AIR POLLUTION Credits https www youtube comwatch vUc

AIR POLLUTION

Credits: https: //www. youtube. com/watch? v=Uc. Wpk. WBX 04 E

What is air pollution? • Air pollution is probably one of the most serious environmental problems confronting our civilization today. • Air pollution means the presence of chemicals or compounds in the air which are usually not present and which lower the quality of the air or cause detrimental changes to the quality of life (such as the damaging of the ozone layer or causing global warming).

Atmosphere As A Resource • Atmospheric Composition • • Nitrogen 78. 08% Oxygen 20. 95% Argon 0. 93% Carbon dioxide 0. 04% • Ecosystem services • Blocks UV radiation • Moderates the climate • Redistributes water in the hydrologic cycle

TYPES AND SOURCES OF AIR POLLUTION • Air pollutants are airborne substances (either solids, liquids, or gases) that occur in concentrations high enough to threaten the health of people and animals, to harm vegetation and structures, or to toxify a given environment. • They come from natural sources and human activities: 1. Natural: dust, volcano, forest fire, ocean waves, … 2. Human: a) fixed sources (power plants, homes, …) b) mobile sources (cars, ships, …)

MAN MADE SOURCES OF AIR POLLUTION • Mainly from: • Combustion Processes • Chemical Processes • Petroleum Operations • Metallurgical processes • Mineral Processing • Agricultural Activities • Nuclear Energy Programmes

• TWO CATEGORIES/ TYPES 1. PRIMARY AIR POLLUTANTS 2. SECONDARY AIR POLLUTANTS.

PRIMARY & SECONDARY POLLUTANTS • 1) The primary pollutants are “directly” emitted from the processes such as fossil fuel consumption, Volcanic eruption and factories. The major primary pollutants are Oxides of Sulphur, Oxides of Nitrogen, Oxides of Carbon, Particulate Matter, Methane, Ammonia, Chlorofluorocarbons, Toxic metals. • 2) The secondary pollutants are not emitted directly. The secondary pollutants form when the primary pollutants react with themselves or other components of the atmosphere. Most important secondary level Air Pollutants are Ground Level Ozone, Smog, Sulphuric Acid or acid mist, Formaldehyde, PAN .

Major Air Pollutants

PRIMARY AIR POLLUTANTS • 1. OXIDES OF SULPHUR • H 2 S and SO 2 are the main gases. • SOURCES OF SO 2 • Combustion of fossil fuels especially coal, petroleum products etc in the refineries, chemical plants, smelting of ores etc. • Thermal power plants accounts for about 40% of SO 2 emission.

Characteristics of SO 2 • Colourless gas with a sharp pungent odour. • Moderately soluble in water and forms weakily acidic H 2 SO 3 • Oxidised slowly in clean air to Sulphur trioxide. • In polluted atmosphere, SO 2 reacts photo chemically with other pollutants to form SO 3, H 2 SO 4 and salts of sulphuric acid.

2. Oxides of Nitrogen • Most of the Nitrogen Oxides are produced due to high temperature combustion. In the cities the brown haze dome above the cities is mostly because of the Nitrogen Oxides. • Main sources are coal, natural gas and motor fuel combustion. • Also found in effluents from industries where nitric acid is used or produced. • The most important gases are Nitrogen dioxide(NO 2), Nitric Oxide(NO).

Characteristics of NOX • NO 2 and NO are the most common gases. • NO 2 is reddish brown in colour. • NO and NO 2 forms a brown haze when high in concentration. • Major sources are automobile exhaust and nitric acid manufacturing plants. • NO 2 combines with water molecules to form acid rain.

Carbon Oxides and Hydrocarbons • Carbon Oxides • Gases carbon monoxide (CO) and carbon dioxide (CO 2) • Greenhouse gases • Hydrocarbons • Diverse group of organic compounds that contain only hydrogen and carbon (ex: CH 4 - methane) • Some are related to photochemical smog and greenhouse gases

