Alberta Health Services Provincial Picture 1 Alberta Blood

Alberta Health Services Provincial Picture

1. Alberta Blood Office (ABO) and CTR – On hold with recent provincial budget changes 2. Provincial recycling of PCC – all outdating product recycled into either Edmonton or Calgary with complete utilization • All jurisdictions except Calgary zone follow NAC recommendations 3. Provincial TM website live April 1 st – standardized TM documents, algorithms and clinical resources • http: //www. albertahealthservices. ca/Lab. Services/wf-lab-clin-tmtrxn-algrthm. pdf – Patient resources hosted on a secondary public site

3. Province wide stakeholder committee for reversal of NOAC (ER, Neuro, Surg, Anesth, Hem, DI, Crit Care) 1. DTIs & major bleeding • • Consider PCC 50 u/kg or FEIBA 50 iu/kg If no PCC/FEIBA to be given then Tx. A (10 mg/kg IV) 2. anti-Xa & major bleeding • • Consider PCC 50 u/kg If no PCC to be given then Tx. A (10 mg/kg IV) 3. For both categories and moderate bleeding – recommending only Tx. A

AHS- Calgary Zone M. Taher Rad

Switching to CMV-Safe Blood Products (1) • 1. Currently all adult hematology-oncology and BMT patients (excluding patients on purine analogs) will receive CMV Safe-Non-Irradiated blood components. • 2. Patients on purine analog medication will get CMV Safe-Irradiated blood components. Here is the final list of drugs for irradiated product needs (from literature review and physician input): Ø Ø Ø Ø Ø ATG-anti-thymocyte globulin Bendamustine Clofarabine Alemtuzumab Deoxycoformicin Fludarabine Cladribine Pentostatin Busulfan

Switching to CMV-Safe blood products (2) • Non-Haematological oncology patients will be transfused with routine blood components "CMV-Safe/Non-irradiated". • Our policy regarding transfusion of paediatric oncology patients as well as neonates requiring CMV-Negative-Irradiated blood components has not been changed and we will continue to provide those patients with CMV-Negative-Irradiated blood components at this stage. • Solid organ transplant patients will receive CMV Safe-Non-Irradiated blood components otherwise there is specific request by attending physicians for CMV-Negative/irradiated blood components, however these patients are required to be Donor CMV-Neg/Recipient CMV-Neg (D-/R-) supported by a recent serological testing. • We will continue to carry 5 -10% inventory of CMV-Neg-Irradiated platelets for immune deficient and "just in case" patients. We don't have on site irradiator which creates some limitations.

SCM-TM Redesign Project • For TM Order sets, we have created an interactive environment within HIS (SCM). • Important medications correlated to blood components has been linked to blood order set. Ø Linking Purine analog drugs the only available option for patient with this medication will be Irradiated blood components Ø Warfarin, Coumadin, Tranexamic acid and Dabigatran will be linked to PCC. Ø Aspirin, NSAID and Anti-platelet will be linked to Platelet order set.
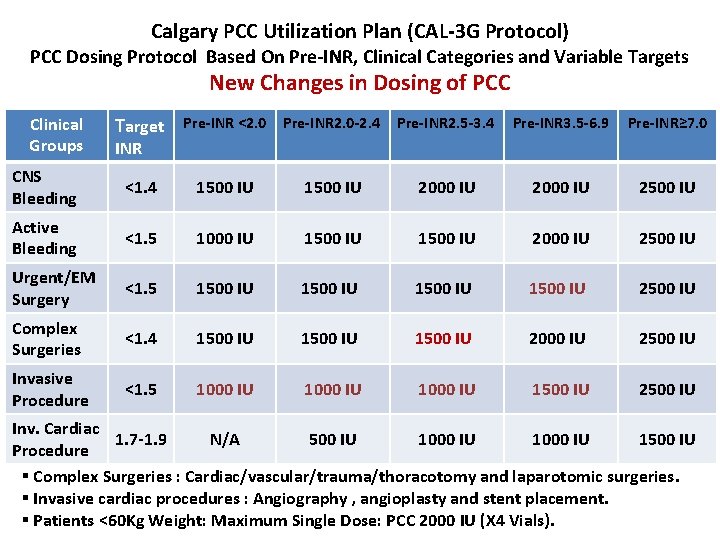
Calgary PCC Utilization Plan (CAL-3 G Protocol) PCC Dosing Protocol Based On Pre-INR, Clinical Categories and Variable Targets New Changes in Dosing of PCC Target Pre-INR <2. 0 Pre-INR 2. 0 -2. 4 Pre-INR 2. 5 -3. 4 Pre-INR 3. 5 -6. 9 Pre-INR≥ 7. 0 CNS Bleeding <1. 4 1500 IU 2000 IU 2500 IU Active Bleeding <1. 5 1000 IU 1500 IU 2000 IU 2500 IU Urgent/EM Surgery <1. 5 1500 IU 2500 IU Complex Surgeries <1. 4 1500 IU 2000 IU 2500 IU Invasive Procedure <1. 5 1000 IU 1500 IU 2500 IU Clinical Groups INR Inv. Cardiac 1. 7 -1. 9 N/A 500 IU 1000 IU 1500 IU Procedure § Complex Surgeries : Cardiac/vascular/trauma/thoracotomy and laparotomic surgeries. § Invasive cardiac procedures : Angiography , angioplasty and stent placement. § Patients <60 Kg Weight: Maximum Single Dose: PCC 2000 IU (X 4 Vials).

