Al Aluminum extraction from Feldspar by Robert Dyck

Al Aluminum extraction from Feldspar by Robert Dyck Ardeco Consulting Ltd.

Traditional ore Aluminum ore on Earth is bauxite Bauxite is formed by tropical rain forests: • igneous minerals weathered by water • tropical ecosystem depletes their needs viron oxide, silicon dioxide, aluminum oxide We aren’t going to find a rainforest on Mars.

MGS-TES
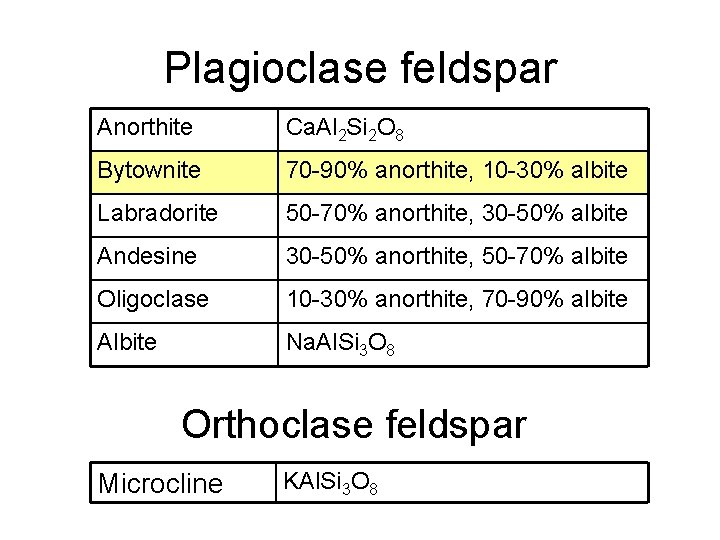
Plagioclase feldspar Anorthite Ca. Al 2 Si 2 O 8 Bytownite 70 -90% anorthite, 10 -30% albite Labradorite 50 -70% anorthite, 30 -50% albite Andesine 30 -50% anorthite, 50 -70% albite Oligoclase 10 -30% anorthite, 70 -90% albite Albite Na. Al. Si 3 O 8 Orthoclase feldspar Microcline KAl. Si 3 O 8

Traditional smelting • Bayer process extracts/purifies alumina from ore – chemical • Sodium hydroxide, carbon dioxide • Hall-Héroult process de-oxidizes alumina into metal – electrolysis • cryolite
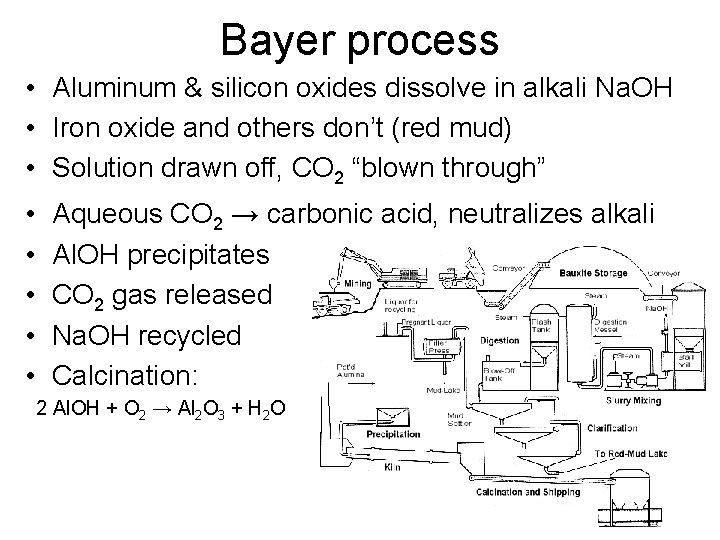
Bayer process • Aluminum & silicon oxides dissolve in alkali Na. OH • Iron oxide and others don’t (red mud) • Solution drawn off, CO 2 “blown through” • • • Aqueous CO 2 → carbonic acid, neutralizes alkali Al. OH precipitates CO 2 gas released Na. OH recycled Calcination: 2 Al. OH + O 2 → Al 2 O 3 + H 2 O
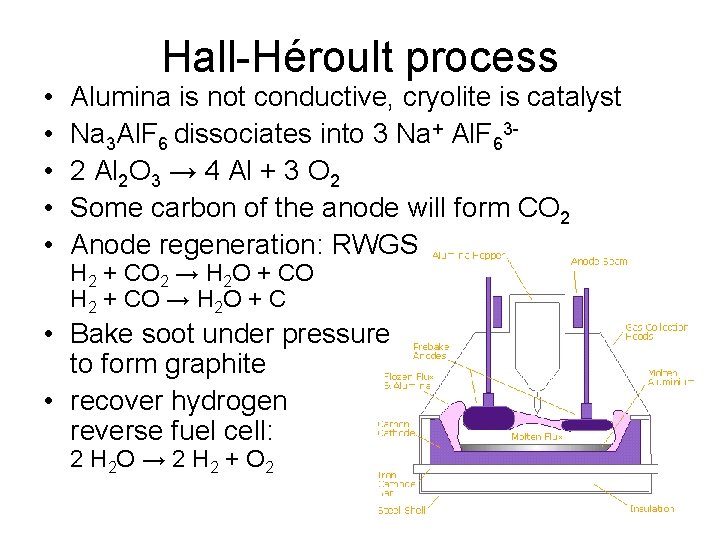
• • • Hall-Héroult process Alumina is not conductive, cryolite is catalyst Na 3 Al. F 6 dissociates into 3 Na+ Al. F 632 Al 2 O 3 → 4 Al + 3 O 2 Some carbon of the anode will form CO 2 Anode regeneration: RWGS H 2 + CO 2 → H 2 O + CO H 2 + CO → H 2 O + C • Bake soot under pressure to form graphite • recover hydrogen reverse fuel cell: 2 H 2 O → 2 H 2 + O 2

