Aim Students will be able to understand why

Aim: Students will be able to understand why gases behave the way they do by examining the Kinetic Molecular Theory Do Now: What is the total number of joules absorbed by 65. 0 g of water when the temperature of the water is raised from 25. 0°C to 40. 0°C?

Do Now q = m. C∆T q = (65. 0 g)(4. 18 J/g·°C)(15. 0°C) q = 4076 J

Gases Have you ever seen a hot air balloon? Have you ever opened a shaken bottle of soda? Why do they work the way they do?

In order to explain WHY gases work the way they do… Scientists have come up with the….

KINETIC MOLECULAR THEORY

The Kinetic Molecular Theory describes the behavior of gases • The KMT (kinetic molecular theory) describes the relationships among pressure, volume, temperature, velocity, frequency, and force of collisions.

Kinetic Molecular Theory • Particles in an ideal gas… – have no volume. – have elastic collisions (there is no loss of KE) – are in constant, random, straight-line motion. – don’t attract or repel each other. – have an avg. KE directly related to Kelvin temperature

Now that you know the laws that make up the Kinetic Molecular Theory • You are ready to understand the relationships that make up the KMT. – Relationship between pressure and # of particles – Relationship of pressure and volume of gas – Relationship of temperature and pressure of gas – Relationship of temperature and volume of gas – Relationship of temperature and velocity

Pressure • What is it? !

Pressure • Pressure is defined as the force the gas exerts on a given area of the container in which it is contained. The SI unit for pressure is the Pascal, Pa. • Pressure = Force Area Units of pressure = atmospheres (atm), torr, millimeters of Mercury (mm. Hg), and kilopascals (k. Pa) Normal atmospheric pressure is 760 torr, 760 mm. Hg, 1 atm, and 101. 3 k. Pa

Pressure vs. Number of gas particles • What happens when you shake a full bottle of soda? What happens when you open it after shaking it?

Pressure vs. # of gas particles The pressure in the soda bottle builds up and if opened, the contents in the bottle rush out.

Pressure vs. # of gas particles Think about it! Would a partially filled bottle of soda create a greater amount of pressure when shaken compared to a full bottle of soda?

Pressure vs. # of gas particles • A filled bottle of soda would create more pressure when shaken due to the number of gas particles present inside and the increased number of collisions between those particles. • Gas particles not only collide with each other, but also with the walls of their container. • As the number of gas particles increases, the pressure increases.

Pressure vs. # of gas particles • Basically, a container with more gas particles would have a greater pressure than a container with very few gas particles. LESS Particles LESS PRESSURE MORE Particles MORE PRESSURE

Boyle’s Law • States that there is an inverse relationship between the volume and pressure of a gas.

Relationship of pressure and volume of a gas: Boyle’s Law • Volume refers to the amount of space an object takes up. • For example, think about this classroom. It’s volume is comfortable for us. Now, what would happen to us (the particles) if the walls began to close in? • Answer this question in terms of pressure!

Pressure vs. Volume: Boyle’s Law • If the walls began to close in, particles would collide more with each other, causing pressure to increase. • So, as volume decreases, pressure increases. PV = k (a constant) P 1 V 1 = P 2 V 2

Example of Boyle’s Law • Suppose that you have 5. 00 liters of a gas at 1. 00 atm pressure, and then you decrease the volume to 2. 00 liters. What is the new pressure?

• Suppose that you have 5. 00 liters of a gas at 1. 00 atm pressure, and then you decrease the volume to 2. 00 liters. What is the new pressure? • • P 1 V 1 = P 2 V 2 (1. 00 atm)(5. 00 liters) = P 2 (2. 00 liters) Divide both sides by 2. 00 liters to find P 2 5. 00 atm·liters = P 2 (2. 00 liters) 2. 00 liters

• Suppose that you have 5. 00 liters of a gas at 1. 00 atm pressure, and then you decrease the volume to 2. 00 liters. What is the new pressure? • 5. 00 atm·liters = P 2 (2. 00 liters) 2. 00 liters • 2. 50 atm = P 2

Relationship of Temperature and Pressure of a Gas: Gay-Lussac’s Law • Recall: the temperature of a substance is the measure of the average kinetic energy of its particles. • As temperature rises, what happens to the kinetic energy? ?

Relationship of Temperature and Pressure of a Gas : Gay-Lussac’s Law • Yes, as temperature rises, the kinetic energy of particles increases. • As kinetic energy increases, the particles hit the walls of the container more frequently and with greater force. • So, as temperature increases, pressure increases.

