AL Amyloidosis Dr John Quinn Beaumont HospitalRCSI Case

AL Amyloidosis Dr. John Quinn Beaumont Hospital/RCSI

Case Presentation � 48 year old female � May 2013 � 18 month history � Fatigue, weight loss, tongue swelling, ankle swelling � GP – urine – 4+ proteinuria � Referred to Nephrology Clinic � Cr 197

Case Presentation � TUP in 24 hours – 5. 6 g � Albumin 19 � Raised Cholesterol Diagnosis: Nephrotic Syndrome

Case Presentation � FBC – NAD � SPEP and UPEP – small lambda paraprotein � Skeletal survey – no lytic disease � BMA – 5% plasma cells � Trephine – 5 -10% plasma cells � Serum free light chain ratio: Lambda 342: Kappa 44. 8 � Ratio: 0. 13 � Echo – NAD � NTpro. BNP and Tn. I - Normal

Case Presentation � Renal Biopsy – Amyloidosis – Likely AA? � Biopsy sent to NAC at Royal Free Hospital � Diagnosis confirmed as AL Amyloidosis � Initial Treatment: Cyclophosphamide and Dexamethasone � Plan to add 3 rd drug depending on tolerability

Case Presentation � Good symptomatic response to dexamethasone � Discharged – but readmitted 4 days later � Collapsed every time she stood up � Severe autonomic neuropathy � Therefore, avoid bortezomib � Added increasing doses of fludrocortisone - mineralocorticoid � Subsequently midodrine 2. 5 mg tds – alpha-1 -receptor agonist

Case Presentation � Allowed cautious diuresis � Thalidomide 50 mg added – 1 month after diagnosis � Warfarin thromboprophylaxis � 6 weeks following diagnosis – albumin 17 g/dl, TUP 6. 8 g/24 hours, creatinine 190 � BP lying 103/64 � BP standing 63/34 � Wheelchair-dependent

Case Presentation � 8 weeks after diagnosis: Normal SFLCr 1. 6 � Starting to manage day leave � BP-stabilising � Discharged � F/up in day ward � Completed a total of 4 cycles of CTD as complete clonal response � Only now fit to travel to NAC in UK � SAP scan – liver + spleen amyloid only

Case Presentation � Gradual improvement in symptoms over 6 months � Meds gradually withdrawn � Now 2. 5 years post diagnosis � Albumin 37 � Cr 109 � Normal SFLCr � Off all meds � Asymptomatic, working full time

