Air Pollution Chemistry Introduction Air Pollutants Carbon monoxide

Air Pollution Chemistry Introduction

Air Pollutants • Carbon monoxide (CO) - Primary • Sulfur dioxide (SO 2) - Primary • Nitrogen dioxide (NO 2) - Primary and Secondary • Ozone (O 3) - Secondary • Particulate matter < 10 um in diameter (PM 10) - Primary and Secondary

Atmospheric Processes • Emission • Dispersion –Wind –Inversion • Transport –Transformation –Deposition

Hydrocarbon Combustion • Ideally Fuel + Air => Products HC+O 2 => CO 2+H 2 O • Too Simple

Methane (CH 4) Combustion • CH 4 +O 2+Inert (N 2 or Ar) – Requires over 170 reactions and over 30 compounds • If add NOx chemistry – Requires over 275 reactions and over 45 compounds

Incomplete Combustion • Intermediate Compound Emissions – Formaldehyde and Acetylene – More toxic or reactive – Precursors to secondary pollutants (e. g. , ozone and particulate)

Oxygenated Compounds • Ethers -- R-O-R CH 3 -O-C(CH 3)3 = MTBE • Multifunctional Carbonyls – More than one oxygenated functional group on same molecule
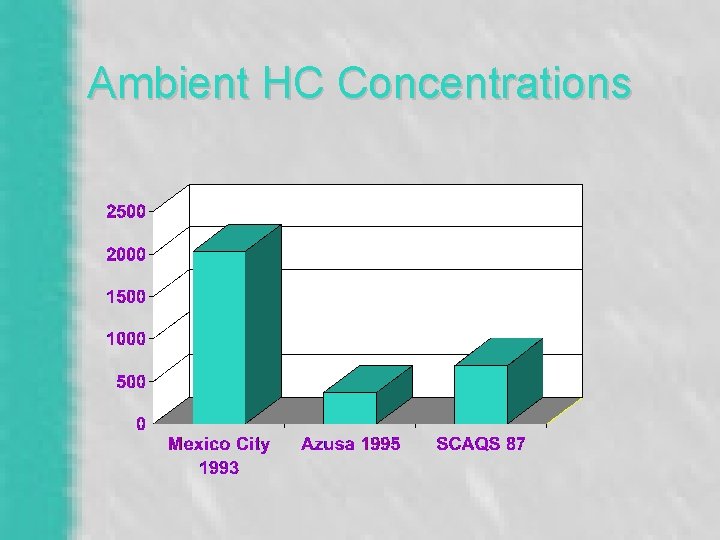
Ambient HC Concentrations

Ambient Concentrations of HC Species

Outline • Ozone Formation • Hydrocarbon Reactivity • Particulate Matter Formation • Conclusions

Nitrogen Cycle Only a few minutes per cycle: O + O 2 + M O 3 + M (fast) M is the “third body”, primarily N 2 and O 2 NO 2 + hv NO + O hv is ultraviolet radiation (k 1) O 3 + NO NO 2 + O 2 (k 3) ozone “scavenging” reaction
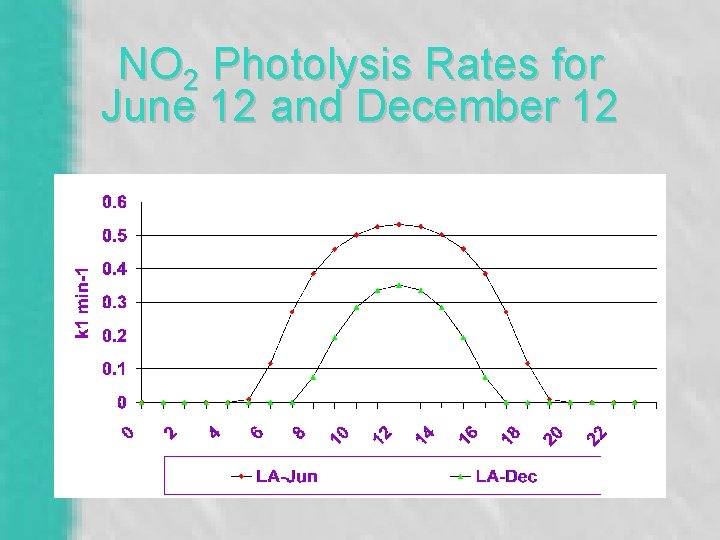
NO 2 Photolysis Rates for June 12 and December 12
![Photostationary State [O 3] = {k 1 / k 3}. {[NO 2] / [NO]} Photostationary State [O 3] = {k 1 / k 3}. {[NO 2] / [NO]}](http://slidetodoc.com/presentation_image_h2/01c8e0040f936d033b8b98c3f37a5d38/image-13.jpg)
Photostationary State [O 3] = {k 1 / k 3}. {[NO 2] / [NO]} NOx emissions < 90% NO. [O 3] = {10}. {1 / 10} [O 3] = 1 ppb

Fast Radical Transfer Reaction • Oxidizes NO to NO 2, allowing O 3 to build up. RO 2 + NO RO + NO 2 • R is any hydrocarbon fragment. 100’s of hydrocarbons 1000’s of reactions • Takes hours to affect PSS.

