Alcohols and Phenols By Dr Gehan Mostafa Kotb

Alcohols and Phenols By Dr. Gehan Mostafa Kotb Date/time: Sunday, 22/3 from 12 to 2 pm and Thursday 26/3, from 12 to 2 pm 1

Introduction Alcohol is a compound that has a hydroxyl group bonded to a saturated, sp 3 -hybridized carbon atom, R—OH. Phenol has a hydroxyl group bonded to an aromatic ring, Ar—OH.
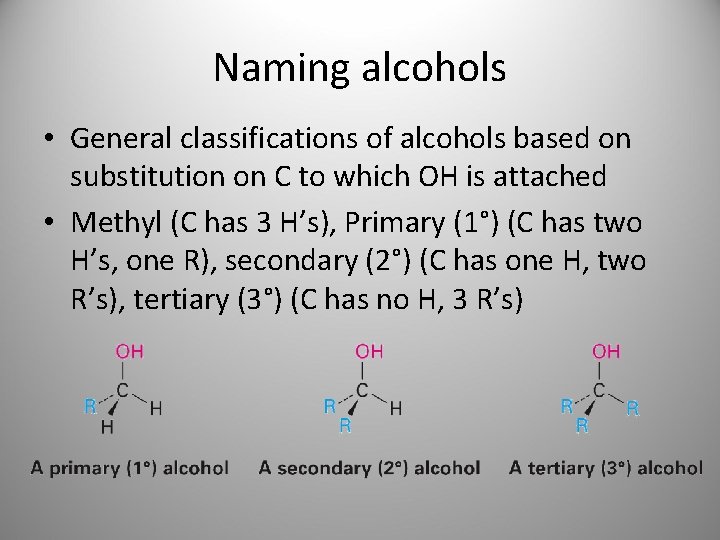
Naming alcohols • General classifications of alcohols based on substitution on C to which OH is attached • Methyl (C has 3 H’s), Primary (1°) (C has two H’s, one R), secondary (2°) (C has one H, two R’s), tertiary (3°) (C has no H, 3 R’s)

Classify each alcohol as (P) primary, (S) secondary, or (T) tertiary. OH | 1. CH 3—CH—CH 2—CH 3 2. CH 3—CH 2—OH OH | 3. CH 3—CH 2—CH 3 | CH 3 4

Naming alcohols • Start with the parent chain and replace the hydrocarbon –e ending with –ol • Number the hydrocarbon chain. The carbon containing the –OH group should have the lowest number. • Number the position of the –OH group • Number and name other side chains or functionalities using IUPAC rules 1 -butanol 2 -methyl-1 -propanol

Naming alcohols • that are common names use the name of the alkyl group followed by alcohol. Formula IUPAC Common Name CH 4 methane CH 3─OH methanol CH 3─CH 3 ethane CH 3─CH 2─OH ethanol methyl alcohol

• Compounds containing more than one OH group are named diols, triols, etc. – Note that the final e of the parent alkane name is retained.
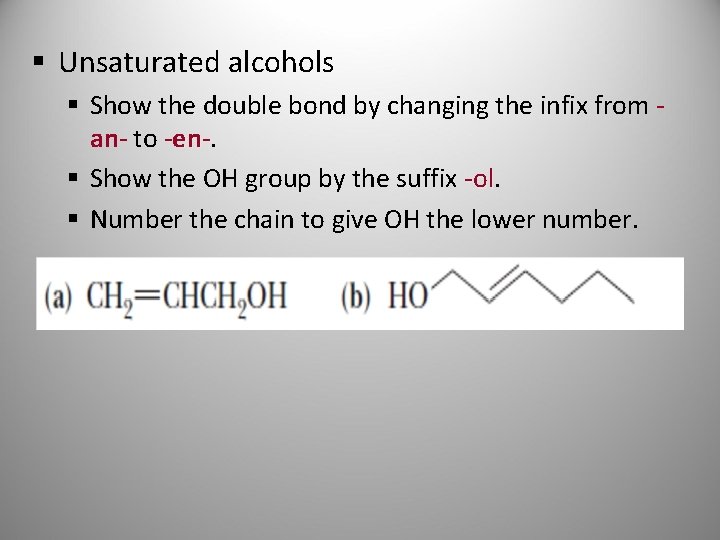
§ Unsaturated alcohols § Show the double bond by changing the infix from an- to -en-. § Show the OH group by the suffix -ol § Number the chain to give OH the lower number.

