Aim How to describe the different types of

Aim: How to describe the different types of solutions

Review how stuff dissolve • How does an ionic compound such as salt dissolve in water? • How does a molecular compound such as sugar dissolve in water
Review temperature and solubility 1. Increasing temperature of a solution ________the solubility of a solid. 2. Increasing temperature of a solution ________ the solubility of a gas. 3. Increasing pressure on a solution ________ the solubility of a gas.

Making Iced Tea • The next time you add sugar to unsweetened tea, take notice of the changes to the solution in the following scenarios. 1. You add the first packet of sugar, and stir until it all dissolves with no sugar settled on the bottom. 2. You add a second packet of sugar and it takes a little more time for the sugar to dissolve. After stirring for a minute, you realize that there are still some sugar crystals at the bottom of the glass. 3. After tasting your tea, you decide it is still not sweet enough and add more sugar. However, it all settles at the bottom, and regardless of how much you stir, the crystals stay at the bottom of the cup. • What can you do to help the sugar crystals dissolve?

Types of Solutions • In each of the three steps above, you created one of the following types of solutions: saturated, unsaturated, and supersaturated

Types of Solutions • Unsaturated Solution- the solution has a small amount of solute dissolved at a certain temperature • More solute can be added to the solution and still dissolve • Saturated Solution – the solution has as much solute as it can hold at a certain temperature • The maximum amount of solute that can completely dissolve in a solution • Supersaturated Solution – the solution has more solute that it can hold at a certain temperature



Supersaturated Solution (this picture is showing the addition of 100 g of glucose to 100 ml of water at 25 0 C) Note: at 250 C, only 91 g of glucose will dissolve in 100 ml of water

