Air Force Research Laboratory Ion Pair Structure and
![Air Force Research Laboratory Ion Pair Structure and Photodissociation Dynamics of Ionic Liquid [emim][Tf Air Force Research Laboratory Ion Pair Structure and Photodissociation Dynamics of Ionic Liquid [emim][Tf](https://slidetodoc.com/presentation_image_h/247a2b3e3c75c9bd96b4514d7e415dc9/image-1.jpg)
Air Force Research Laboratory Ion Pair Structure and Photodissociation Dynamics of Ionic Liquid [emim][Tf 2 N] 19 June 2014 Jaime A. Stearns, Russell Cooper, a David Sporleder, a Alexander Zolotb Space Vehicles Directorate Air Force Research Lab Integrity Service Excellence a. National b. Boston Jerry Boatz Aerospace Systems Directorate Air Force Research Lab Research Council Associate College Institute for Scientific Research 1

Air Force Research Lab • “leading the discovery, development, and integration of warfighting technologies for our air, space, and cyberspace forces” • ~10, 000 government employees and contractors • ~$2 billion budget • 8 technical directorates + AFOSR • Space Vehicles (1000 people, $378 million) • Aerospace Systems (1800 people) • • Boston College National Research Council Collaborations with academia Space Scholar Program 2
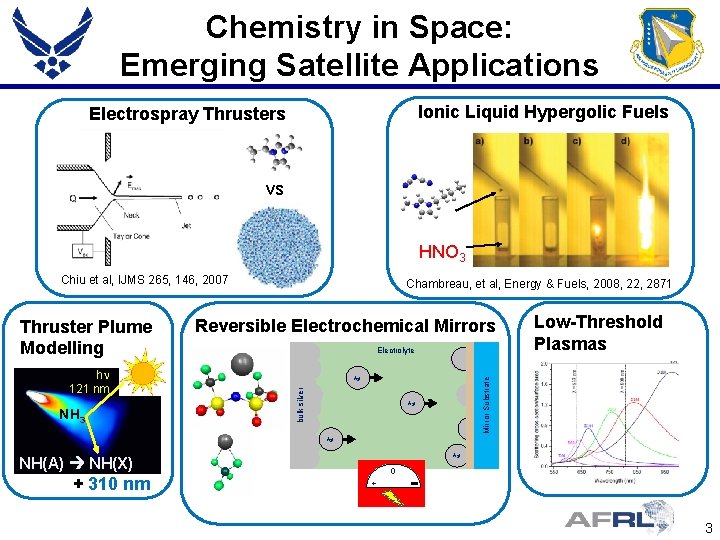