Naming Phenols • Use “phenol” as the parent hydrocarbon name, not benzene • Name substituents on aromatic ring by their position from OH

Physical properties of alcohols § Polarity of the C-O-H bond in an alcohol. § They interact with themselves and with other polar compounds by dipole -dipole interactions. § Dipole-dipole interaction: The attraction between the positive end of one dipole and the negative end of another. § Alcohols have much higher boiling points than similar alkanes and alkyl halides

Physical properties of alcohols water insoluble! (except for alcohols of three carbons or less) CH 3 CH 2 CH 2 CH 2 -OH hydrophobic hydrophilic

Physical properties of alcohols • We already saw that the boiling points of alkanes increase with increasing chain length. The same is true for alcohols. • Alcohols with more than one hydroxyl group (polyhydroxy alcohols) have higher boiling points than monoydroxy alcohols.
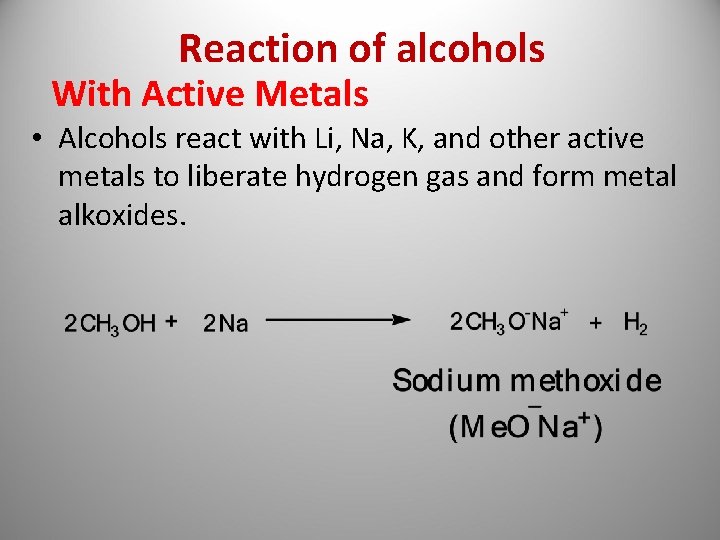
Reaction of alcohols With Active Metals • Alcohols react with Li, Na, K, and other active metals to liberate hydrogen gas and form metal alkoxides.
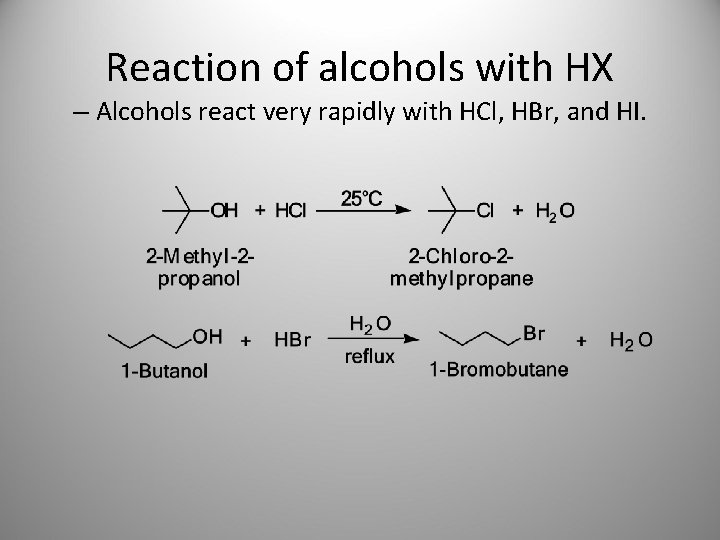
Reaction of alcohols with HX – Alcohols react very rapidly with HCl, HBr, and HI.

