AL Amyloidosis and renal complications Alex Legg Ph



















- Slides: 53

AL Amyloidosis and renal complications Alex Legg Ph. D Scientific Affairs Manager The Binding Site alex. legg@bindingsite. com Distributor in Poland BIOKOM beata. olsz@biokom. com. pl

Why are FLCs associated with kidney disease? In plasma cell dyscrasias toxic monoclonal FLCs are produced: Light chain physico-chemical properties organisation of light chain aggregates Location of deposits Characteristic organ/tissue injury

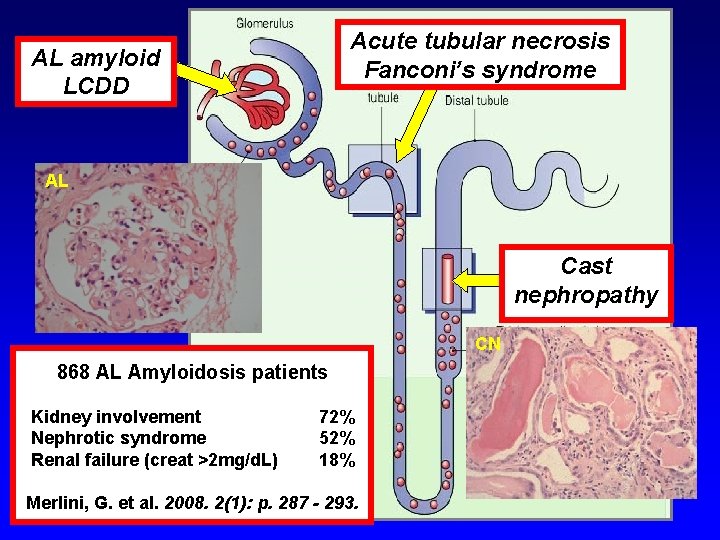
Acute tubular necrosis Fanconi’s syndrome AL amyloid LCDD AL Cast nephropathy CN 868 AL Amyloidosis patients Kidney involvement Nephrotic syndrome Renal failure (creat >2 mg/d. L) 72% 52% 18% Merlini, G. et al. 2008. 2(1): p. 287 - 293.

AL Amyloidosis Diagnosis Monoclonal Protein Investigations Serum electrophoresis: SPE + s. IFE + Urine electrophoresis: UPE + u. IFE and/or? Serum FLC assay

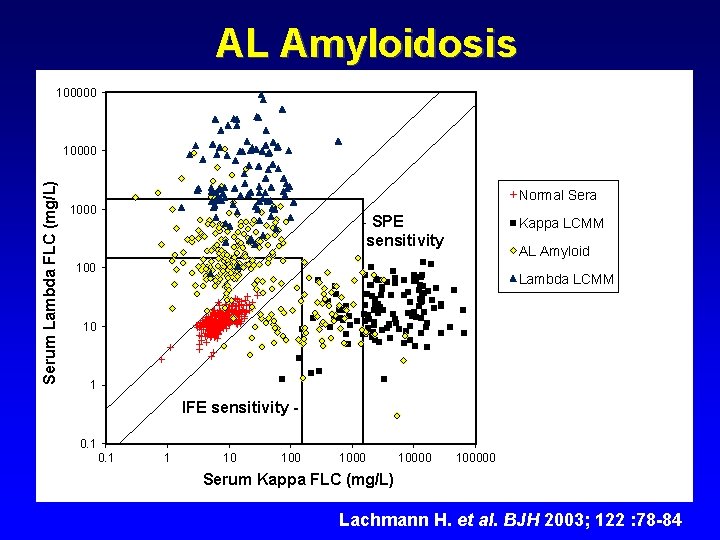
AL Amyloidosis 100000 Serum Lambda FLC (mg/L) 10000 Normal Sera 1000 - SPE sensitivity Kappa LCMM AL Amyloid 100 Lambda LCMM 10 1 IFE sensitivity 0. 1 1 10 100000 Serum Kappa FLC (mg/L) Lachmann H. et al. BJH 2003; 122 : 78 -84

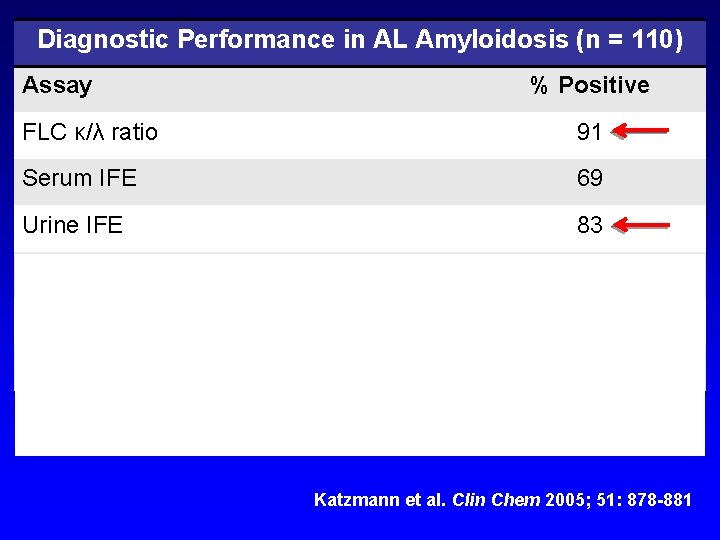
Diagnostic Performance in AL Amyloidosis (n = 110) Assay % Positive FLC κ/λ ratio 91 Serum IFE 69 Urine IFE 83 Serum IFE + urine IFE 95 FLC κ/λ ratio + serum IFE 99 FLC κ/λ ratio + serum IFE + urine IFE 99 ‘Urine IFE did not add any additional information. ’ Katzmann et al. Clin Chem 2005; 51: 878 -881

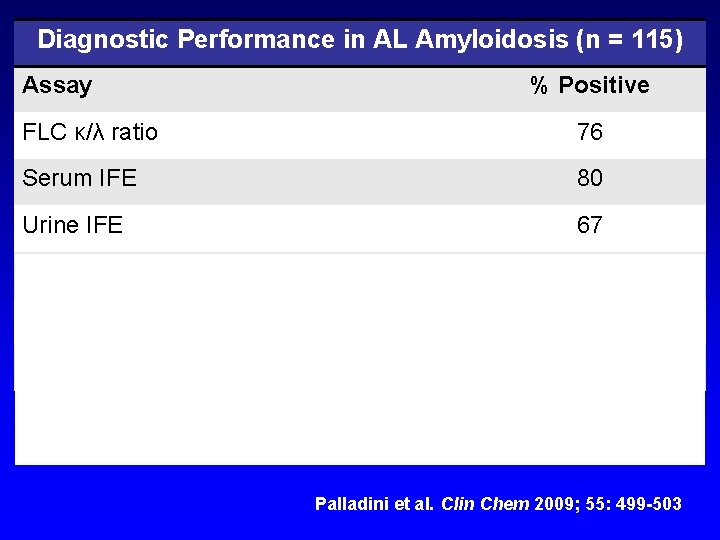
Diagnostic Performance in AL Amyloidosis (n = 115) Assay % Positive FLC κ/λ ratio 76 Serum IFE 80 Urine IFE 67 Serum IFE + urine IFE 96 FLC κ/λ ratio + serum IFE + urine IFE 100 All three assays are complementary Palladini et al. Clin Chem 2009; 55: 499 -503