Chemistry in Space: Emerging Satellite Applications Ionic Liquid Hypergolic Fuels Electrospray Thrusters vs HNO 3 Chiu et al, IJMS 265, 146, 2007 NH 3 Electrolyte Ag+ A g + Ag+ NH(A) NH(X) + 310 nm Low-Threshold Plasmas Mirror Substrate hn 121 nm Reversible Electrochemical Mirrors bulk silver Thruster Plume Modelling Chambreau, et al, Energy & Fuels, 2008, 22, 2871 0 + 3
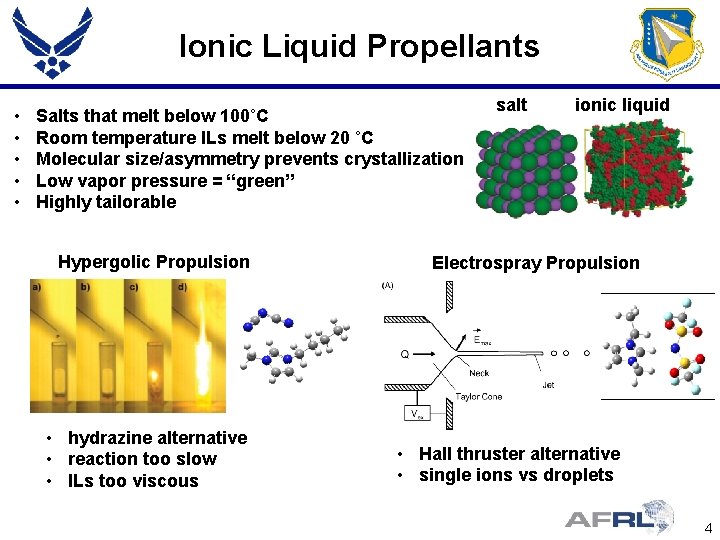
Ionic Liquid Propellants • • • Salts that melt below 100˚C Room temperature ILs melt below 20 ˚C Molecular size/asymmetry prevents crystallization Low vapor pressure = “green” Highly tailorable Hypergolic Propulsion • hydrazine alternative • reaction too slow • ILs too viscous salt ionic liquid Electrospray Propulsion • Hall thruster alternative • single ions vs droplets 4
![[emim][Tf 2 N] • Typical IL tested in EP systems • High decomposition temp: [emim][Tf 2 N] • Typical IL tested in EP systems • High decomposition temp:](http://slidetodoc.com/presentation_image_h/247a2b3e3c75c9bd96b4514d7e415dc9/image-5.jpg)
[emim][Tf 2 N] • Typical IL tested in EP systems • High decomposition temp: Tonset=455˚C [1] • Vapor pressure: 4 x 10 -4 torr @201˚C [2] • Interesting computational question [3] MP 2 minimum (BE=355 k. J/mol) DFT minimum (BE=315 k. J/mol) [1] Ngo, H. L. , et al. Thermochim. Acta 2000, 357 -358, 97 -102. [2] Zaitsau, D. H. , et al. J. Phys. Chem. A 2006, 110, 7303 -7306. [3] Boatz, J, personal communication 5
