ALCOHOL ETHER part II By Mdm Rohazita Bahari

ALCOHOL & ETHER (part II) By Mdm Rohazita Bahari Semester II 2019/2020 ERT 116, Organic Chemistry II
INTRODUCTION
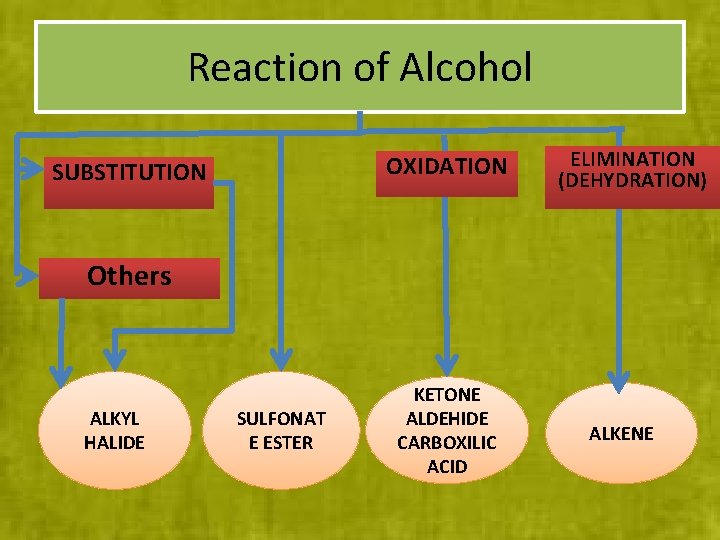
Reaction of Alcohol OXIDATION SUBSTITUTION ELIMINATION (DEHYDRATION) Others ALKYL HALIDE SULFONAT E ESTER KETONE ALDEHIDE CARBOXILIC ACID ALKENE

Alcohol • Alcohol not reactive in nucleophilic substitution or elemination reaction since hydroxide is strong base (poor leaving group) : : key point: : • In substitution reaction of alcohol involve nucleophilic substitution of alcohol to convert strong base (hydroxide) to weaker base (good basic leaving group)

nucleophilic substitution? ? • Involve attack of a positively charged atom or group by nucleophile Electrophilic substitutions? ? • Involve displacement of functional group by an electrophile

Nucleophiles Molecules that contain atoms with lone pairs, which can donate electrons. Often these are negatively charged. Almost all the time they contain elements from groups 15– 17 of the periodic table, since those have lone pairs. ØExamples:

