Aim How to read Table I heat of

Aim: How to read Table I heat of reaction? Do Now: Discuss homework answers in pairs.

During the demo, answer the following questions 1. Will Na. OH(s) dissolve in water when mixed? Which table from the reference table do you have to use? 2. What is the initial temperature of the water? 3. What is the final temperature of the water? 4. What happened to the temperature of the water? 5. Did the water go through an exothermic or endothermic process? 6. Where did the water gain the heat from? 7. Go to Table I. Find the reaction that shows Na. OH dissolving in water. How does this equation support your answer to #6.
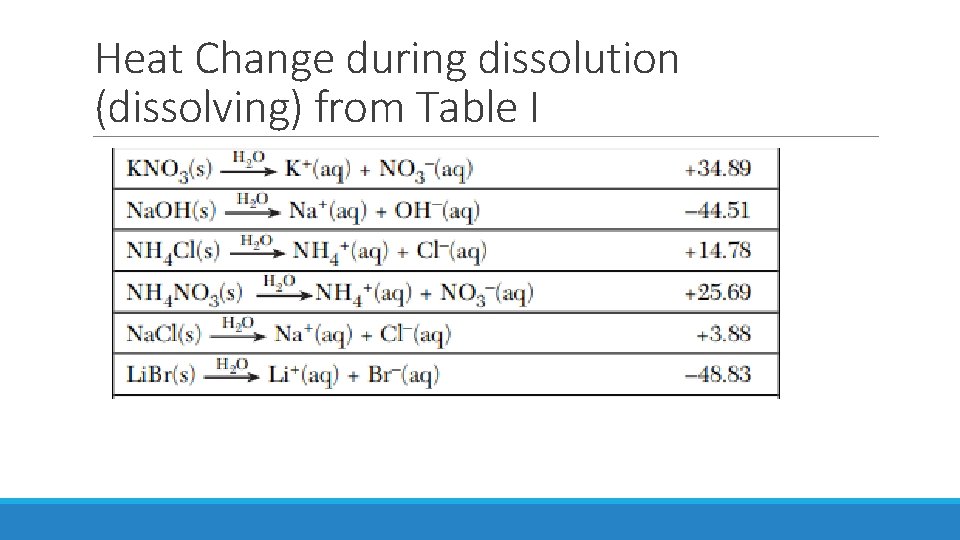
Heat Change during dissolution (dissolving) from Table I
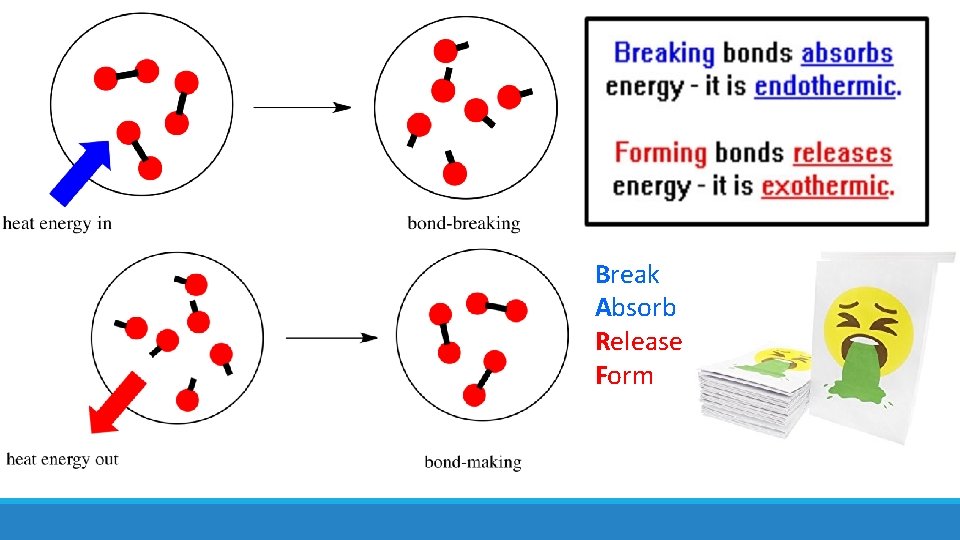
Break Absorb Release Form

