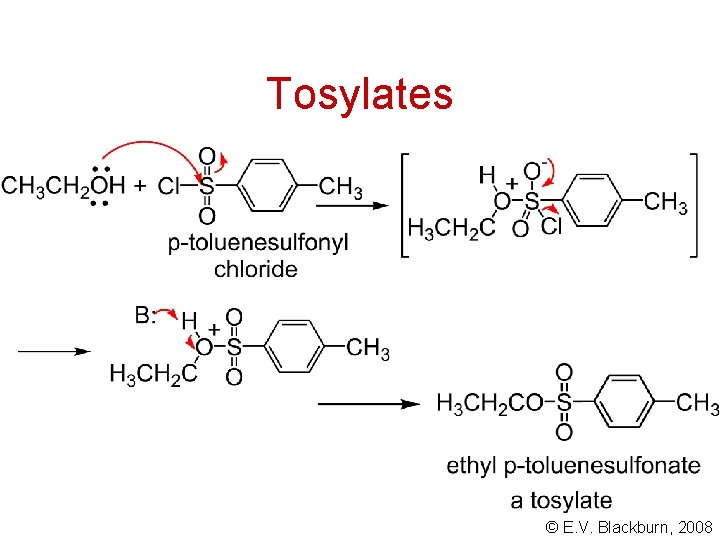
Tosylates © E. V. Blackburn, 2008
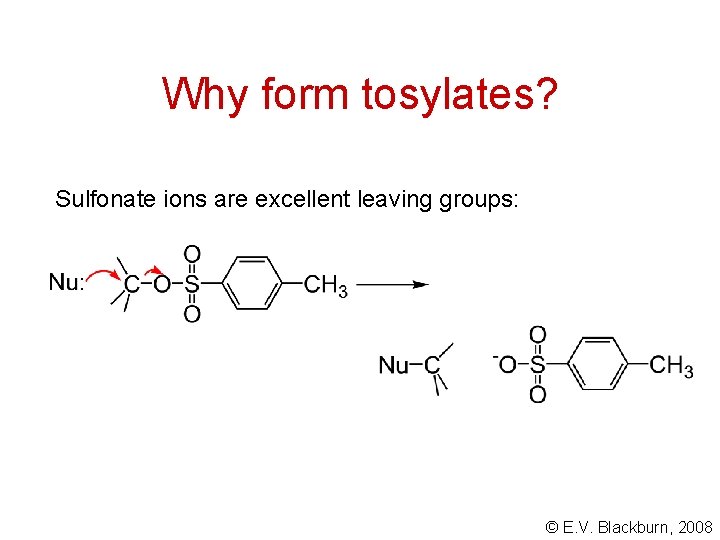
Why form tosylates? Sulfonate ions are excellent leaving groups: © E. V. Blackburn, 2008
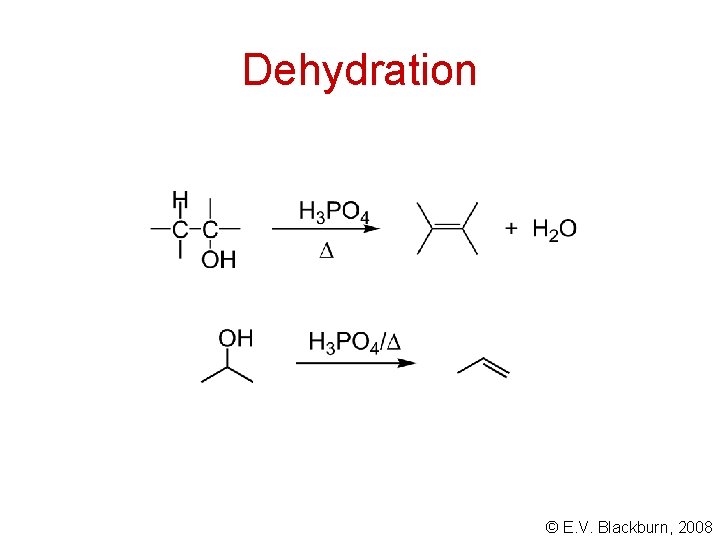
Dehydration © E. V. Blackburn, 2008
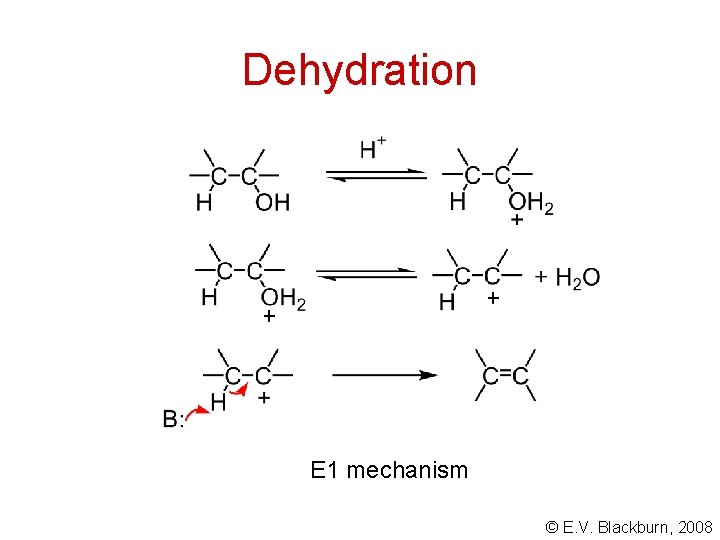
Dehydration E 1 mechanism © E. V. Blackburn, 2008
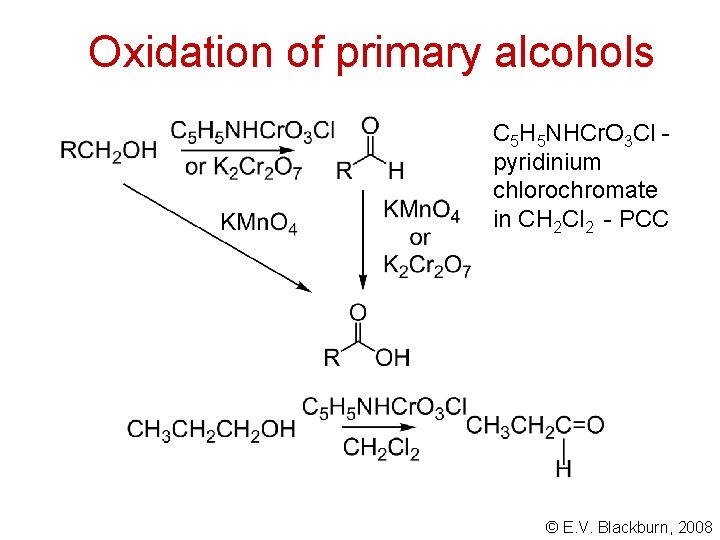
