Air Conditioners 1 Air Conditioners Air Conditioners 2




























- Slides: 29

Air Conditioners 1 Air Conditioners

Air Conditioners 2 Introductory Question n If you operate a window air conditioner on a table in the middle of a room, the average temperature in the room will A. become colder become hotter stay the same B. C.

Air Conditioners 4 5 Questions about Air Conditioners Why doesn’t heat flow from cold to hot? n Why does an air conditioner need electricity? n How does an air conditioner cool room air? n What role does the electricity play? n How does an air conditioner heat outdoor air? n

Air Conditioners 5 Question 1 n Why doesn’t heat flow from cold to hot? Does such heat flow violate the laws of motion? n Does such heat flow violate some other laws? n

Air Conditioners 6 Laws Governing Heat Flow n The four laws of thermodynamics are the rules governing thermal energy flow n and establish the relationships between n n disordered (thermal) energy and ordered energy n heat and work

Air Conditioners 7 0 th Law of Thermodynamics The law about thermal equilibrium: “If two objects are in thermal equilibrium with a third object, then they are in thermal equilibrium with each other. ” n

Air Conditioners 8 1 st Law of Thermodynamics The law about conservation of energy “Change in internal energy equals heat in minus work out” n where: n Internal energy: thermal + stored energies n Heat in: heat transferred into object n Work out: external work done by object n

Air Conditioners 9 Order versus Disorder n Converting ordered energy into thermal energy involves events that are likely to occur n is easy to accomplish and often happens n n Converting thermal energy into ordered energy involves events that are unlikely to occur n is hard to accomplish and effectively never happens n n Statistically, ordered always becomes disordered

Air Conditioners 10 Entropy n Entropy is the measure of an object’s disorder n Includes both thermal and structural disorders An isolated system’s entropy never decreases n But entropy can move or be transferred n

Air Conditioners 11 2 nd Law of Thermodynamics n The law about disorder (entropy) “Entropy of a thermally isolated system never decreases”

Air Conditioners 12 3 rd Law of Thermodynamics n The law about entropy and temperature “An object’s entropy approaches zero as its temperature approaches absolute zero”

Air Conditioners 13 More on the 2 nd Law n According to the 2 nd Law: Entropy of a thermally isolated system can’t decrease n But entropy can be redistributed within the system n Part of the system can become hotter while another part becomes colder! n n Exporting entropy is like throwing out trash!

Air Conditioners 14 Natural Heat Flow One unit of thermal energy is more disordering to a cold object than to a hot object n When heat flows from hot object to cold object, n n the hot object’s entropy decreases and the cold object’s entropy increases, n so the overall entropy of the system increases n and total energy is conserved n n Laws of motion and thermodynamics satisfied

Air Conditioners 15 Unnatural Heat Flow n When heat flows from cold object to hot object, the cold object’s entropy decreases, n and the hot object’s entropy increases n so the overall entropy of the system decreases n although total energy is conserved n n The 2 nd law of thermodynamics is violated To save 2 nd law, we need more entropy! n Ordered energy must become disordered energy! n

Air Conditioners 16 Question 2 n Why does an air conditioner need electricity?

Air Conditioners 17 Heat Machines n Air conditioners use work to transfer heat from cold to hot n are a type of “heat pump” n

Air Conditioners 18 Air conditioners (Part 1) n An air conditioner moves heat from cold room air to hot outside air n moves heat against its natural flow n must convert ordered energy into disordered energy n so as not to decrease the world’s total entropy! n uses a “working fluid” (chlorofluorocarbons –freon -, hydrofluorocarbons)to transfer heat n n This fluid absorbs heat from cool room air n This fluid releases heat to warm outside air