Electrophiles Molecules that contain atoms with empty orbital, which can accommodate electrons. Typically, these are positively charged. ØExamples: Boron has only 6 valence electrons. BF 3 is a Lewis acid.
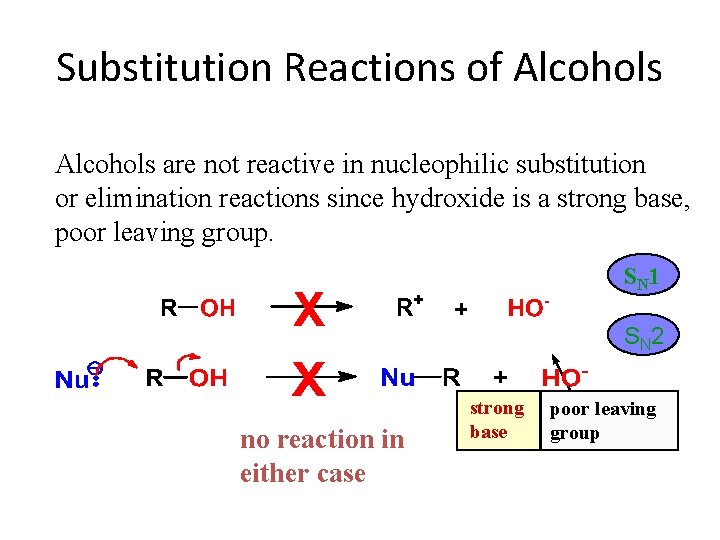
Substitution Reactions of Alcohols are not reactive in nucleophilic substitution or elimination reactions since hydroxide is a strong base, poor leaving group. S N 1 S N 2 no reaction in either case strong base poor leaving group
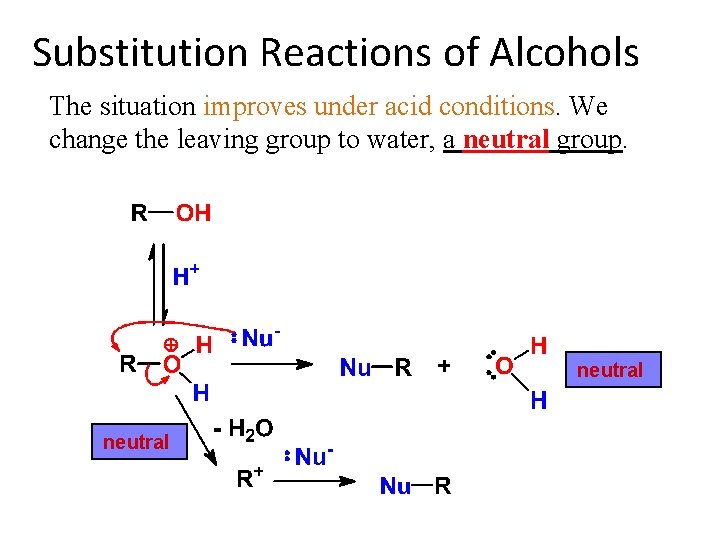
Substitution Reactions of Alcohols The situation improves under acid conditions. We change the leaving group to water, a neutral group. neutral
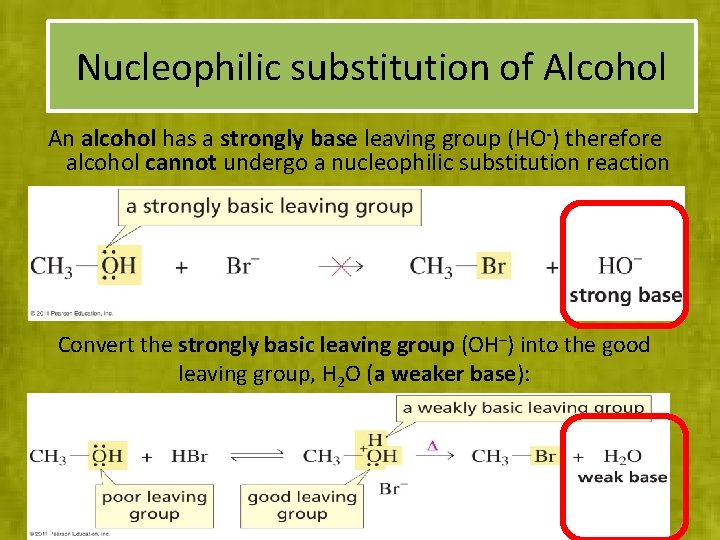
Nucleophilic substitution of Alcohol An alcohol has a strongly base leaving group (HO-) therefore alcohol cannot undergo a nucleophilic substitution reaction Convert the strongly basic leaving group (OH–) into the good leaving group, H 2 O (a weaker base):
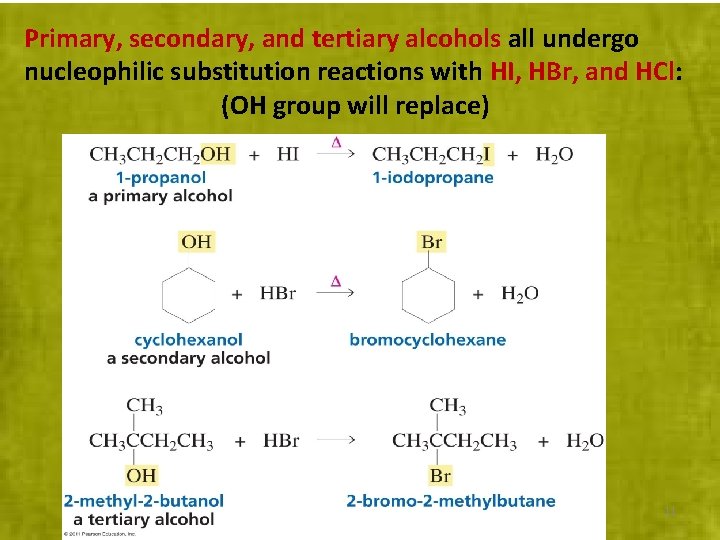
Primary, secondary, and tertiary alcohols all undergo nucleophilic substitution reactions with HI, HBr, and HCl: (OH group will replace) 11

SN 1 reaction • The SN 1 reaction is a substitution reaction in organic chemistry. "SN" stands for nucleophilic substitution and the "1" represents the fact that the ratedetermining step is unimolecular. • It involves a carbocation intermediate and is commonly seen in reactions of secondary or tertiary alkyl halides or, under strongly acidic conditions, with secondary or tertiary alcohols.

SN 2 reaction • The SN 2 reaction (known as bimolecular substitution nucleophilic) is a type of nucleophilic substitution, where a lone pair from a nucleophilic attacks an electron deficient electrophilic center and bonds to it, expelling another group called a leaving group. Thus the incoming group replaces the leaving group in one step. • Since two reacting species are involved in the slow, ratedetermining step of the reaction, this leads to the name bimolecular nucleophilic substitution, or SN 2. • The somewhat more transparently named analog to SN 2 among inorganic chemists is the interchange mechanism.
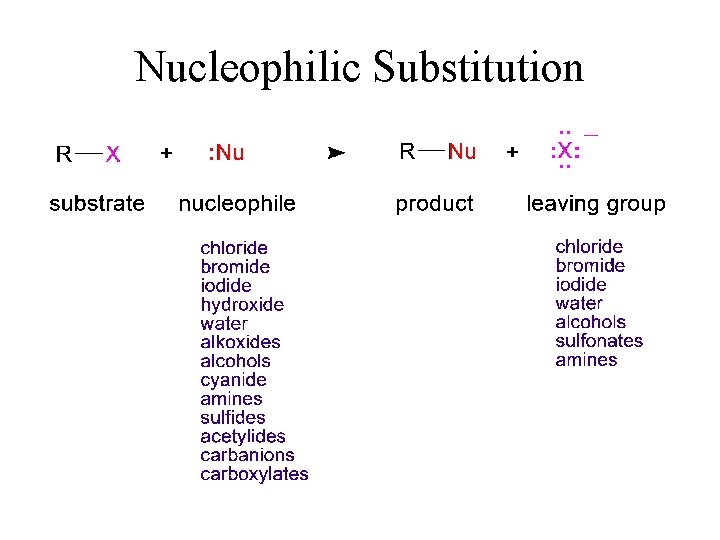
Nucleophilic Substitution