Oxidation of primary alcohols C 5 H 5 NHCr. O 3 Cl pyridinium chlorochromate in CH 2 Cl 2 - PCC © E. V. Blackburn, 2008
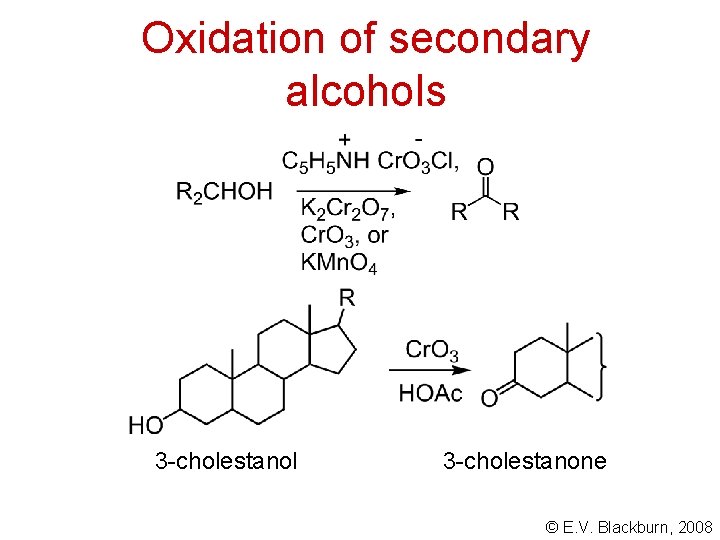
Oxidation of secondary alcohols 3 -cholestanol 3 -cholestanone © E. V. Blackburn, 2008
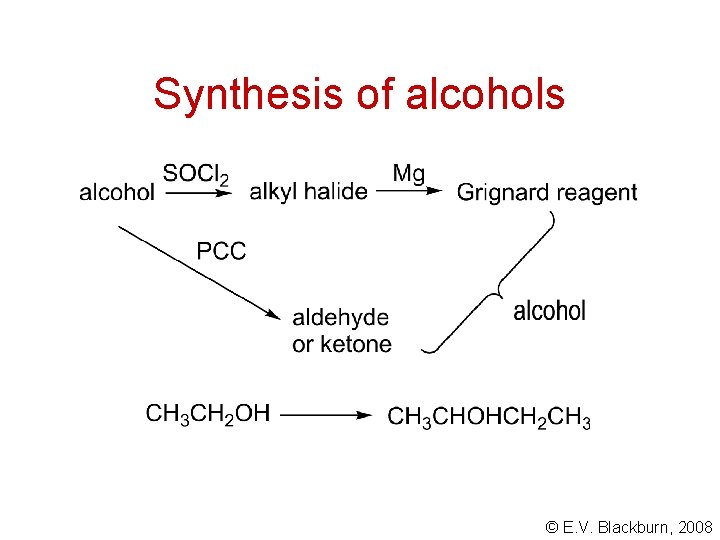
Synthesis of alcohols © E. V. Blackburn, 2008
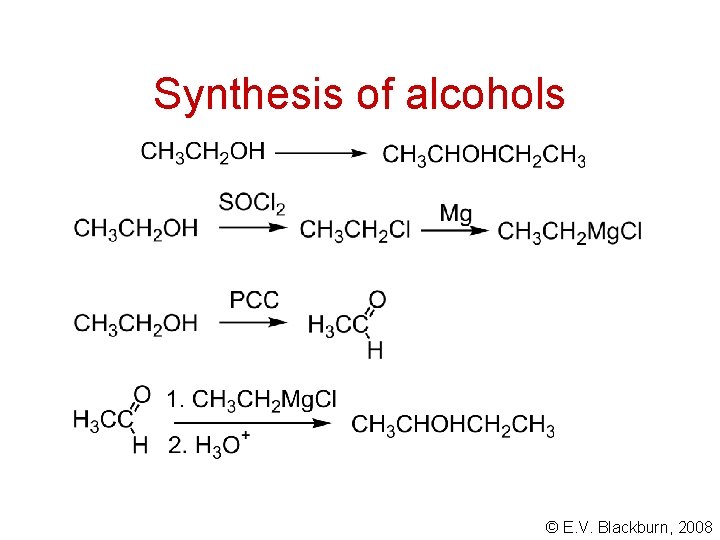
Synthesis of alcohols © E. V. Blackburn, 2008