AL Amyloidosis Guidelines Summary Screening Publication Screening IMWG for s. FLC analysis s. IFE + s. FLC + u. IFE Dispenzieri, A. , et al. Leukemia, 2009. 23(2): p. 215 -24. BCSH AL Amyloidosis guidelines Bird, J. M. , et al. Br J Haematol, 2004. 125(6): p. 681 -700. s. IFE + s. FLC + u. IFE

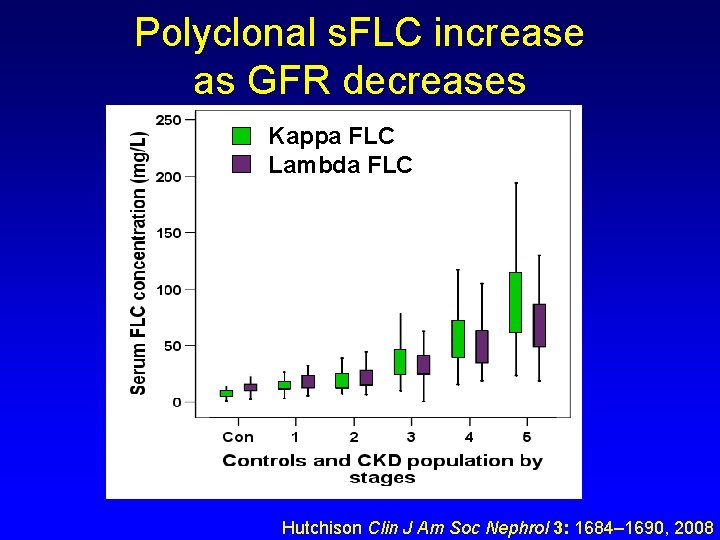
Polyclonal s. FLC increase as GFR decreases Kappa FLC Lambda FLC Hutchison Clin J Am Soc Nephrol 3: 1684– 1690, 2008

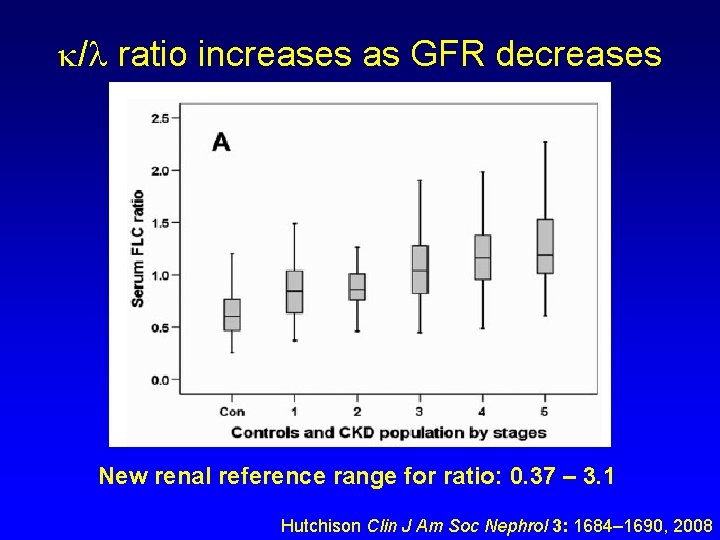
/ ratio increases as GFR decreases New renal reference range for ratio: 0. 37 – 3. 1 Hutchison Clin J Am Soc Nephrol 3: 1684– 1690, 2008

Can s. FLC assays be used to diagnose multiple myeloma in patients with renal failure? • Audit of 142 patients with new dialysis dependent acute renal failure • 41 / 142 patients with multiple myeloma Hutchison et al. BMC Nephrology 2008, 9: 11

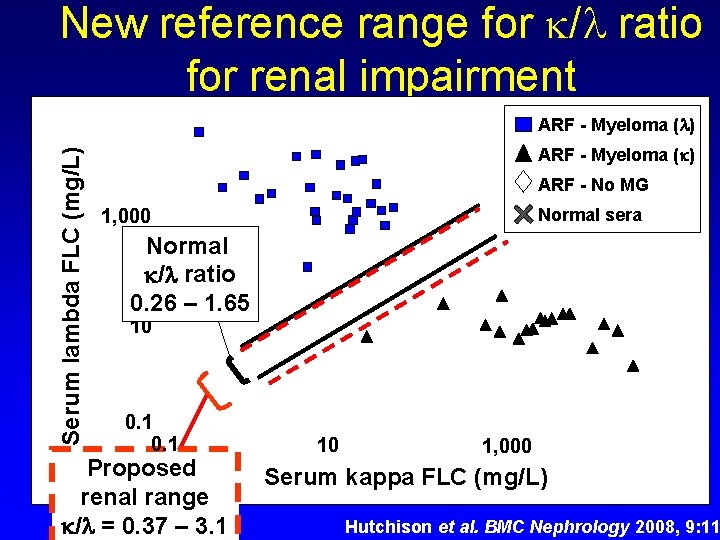
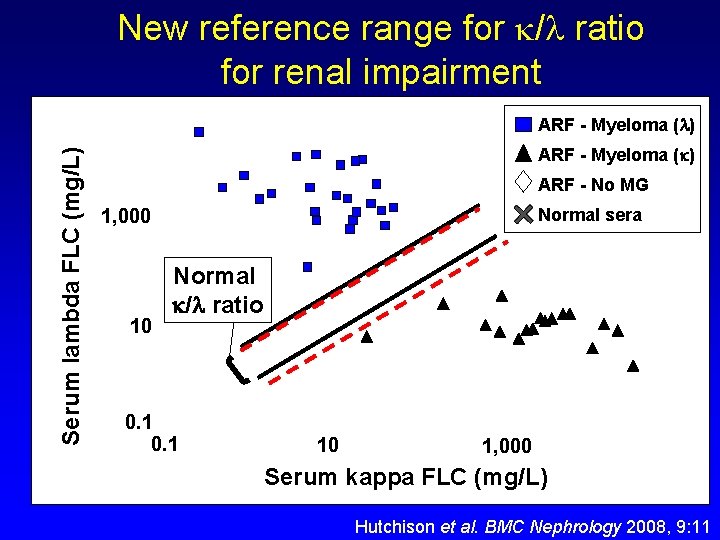
New reference range for / ratio for renal impairment Serum lambda FLC (mg/L) ARF - Myeloma ( ) ARF - No MG 1, 000 Normal sera Normal / ratio 0. 26 – 1. 65 10 0. 1 Proposed renal range / = 0. 37 – 3. 1 10 1, 000 Serum kappa FLC (mg/L) Hutchison et al. BMC Nephrology 2008, 9: 11

New reference range for / ratio for renal impairment 1. Interpret s. FLC results in the context of clinical findings and other laboratory tests… including renal function 2. If patient has renal impairment, then renal reference range ( / = 0. 37 – 3. 1) may be applicable 3. Renal reference range improves diagnostic specificity without changing diagnostic sensitivity