Enthalpy (H) is a measure of the heat in the system. The heat released or absorbed in a chemical reaction, heat of reaction, is the same as the change in enthalpy ΔH. ΔH = H (products) – H (reactants) Table I has ΔH for various reactions
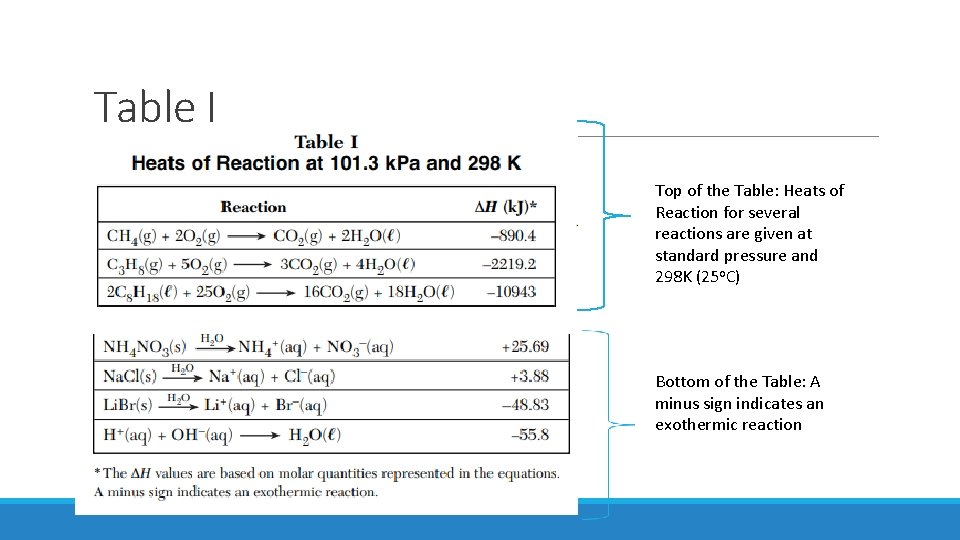
Table I Top of the Table: Heats of Reaction for several reactions are given at standard pressure and 298 K (25 o. C) Bottom of the Table: A minus sign indicates an exothermic reaction

Thermochemical Equations In thermochemical equations, the enthalpy change can be written as either a reactant or a product.

Thermochemical Equations for Endothermic reactions. In an endothermic process, heat is absorbed/used during a chemical reaction. That is the energy of the products is greater than the energy of the reactants. N 2(g) + O 2 (g) ---> 2 NO(g) ; ΔH= +182. 6 KJ Can be written as 182. 6 KJ + N 2(g) + O 2(g) ---> 2 NO(g)

Thermochemical equations for Exothermic Reactions In an exothermic process, heat is released/produced during a chemical reaction. That is the energy of the products is less than the energy of the reactants. CH 4(g) + 2 O 2 (g) --> CO 2 (g) + 2 H 2 O(l) ; ΔH= -890. 4 KJ Can be written as CH 4(g) + 2 O 2(g) – 890. 4 k. J --> CO 2(g) + 2 H 2 O(l) + 890. 4 k. J ◦ CH 4(g) + 2 O 2(g) --> CO 2(g) + 2 H 2 O(l) + 890. 4 KJ

Reverse Reaction Find the heat of reaction for the reverse of the given reaction 2 SO 2 (g) + O 2 (g) 2 SO 3 ΔH= -196 k. J

Important features of ΔH ΔH is directly proportional to the amount of reactant consumed. 2 SO 2 (g) + O 2 (g) 2 SO 3 ΔH= -196 k. J What is ΔH for the reaction of 4 mole of SO 2(g) with 2 mol O 2(g)?

Problem 1 Given the reaction: 2 CO(g) + O 2 (g) 2 CO 2 (g) ΔH = -566. 0 KJ How much heat would be released if 4 moles of carbon monoxide were consumed by oxygen?

Problem 2 Given the reaction: 2 H 2 (g) + O 2(g) 2 H 2 O (l) ΔH = -571. 6 KJ Calculate ΔH for the following reaction H 2(g) + 1/2 O 2(g) H 2 O (l)
- Slides: 13