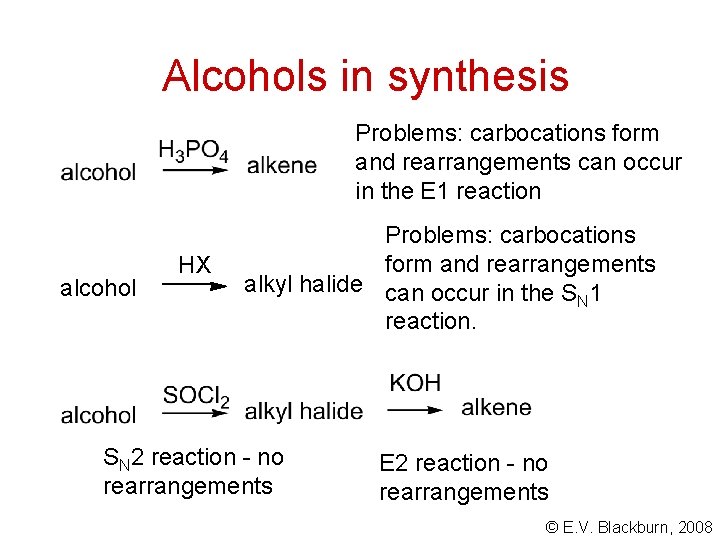
Alcohols in synthesis Problems: carbocations form and rearrangements can occur in the E 1 reaction alcohol HX Problems: carbocations form and rearrangements alkyl halide can occur in the S 1 N reaction. SN 2 reaction - no rearrangements E 2 reaction - no rearrangements © E. V. Blackburn, 2008
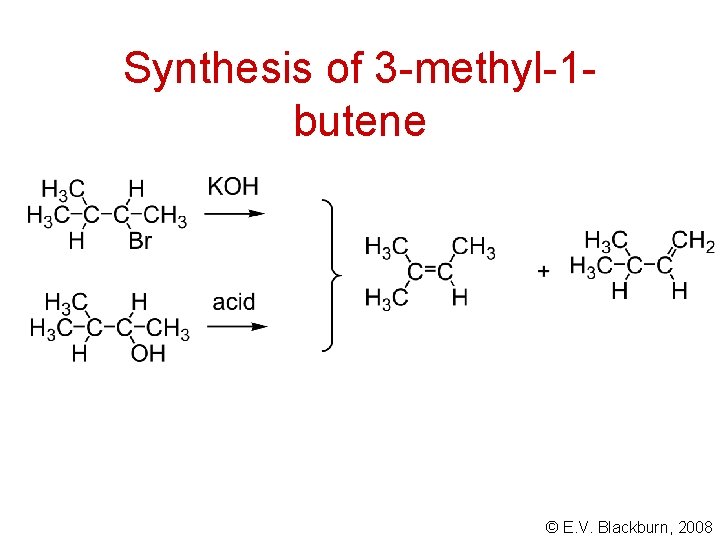