Bytownite to Alumina • Bytownite is an alkali feldspar so an alkali like sodium hydroxide will not dissolve it. Hydrochloric acid will. • 1. 8 Al 3+, 0. 8 Ca 2+, 0. 2 Na+, 2. 2 Si. O 2 • Ammonia will raise p. H, and evaporate when released • Ammonium chloride p. H 5. 0 @ 10% • Al. OH will between p. H 4. 5 and 9. 2 • Al. OH can be calicinated: 2 Al. OH + O 2 → Al 2 O 3 + H 2 O • Na. Cl, KCl, Ca. Cl 2 are salts, they will remain in solution • Silica scale is a problem in the Bayer process it will be more so with bytownite
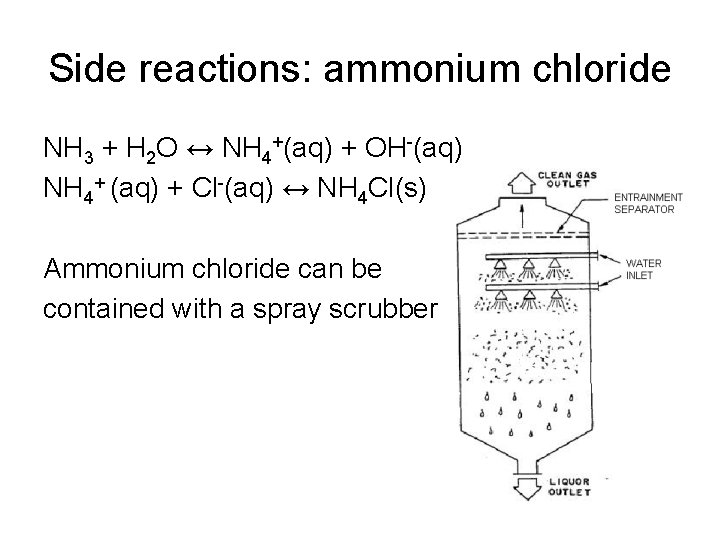
Side reactions: ammonium chloride NH 3 + H 2 O ↔ NH 4+(aq) + OH-(aq) NH 4+ (aq) + Cl-(aq) ↔ NH 4 Cl(s) Ammonium chloride can be contained with a spray scrubber
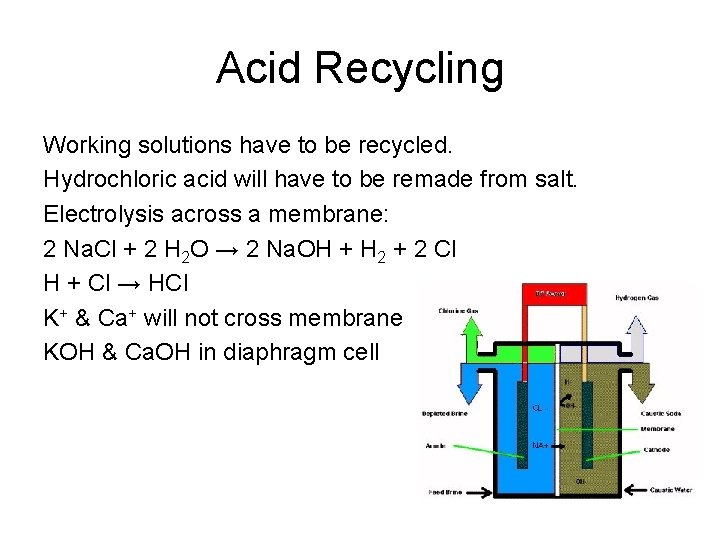
Acid Recycling Working solutions have to be recycled. Hydrochloric acid will have to be remade from salt. Electrolysis across a membrane: 2 Na. Cl + 2 H 2 O → 2 Na. OH + H 2 + 2 Cl H + Cl → HCl K+ & Ca+ will not cross membrane KOH & Ca. OH in diaphragm cell
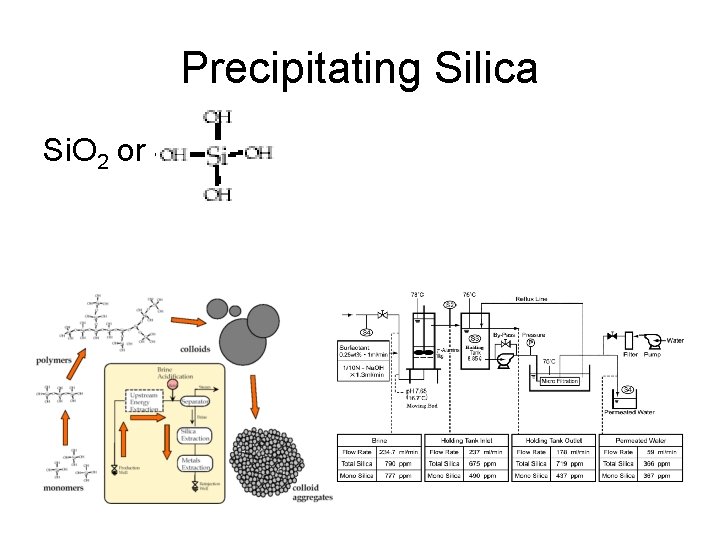
Precipitating Silica Si. O 2 or Si(OH)4
- Slides: 11