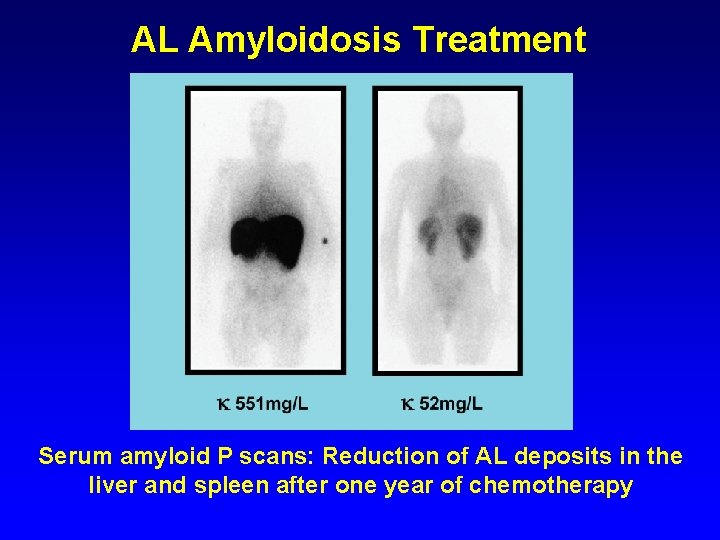
AL Amyloidosis Treatment Serum amyloid P scans: Reduction of AL deposits in the liver and spleen after one year of chemotherapy

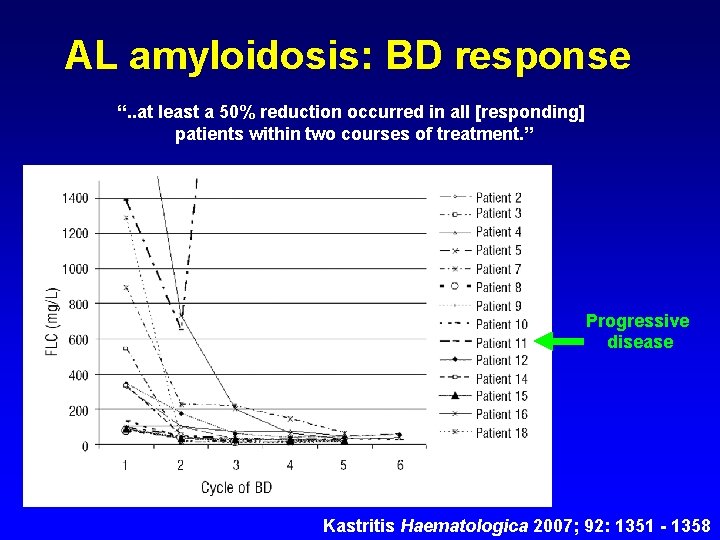
AL amyloidosis: BD response “. . at least a 50% reduction occurred in all [responding] patients within two courses of treatment. ” Progressive disease Kastritis Haematologica 2007; 92: 1351 - 1358

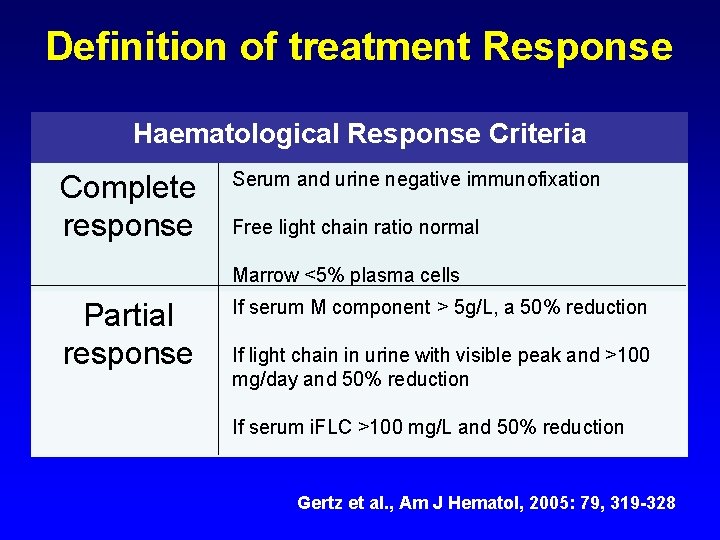
Definition of treatment Response Haematological Response Criteria Complete response Serum and urine negative immunofixation Free light chain ratio normal Marrow <5% plasma cells Partial response If serum M component > 5 g/L, a 50% reduction If light chain in urine with visible peak and >100 mg/day and 50% reduction If serum i. FLC >100 mg/L and 50% reduction Gertz et al. , Am J Hematol, 2005: 79, 319 -328

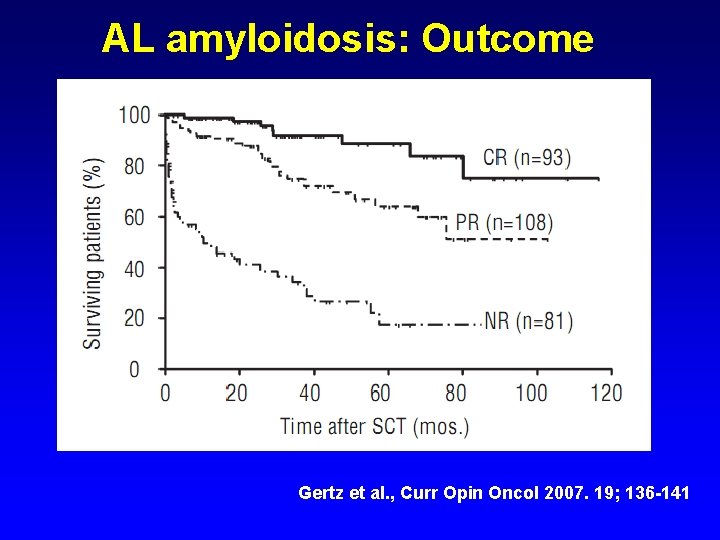
AL amyloidosis: Outcome Gertz et al. , Curr Opin Oncol 2007. 19; 136 -141

AL Amyloidosis Guidelines Summary Monitoring Publication Monitoring IMWG for s. FLC analysis s. FLC essential Dispenzieri, A. , et al. Leukemia, 2009. 23(2): p. 215 -24. BCSH AL Amyloidosis guidelines Bird, J. M. , et al. Br J Haematol, 2004. 125(6): p. 681 -700. International Consensus Opinion Gertz, M. A. , et al. , Am J Hematol, 2005. 79(4): p. 319 -28. (Recommended for LCDD) s. FLC recommended

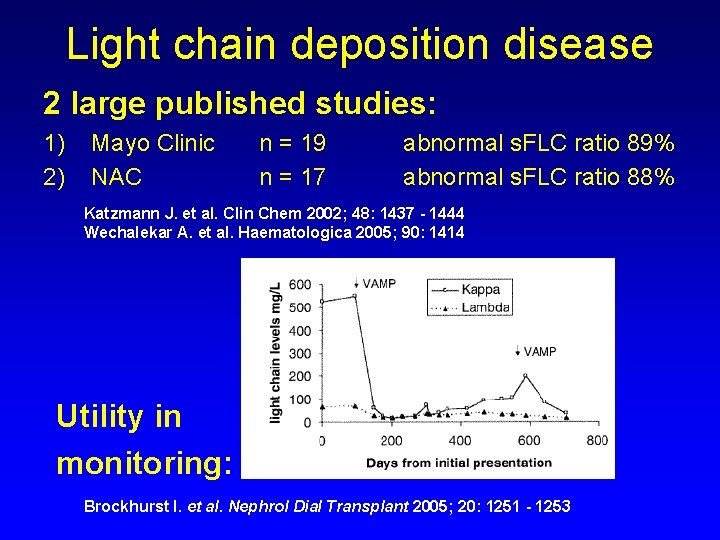
Light chain deposition disease 2 large published studies: 1) 2) Mayo Clinic NAC n = 19 n = 17 abnormal s. FLC ratio 89% abnormal s. FLC ratio 88% Katzmann J. et al. Clin Chem 2002; 48: 1437 - 1444 Wechalekar A. et al. Haematologica 2005; 90: 1414 Utility in monitoring: Brockhurst I. et al. Nephrol Dial Transplant 2005; 20: 1251 - 1253