Hydrocarbon Oxidation Cycle The hydroxyl radical begins cycle, OH + HCHO H 2 O + HCO + O 2 HO 2 + CO HO 2 + NO NO 2 + OH and is returned at end of cycle.

Sources of Radicals Formaldehyde photolysis HCHO + hv + O 2 HO 2 + HCO Ozone photolysis O 3 + hv + H 20 2 OH + O 2 Nitrous acid photolysis HONO + hv OH + NO

NOx and Radical Sink Reaction OH + NO 2 HNO 3 (nitric acid) • Limits ozone formation. • Increases acid deposition and particulate matter.

Typical Pattern of O 3 Formation • Before sunrise – No NO 2 photolysis, radicals, or O 3 • During the day – Radicals created, convert NO to NO 2 – NO 2/NO ratio increases, O 3 builds up – NO 2 decreases via sink reaction, limiting O 3 • After sunset – Fresh NO emissions scavenge O 3
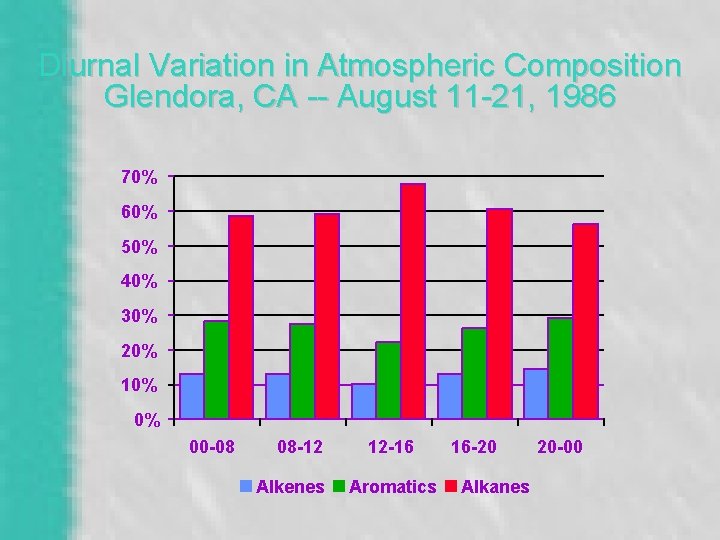
Diurnal Variation in Atmospheric Composition Glendora, CA -- August 11 -21, 1986 70% 60% 50% 40% 30% 20% 10% 0% 00 -08 08 -12 Alkenes 12 -16 Aromatics 16 -20 Alkanes 20 -00

Control Implications • Role of NOx – Required for ozone formation – Shorter lifetime than hydrocarbons – Emissions inhibit O 3 near sources – Emissions limit O 3 downwind • Role of hydrocarbons – Greatly enhance the build-up of O 3 from NOx

Control Implications (Cont’d) • Near NOx source areas – Hydrocarbon control required • Areas affected by transport – NOx control generally required • Large urban areas – Control both NOx and hydrocarbons
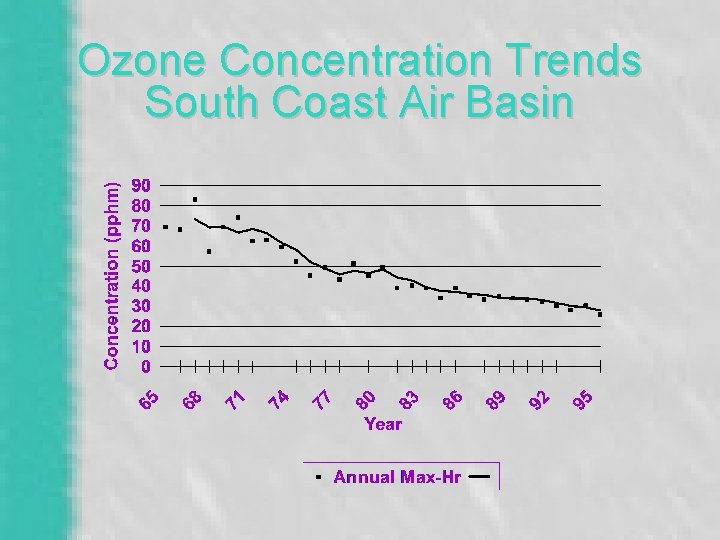
Ozone Concentration Trends South Coast Air Basin