Audit on Fibrinogen – NAC email • 95% of Fibrinogen concentrate has been utilized was due to three patients with congenital fibrinogen disorders ØOne case of Afibrinogenemia ØTwo cases of Dysfibrinogenemia • Clinical indication: ØSame as very where else? ? ØNo concentrated fibrinogen used for acute acquired hypofibrinogenemia

AHS- Edmonton Zone S. Nahirniak

EZ updates • IVIG changes – Gammagard Liquid = 29% of our product utilization • Hereditary Angioedema patients – Two patients in ICU recently • Blood “safe” fridges – Approval to move forward with purchase for Outpatient clinic and PICU – Request from STARs to place in their bases • Blood Product costing study – NAIT, AHS and AH collaboration due to start towards end of May 2013
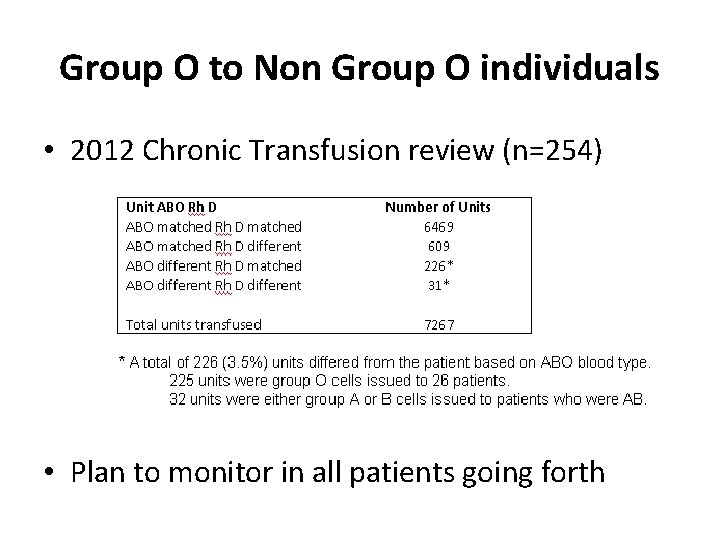
Group O to Non Group O individuals • 2012 Chronic Transfusion review (n=254) • Plan to monitor in all patients going forth
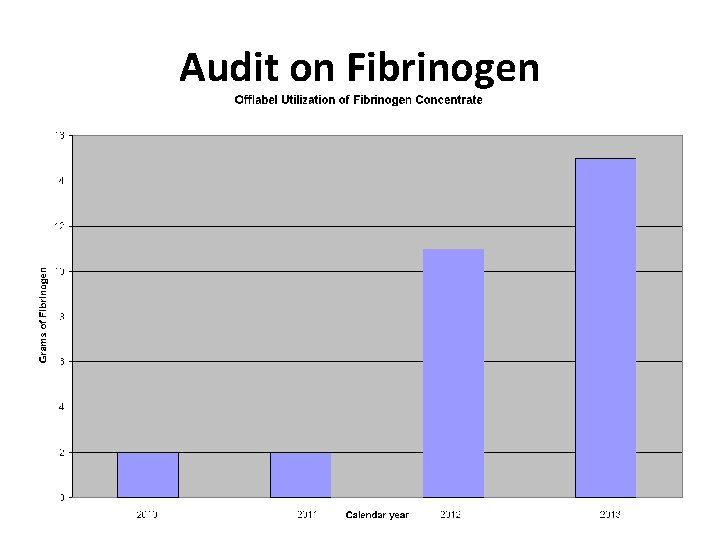
Audit on Fibrinogen
- Slides: 13