Relationship of Temperature and Pressure of a Gas: Gay-Lussac’s Law • Pressure is directly proportional to temperature. • P = k. T or P/T = k • P 1 = P 2 T 1 T 2

Example of Gay-Lussac’s Law As you have a tank of gas at 800 torr pressure and a temperature of 250 Kelvin, and it’s heated to 400 Kelvin, what is the new temperature? P 1 = P 2 T 1 T 2

• As you have a tank of gas at 800 torr pressure and a temperature of 250 Kelvin, and it’s heated to 400 Kelvin, what is the new temperature? P 1 = P 2 T 1 T 2 • (800 torr) = P 2 250 K 400 K (800 torr)(400 K) = P 2 = 1280 torr 250 K

Relationship between temperature and volume of a gas – Charles’s Law • At constant pressure (meaning, pressure does not change), as temperature increases, the molecules push harder against the walls of a container causing volume to increase. • As temperature increases, volume increases. V/T = b (where b is a constant) V 1/T 1 = b

Example of Charles’s Law • Suppose you live in Alaska and are outside in the middle of winter where the temperature is -23°C. You blow up a balloon so that it has a volume of 1. 00 liter. You then take it inside your home where the temperature is a toasty 27°C. What is the new volume of the balloon? • Charles’s Law: V 1/T 1 = V 2/T 2

• Suppose you live in Alaska and are outside in the middle of winter where the temperature is -23°C. You blow up a balloon so that it has a volume of 1. 00 liter. You then take it inside your home where the temperature is a toasty 27°C. What is the new volume of the balloon? • V 1/T 1 = V 2/T 2 (temp must be expressed in K) -23°C + 273 = 250 K • (1. 00 liter) = V 2 27°C + 273 = 300 K (250 K) (300 K)

• V 1/T 1 = V 2/T 2 • (1. 00 liter) = V 2 (250 K) (300 K) (1. 00 liter)(300 K) = V 2 = 1. 20 liters 250 K

Relationship of Temperature and Velocity • As temperature increases, the kinetic energy increases. • Velocity refers to the speed of the particles • The higher the temperature, the greater the average velocity of the particles

Gas variables Ø VOLUME (V) Ø AMOUNT (n) – UNITS OF VOLUME (L) – UNITS OF AMOUNT (MOLES) Ø TEMPERATURE (T) – UNITS OF TEMPERATURE (K) Ø PRESSURE (P) – UNITS OF PRESSURE (mm. Hg) – UNITS OF PRESSURE (k. Pa) – UNITS OF PRESSURE (atm) – UNITS OF PRESSURE (torr)

A little review Ø BOYLE’S LAW – PRESSURE & VOLUME – AS P THEN V – AT CONSTANT T, n P 1 V 1 = P 2 V 2

A Little review ØGAY-LUSSAC’S LAW: – TEMPERATURE & PRESSURE – AS P THEN T – AT CONSTANT V, n P 1 T 1 = P 2 T 2

A Little review ØCHARLES’ LAW: – TEMPERATURE & VOLUME – AS T THEN V – AT CONSTANT P, n V 1 T 1 = V 2 T 2

Another step up… Ø If we combine all of the relationships from the 3 laws covered thus far (Boyle’s, Charles’s, and Gay-Lussac’s) we can develop a mathematical equation that can solve for a situation where 3 variables change : PV=k 1 V/T=k 2 P/T=k 3

Combined gas law ØAMOUNT IS HELD CONSTANT ØIS USED WHEN YOU HAVE A CHANGE IN VOLUME, PRESSURE, OR TEMPERATURE P 1 V 1 =k T 1 P 2 V 2 =k T 2

Combined gas law Ø AMOUNT IS HELD CONSTANT Ø IS USED WHEN YOU HAVE A CHANGE IN VOLUME, PRESSURE, OR TEMPERATURE P 1 V 1 P 2 V 2 = T 1 T 2 P 1 V 1 T 2 = P 2 V 2 T 1