Particulate Matter Formation • NOx, SOx + OH to acids – Also in fog and clouds • NH 3, salts + acids to nitrates, sulfates • Organic aerosols – Large alkenes + O 3 – Large alkanes + OH – Aromatics + OH

PARTICLES IN THE ATMOSPHERE • ORIGINS, EMISSIONS, & CHEMISTRY • PHYSICAL PROPERTIES • DEPOSITION AND TRANSPORT • “ACID RAIN” • HAZE & VISIBILITY • CONCENTRATION DYNAMICS

Particles Origins – Primary Particles • Mechanical Processes • Chemical Processes – Weathering – Combustion – Secondary Particles • Precursors & Catalysts • Reaction Rates • Growth and Volatility

Primary Particles – Combustion & Process Venting • Open Combustion • “Stack” & Tailpipe Emissions – Urban & Industrial • Cutting, Sand Blasting, Abrasion (Tires, Brakes, etc. ) – Natural • Soil, Vegetation, Evaporation, Fire, Volcanoes, etc.

Combustion & Process Venting – Combustion • Carbon - Organic & Elemental (Fuels) • “Ash” - “Inert Contaminants” (Metals, Soil, etc. ) – Vapors • Coking & Cooking (Organics, sulfur, etc. ) • Metals (Foundries, Welding, etc. ) • Non-Metallic (Fluxes, Salts, etc. )

“Fugitive” Material – Soil – Roads – Material Handling and Storage – Blasting & Grinding – Drift

Secondary Particles – Oxidation Pathways • Gas Phase Conversion • Aqueous Conversion • Catalysis – Particle Growth and Decay • Nucleation • Condensation and Accumulation • Vaporization

Sulfate Aerosol Formation – Gas Phase • Oxidation (OH, O 2) of H 2 S & SO 2 • O 3 Catalysis – Aqueous Conversion • Oxidation (OH) & Catalysis (Mn)

Natural Sulfate Aerosols – Sulfuric Acid H 2 SO 4 – Organo - Sulfate – Mg. SO 4 - Sea Salt (6%) – Ca. SO 4 - Gypsum – Ammonium Compounds • Ammonium Sulfate (NH 4)2 SO 4 • Ammonium Bisulfate (NH 4)HSO 4

Anthropogenic Sulfate Aerosols – Sulfuric Acid H 2 SO 4 – Organo - Sulfate – Ca. SO 4 - Scrubbers, Fugitives – Zn, Fe, Cu SO 4 - Smelting, etc. – Ammonium Compounds • Ammonium Sulfate (NH 4)2 SO 4 • Ammonium Bisulfate (NH 4)HSO 4

Nitrate Aerosols – Reversible Reactions – T, RH, Pp (NH 3, HNO 3) • Generally Winter - Cold, Wet • Ammonia Forcing (Chino example) • Reactions (e. g. Sea Salt) • Growth on Pre-existing Particles