Types of Solutions • Return to the steps for making tea. • Label each step as either a saturated, unsaturated, or supersaturated solution
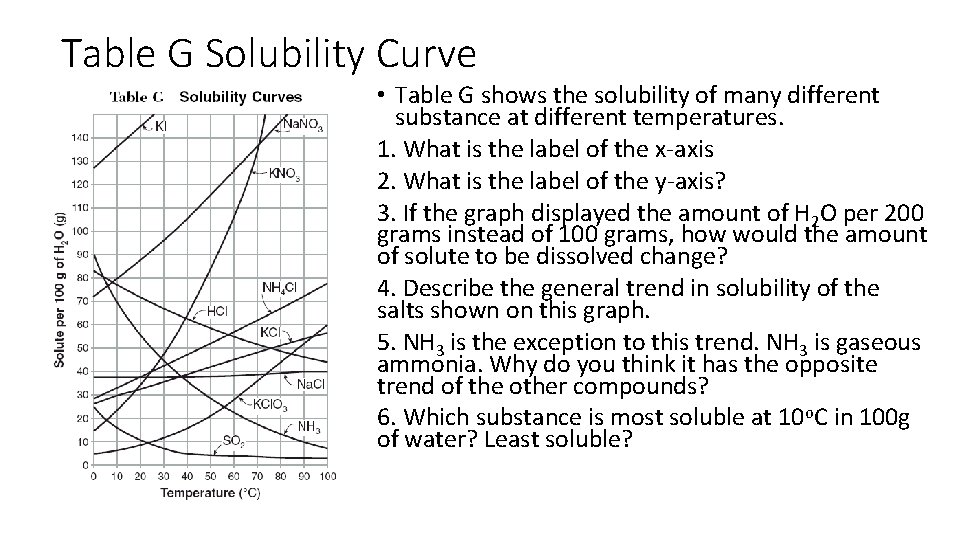
Table G Solubility Curve • Table G shows the solubility of many different substance at different temperatures. 1. What is the label of the x-axis 2. What is the label of the y-axis? 3. If the graph displayed the amount of H 2 O per 200 grams instead of 100 grams, how would the amount of solute to be dissolved change? 4. Describe the general trend in solubility of the salts shown on this graph. 5. NH 3 is the exception to this trend. NH 3 is gaseous ammonia. Why do you think it has the opposite trend of the other compounds? 6. Which substance is most soluble at 10 o. C in 100 g of water? Least soluble?