Amyloidosis � A protein problem! � Increasing number of diagnoses � Cardiac MRI � Serum Free Light Chain Test � Survival improving for patients with AL Amyloidosis
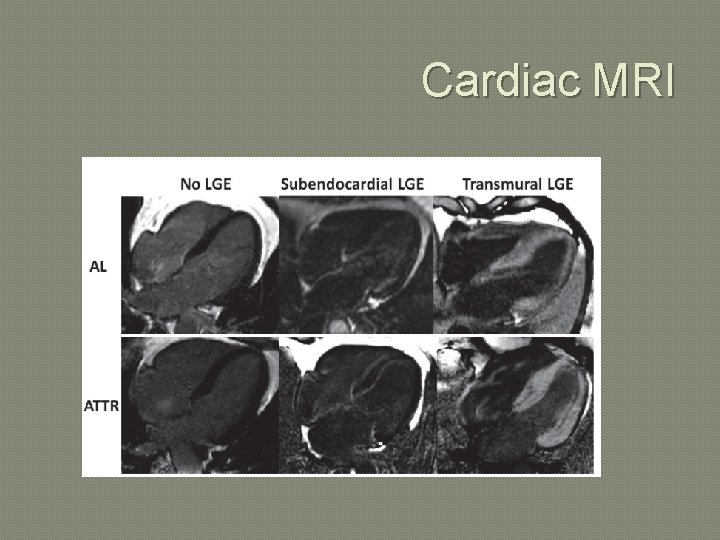
Cardiac MRI
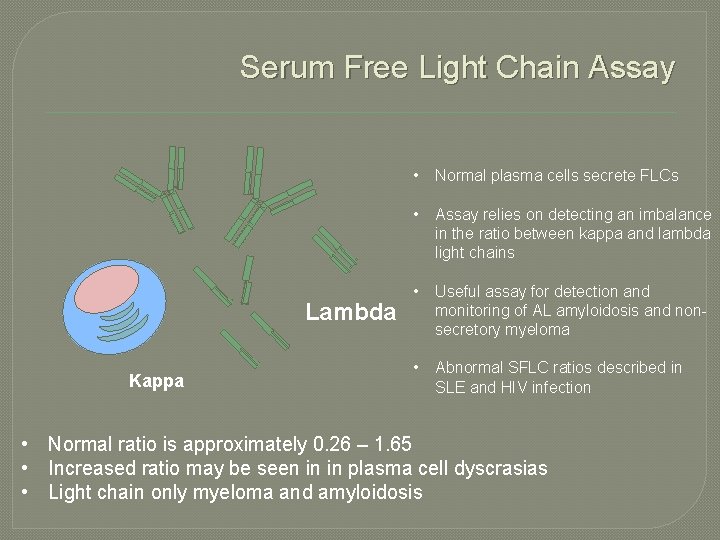
Serum Free Light Chain Assay Lambda Kappa • Normal plasma cells secrete FLCs • Assay relies on detecting an imbalance in the ratio between kappa and lambda light chains • Useful assay for detection and monitoring of AL amyloidosis and nonsecretory myeloma • Abnormal SFLC ratios described in SLE and HIV infection • Normal ratio is approximately 0. 26 – 1. 65 • Increased ratio may be seen in in plasma cell dyscrasias • Light chain only myeloma and amyloidosis
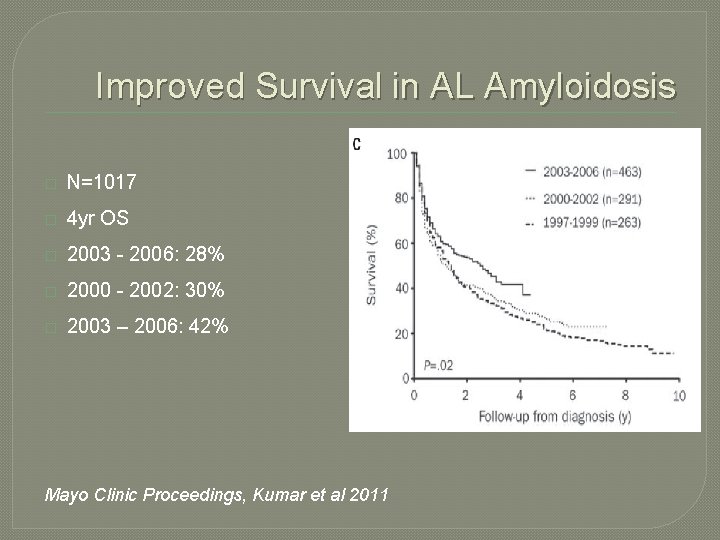
Improved Survival in AL Amyloidosis � N=1017 � 4 yr OS � 2003 - 2006: 28% � 2000 - 2002: 30% � 2003 – 2006: 42% Mayo Clinic Proceedings, Kumar et al 2011

Introduction � Amyloidosis is a rare systemic disorder � Mis-folding of aberrant precursor proteins � Unstable aggregates in a Beta-pleated structure � Fibrils are deposited in organs affecting structure and function � The unstable protein may be hereditary or acquired