Secondary Hydrocarbons – Phase Determinants • Concentration • Temperature • O 3 Catalysis – Episodic in Ozone Season
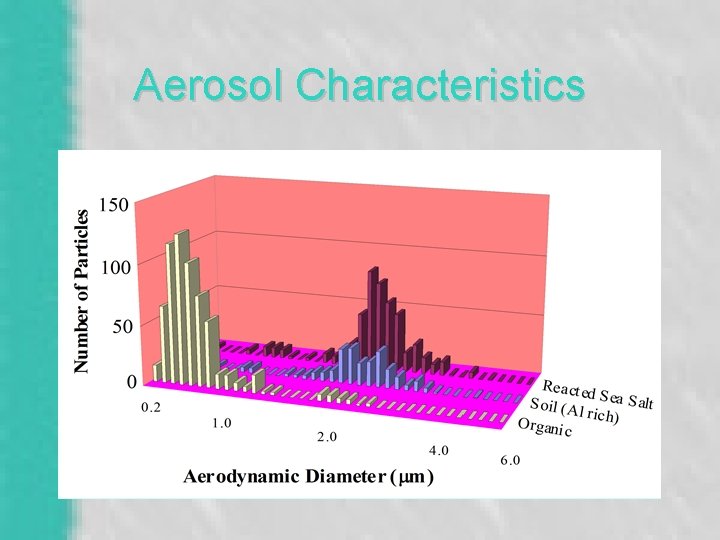
Aerosol Characteristics
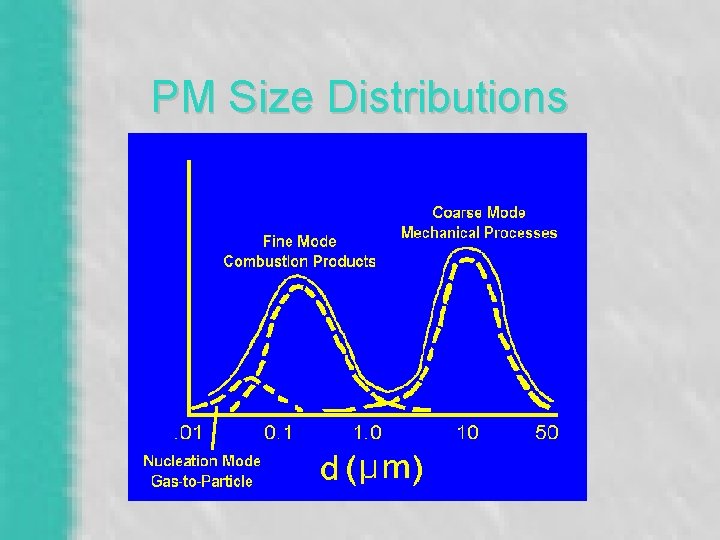
PM Size Distributions
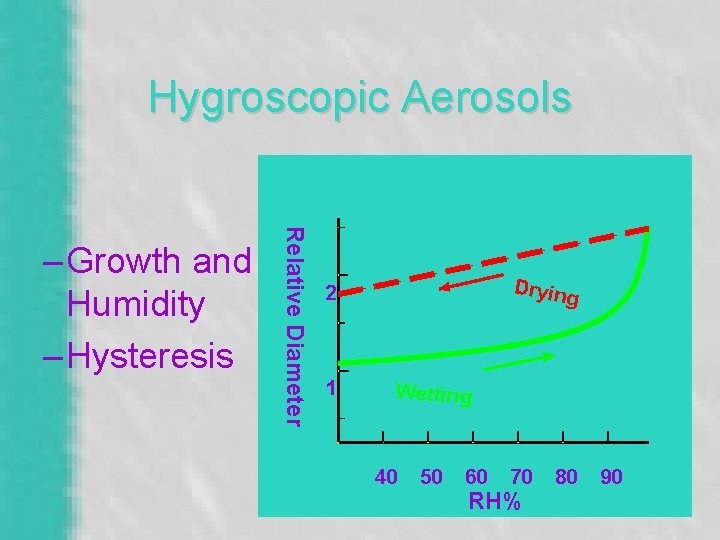
Hygroscopic Aerosols Relative Diameter – Growth and Humidity – Hysteresis Drying 2 1 Wetting 40 50 60 70 RH% 80 90
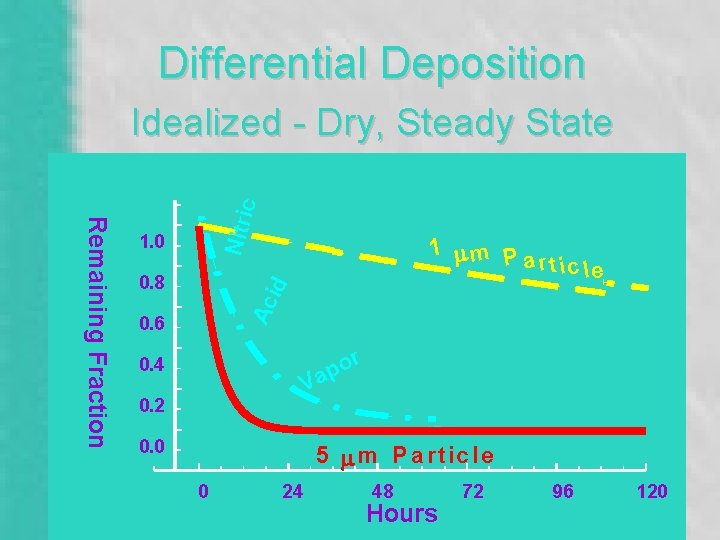
Differential Deposition 1. 0 id 0. 8 1 mm P a rtic le Ac Remaining Fraction Nitri c Idealized - Dry, Steady State 0. 6 r o ap 0. 4 V 0. 2 0. 0 5 m m P a r tic le 0 24 48 Hours 72 96 120
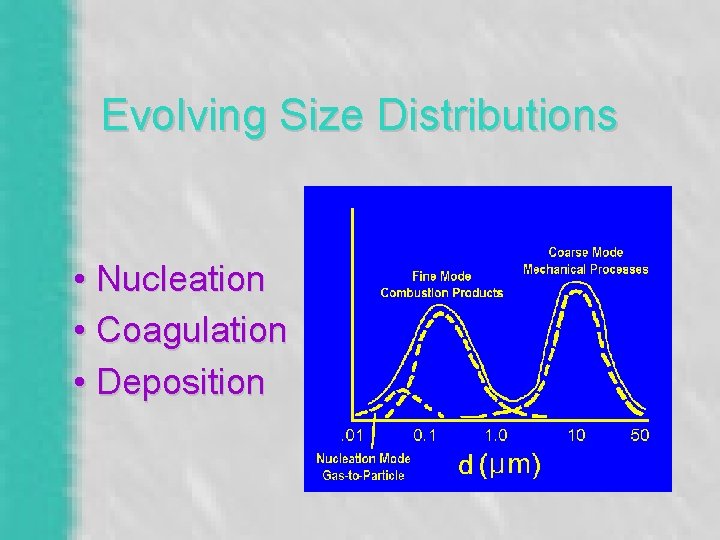
Evolving Size Distributions • Nucleation • Coagulation • Deposition