Synthesis of 3 -methyl-1 butene © E. V. Blackburn, 2008
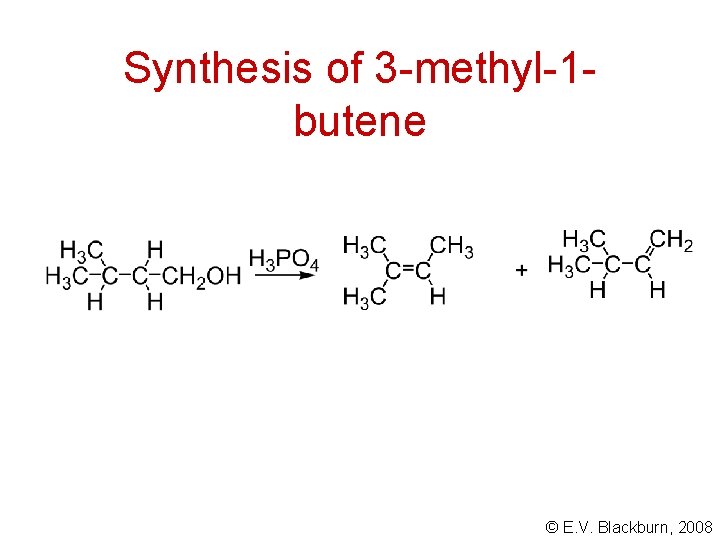
Synthesis of 3 -methyl-1 butene © E. V. Blackburn, 2008
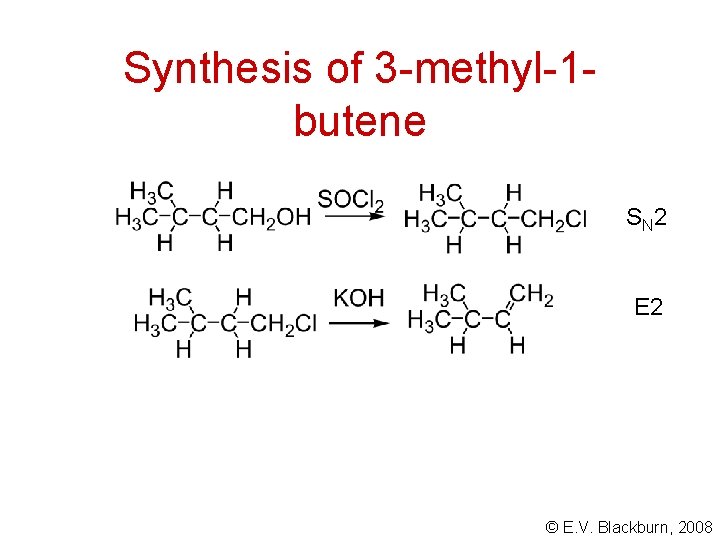
Synthesis of 3 -methyl-1 butene S N 2 E 2 © E. V. Blackburn, 2008

Ethers Structure: R-O-R, Ar-O-R, or Ar-O-Ar nomenclature Name the two groups bonded to the oxygen and add the word ether. CH 3 CH 2 OCH 2 CH 3 - diethyl ether © E. V. Blackburn, 2008