Introduction � AL amyloidosis is most common � Amyloidogenic protein is a monoclonal light chain secreted by an underlying plasma cell dyscrasia � Amyloidosis caused by deposition of misfolded transthyretin is next most common � Others include misfolding of lysozyme and gelsolin � Localised AL amyldosis – amyloid deposits at a single site – bladder, skin, larynx, lung
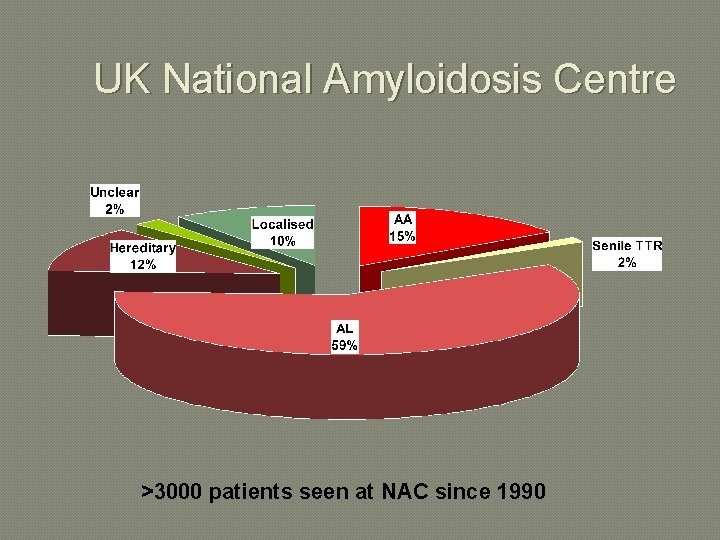
UK National Amyloidosis Centre >3000 patients seen at NAC since 1990
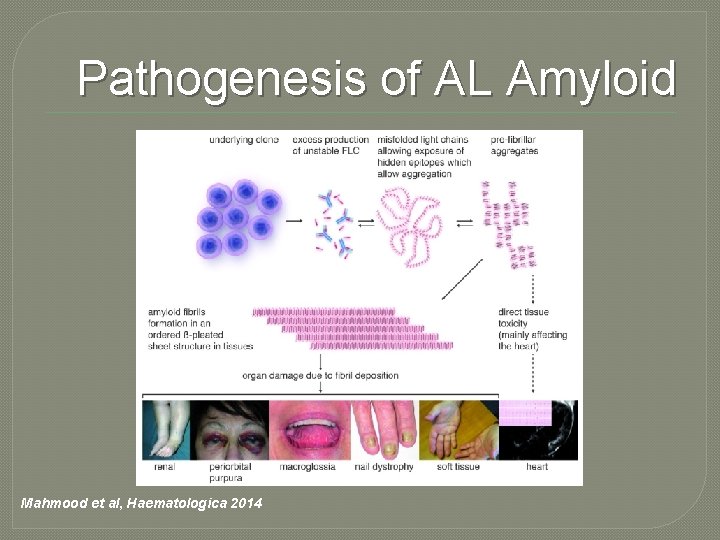
Pathogenesis of AL Amyloid Mahmood et al, Haematologica 2014
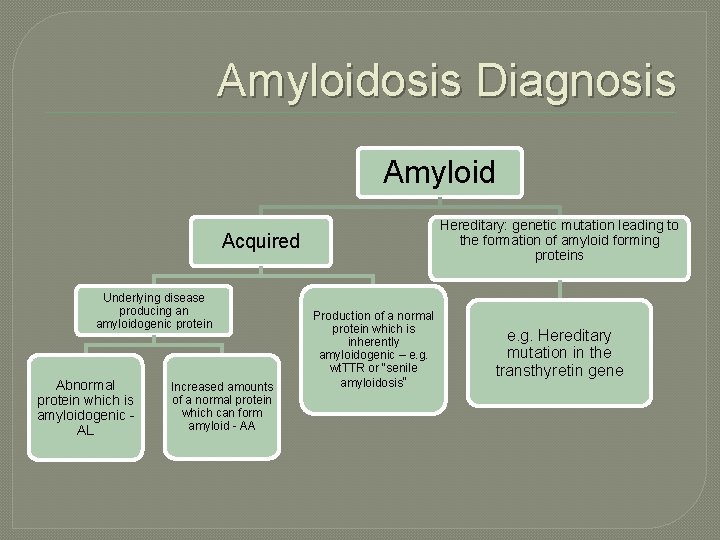
Amyloidosis Diagnosis Amyloid Hereditary: genetic mutation leading to the formation of amyloid forming proteins Acquired Underlying disease producing an amyloidogenic protein Abnormal protein which is amyloidogenic AL Increased amounts of a normal protein which can form