Reaction of alcohols with PBr 3 • An alternative method for the synthesis of bromoalkanes is reaction of an alcohol with phosphorous tribromide (PBr 3). 3 3
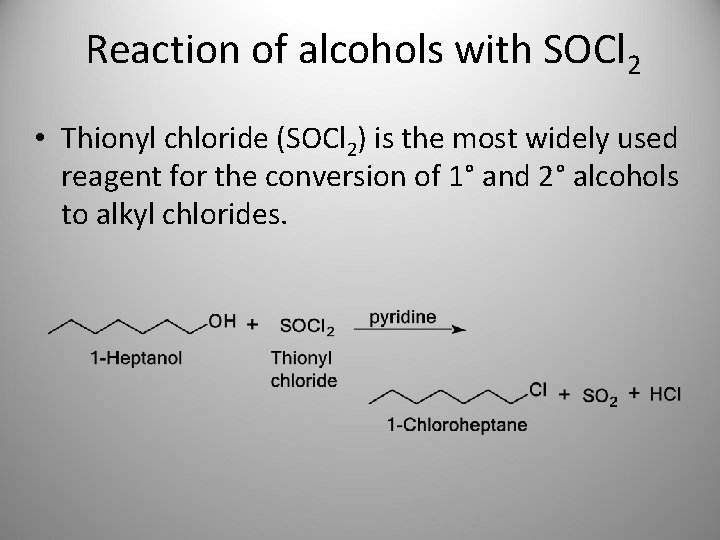
Reaction of alcohols with SOCl 2 • Thionyl chloride (SOCl 2) is the most widely used reagent for the conversion of 1° and 2° alcohols to alkyl chlorides.

Dehydration of R-OH • An alcohol can be converted to an alkene by acid-catalyzed dehydration. – 1° alcohols must be heated at high temperature in the presence of an acid catalyst, such as H 2 SO 4 or H 3 PO 4. – 2° alcohols undergo dehydration at somewhat lower temperatures. – 3° alcohols often require temperatures at or only slightly above room temperature.
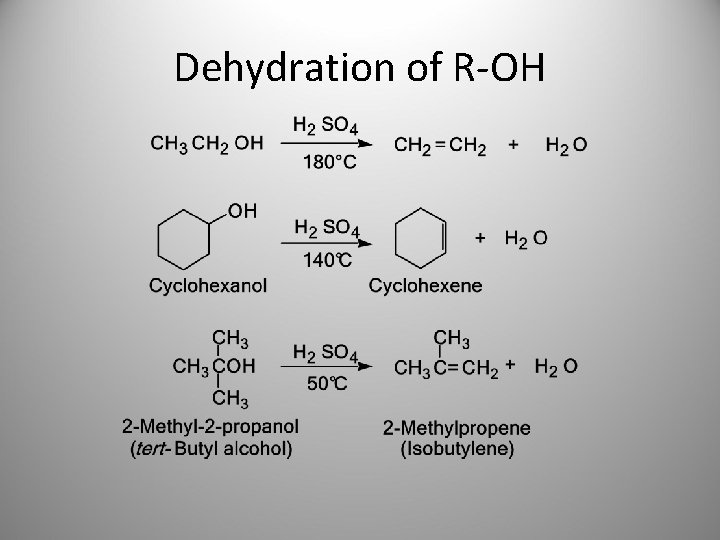
Dehydration of R-OH

Dehydration of R-OH – Where isomeric alkenes are possible, the alkene having the greater number of substituents on the double bond (the more stable alkene) usually predominates Saytzeff’s rule (Zaitsev rule).