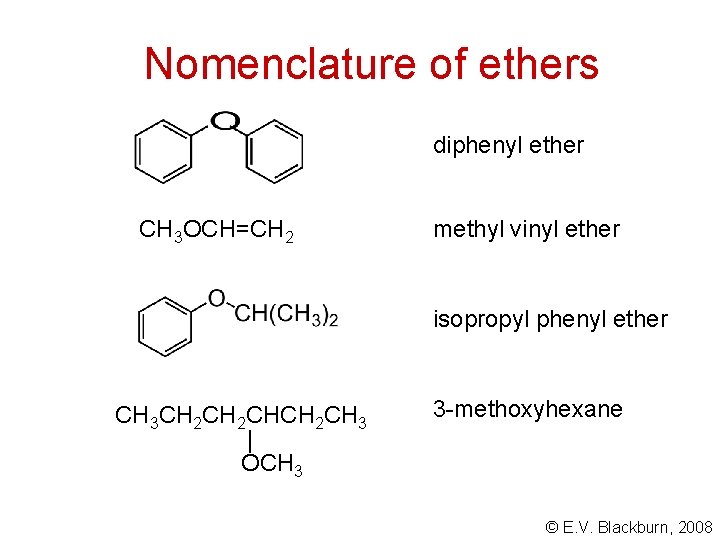
Nomenclature of ethers diphenyl ether CH 3 OCH=CH 2 methyl vinyl ether isopropyl phenyl ether CH 3 CH 2 CHCH 2 CH 3 | OCH 3 3 -methoxyhexane © E. V. Blackburn, 2008
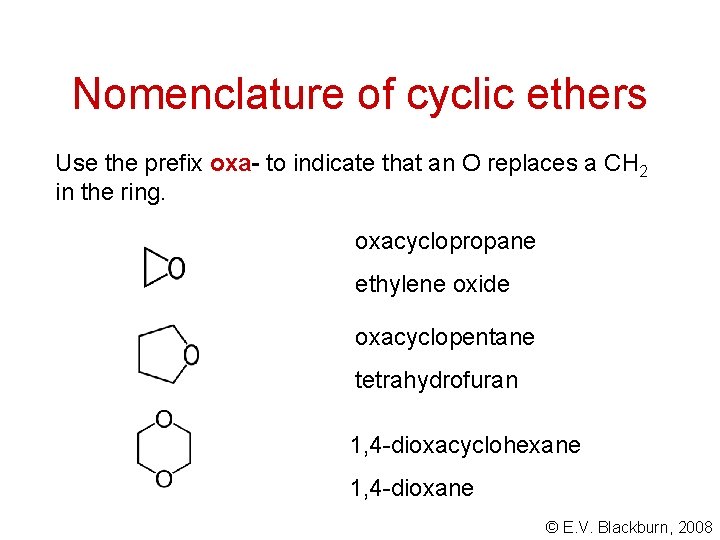
Nomenclature of cyclic ethers Use the prefix oxa- to indicate that an O replaces a CH 2 in the ring. oxacyclopropane ethylene oxide