amyloid - AA Production of a normal protein which is inherently amyloidogenic – e. g. wt. TTR or “senile amyloidosis” e. g. Hereditary mutation in the transthyretin gene

Amyloid deposits Amyloid fibrils + Heparan/dermatan sulphate + Serum amyloid P component (SAP)

When to suspect Amyloidosis Symptoms are often non-specific! � Lethargy, fatigue � Weight loss � Peripheral oedema � Unexplained heart failure � Alternating diarrhoea/constipation � Peripheral/autonomic neuropathy � Postural Hypotension � Purpura � Macroglossia
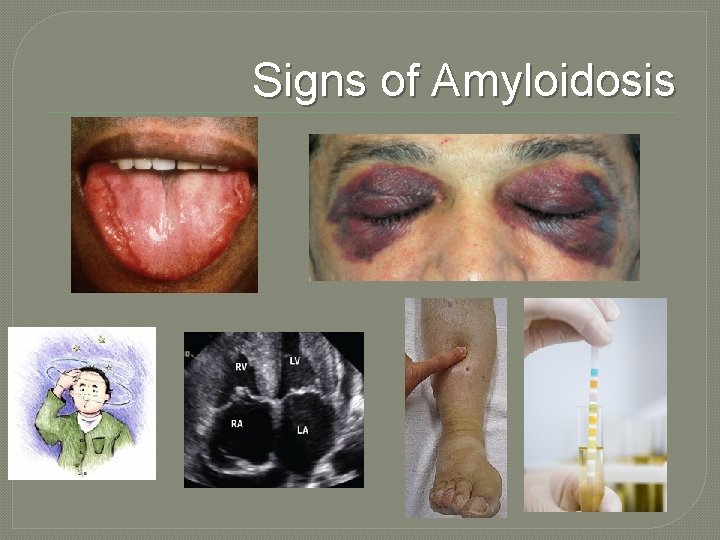
Signs of Amyloidosis
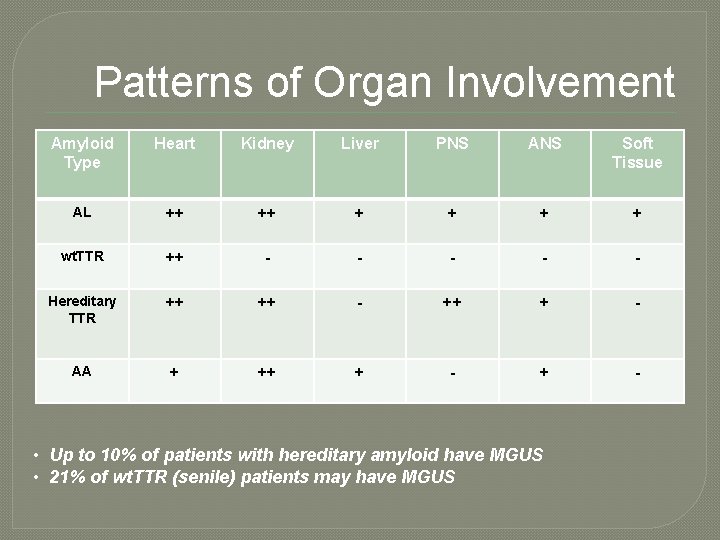
Patterns of Organ Involvement Amyloid Type Heart Kidney Liver PNS ANS Soft Tissue AL ++ ++ + + wt. TTR ++ - - - Hereditary TTR ++ ++ - ++ + - AA + ++ + - • Up to 10% of patients with hereditary amyloid have MGUS • 21% of wt. TTR (senile) patients may have MGUS