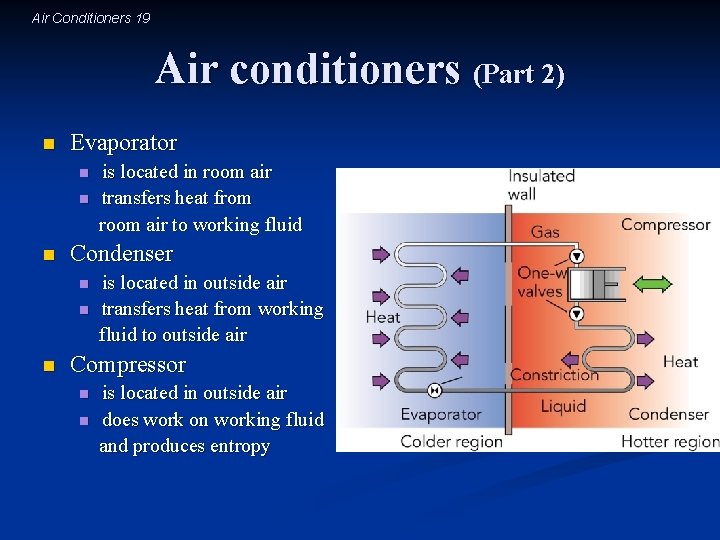
Air Conditioners 19 Air conditioners (Part 2) n Evaporator n n n Condenser n n n is located in room air transfers heat from room air to working fluid is located in outside air transfers heat from working fluid to outside air Compressor n n is located in outside air does work on working fluid and produces entropy

Air Conditioners 20 Question 3 n How does an air conditioner cool room air?

Air Conditioners 21 The Evaporator (Part 1) n The evaporator is a long, wide metal pipe n n The working fluid n n n pipe is heat exchanger between air and working fluid arrives as a high pressure, room temperature liquid but loses pressure passing through a constriction and enters the evaporator as a low pressure liquid Loss of pressure destabilizes the liquid phase The liquid working fluid begins to evaporate!

Air Conditioners 22 The Evaporator (Part 2) n Working fluid evaporates in the evaporator n n Heat flows from the hot room air to the cold gas Working fluid leaves the evaporator n n n Fluid absorbs thermal energy while evaporating, so it transforms into a cold gas as a low density gas near room temperature and carries away some of the room’s thermal energy Heat has left the room!

Air Conditioners 23 Question 4 n What role does the electricity play?

Air Conditioners 24 The Compressor n n The compressor increases density of a gas Working fluid n n Working fluid leaves the compressor n n n arrives as a low density gas near room temperature, has work done on it by the compressor, and experiences a rise in temperature as a result. as a hot, high density gas and carries away electric energy as thermal energy Ordered energy has become disordered energy!

Air Conditioners 25 Question 5 n How does an air conditioner heat outdoor air?

Air Conditioners 26 The Condenser (Part 1) n The condenser is a long, narrow metal pipe n n The working fluid n n pipe is heat exchanger between air and working fluid arrives as a hot, high density gas but begins to lose heat to the cooler outdoor air Loss of heat destabilizes the gaseous phase The gaseous working fluid begins to condense!

Air Conditioners 27 The Condenser (Part 2) n Working fluid condenses in the condenser n n Working fluid leaves the condenser n n n Fluid releases thermal energy while condensing, so it transforms into a hot liquid and even more heat flows from fluid into outside air as high-pressure room-temperature liquid having released some of the room’s thermal energy Heat has reached the outside air!

Air Conditioners 28 Air Conditioner Overview n Evaporator – located in room air n n Compressor – located in outside air n n transfers heat from room air to working fluid does work on fluid, so working fluid gets hotter Condenser – located in outside air n transfers heat from working fluid to outside air, n n including thermal energy extracted from inside air and thermal energy added by compressor

Air Conditioners 29 Introductory Question (Revisited) n If you operate a window air conditioner on a table in the middle of a room, the average temperature in the room will A. become colder become hotter stay the same B. C.

Air Conditioners 30 Summary about Air Conditioners They pump heat from cold to hot n They don’t violate thermodynamics n They convert ordered energy to thermal energy n