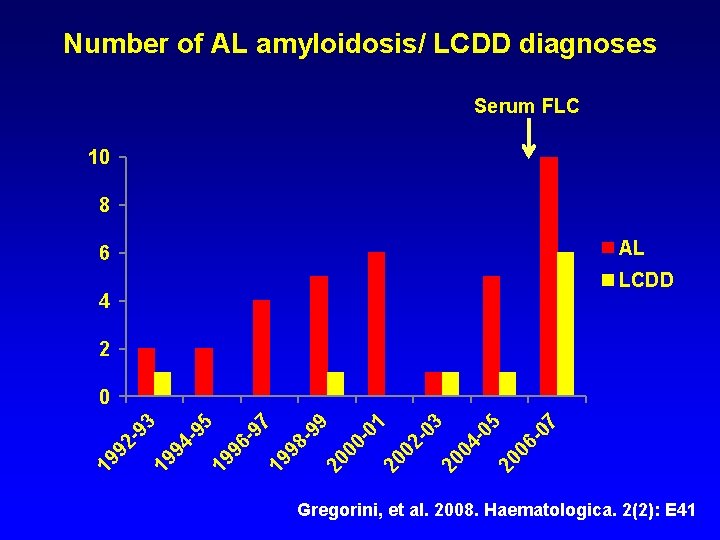
Number of AL amyloidosis/ LCDD diagnoses Serum FLC 10 8 AL 6 LCDD 4 2 697 19 98 -9 9 20 00 -0 1 20 02 -0 3 20 04 -0 5 20 06 -0 7 19 9 95 19 94 - 19 92 - 93 0 Gregorini, et al. 2008. Haematologica. 2(2): E 41

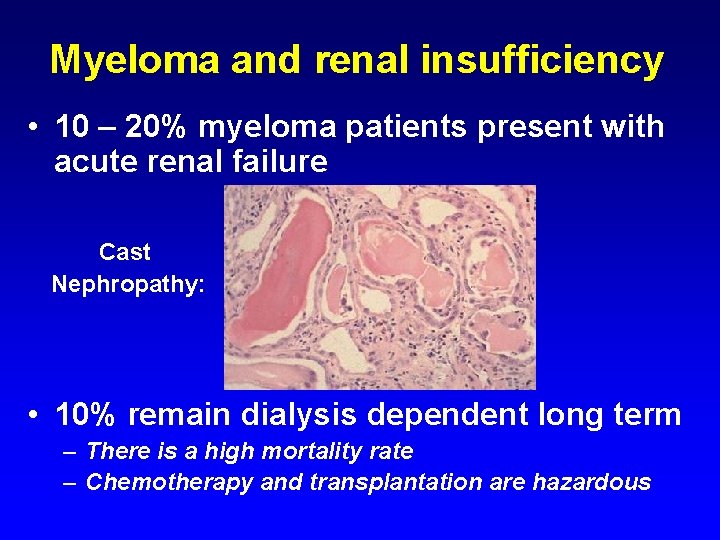
Myeloma and renal insufficiency • 10 – 20% myeloma patients present with acute renal failure Cast Nephropathy: • 10% remain dialysis dependent long term – There is a high mortality rate – Chemotherapy and transplantation are hazardous

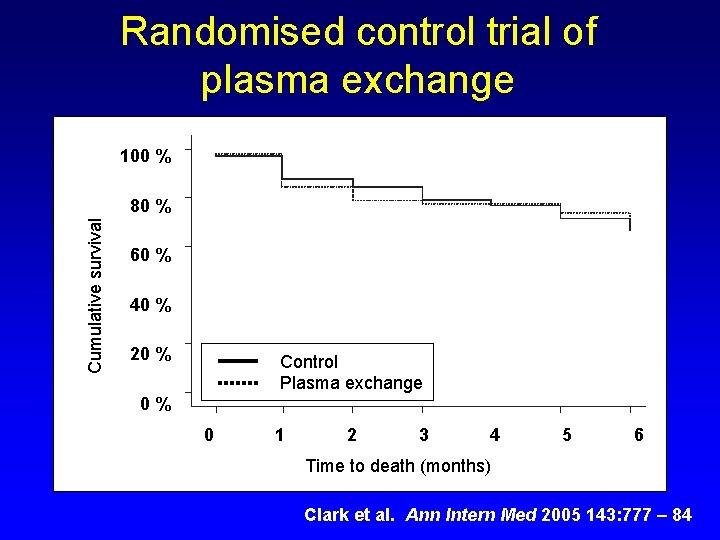
Light chain removal strategies for cast nephropathy 1. Plasma exchange • Used since 1980 s 2. Haemodialysis • New treatment strategy

Plasma exchange to remove s. FLCs Challenges: 1. >80% of FLCs are extravascular. 2. PE procedures are of limited frequency & duration (typically 6 x 1. 5 hour sessions over 2 weeks) Typical recovery rates: 10 - 20%.

Randomised control trial of plasma exchange 100 % Cumulative survival 80 % 60 % 40 % 20 % Control Plasma exchange 0% 0 1 2 3 4 5 6 Time to death (months) Clark et al. Ann Intern Med 2005 143: 777 – 84

Haemodialysis to remove s. FLCs • 7 dialysers evaluated in vitro for filtration efficiency • The Gambro HCO 1100* was the most efficient at removing FLC * Available in Poland Hutchison, CA. et al. JASN 2007; 18: 886 -895

Distribution of filter pore sizes High Flux High Cut-Off Plasma Filter Pore size [ m] Size of albumin

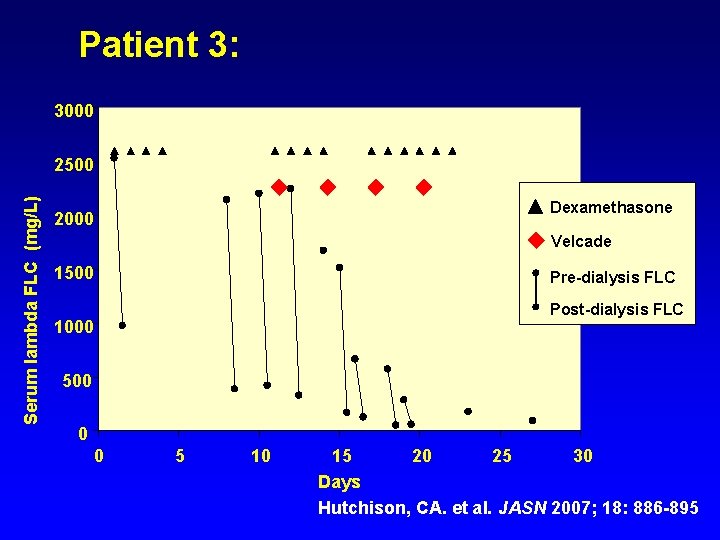
Patient 3: 3000 Serum lambda FLC (mg/L) 2500 Dexamethasone 2000 Velcade 1500 Pre-dialysis FLC Post-dialysis FLC 1000 500 0 0 5 10 15 20 25 30 Days Hutchison, CA. et al. JASN 2007; 18: 886 -895