Diagnosis

Diagnosing AL Amyloidosis �Confirm presence of Amyloid – this requires a biopsy �Confirm AL Amyloid and not another subtype – this requires considerable expertise �If AL Amyloid confirmed – need plasma cell disorder work-up and to exclude Myeloma and Systemic W/Up
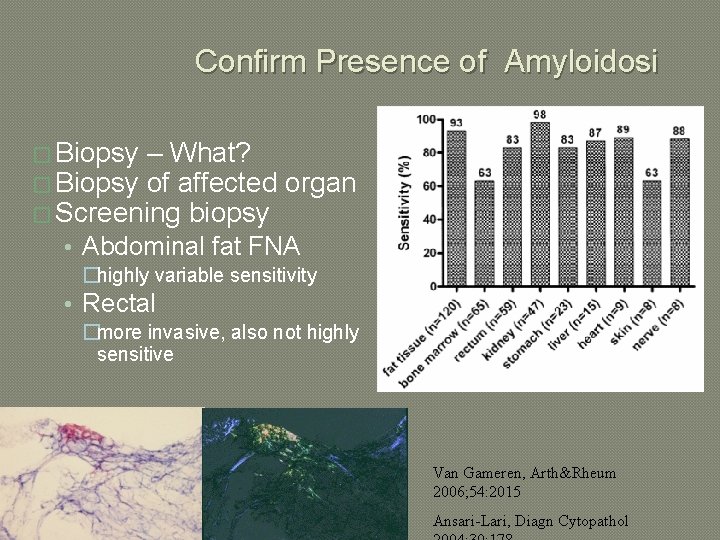
Confirm Presence of Amyloidosi � Biopsy – What? � Biopsy of affected organ � Screening biopsy • Abdominal fat FNA �highly variable sensitivity • Rectal �more invasive, also not highly sensitive Van Gameren, Arth&Rheum 2006; 54: 2015 Ansari-Lari, Diagn Cytopathol

Confirm AL Subtype � Essential � NB – Refer Case to NAC in London � Mass Spectrometry � Immuno-electromicroscopy � Immunohistochemistry � DNA Analysis NEJM 2002
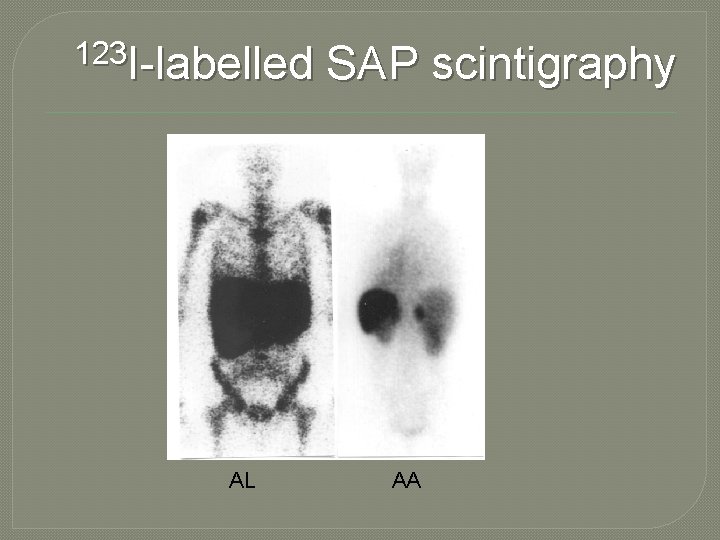
123 I-labelled AL SAP scintigraphy AA