Size - Origin Relationships – Primary • Mechanical Particles • Weathering Particles • Combustion Particles – Secondary • Gas-Phase Nucleation • Dry Particle Growth • Aqueous Conversion

Clouds, Fog & Rain – Stimulated Condensation – Selective Rain-out • Cloud Scavenging • Inertial Scavenging – Evaporation & Residual Aerosol • Grand Canyon Example

Aerosol History – Aerosol - Emission Relationships are Non-Linear – Air Mass History Determines Aerosol Properties

Deposition & Transport
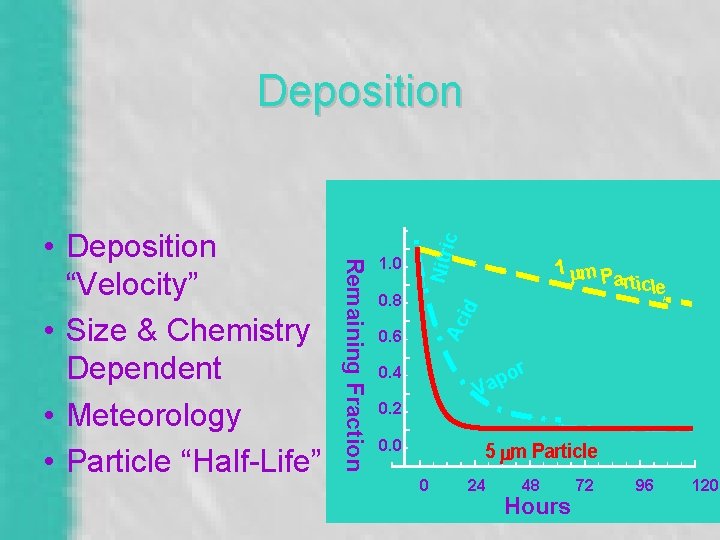
1. 0 0. 8 1 mm P article Ac id Remaining Fraction • Deposition “Velocity” • Size & Chemistry Dependent • Meteorology • Particle “Half-Life” Nitri c Deposition 0. 6 or p Va 0. 4 0. 2 0. 0 5 mm Particle 0 24 48 Hours 72 96 120
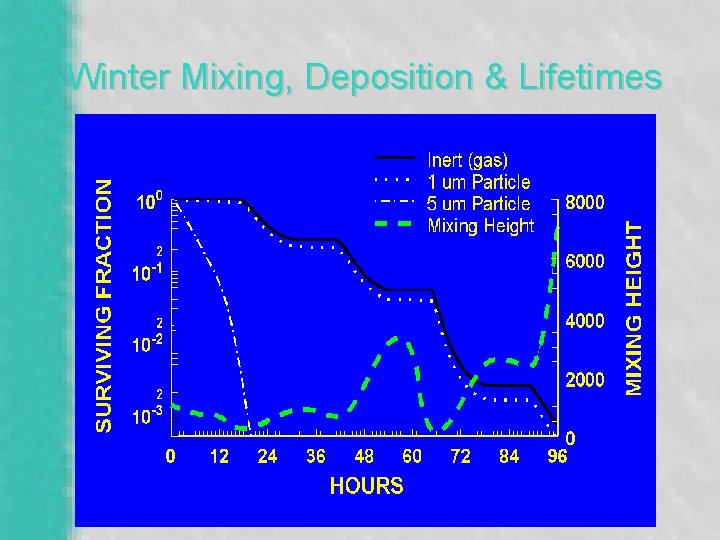
Winter Mixing, Deposition & Lifetimes

Transport – Source Areas & Receptors – Prevailing Winds – Mixing Height & Topography – Distance, Time, and Aerosol Evolution • Secondary Formation / Dissociation • Dispersion • Deposition

ACID DEPOSITION “ACID RAIN”