Oxidation: 1° R-OH • Oxidation of a primary alcohol gives an aldehyde or a carboxylic acid, depending on the experimental conditions.
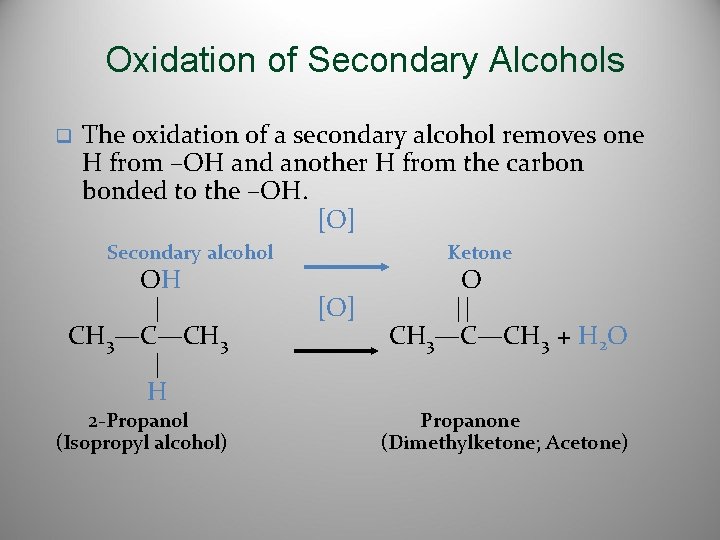
Oxidation of Secondary Alcohols q The oxidation of a secondary alcohol removes one H from –OH and another H from the carbon bonded to the –OH. [O] Secondary alcohol OH | CH 3—C—CH 3 | H 2 -Propanol (Isopropyl alcohol) Ketone [O] O || CH 3—C—CH 3 + H 2 O Propanone (Dimethylketone; Acetone)
![Oxidation of Tertiary Alcohols q Tertiary alcohols are resistant to oxidation. [O] Tertiary alcohols Oxidation of Tertiary Alcohols q Tertiary alcohols are resistant to oxidation. [O] Tertiary alcohols](http://slidetodoc.com/presentation_image_h2/3a648d2cd9aff09a219f6b93724449b0/image-22.jpg)
Oxidation of Tertiary Alcohols q Tertiary alcohols are resistant to oxidation. [O] Tertiary alcohols no reaction OH | [O] CH 3—C—CH 3 no product | CH 3 no H on the C-OH to oxidize 2 -Methyl-2 -propanol
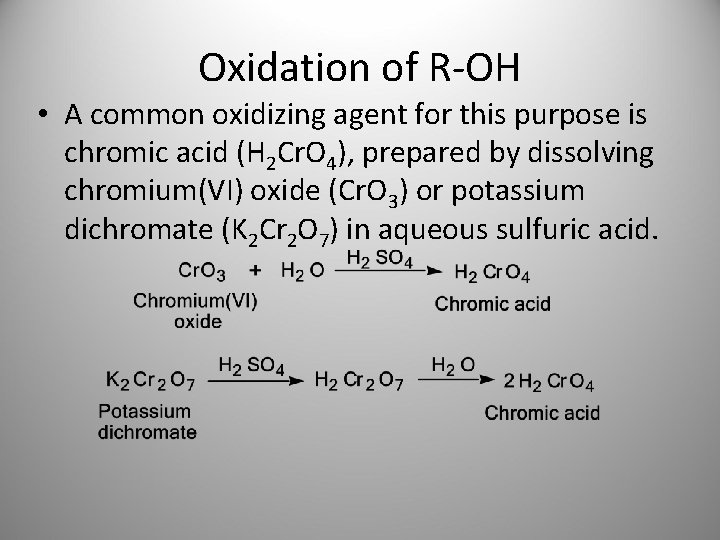
Oxidation of R-OH • A common oxidizing agent for this purpose is chromic acid (H 2 Cr. O 4), prepared by dissolving chromium(VI) oxide (Cr. O 3) or potassium dichromate (K 2 Cr 2 O 7) in aqueous sulfuric acid.
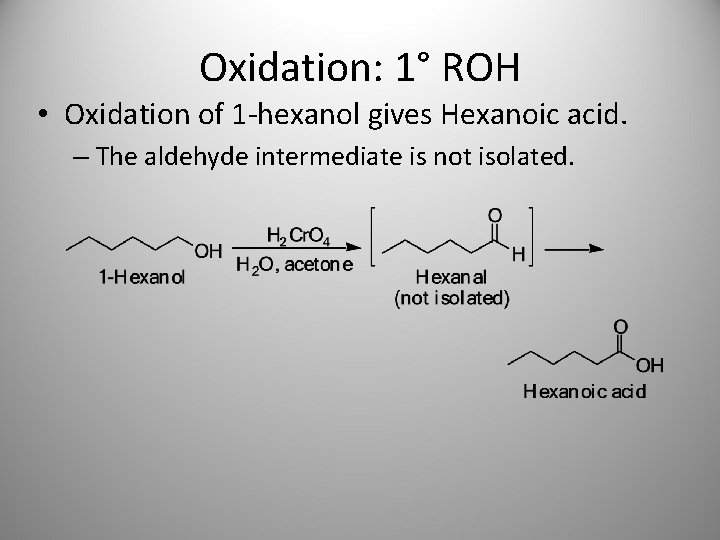
Oxidation: 1° ROH • Oxidation of 1 -hexanol gives Hexanoic acid. – The aldehyde intermediate is not isolated.