Plasma Cell Disorder Work-Up � FBC + Biochem + CPM � Albumin � SPEP � UPEP � SFLCr � 24 Hour TUP � BMA + Biopsy � Skeletal Survey � CT if Ig. M paraprotein � Staging Investigations: NT-Pro. BNP � Echo +/- Cardiac MRI � US abdomen if ? liver involvement � Nerve conduction studies � SAP scan at NAC and Troponin T
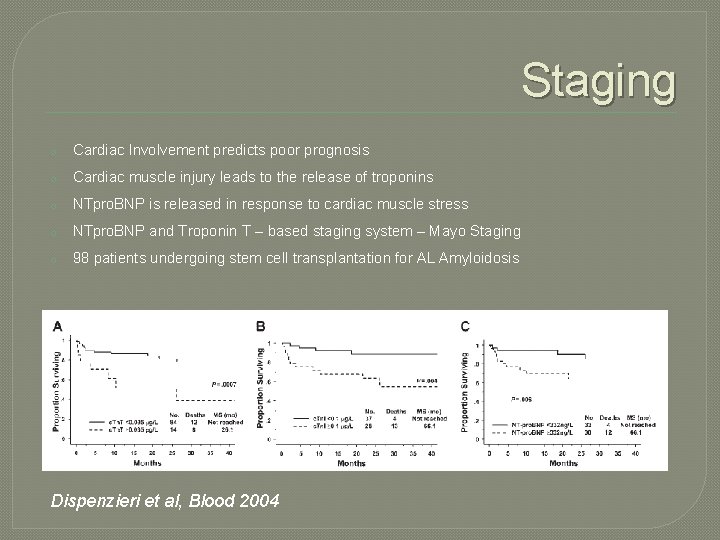
Staging o Cardiac Involvement predicts poor prognosis o Cardiac muscle injury leads to the release of troponins o NTpro. BNP is released in response to cardiac muscle stress o NTpro. BNP and Troponin T – based staging system – Mayo Staging o 98 patients undergoing stem cell transplantation for AL Amyloidosis Dispenzieri et al, Blood 2004
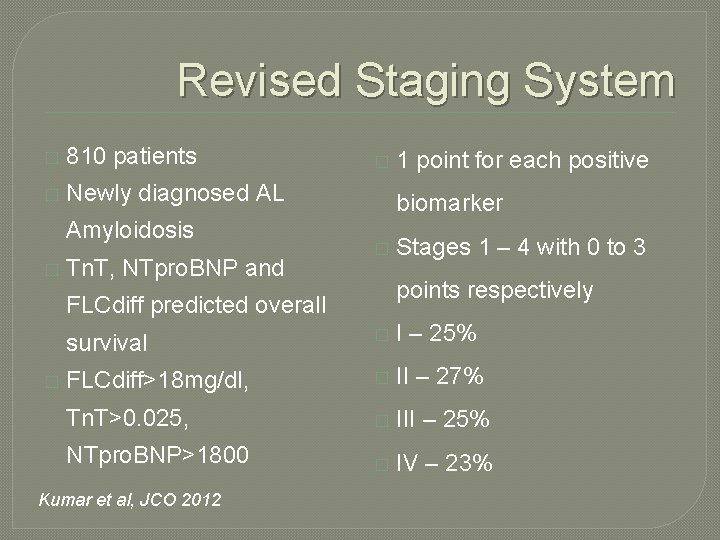
Revised Staging System � 810 patients � Newly diagnosed AL Amyloidosis � Tn. T, NTpro. BNP and � biomarker � Stages 1 – 4 with 0 to 3 points respectively FLCdiff predicted overall � 1 point for each positive survival � I – 25% FLCdiff>18 mg/dl, � II – 27% Tn. T>0. 025, � III – 25% NTpro. BNP>1800 � IV – 23% Kumar et al, JCO 2012
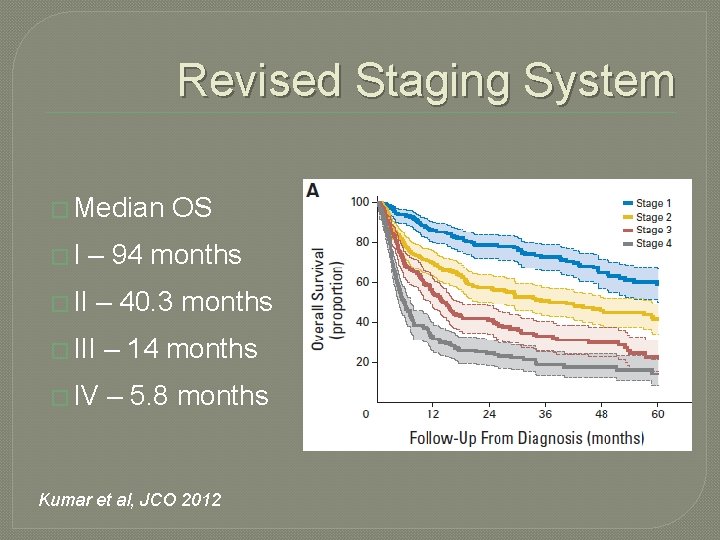
Revised Staging System � Median �I OS – 94 months � II – 40. 3 months � III – 14 months � IV – 5. 8 months Kumar et al, JCO 2012
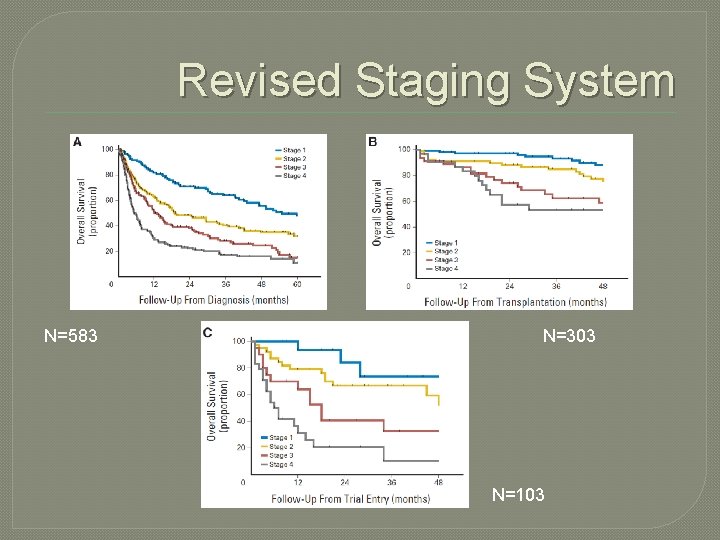
Revised Staging System N=583 N=303 N=103