Acid Formation – Sulfuric Acid – Nitric Acid – Organic Acids

Acid Transport & Deposition – Acid Rain • Local or Upwind Emissions • Cloud Scavenging and Precipitation • Acid Fog – Dry Deposition • Upwind Emissions • Wet or Dry Oxidation • Concentration and Residence
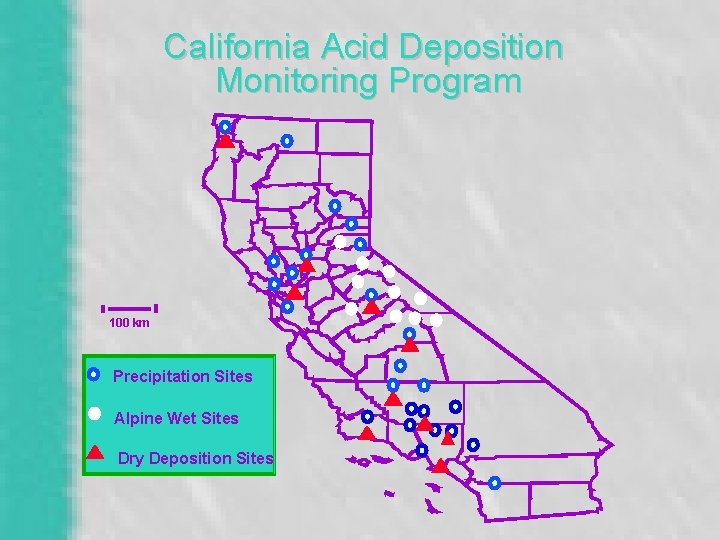
California Acid Deposition Monitoring Program 100 km Precipitation Sites Alpine Wet Sites Dry Deposition Sites
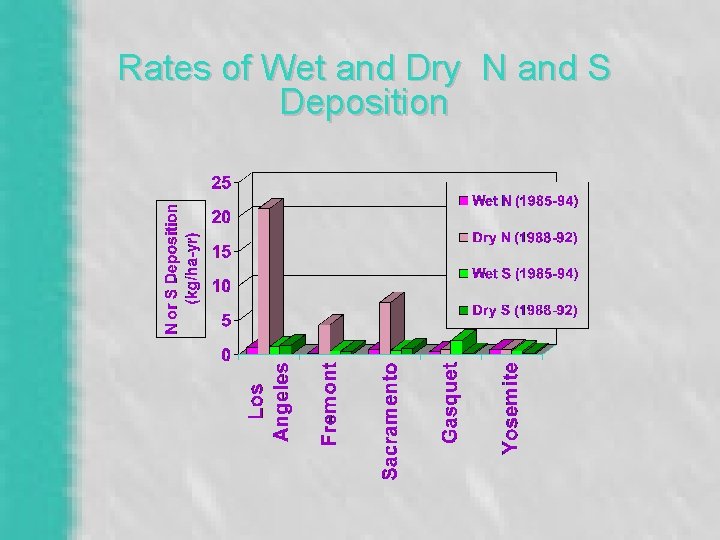
Rates of Wet and Dry N and S Deposition

Annual Dry Deposition 1988 -92

Urban NOx Emissions & Wet and Dry N Deposition

Acid Deposition in California – Nitric Acid Dominates – Patterns of Deposition • Local & Upwind Population – Sensitivity & Buffering • Soils & Water • Lichens, Fish, Amphibians – Resources at Risk • Alpine Lakes

Visibility-Reducing Particles

Vision, Haze & Extinction – Bext = Scatter + Absorption – = Particle + Gas – = Bsp + Bap + Bsg + Bag etc. –