Primary alcohol to produce alkyl halide • primary alcohol react very slow with hydrochloric acid (HCl) because chloride is a weaker nueleophilic than bromide. However Zn. Cl 2 can enhance which Zn. Cl 2 is a powerful lewis acid, known as Lucas reagent.

SN 2 REACTION OF ALCOHOL Primary alcohols undergo SN 2 reactions with hydrogen halides: 16

When HCl is used; SN 2 reaction is slower, but the rate can be increased using Zn. Cl 2 as catalyst. Zn. Cl 2 functions as a Lewis acid that complexes strongly with the lone-pair electrons on oxygen: 17
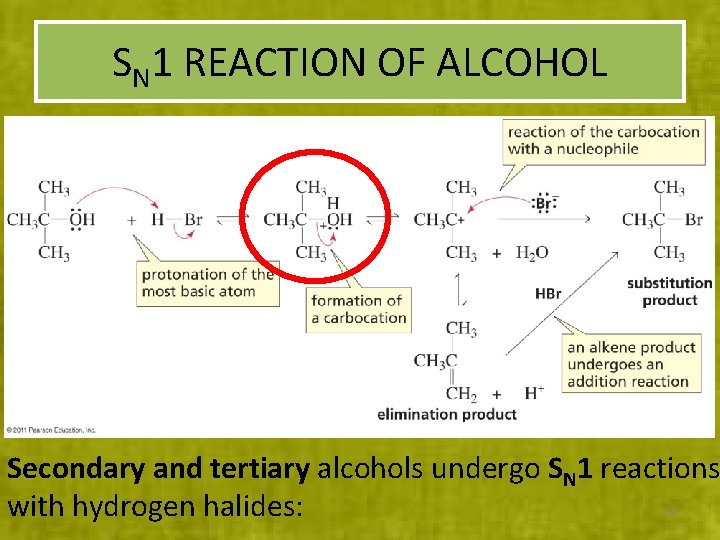
SN 1 REACTION OF ALCOHOL Secondary and tertiary alcohols undergo SN 1 reactions with hydrogen halides: 18
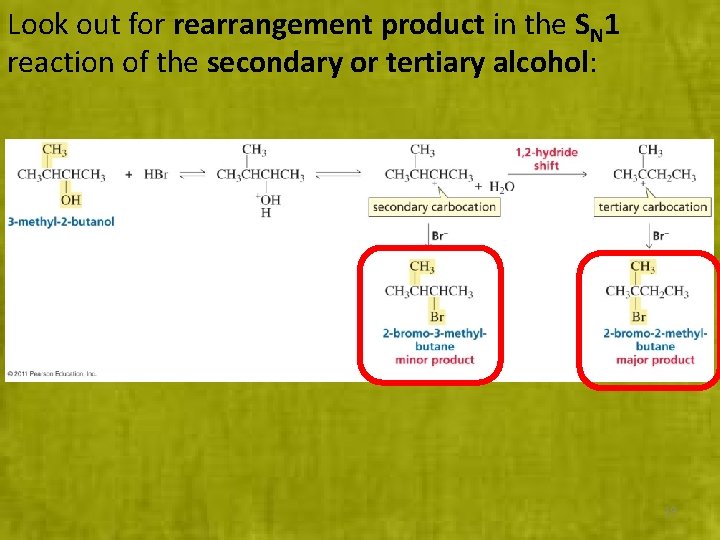
Look out for rearrangement product in the SN 1 reaction of the secondary or tertiary alcohol: 19