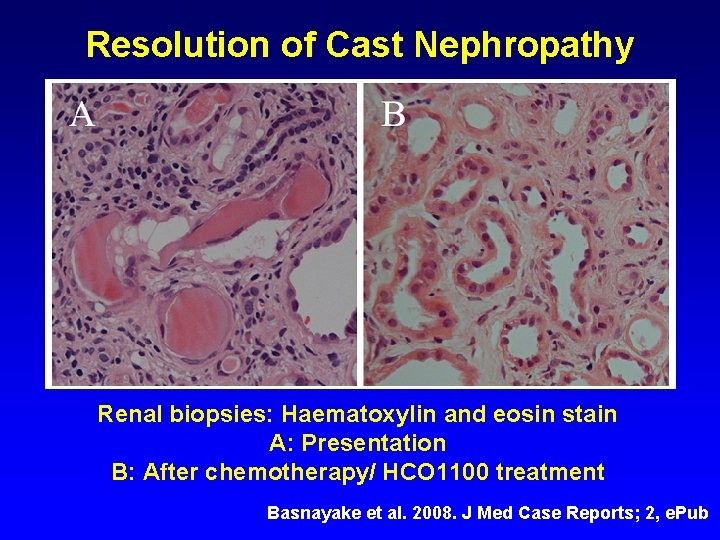
Resolution of Cast Nephropathy Renal biopsies: Haematoxylin and eosin stain A: Presentation B: After chemotherapy/ HCO 1100 treatment Basnayake et al. 2008. J Med Case Reports; 2, e. Pub

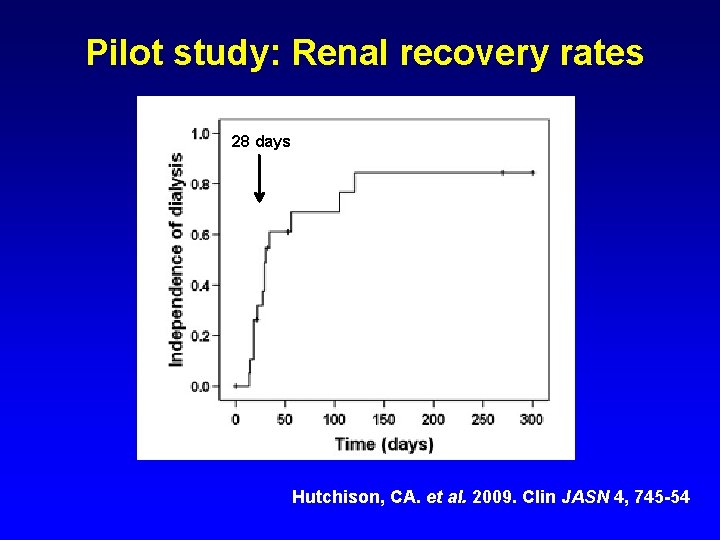
Pilot study: Renal recovery rates 28 days Hutchison, CA. et al. 2009. Clin JASN 4, 745 -54

European Trial of Free Light Chain Removal by Extended Haemodialysis in Cast Nephropathy Contact: Dr Colin Hutchison cah 692@bham. ac. uk AL amyloidosis? Publication in press

Guidelines Summary IMWG 1 BCSH 2 Screening N/A + s. IFE & u. IFE Prognosis International Consensus Opinion 3 - N/A Monitoring 1. Dispenzieri, A. , et al. Leukemia, 2009. 23(2): p. 215 -24 2. Bird, J. M. , et al. Br J Haematol, 2004. 125(6): p. 681 -700 3. Gertz, M. A. , et al. , Am J Hematol, 2005. 79(4): p. 319 -28

Conclusions FLCs in AL amyloidosis: “The introduction of FLC assay has greatly improved the management of patients with AL amyloidosis and is now an essential tool in the care of this disease. ” Prof. G. Merlini 5 th International Symposium, Bath Assembly Rooms Biennial Meeting, 2008 • alex. legg@bindingsite. com

New reference range for / ratio for renal impairment Serum lambda FLC (mg/L) ARF - Myeloma ( ) ARF - No MG 1, 000 10 Normal sera Normal / ratio 0. 1 10 1, 000 Serum kappa FLC (mg/L) Hutchison et al. BMC Nephrology 2008, 9: 11

Patient inclusion criteria • Dialysis dependent renal failure, renal biopsy proven cast nephropathy • Fulfils diagnostic criteria for the diagnosis of symptomatic de novo MM • Abnormal s. FLC ratio and s. FLC > 500 mg/L • Informed consent • Commencement of study within 10 days of presentation

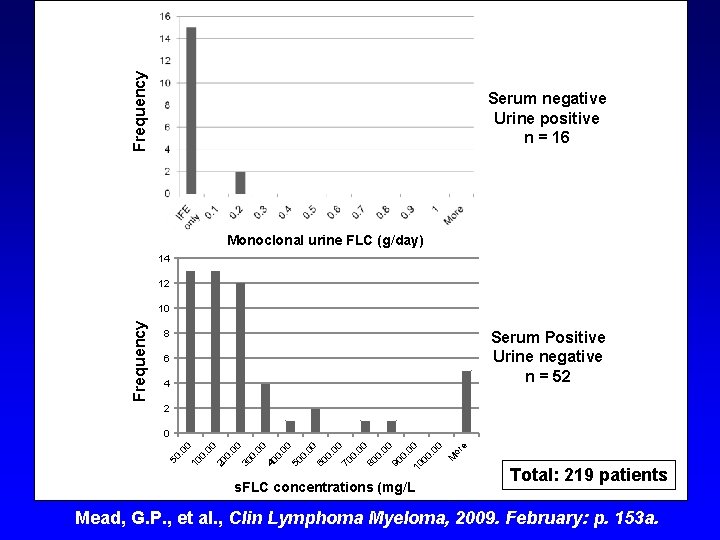
Frequency Serum negative Urine positive n = 16 Monoclonal urine FLC (g/day) 14 12 Frequency 10 8 Serum Positive Urine Series 1 negative n = 52 6 4 2 s. FLC concentrations (mg/L or e M 50 . 0 0 10 0. 00 20 0. 00 30 0. 00 40 0. 00 50 0. 00 60 0. 00 70 0. 00 80 0. 00 90 0. 0 10 0 00. 0 0 0 Total: 219 patients Mead, G. P. , et al. , Clin Lymphoma Myeloma, 2009. February: p. 153 a.

AL amyloidosis: Serum FLC negative and urine positive? Patient X: Serum FLCs before developing AL amyloidosis: Kappa: Lambda: k/l ratio: 10 mg/ L 1 Normal Patient X then develops a very subtle AL amyloidosis tumour Kappa: Lambda: k/l ratio: 12 mg/ L 8 mg/ L 1. 5 Normal This patient would normally be urine negative due to normal kidney function. . .