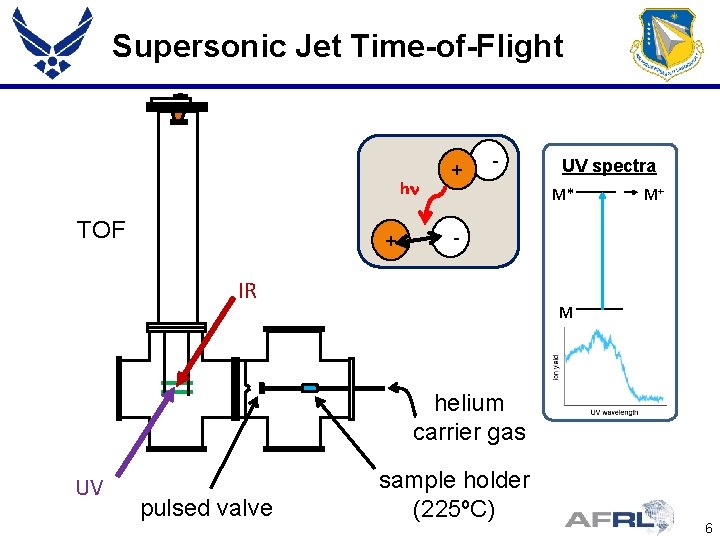
Supersonic Jet Time-of-Flight hn TOF + + - UV spectra M* M+ - IR M helium carrier gas UV pulsed valve sample holder (225ºC) 6
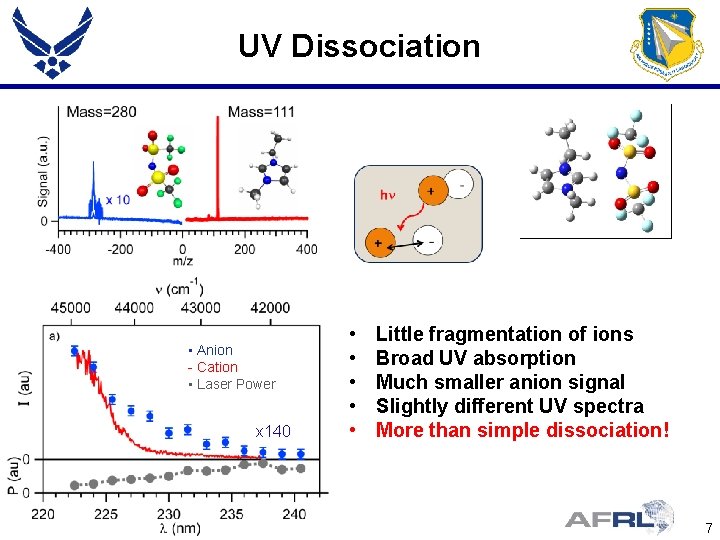
UV Dissociation • Anion - Cation • Laser Power x 140 • • • Little fragmentation of ions Broad UV absorption Much smaller anion signal Slightly different UV spectra More than simple dissociation! 7
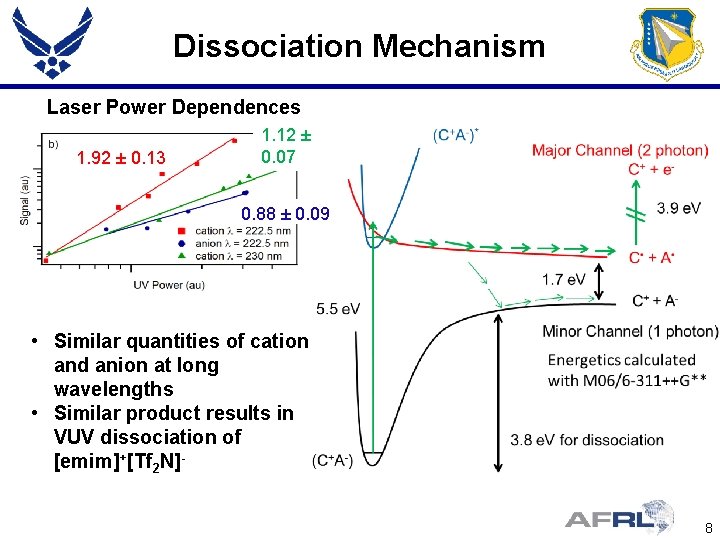
Dissociation Mechanism Laser Power Dependences 1. 92 ± 0. 13 1. 12 ± 0. 07 0. 88 ± 0. 09 • Similar quantities of cation and anion at long wavelengths • Similar product results in VUV dissociation of [emim]+[Tf 2 N]8
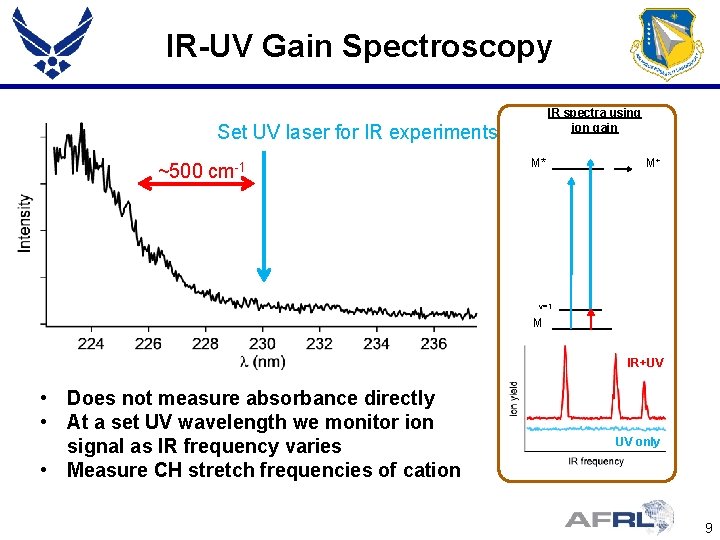
IR-UV Gain Spectroscopy IR spectra using ion gain Set UV laser for IR experiments ~500 cm-1 M* M+ v=1 M IR+UV • Does not measure absorbance directly • At a set UV wavelength we monitor ion signal as IR frequency varies • Measure CH stretch frequencies of cation UV only 9
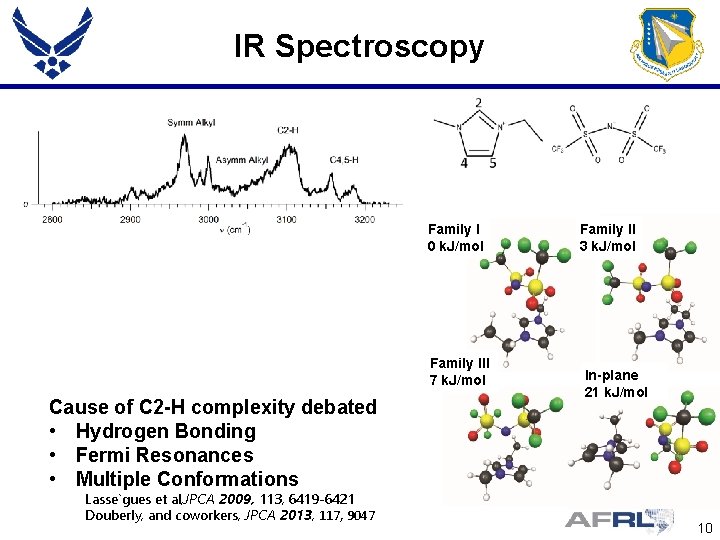
IR Spectroscopy Family I 0 k. J/mol Family III 7 k. J/mol Cause of C 2 -H complexity debated • Hydrogen Bonding • Fermi Resonances • Multiple Conformations Lasse gues et al, JPCA 2009, 113, 6419 -6421 Douberly, and coworkers, JPCA 2013, 117, 9047 Family II 3 k. J/mol In-plane 21 k. J/mol 10