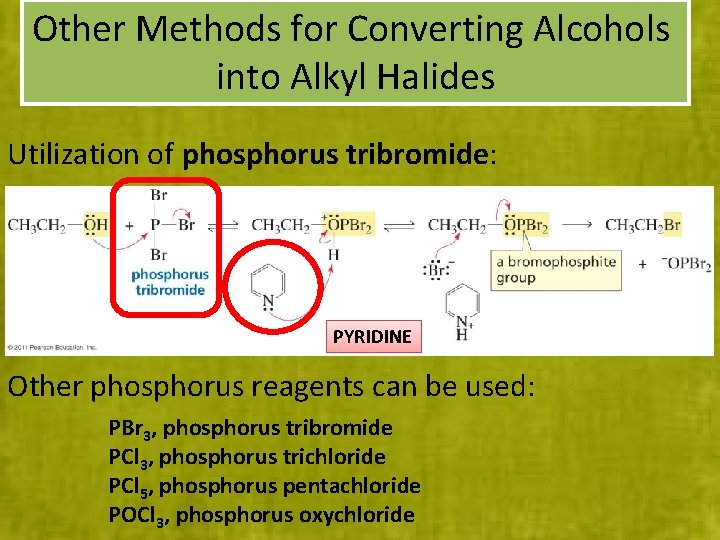
Other Methods for Converting Alcohols into Alkyl Halides Utilization of phosphorus tribromide: PYRIDINE Other phosphorus reagents can be used: PBr 3, phosphorus tribromide PCl 3, phosphorus trichloride PCl 5, phosphorus pentachloride POCl 3, phosphorus oxychloride
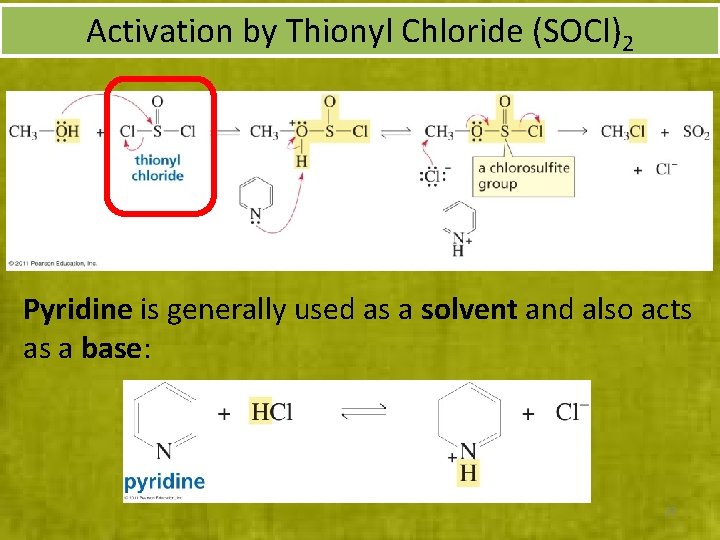
Activation by Thionyl Chloride (SOCl)2 Pyridine is generally used as a solvent and also acts as a base: 22