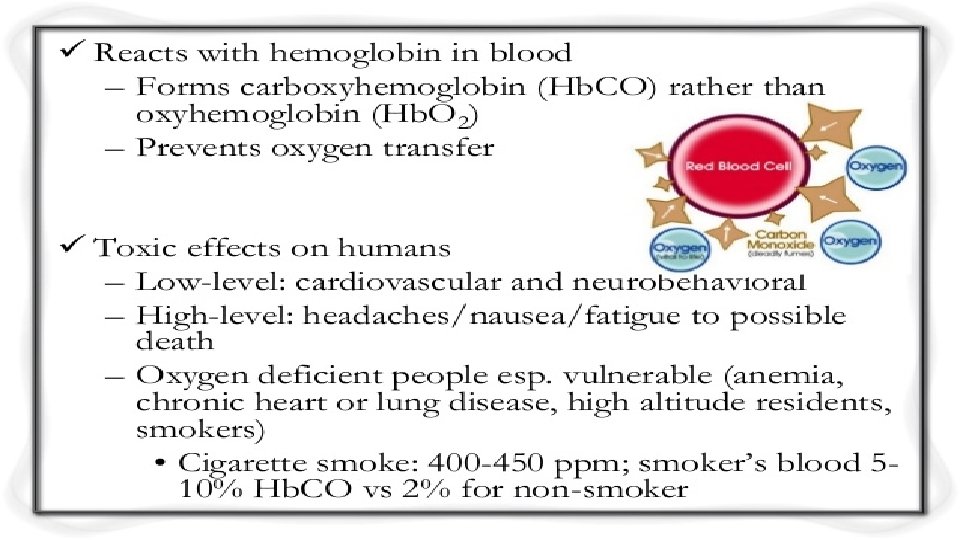
3. Oxides of Carbon • (1) Carbon Monoxide, which is colourless, odourless and non irritating but very poisonous gas is the product of incomplete combustion of the natural gas, coal or wood. The vehicle exhaust is the major source of CO. • (2)The Carbon Dioxide is associated with the Ocean Acidification and is emitted from combustion, factories and respiration of living organisms. • (3) Volatile Organic Compounds or VOCs which are methane (CH 4) and non -methane (NMVOCs). Methane is a GHG which contributes to Global Warming. The NMVOCs include the aromatic compounds such as Benzene, Toluene, Xylene which are proved or suspected carcinogens.

Particulate Material • Thousands of different solid or liquid particles suspended in air • Includes: soil particles, soot, lead, asbestos, sea salt, and sulfuric acid droplets • Dangerous for 2 reasons • May contain materials with toxic or carcinogenic effects • Extremely small particles can become lodged in lungs

SECONDARY POLLUTANTS • 1. Ground Level Ozone The most important secondary pollutant is the Ground Level Ozone or Tropospheric Ozone. Emissions from industrial facilities and electric utilities, motor vehicle exhaust, gasoline vapours, and chemical solvents are some of the major sources . Ground Level Ozone forms due to reactions of the NOx, Carbon Monoxide and VOCs in presence of sunlight.

. . Contd. • 2. Smog • Another most important secondary pollutant is the Smog, which has made up of Smoke and Fog. Traditionally, the smog has resulted from large amounts of coal burning in an area caused by a mixture of smoke and sulphur dioxide. Now-a-days, the Vehicle emissions and Industrial emissions that are acted on in the atmosphere by ultraviolet light from the sun to form secondary pollutants that also combine with the primary emissions to form photochemical smog.

Urban Air Pollution • Photochemical Smog (ex: Los Angeles below) • Brownish-orange haze formed by chemical reactions involving sunlight, nitrogen oxide, and hydrocarbons
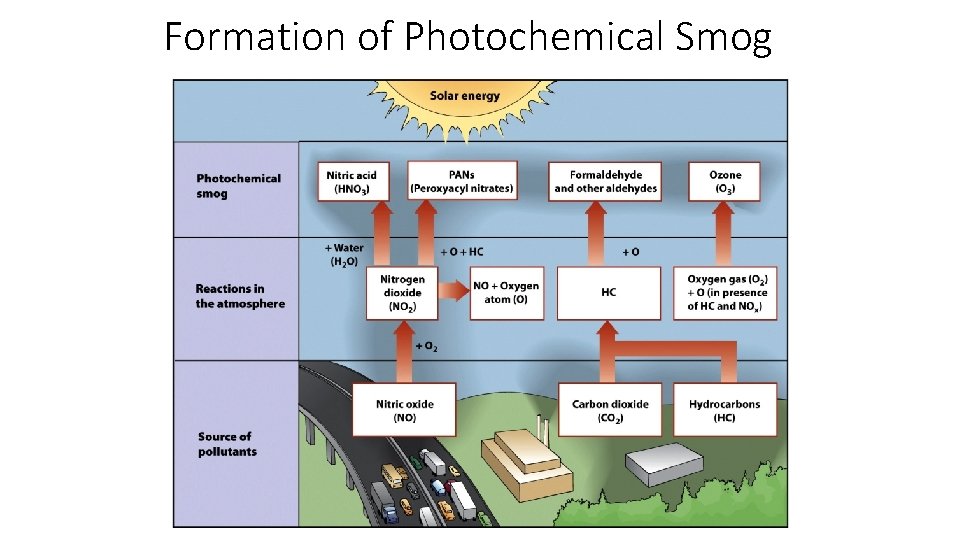
Formation of Photochemical Smog

…. Contd. • Sulphuric Acid • In polluted atmosphere, SO 2 reacts photo chemically with other pollutants to form SO 3, H 2 SO 4 and salts of sulphuric acid.

Impacts of Air Pollution • On Humans • On Vegetation • On Environment.










EFFECT ON PLANTS


EFFECT ON ENVIRONMENT • Deteriorates the metallic surfaces, glass surfaces, painted surfaces. • Damages paper and fabric. • Damages building stones, discolouration to walls, windows etc. • Deterioration of materials. • Aesthetic losses • Effect on climate
- Slides: 35