“Extinction” – Bouguer’s Law (1760) – I = I 0 * e -(Bext * r) – Absorption (Babs) • Chemical Composition & Mass (PM 2. 5) – Scattering (Bscat) • Strong Size Dependence - Mie Theory – Rayleigh Scattering (Bsg)
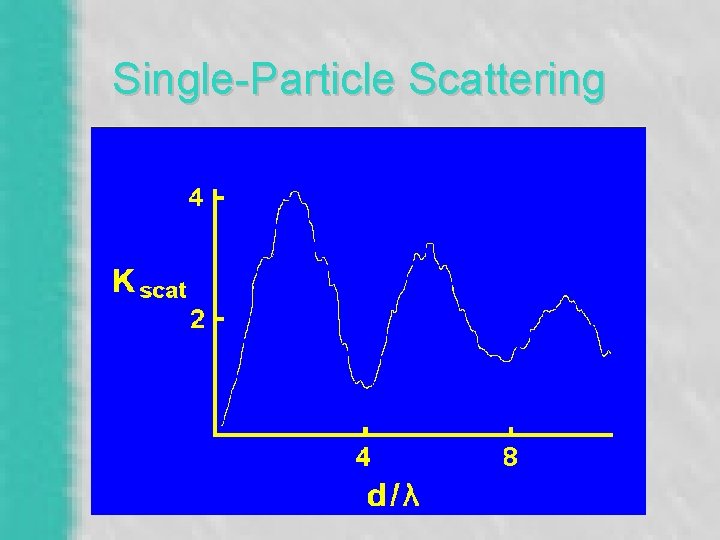
Single-Particle Scattering
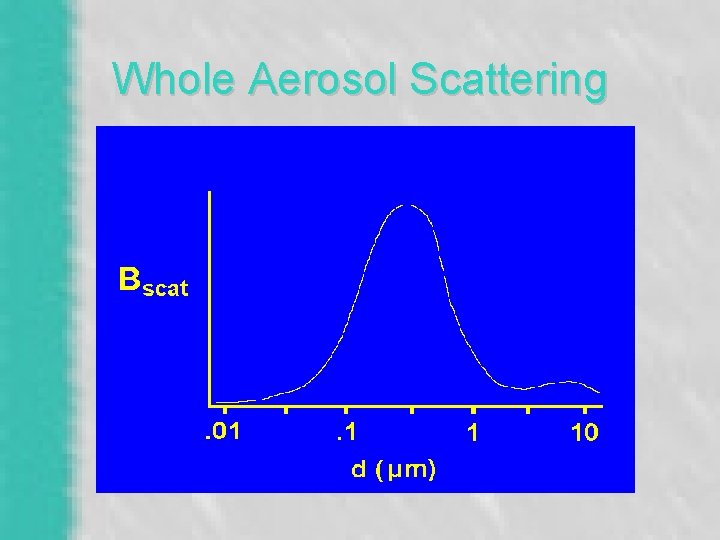
Whole Aerosol Scattering

“Visual Range” – Human Observation of “the greatest distance at which a large dark object can be discerned against the background sky around the majority of the horizon. ” – Nominal: I = 0. 02 I 0 (2% contrast) – Koschmieder’s Law: Vr = 3. 912/Bext (2%) – Real Targets and Observers 3% to 10% – Conditions Rarely Met in Practice

Scenic Quality Esthetic Appeal vs Liminal Perception

Visibility in the US.
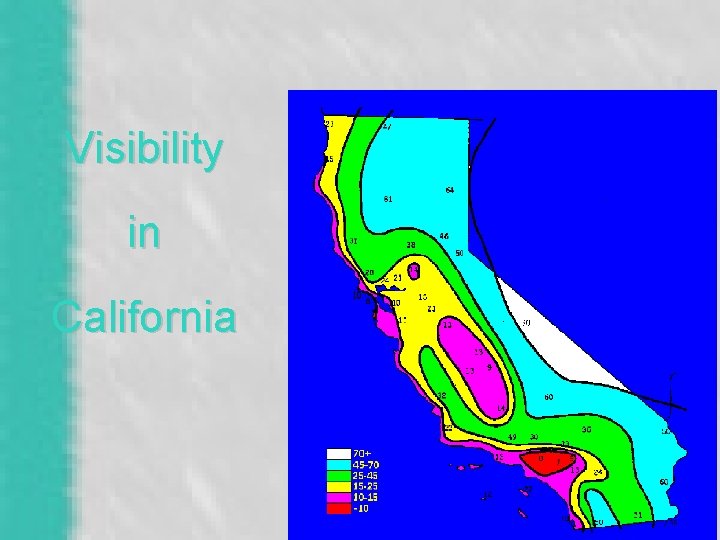
Visibility in California

Variation of PM & Visibility – Seasonal – Diurnal – Transient vs Persistent – Coarse vs Fine
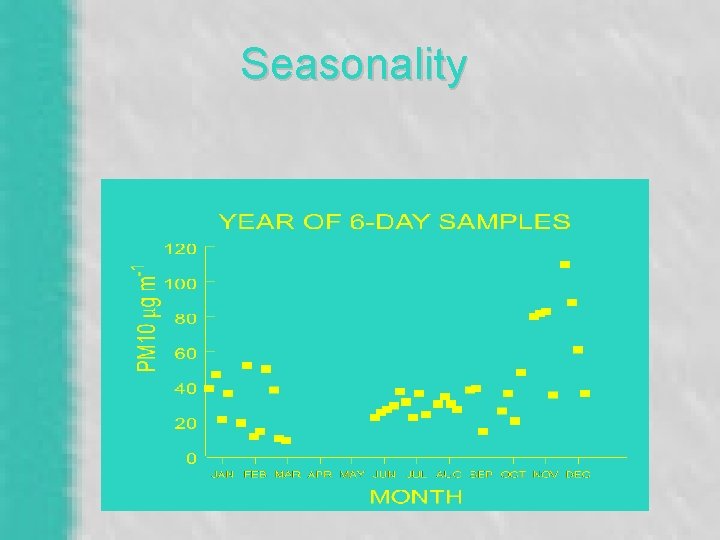
Seasonality