Summary: Converting of Alcohols to Alkyl Halides Recommended procedures: 23
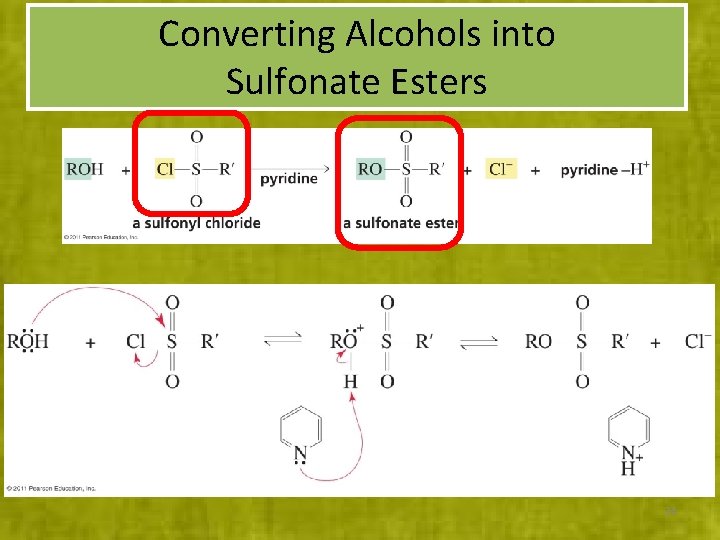
Converting Alcohols into Sulfonate Esters 24
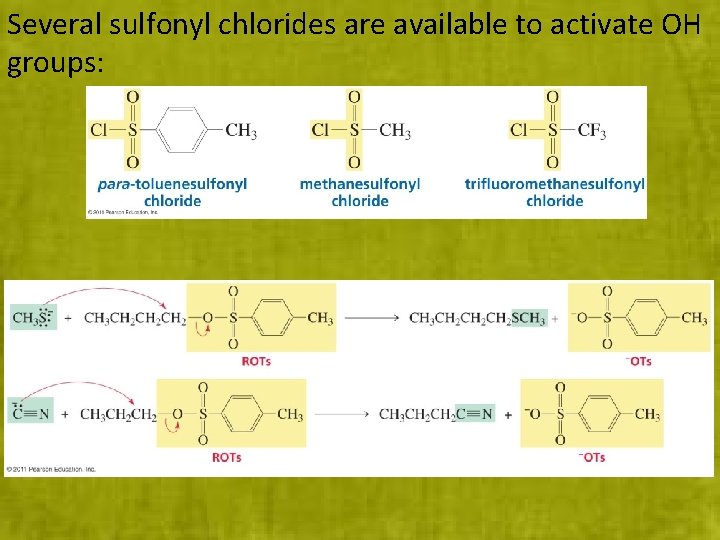
Several sulfonyl chlorides are available to activate OH groups:
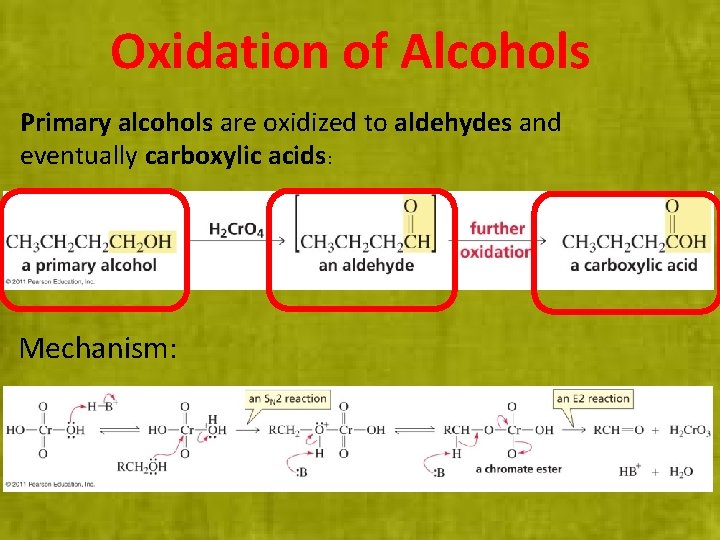
Oxidation of Alcohols Primary alcohols are oxidized to aldehydes and eventually carboxylic acids: Mechanism:
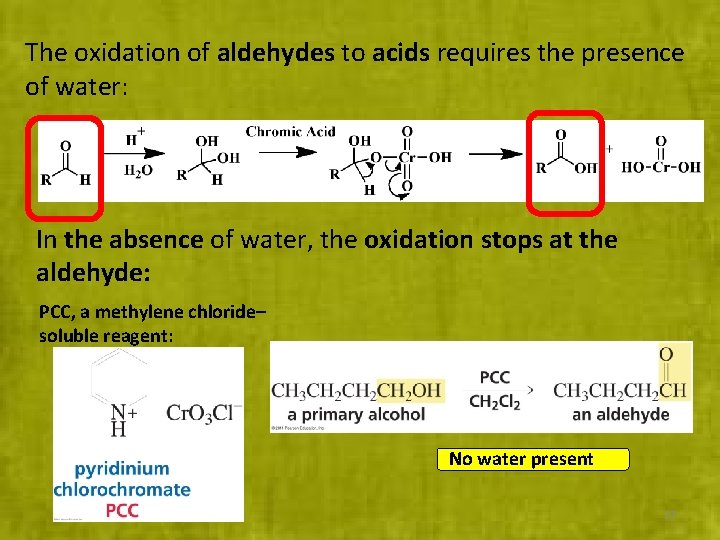
The oxidation of aldehydes to acids requires the presence of water: In the absence of water, the oxidation stops at the aldehyde: PCC, a methylene chloride– soluble reagent: No water present 27
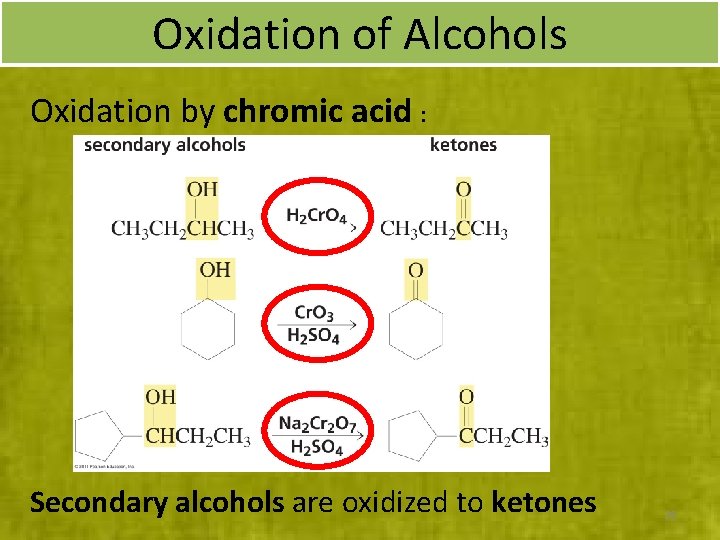
Oxidation of Alcohols Oxidation by chromic acid : Secondary alcohols are oxidized to ketones 28
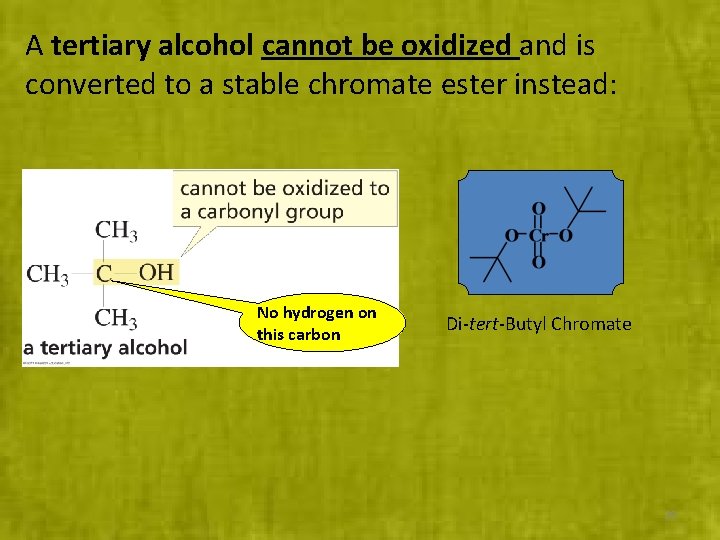
A tertiary alcohol cannot be oxidized and is converted to a stable chromate ester instead: No hydrogen on this carbon Di-tert-Butyl Chromate 29
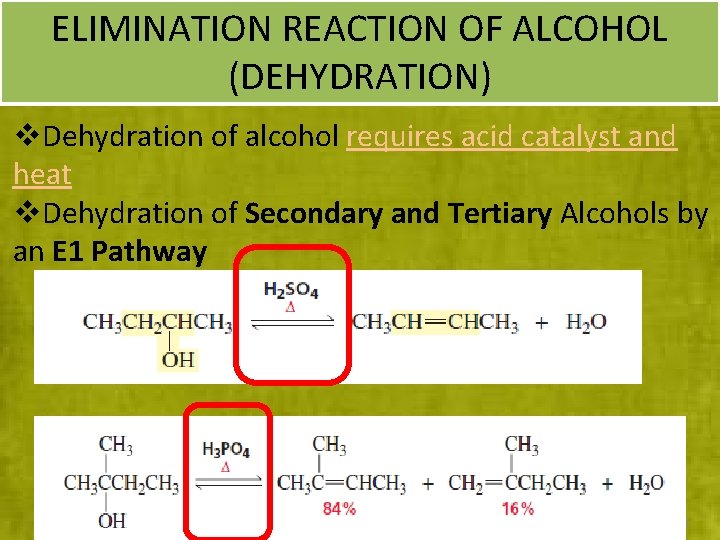
ELIMINATION REACTION OF ALCOHOL (DEHYDRATION) v. Dehydration of alcohol requires acid catalyst and heat v. Dehydration of Secondary and Tertiary Alcohols by an E 1 Pathway 30 30