Treating AL Amyloidosis Reduce the number of clonal plasma cells Reduce the production of abnormal light chains Slow or stop production of new amyloid Gradual regression of existing amyloid deposits

NEJM, 2007
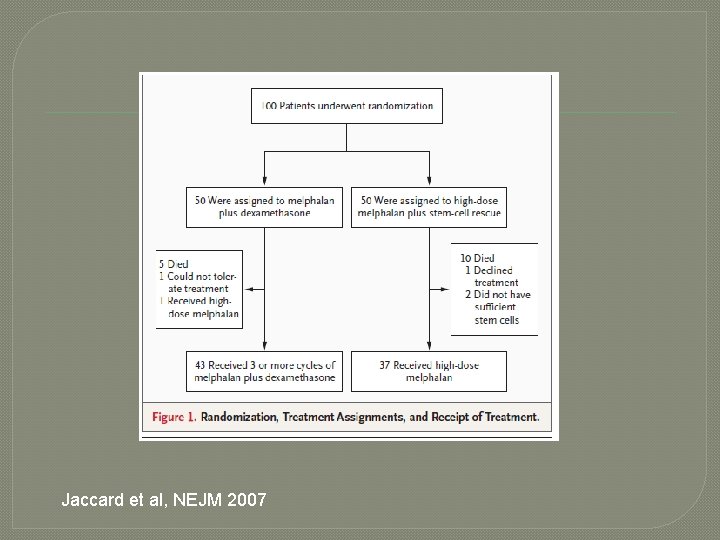
Jaccard et al, NEJM 2007

Jaccard et al, NEJM 2007 � 29/37 patients who received HDT survived to >3 months post-auto �Haematologic response rates were 68% for MD and 66% for HDT �Median time to progression was similar in both groups
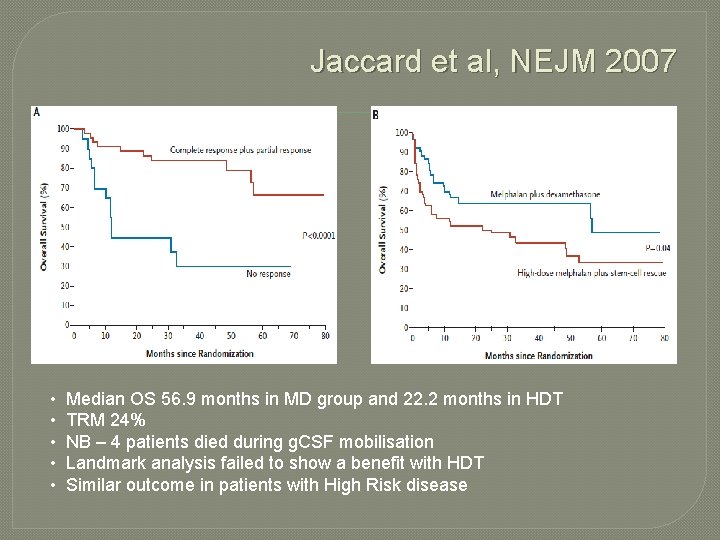
Jaccard et al, NEJM 2007 • • • Median OS 56. 9 months in MD group and 22. 2 months in HDT TRM 24% NB – 4 patients died during g. CSF mobilisation Landmark analysis failed to show a benefit with HDT Similar outcome in patients with High Risk disease
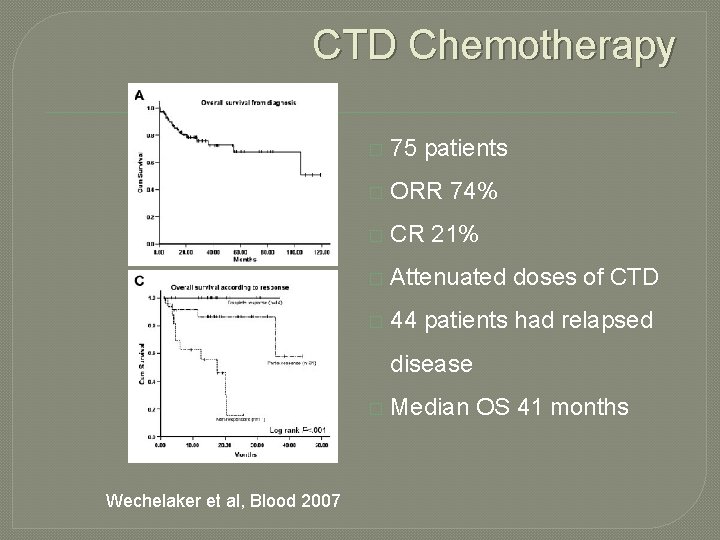
CTD Chemotherapy � 75 patients � ORR 74% � CR 21% � Attenuated doses of CTD � 44 patients had relapsed disease � Wechelaker et al, Blood 2007 Median OS 41 months
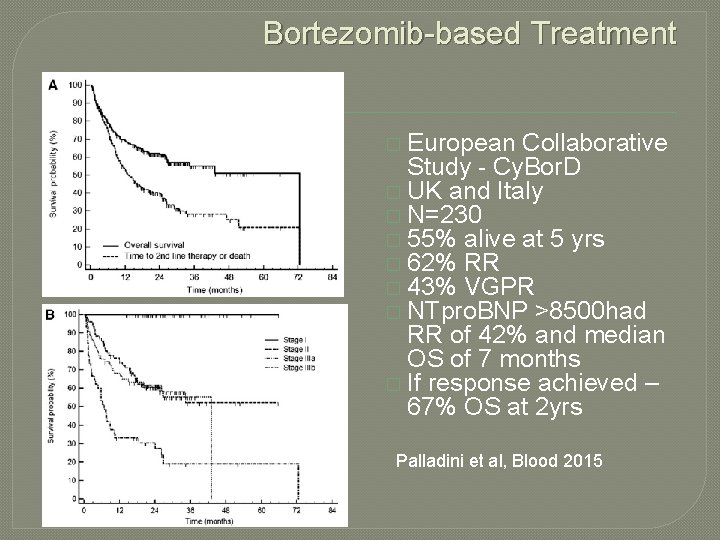
Bortezomib-based Treatment � European Collaborative Study - Cy. Bor. D � UK and Italy � N=230 � 55% alive at 5 yrs � 62% RR � 43% VGPR � NTpro. BNP >8500 had RR of 42% and median OS of 7 months � If response achieved – 67% OS at 2 yrs Palladini et al, Blood 2015
Mahmood S et al. Haematologica 2014; 99: 209 -221

Response Assessment
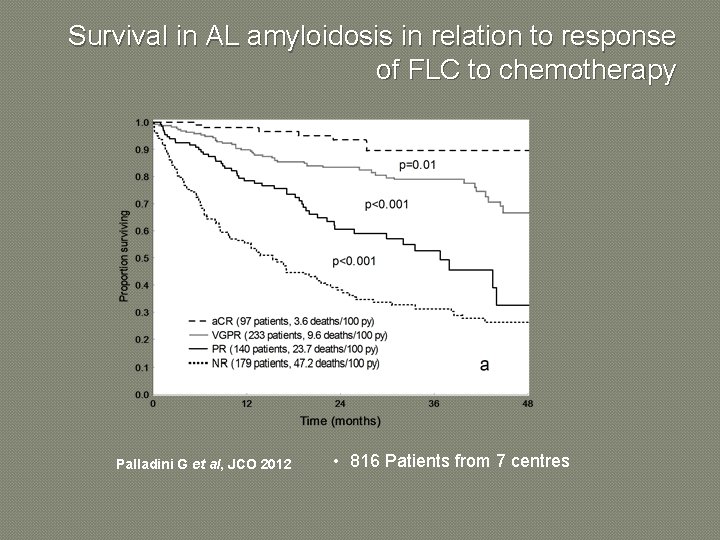
Survival in AL amyloidosis in relation to response of FLC to chemotherapy Palladini G et al, JCO 2012 • 816 Patients from 7 centres