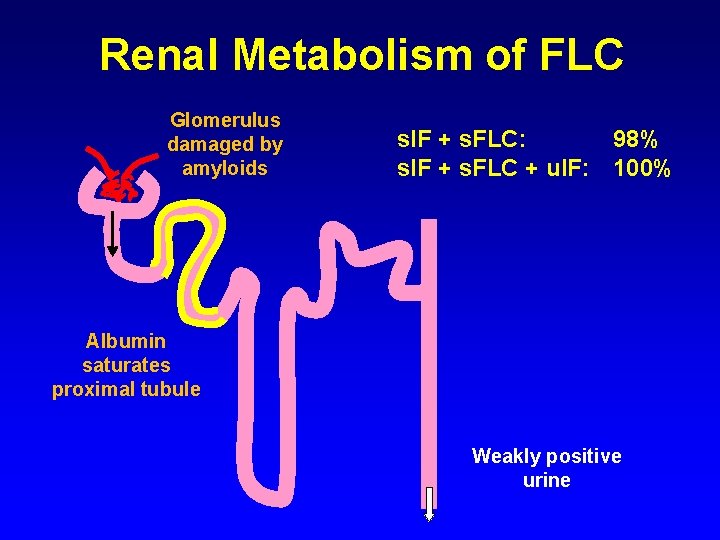
Renal Metabolism of FLC Glomerulus damaged by amyloids s. IF + s. FLC: 98% s. IF + s. FLC + u. IF: 100% Albumin saturates proximal tubule Weakly positive urine

Randomised and controlled 90 Patients recruited Randomisation Control Arm HD 45 Patients Standard high-flux HD Research Arm HD 45 Patients Extended HD on HCO 1100 ‘Modified PAD regimen’ Chemotherapy (P) VELCADE™ (bortezomib) iv (A) Adriamycin (Doxorubicin) iv (D) Dexamethasone oral Assess outcome 1. 0 mg/m 2 9. 0 mg/m 2 40 mg

Trial time course Day 0 1 Research √ arm HD (6) (Hours) Chemo s. FLC measured √ 2 3 4 √ √ (8) 5 6 7 √ √ √ (8) (8) V V A A D D √ √ √ 8 √ 9 10 11 12 13 14 15 16 17 18 19 20 21 22 onwards √ √ (8) V √ (8) √ (8) V √ √ √ s. FLC measured • at assessment • pre dialysis • post dialysis • non-dialysis Accord. to clin need (6) As per PAD protocol D* D* √ √ (8) D* D* √ √ √ Run within 24 hours Run once /week

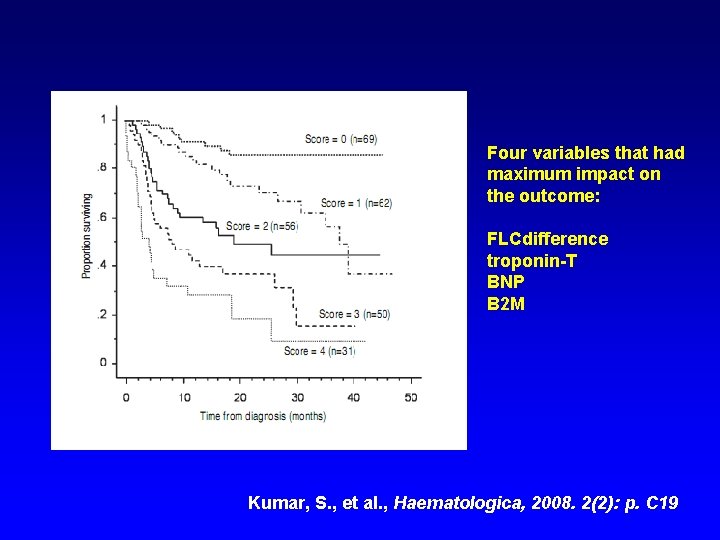
Four variables that had maximum impact on the outcome: FLCdifference troponin-T BNP B 2 M Kumar, S. , et al. , Haematologica, 2008. 2(2): p. C 19

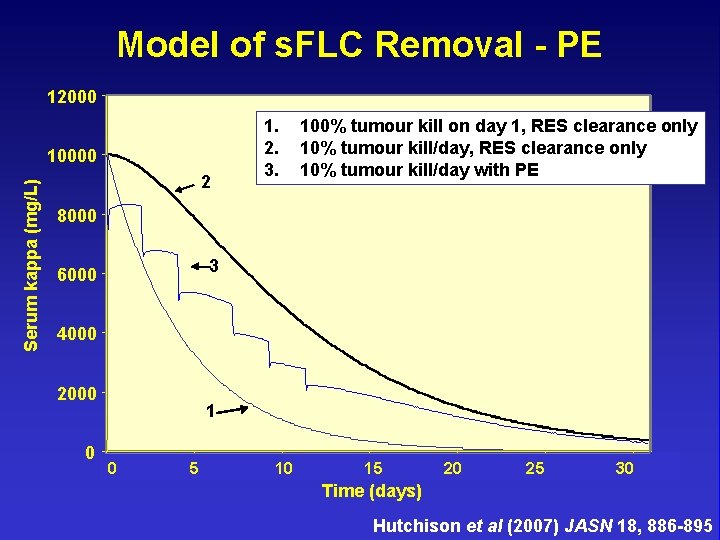
Model of s. FLC Removal - PE 12000 1. 2. 3. Serum kappa (mg/L) 10000 2 100% tumour kill on day 1, RES clearance only 10% tumour kill/day with PE 8000 3 6000 4000 2000 0 1 2 4 5 5 6 7 8 10 11 9 12 13 1515 14 17 18 20 20 19 21 22 24 25 25 26 27 3030 28 Time (days) Hutchison et al (2007) JASN 18, 886 -895

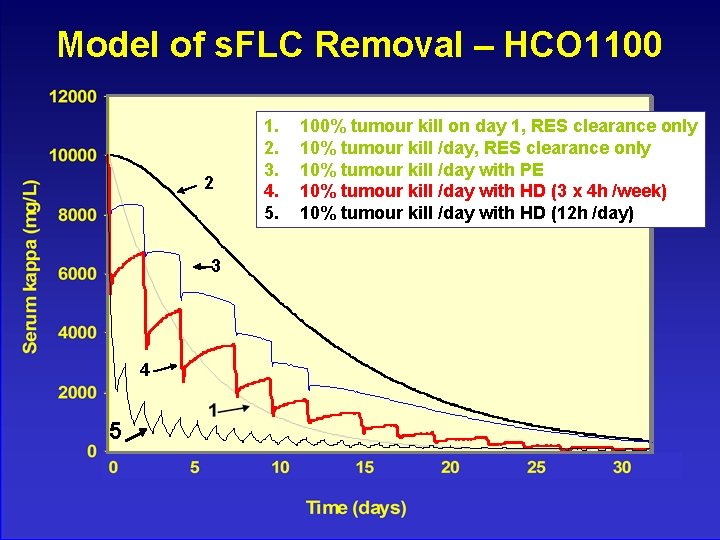
Model of s. FLC Removal – HCO 1100 2 3 4 5 1. 2. 3. 4. 5. 100% tumour kill on day 1, RES clearance only 10% tumour kill /day with PE 10% tumour kill /day with HD (3 x 4 h /week) 10% tumour kill /day with HD (12 h /day)