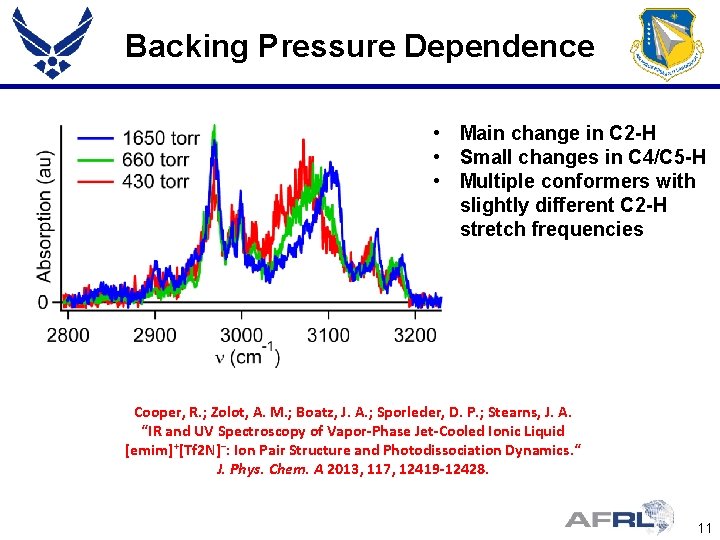
Backing Pressure Dependence • Main change in C 2 -H • Small changes in C 4/C 5 -H • Multiple conformers with slightly different C 2 -H stretch frequencies Cooper, R. ; Zolot, A. M. ; Boatz, J. A. ; Sporleder, D. P. ; Stearns, J. A. “IR and UV Spectroscopy of Vapor-Phase Jet-Cooled Ionic Liquid [emim]+[Tf 2 N]−: Ion Pair Structure and Photodissociation Dynamics. “ J. Phys. Chem. A 2013, 117, 12419 -12428. 11
![Conclusions • [emim][Tf 2 N] has two photodissociation mechanisms • IR spectra point to Conclusions • [emim][Tf 2 N] has two photodissociation mechanisms • IR spectra point to](http://slidetodoc.com/presentation_image_h/247a2b3e3c75c9bd96b4514d7e415dc9/image-12.jpg)
Conclusions • [emim][Tf 2 N] has two photodissociation mechanisms • IR spectra point to stacked structure • B 3 LYP incorrectly predicts in-plane structure • Significant interaction between C 2 -H and anion O • Complexity arises from multiple conformers Current work: linking structure with viscosity h = 21. 0 c. P h = 74. 0 c. P 12
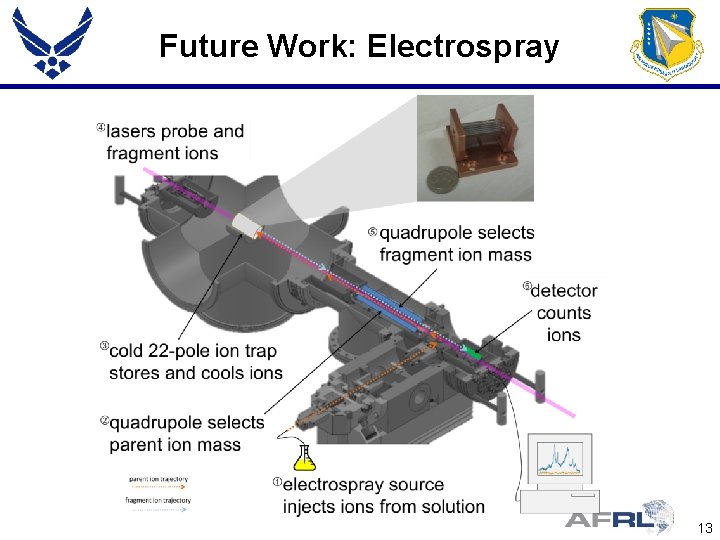
Future Work: Electrospray 13
Acknowledgements AFOSR • Dr. Russell Cooper • Dr. Alex Zolot • Dr. Jerry Boatz • Dr. Ryan Booth • Dr. David Sporleder National Research Council Boston College Institute for Scientific Research Space Scholars Program 14
- Slides: 14