Synoptic “Cycles”
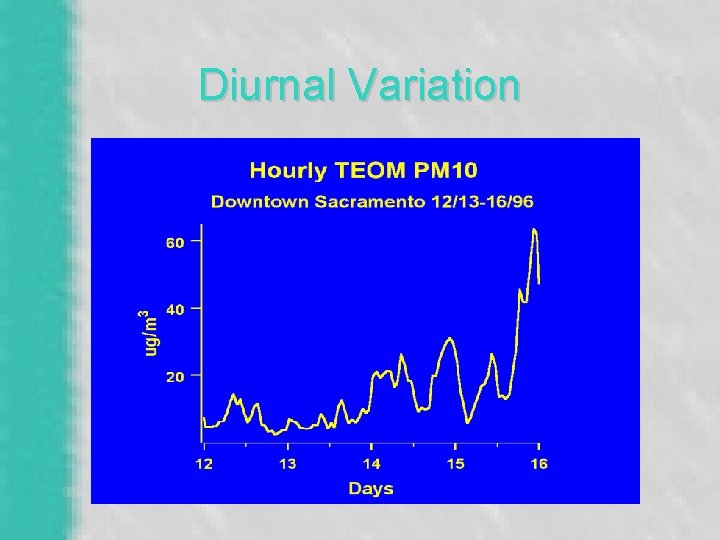
Diurnal Variation

Diurnal Cycles
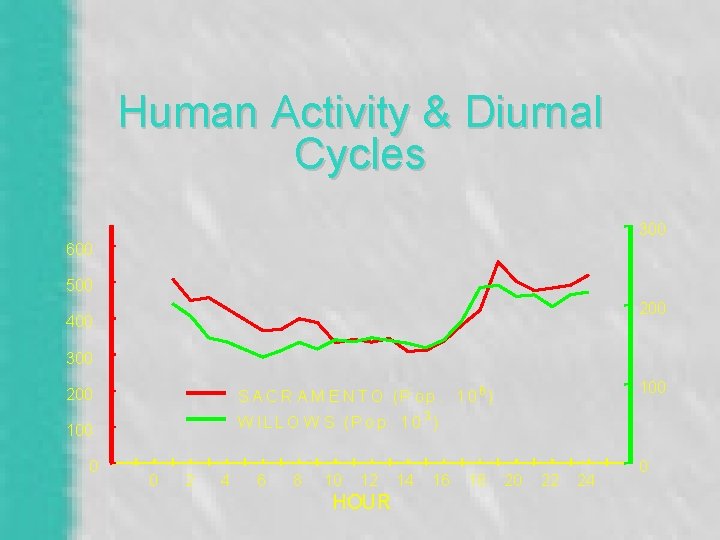
Human Activity & Diurnal Cycles 300 600 500 200 400 300 W IL L O W S ( P o p. 1 0 3 ) 100 0 100 S A C R A M E N T O (P o p. 1 0 6 ) 200 0 2 4 6 8 10 12 HOUR 14 16 18 20 22 24 0

SUMMARY • Sources • Primary & Secondary • Non-Linearity • Regional vs Local Effects • Health Effects, Visibility & Acid Deposition

Air Pollution Chemistry Summary

Combustion Chemistry • Chemistry is very complicated • Intermediate compounds of incomplete combustion are very reactive

Atmospheric Reactions • Steady-state ozone concentration would be only 1 ppb without reactive hydrocarbons • Ozone is trending downward for the LA basin

Hydrocarbons • Hydrocarbon reactivity is a very important concept for control strategies

Particles • Particles come from a large variety of primary and secondary sources • During high ozone episodes, secondary particle formation can be quite large compared to primary

Particles (cont’d) • Size distribution changes continuously within an air mass • Need to know aerosol history to understand deposition impacts

Visibility • Visibility reducing particles and ozone are part of the same system • Depends on population, relative humidity, and fire • Has a strong diurnal pattern

Acid Deposition • Lakes and forests are quite sensitive to acid deposition

Toxic Air Contaminants • Most contaminants are on the list due to their propensity to cause cancer • Major contaminants from mobile sources are: 1, 3 -butadiene, benzene, and benzo[a]pyrene

Toxic Air Contaminants (Cont’d) • Most of vapor-phase mutagenicity in the LA basin comes from nitro-naphthalenes from mobile sources

Global Air Pollution • Compounds with global warming potential have to have long lifetimes - same for ozone depleters

Conclusions • Need to reduce both NOx and hydrocarbon emissions • Control of aldehydes, alkenes, and higher aromatics more effective • RAFs lead to equal O 3 impacts for AFVs • Ozone, particulate matter, acids, and visibility reducing particles part of same chemical system
- Slides: 82