Future Treatments

Future Treatments �Effective treatment reduces production of precursor protein �However regression of preformed amyloid deposits is very slow or does not occur at all � 65% of Amyloid Cases are AL � 20% of patients die within 6 months of diagnosis before delayed effects are realised

�Aim of studies was to stimulate normal phagocytic clearance mechanisms �Pre-treat with CPHPC �Mops up circulating SAP �Then administer humanised monoclonal Ig. G 1 anti-SAP antibody �Activate macrophage destruction of Sapcontaining amyloid deposits

�All amyloid deposits contain SAP �Normal plasma glycoprotein �Binding of SAP to amyloid fibrils is reversible

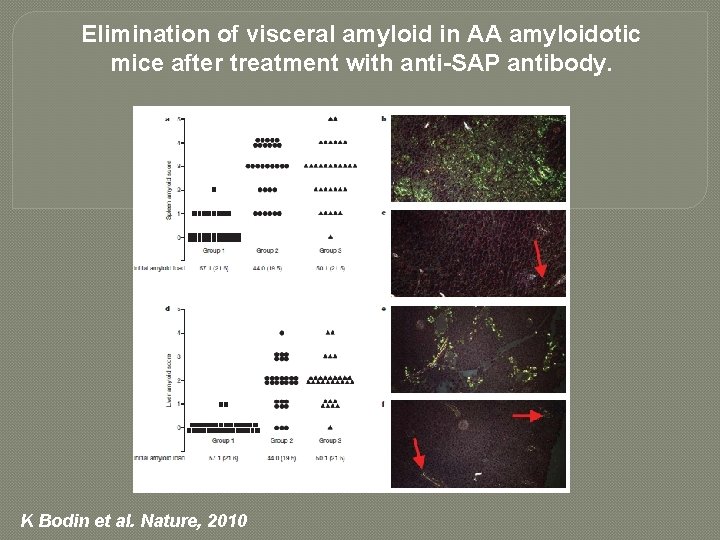
Elimination of visceral amyloid in AA amyloidotic mice after treatment with anti-SAP antibody. K Bodin et al. Nature, 2010


NEJM 2015 � 16 patients � Patients with clinical evidence of cardiac involvement or clinically significant renal or liver involvement were excluded � 3 day infusion of CPHPC � Single � 2 dose of anti-SAP Ma. B AA, 8 AL, 6 other Amyloid

NEJM 2015 �No SAEs �Most patients had mild IRRs which were manageable �Patients who received higher dose of the antibody showed evidence of an acute response �After 42 days � 6/8 patients with liver involvement had reduced liver stiffness measured using a fibroscan

NEJM 2015 � 4 patients had substantial reductions in liver amyloid by SAP-imaging � 1 patient had reduction in renal amyloid on SAP-imaging �Next trial – include patients with significant cardiac and renal dysfunction
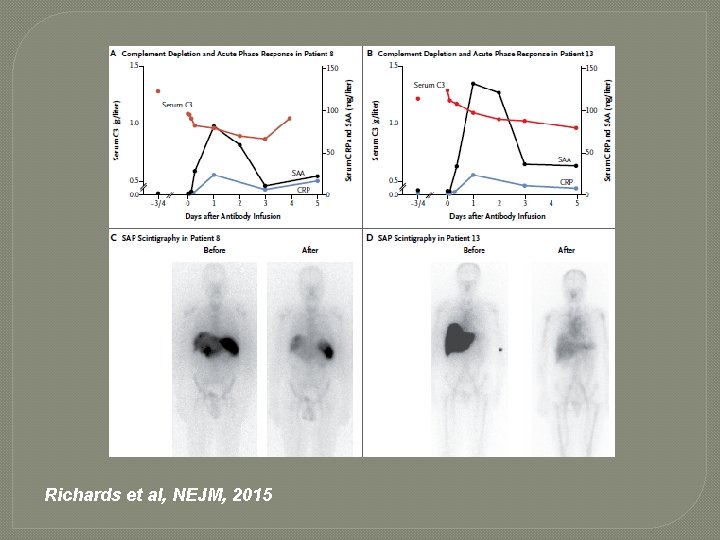
Richards et al, NEJM, 2015

Case Study � 74 yr old man �Previously well �Increasing SOB x 9 months �Extensive investigations �Cardiac MRI – LGE consistent with Amyloid �Referred for evaluation

Case Study � Mild renal impairment � Small lambda paraprotein in serum � SFLCr � BM � No – Lambda 670/Kappa 30 – 20% plasma cells lytic disease in bones � Not anaemic � Next Test? ?

Case Study �Fat pad aspirate – negative for amyloid �Trephine – no amyloid �NTpro. BNP �Tn. I – 0. 25 �Next test? ? – 6415

Conclusions �More amyloid cases diagnosed �Better outcomes with newer agents and supportive care �Cardiac �Follow disease predicts survival disease with LC assay �Individualised treatment approach
- Slides: 57