Synthesis of Alcohols – Reduction of Aldehydes and Ketones • Aldehydes are reduced to give primary alcohols, and ketones are reduced to give secondary alcohols.
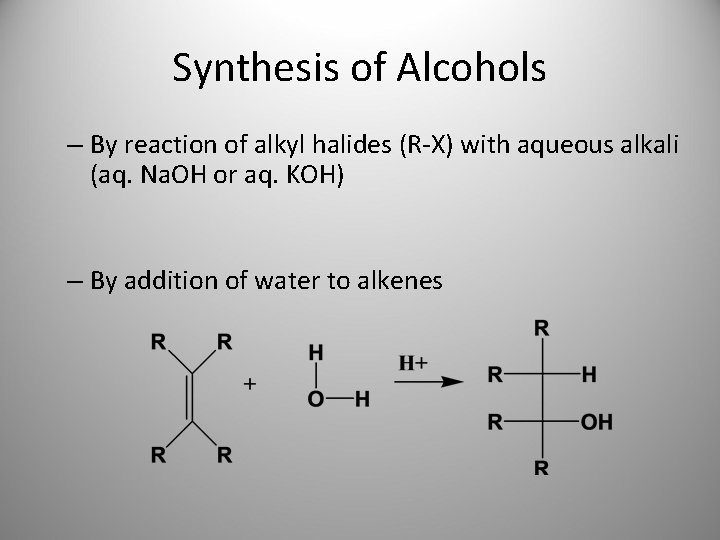
Synthesis of Alcohols – By reaction of alkyl halides (R-X) with aqueous alkali (aq. Na. OH or aq. KOH) – By addition of water to alkenes

Physical properties of Phenols • The OH group of phenols allows hydrogen bonding to other phenol molecules and to water, O-H………O • phenols are polar, • phenols are water soluble, • phenols are stronger acids than water. • Pure phenol is a white crystalline solid, • Smelling of disinfectant.