oxacyclopentane tetrahydrofuran 1, 4 -dioxacyclohexane 1, 4 -dioxane © E. V. Blackburn, 2008

Williamson synthesis A primary halide is necessary to ensure an SN 2 reaction and not an E 2 elimination. © E. V. Blackburn, 2008
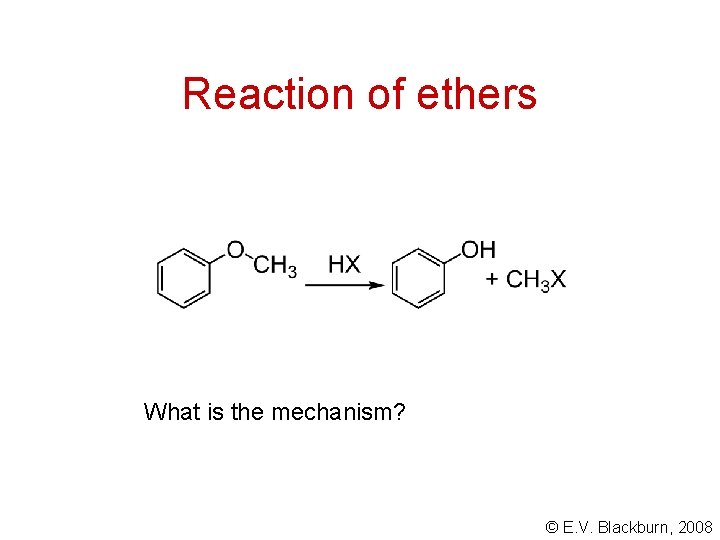
Reaction of ethers What is the mechanism? © E. V. Blackburn, 2008
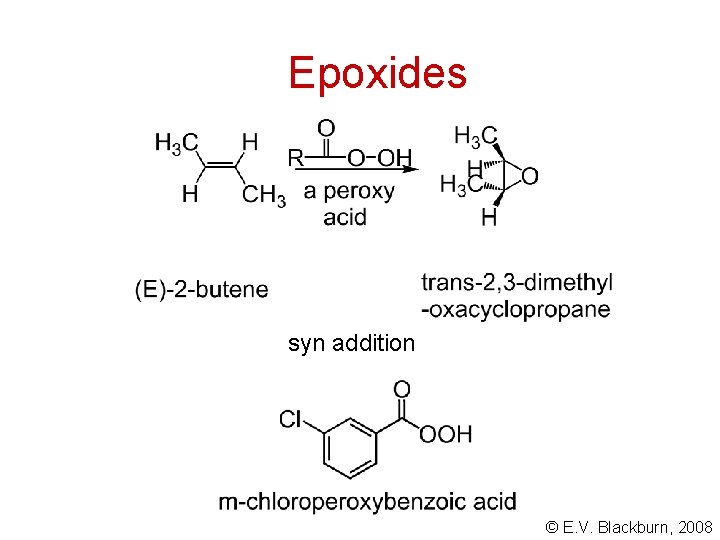
Epoxides syn addition © E. V. Blackburn, 2008