Mechanism of E 1 Dehydration of an Alcohol
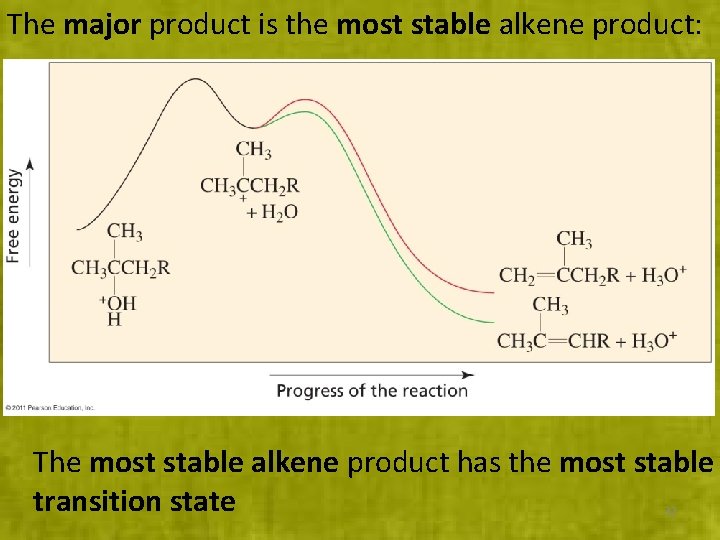
The major product is the most stable alkene product: The most stable alkene product has the most stable transition state 32

The rate of dehydration reflects the ease with which the carbocation is formed: 33