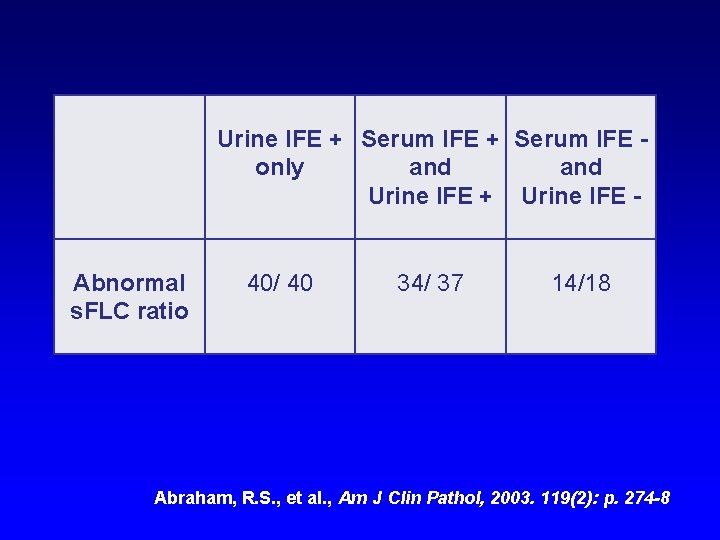
Urine IFE + Serum IFE only and Urine IFE + Urine IFE Abnormal s. FLC ratio 40/ 40 34/ 37 14/18 Abraham, R. S. , et al. , Am J Clin Pathol, 2003. 119(2): p. 274 -8

All urine IFE+ AL amyloidosis patients identified by s. IFE + s. FLC Katzmann, J. A. , et al. , Mayo Clin Proc, 2006. 81(12): p. 1575 -8.

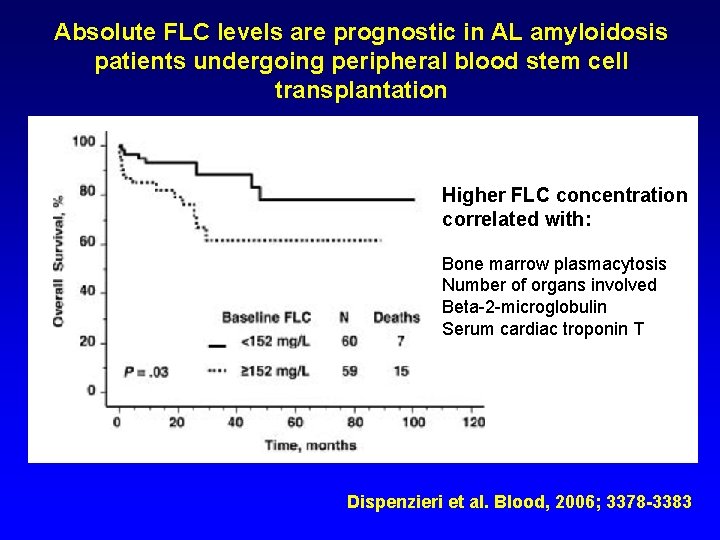
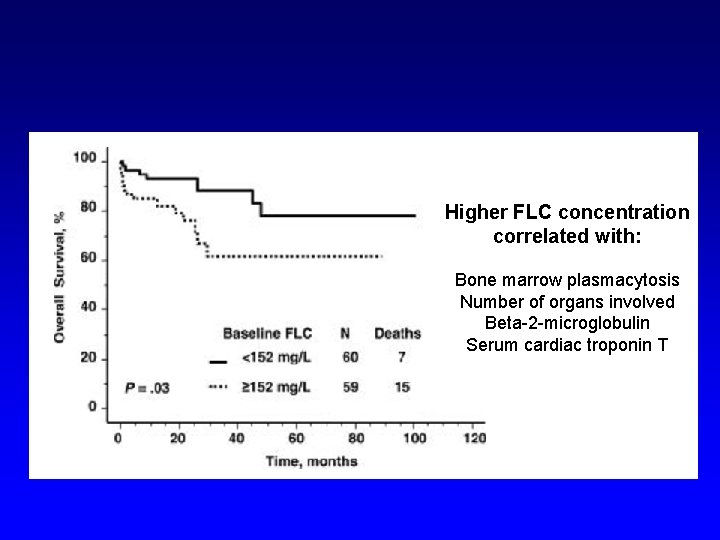
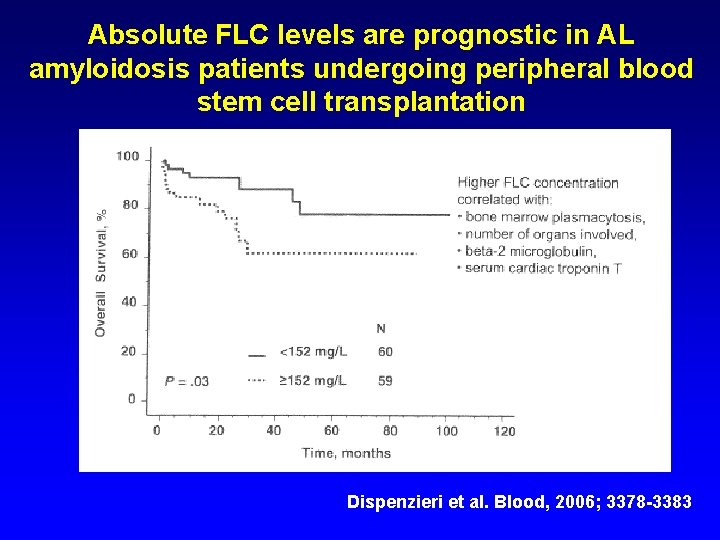
Absolute FLC levels are prognostic in AL amyloidosis patients undergoing peripheral blood stem cell transplantation Higher FLC concentration correlated with: Bone marrow plasmacytosis Number of organs involved Beta-2 -microglobulin Serum cardiac troponin T Dispenzieri et al. Blood, 2006; 3378 -3383

Higher FLC concentration correlated with: Bone marrow plasmacytosis Number of organs involved Beta-2 -microglobulin Serum cardiac troponin T

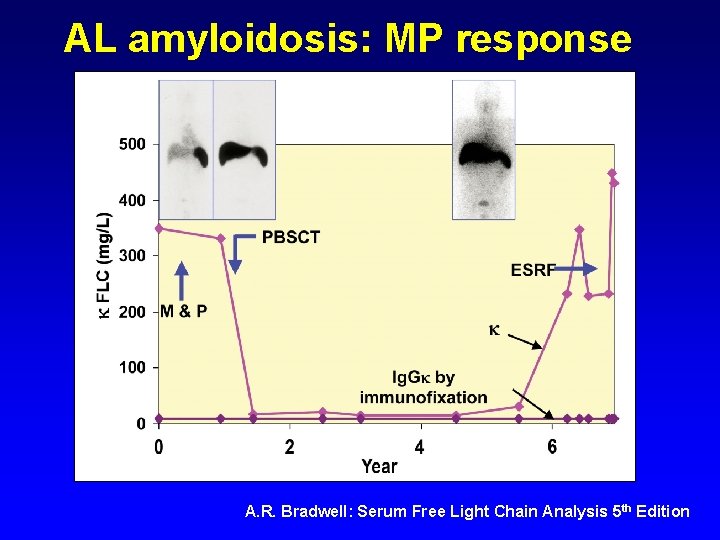
AL amyloidosis: MP response A. R. Bradwell: Serum Free Light Chain Analysis 5 th Edition