Reactions of phenols alcohols phenols 1. HX NR 2. PX 3 NR 3. dehydration NR 4. as acids phenols are more acidic 5. oxidation NR
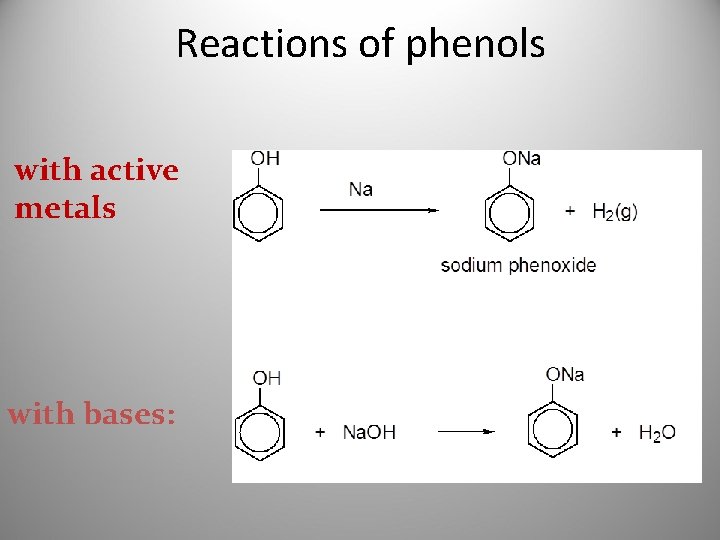
Reactions of phenols with active metals with bases:
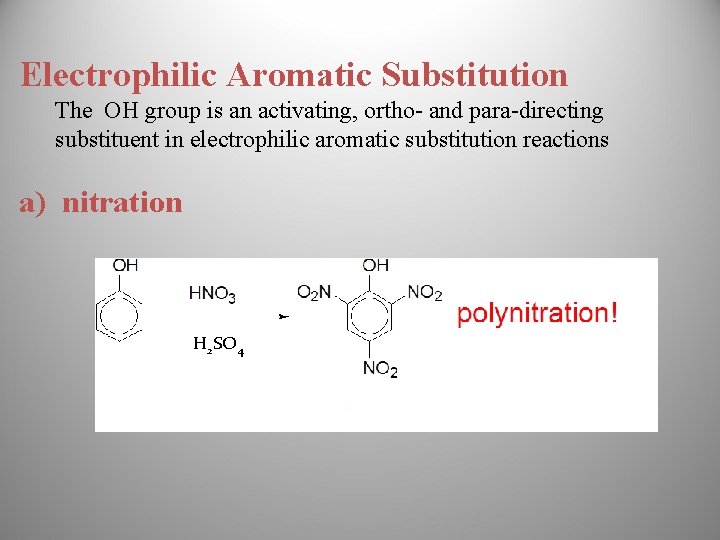
Electrophilic Aromatic Substitution The OH group is an activating, ortho- and para-directing substituent in electrophilic aromatic substitution reactions a) nitration H 2 SO 4
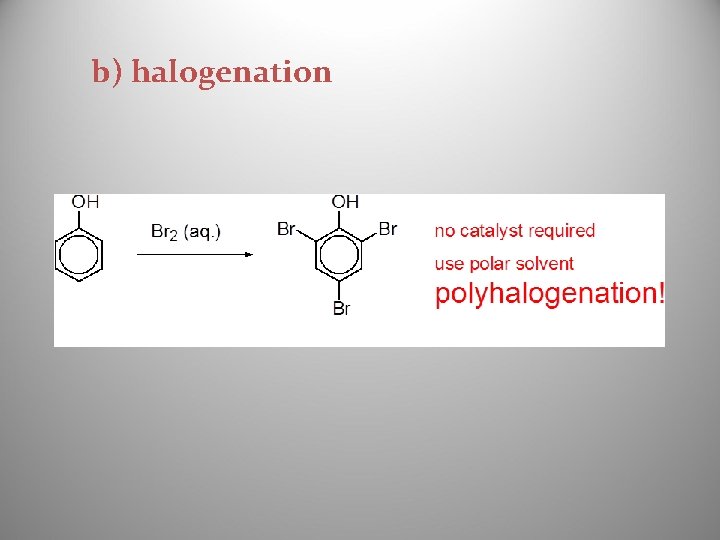
b) halogenation

c) Sulfonation
- Slides: 32