Example problem A GAS WITH A VOLUME OF 4. 0 L AT STP. WHAT IS ITS VOLUME AT 2. 0 ATM AND AT 30°C? P 1 1 atm V 1 4. 0 L T 1 273 K P 2 2. 0 atm V 2 ? T 2 30°C + 273 = 303 K
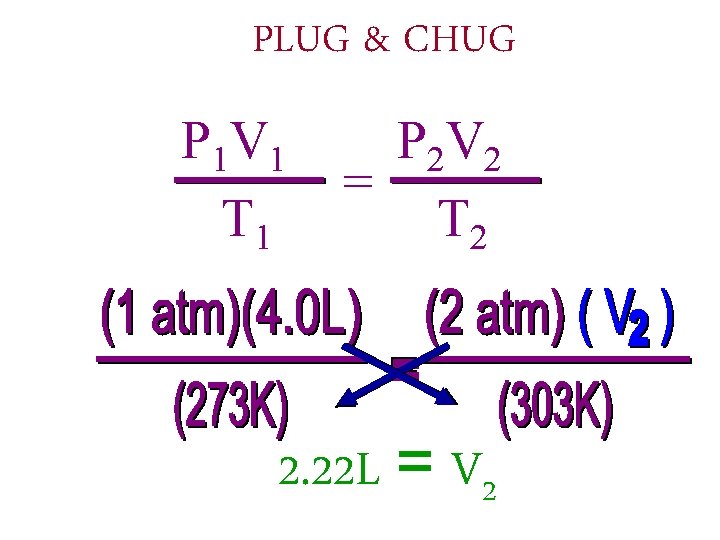
PLUG & CHUG P 1 V 1 T 1 P 2 V 2 = T 2 2. 22 L = V 2

Gas laws deal with one other variable. We have discussed: Pressure Temperature Volume What else do you think would effect HOW a gas behaves?

THE AMOUNT OF PARTICLES OF GAS This leads us to AVOGADRO’S LAW (sounds kind of like “avocado”)
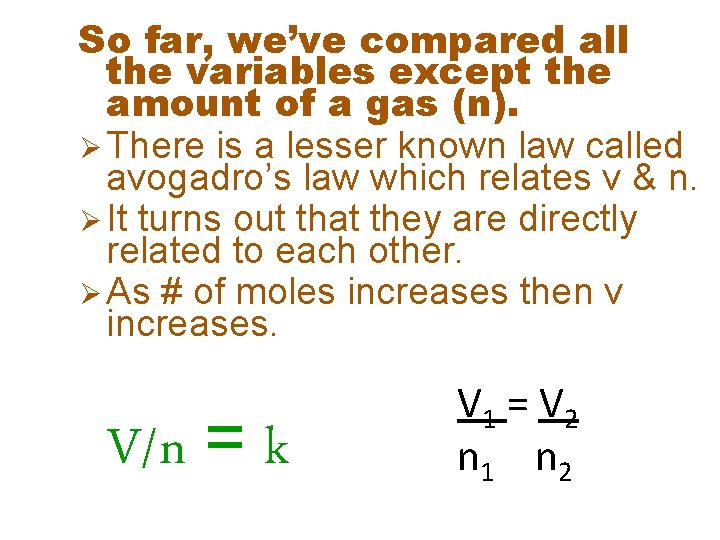
So far, we’ve compared all the variables except the amount of a gas (n). Ø There is a lesser known law called avogadro’s law which relates v & n. Ø It turns out that they are directly related to each other. Ø As # of moles increases then v increases. V/n = k V 1 = V 2 n 1 n 2

This leads us to the:

The Ideal Gas Law • So far we have always held at least one of the variables constant. • Now, we can set up a much more powerful equation which can be derived by combining the proportions provided by the other laws we’ve studied recently.

The Ideal Gas Law Equation PV = n. RT where “R” is the universal gas constant.

The Ideal Gas Law • R is a constant that connects all four variables. • R is dependent on the UNITS of the variables for P, V, and T. • Temp is always in Kelvin Volume is always in Liters Pressure is either in atm, mm. Hg, k. Pa

Ø BECAUSE OF THE DIFFERENT PRESSURE UNITS THERE ARE 3 POSSIBILITIES FOR OUR R L • atm mol • K – IF PRESSURE IS GIVEN IN atm R=. 0821 – IF PRESSURE IS GIVEN IN mm. Hg L • mm. Hg R=62. 4 mol • K L • k. Pa R=8. 314 mol • K – IF PRESSURE IS GIVEN IN k. Pa