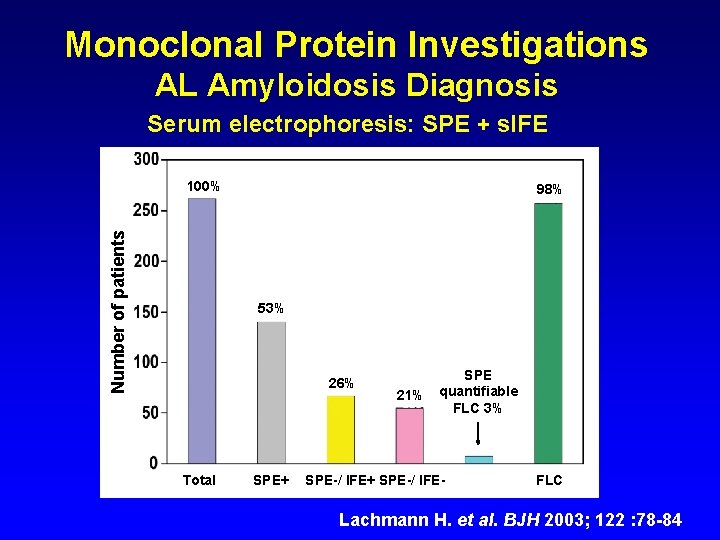
Monoclonal Protein Investigations AL Amyloidosis Diagnosis Serum electrophoresis: SPE + s. IFE Number of patients 100% 98% 53% 26% Total SPE+ 21% SPE quantifiable FLC 3% SPE-/ IFE+ SPE-/ IFE- FLC Lachmann H. et al. BJH 2003; 122 : 78 -84

Absolute FLC levels are prognostic in AL amyloidosis patients undergoing peripheral blood stem cell transplantation Dispenzieri et al. Blood, 2006; 3378 -3383

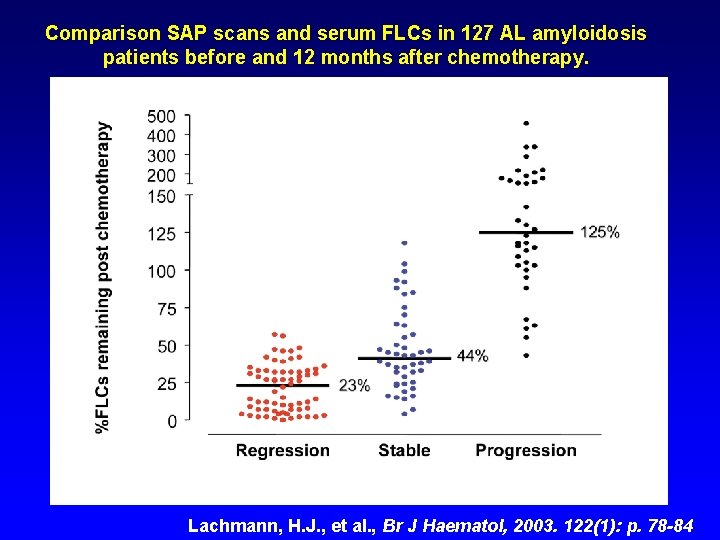
Comparison SAP scans and serum FLCs in 127 AL amyloidosis patients before and 12 months after chemotherapy. Lachmann, H. J. , et al. , Br J Haematol, 2003. 122(1): p. 78 -84

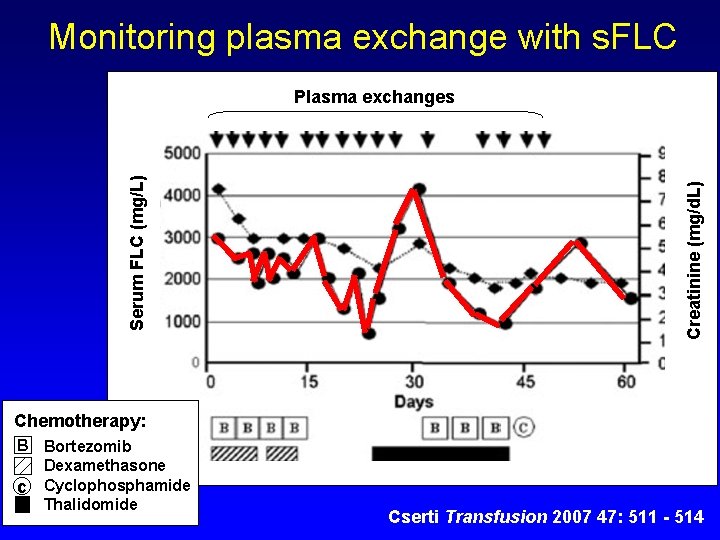
Monitoring plasma exchange with s. FLC Creatinine (mg/d. L) Serum FLC (mg/L) Plasma exchanges Chemotherapy: B Bortezomib Dexamethasone c Cyclophosphamide Thalidomide Cserti Transfusion 2007 47: 511 - 514

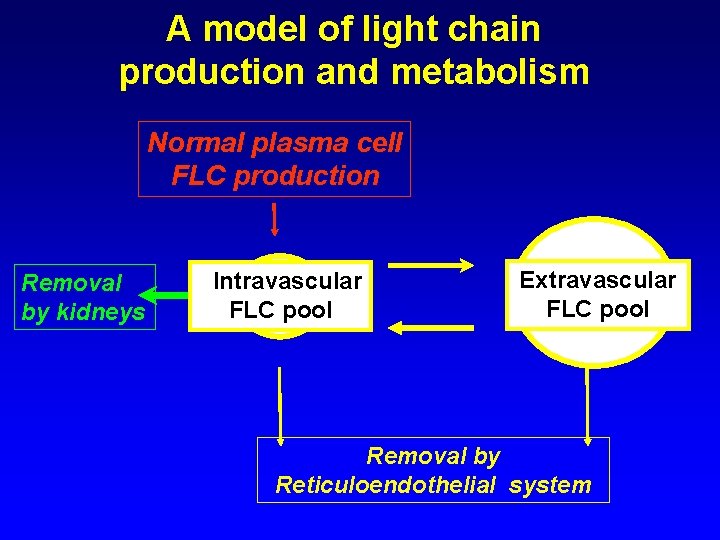
A model of light chain production and metabolism Normal plasma cell FLC production Removal by kidneys Intravascular FLC pool Extravascular FLC pool Removal by Reticuloendothelial system

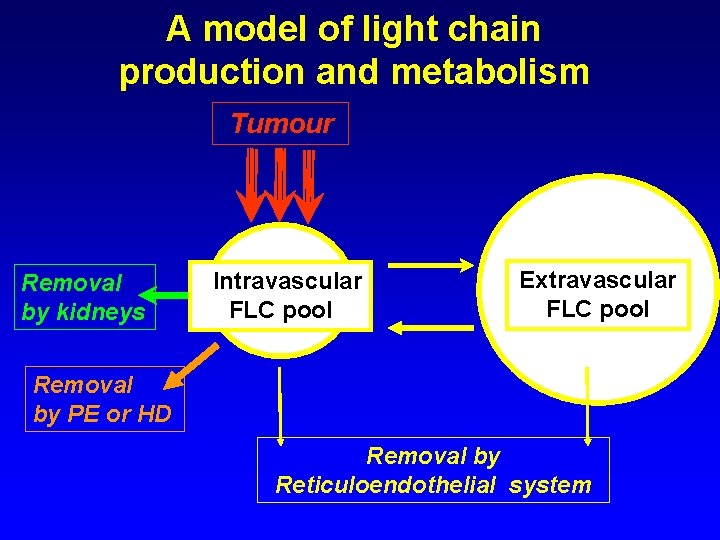
A model of light chain production and metabolism Tumour Removal by kidneys Intravascular FLC pool Extravascular FLC pool Removal by PE or HD Removal by Reticuloendothelial system