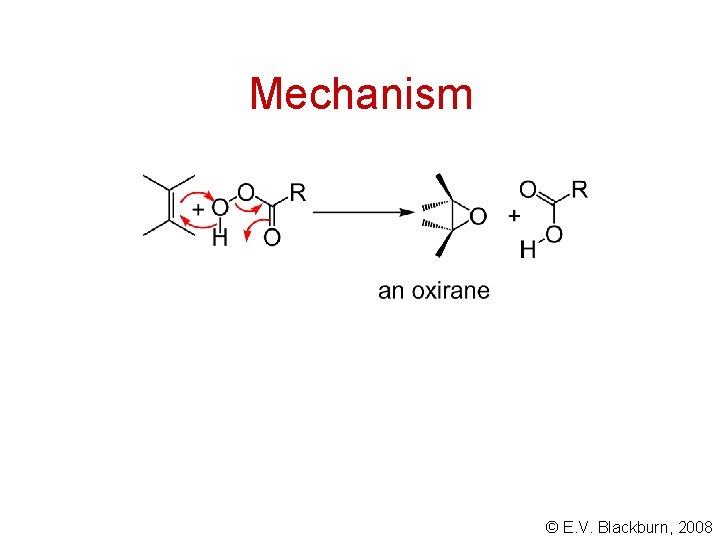
Mechanism © E. V. Blackburn, 2008
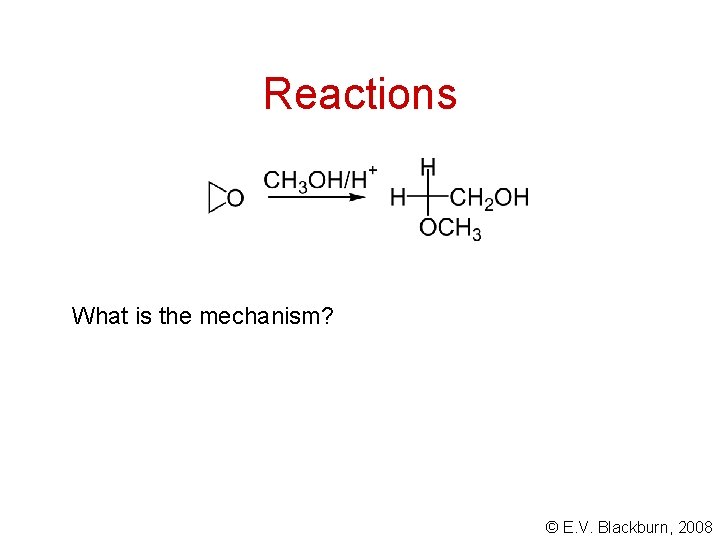
Reactions What is the mechanism? © E. V. Blackburn, 2008
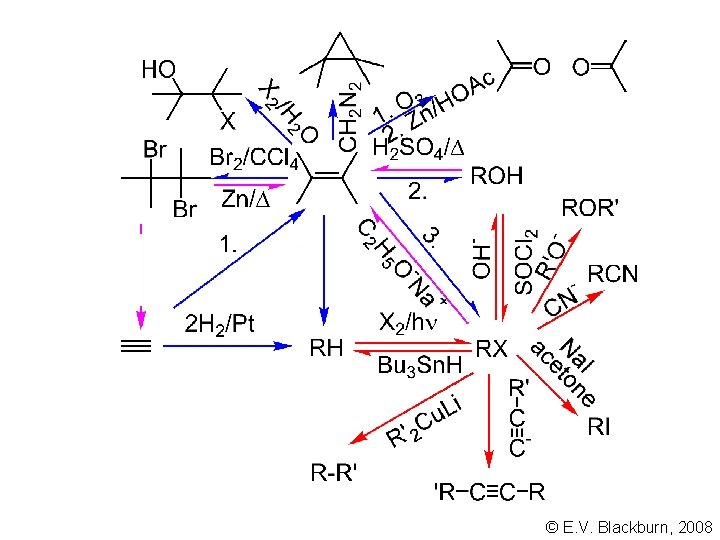
© E. V. Blackburn, 2008

Problems Solomons and Fryhle, 11. 26, 11. 27, 11. 30, 11. 33, 11. 36, 11. 46, 12. 11, 12. 12, 12. 14, 12. 16, 12. 17, and 12. 20 © E. V. Blackburn, 2008
- Slides: 76