Aim How to describe the polarity of a
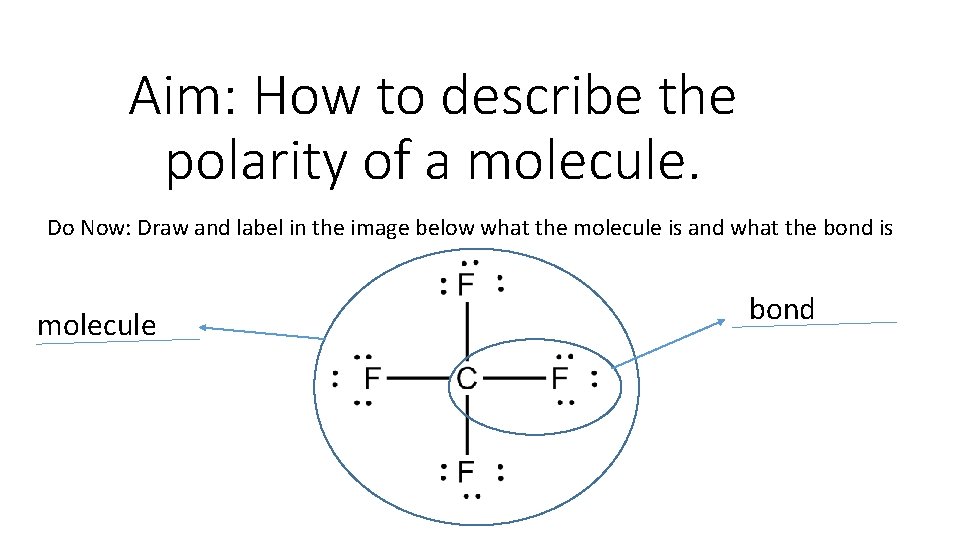
Aim: How to describe the polarity of a molecule. Do Now: Draw and label in the image below what the molecule is and what the bond is molecule bond

Definition of bond and molecule • The force that holds atoms together in collections known as molecules is referred to as a chemical bond. • A chemical bond is the attraction between two atoms.

Polar Molecules • Molecules with a positive and a negative end • Requires two things to be true ¬The molecule must contain polar bonds This can be determined from differences in electronegativity. Asymmetric molecule.

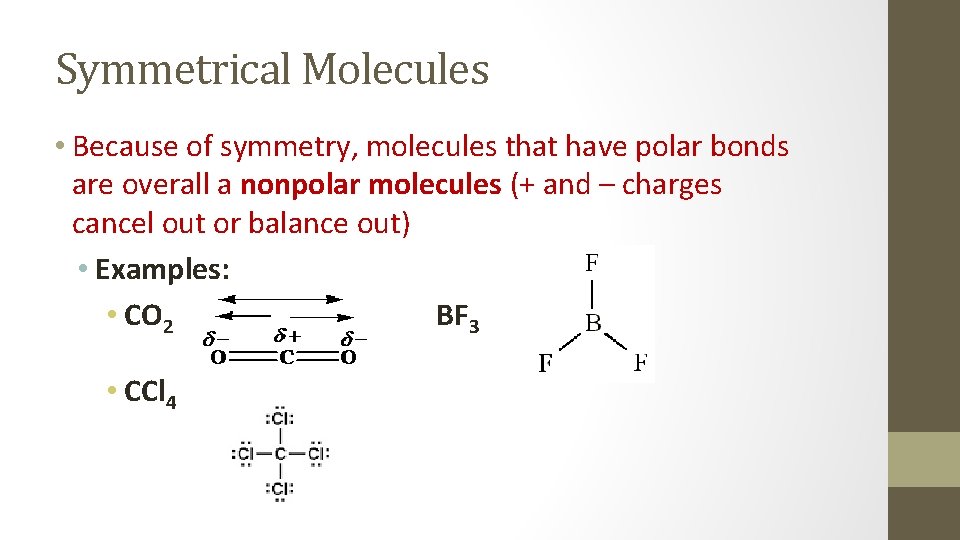
Symmetrical Molecules • Because of symmetry, molecules that have polar bonds are overall a nonpolar molecules (+ and – charges cancel out or balance out) • Examples: • CO 2 BF 3 • CCl 4

Asymmetrical Molecules • If a molecule has polar bonds (and there is no symmetry to cancel out + and – charges), the molecule is polar. • Examples: • H 2 O HCl • NH 3

Dipole l. When there is unequal sharing of electrons a dipole exists l. Dipole is a molecule that has two poles or regions with opposite charges l. A dipole is represented by a dipole arrow pointing towards the more negative end



Is it Polar? (Draw the molecule to determine if its symmetric or asymmetric) • HF • H 2 O • NH 3 • CF 4 • CO 2

Summary • In a polar bond, one atom is more electronegative than the other. • In a nonpolar bond, both atoms have similar electronegativities. • An asymmetric molecule is a polar molecule. • A symmetric molecule is always a nonpolar molecule.
- Slides: 11