Look out for carbocation rearrangement: 34
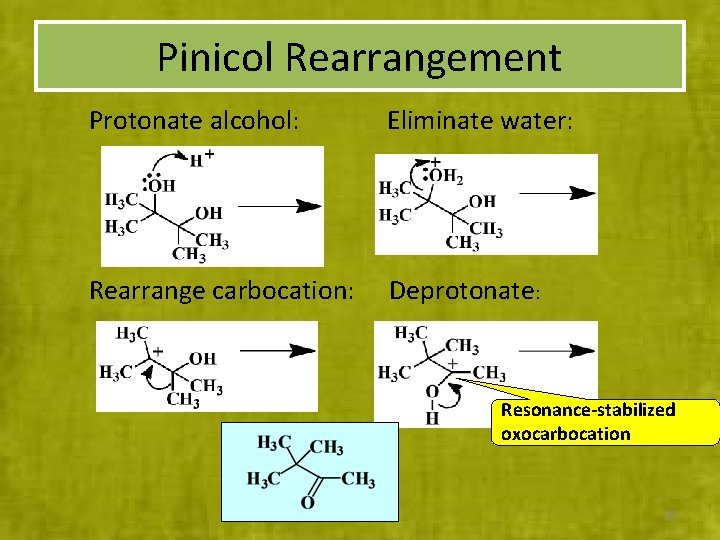
Pinicol Rearrangement Protonate alcohol: Eliminate water: Rearrange carbocation: Deprotonate: Resonance-stabilized oxocarbocation 35
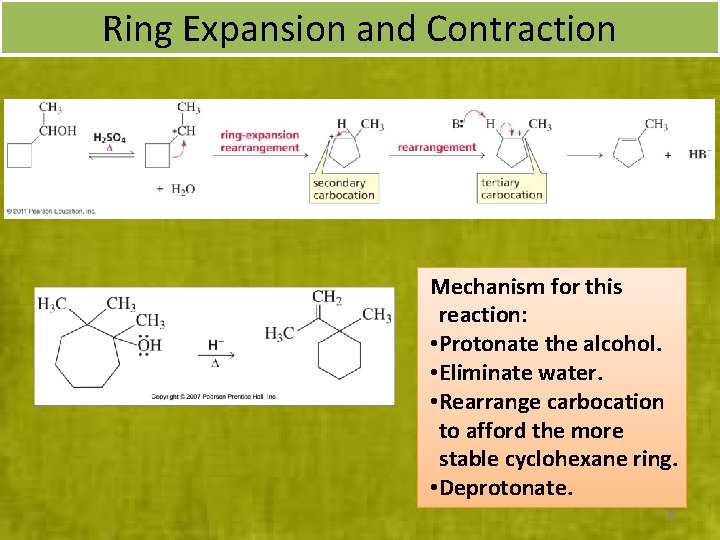
Ring Expansion and Contraction Mechanism for this reaction: • Protonate the alcohol. • Eliminate water. • Rearrange carbocation to afford the more stable cyclohexane ring. • Deprotonate. 36
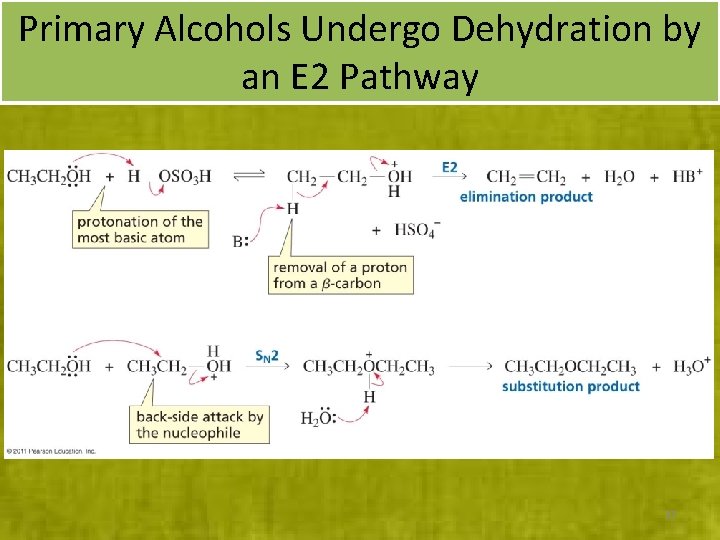
Primary Alcohols Undergo Dehydration by an E 2 Pathway 37
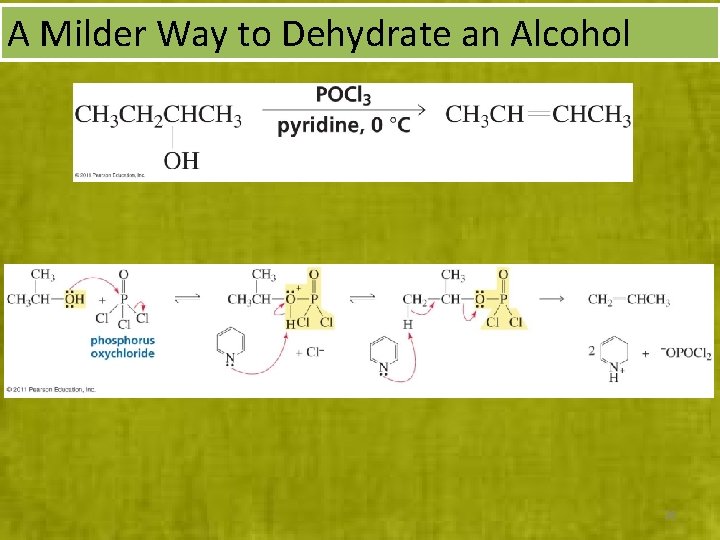
A Milder Way to Dehydrate an Alcohol 38

ETHER

Nucleophilic substitution reaction of Ethers, like alcohols, can be activated by protonation: Ether can undergo nucleophilic substitution with HBr and HI only (HCl cannot be used because Cl- too poor nucleophile
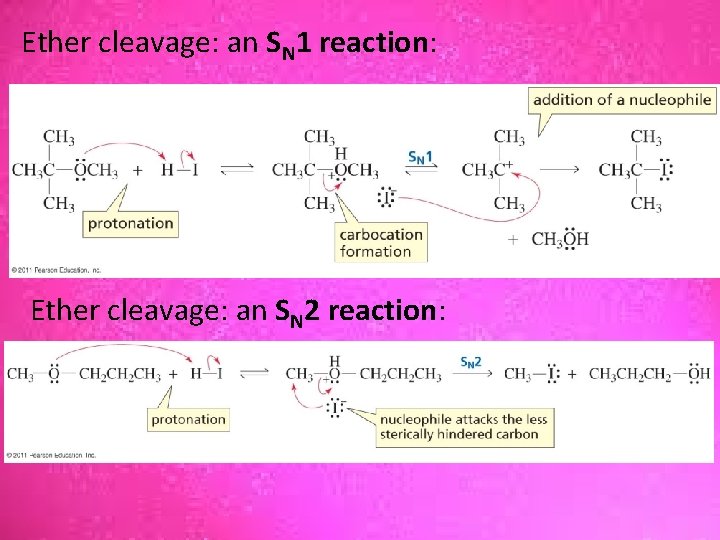
Ether cleavage: an SN 1 reaction: Ether cleavage: an SN 2 reaction:

Reagents such as SOCl 2 and PCl 3 can activate alcohols but not ethers Ethers are frequently used as solvents because only they react with hydrogen halides 42
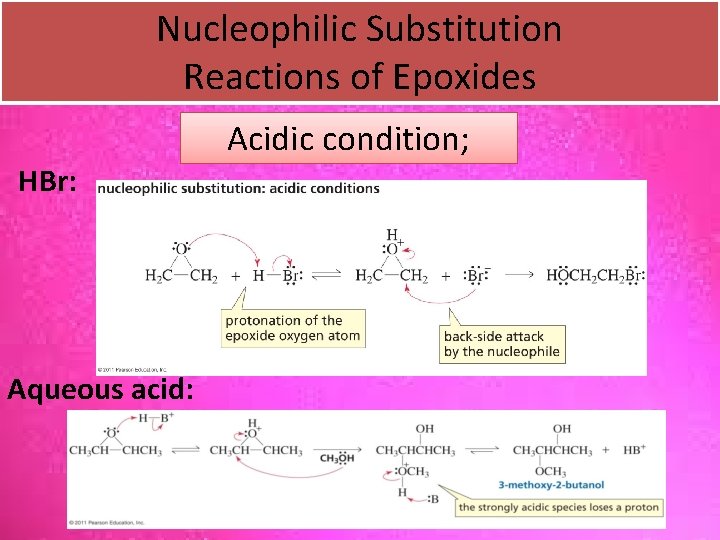
Nucleophilic Substitution Reactions of Epoxides Acidic condition; HBr: Aqueous acid:
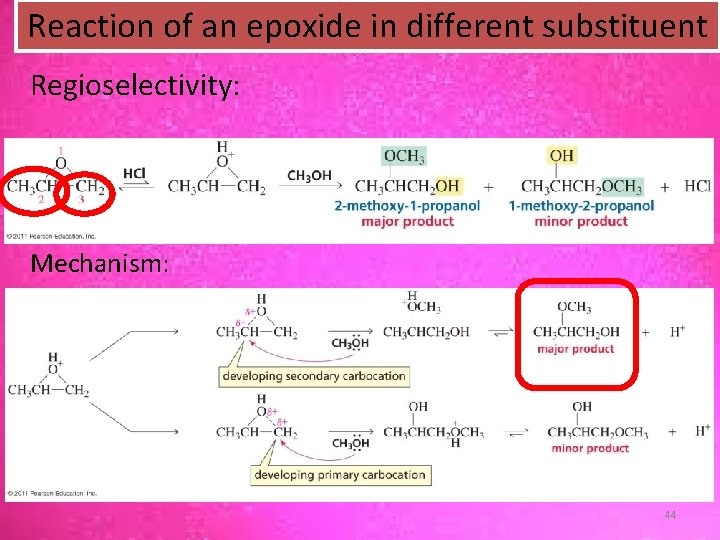
Reaction of an epoxide in different substituent Regioselectivity: Mechanism: 44

Neutral or Basic condition: When a nucleophile attacks an unprotonated epoxide, the reaction is a pure SN 2 reaction: Therefore: 45
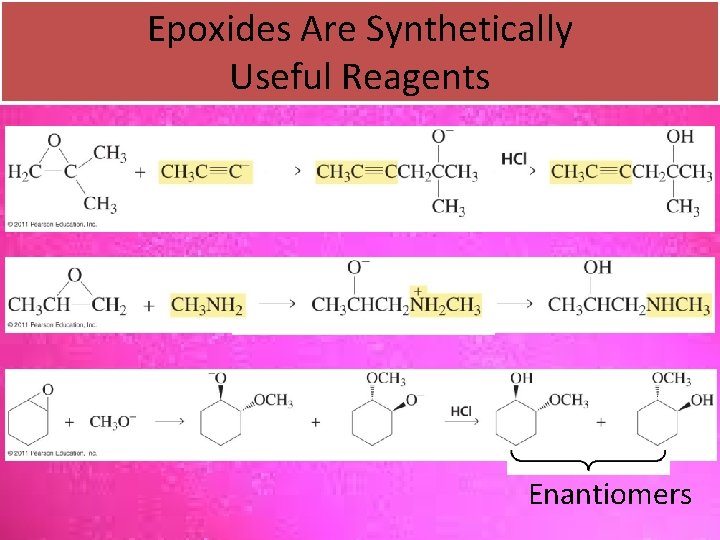
Epoxides Are Synthetically Useful Reagents Enantiomers

CLASS EXERCISE 2 Give the major product of the following reactions:

THE END
- Slides: 48