Using Ideal gas law EG #1: WHAT VOL DOES 9. 45 g OF C 2 H 2 OCCUPY AT STP?
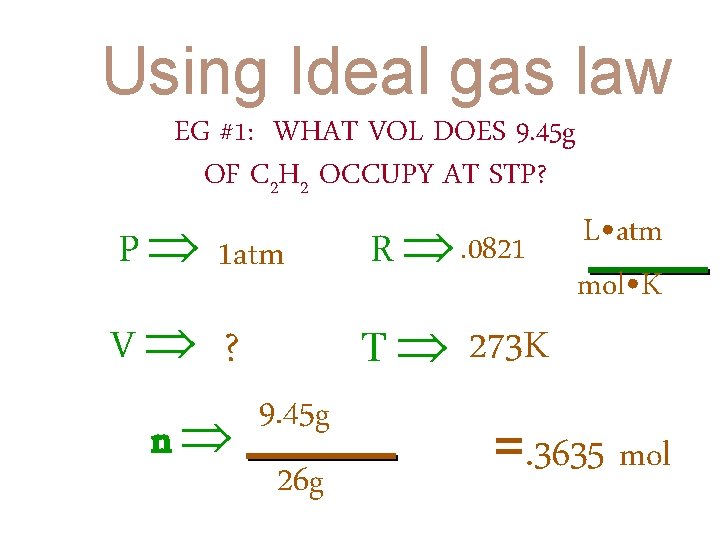
Using Ideal gas law EG #1: WHAT VOL DOES 9. 45 g OF C 2 H 2 OCCUPY AT STP? P 1 atm R . 0821 V ? T 273 K 9. 45 g n 26 g L • atm mol • K =. 3635 mol

PV = n. RT (1. 0 atm) (V) (. 3635 mol) = (. 0821 (1. 0 atm) (V) = L • atm mol • K ) (273 K) (8. 147 L • atm) V = 8. 15 L

Using Ideal gas law EG #2: A CAMPING STOVE PROPANE TANK HOLDS 3000 g OF C 3 H 8. HOW LARGE A CONTAINER WOULD BE NEEDED TO HOLD THE SAME AMOUNT OF PROPANE AS A GAS AT 25°C AND A PRESSURE OF 303 k. Pa?
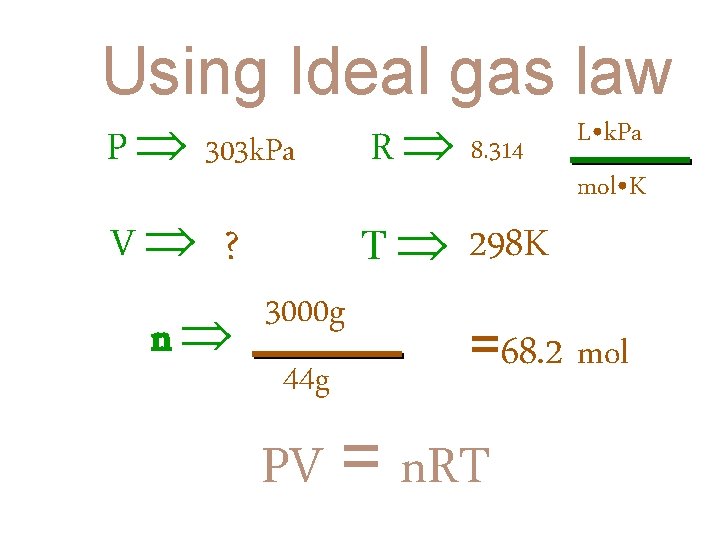
Using Ideal gas law P 303 k. Pa R 8. 314 V ? T 298 K 3000 g n 44 g L • k. Pa mol • K =68. 2 mol PV = n. RT

(303 k. Pa) (V) = (8. 314 (68. 2 mol) (303 k. Pa) (V) = L • k. Pa mol • K ) (298 K) (168, 970. 4 L • k. Pa) V = 557. 7 L

Ideal versus Real Gases • What’s the difference?

What does the term “ideal” mean?

The ideal meal

The ideal weather

The ideal place to be instead of work/school

When something is “ideal” It is the best, perfect option or idea for a particular situation.

Ideal versus Real Gases • The Kinetic Molecular Theory explains the behavior of gases using a model gas called an “ideal” gas. • When the gas laws are used to solve real problems using “real” gases, the results do not always match the results obtained in the lab. • *This is because ideal gas model does not match the behavior of real gases.

Ideal vs. Real

When ideal gases do not match the behavior of real gases It is due to the fact that two of the assumptions made by the KMT are not exactly correct: - gas particles do not attract one another. (in extreme conditions, particles do attract each other) - gas particles do not occupy volume. (while they typically occupy little to no volume, under high pressures, increased concentration of particles means far greater chances of combining)

What makes a gas truly “ideal”? • When it behaves exactly as predicted. • Gases vary from ideal behavior because of 2 factors (increasing mass & increasing polarity) • Gases are most ideal at low pressures and high temperatures.
- Slides: 64