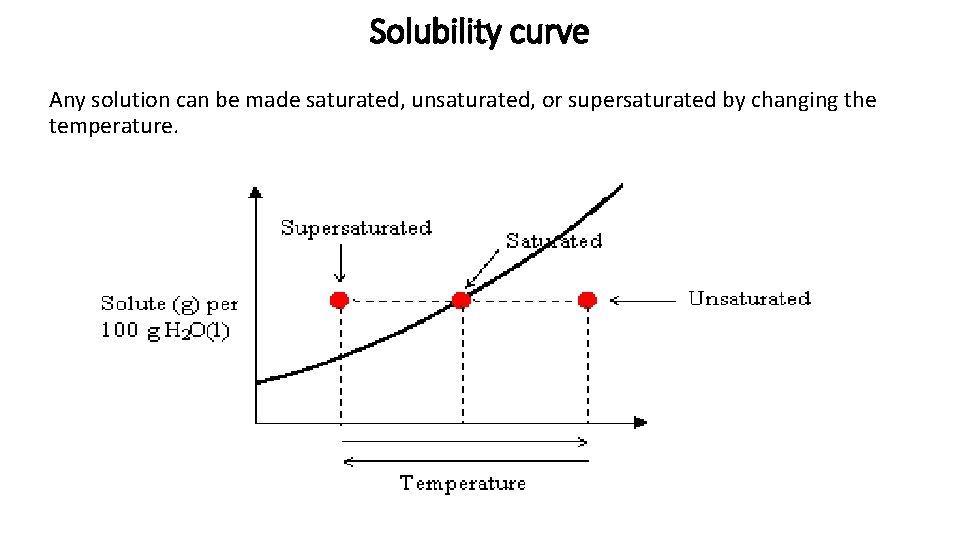
Solubility curve Any solution can be made saturated, unsaturated, or supersaturated by changing the temperature.
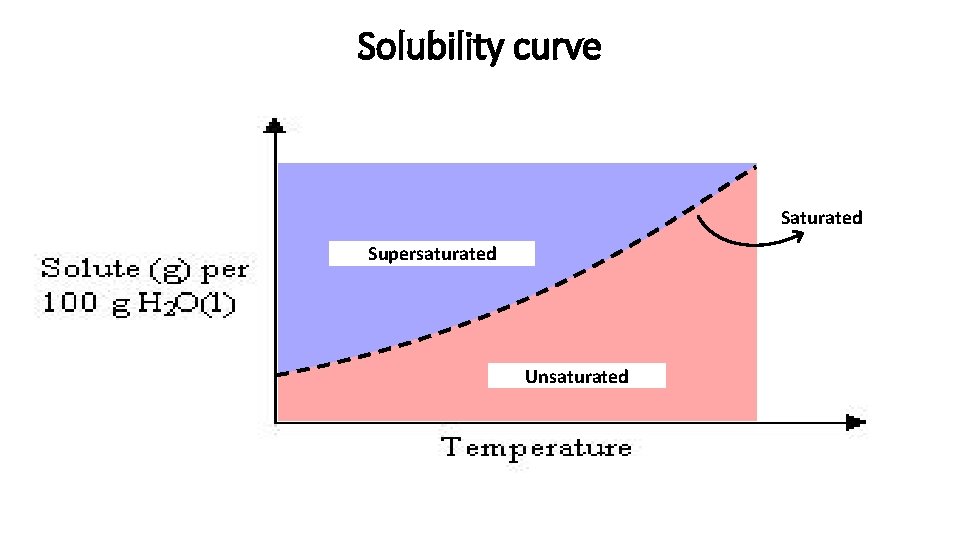
Solubility curve Saturated Supersaturated Unsaturated
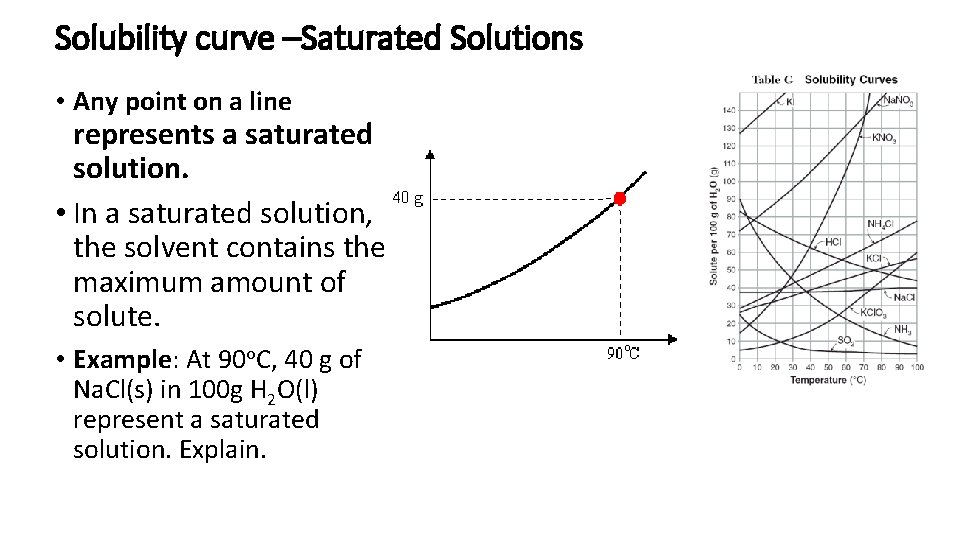
Solubility curve –Saturated Solutions • Any point on a line represents a saturated solution. • In a saturated solution, the solvent contains the maximum amount of solute. • Example: At 90 o. C, 40 g of Na. Cl(s) in 100 g H 2 O(l) represent a saturated solution. Explain.
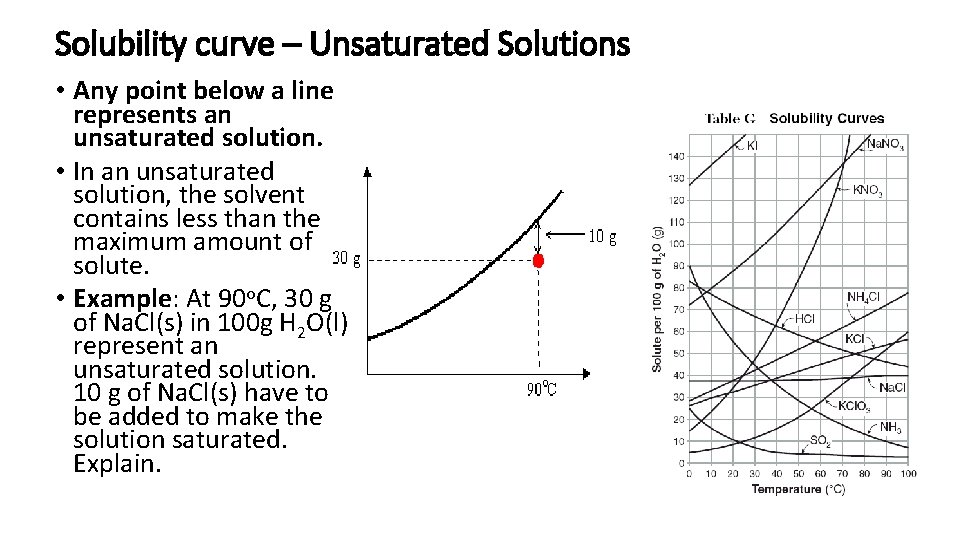
Solubility curve – Unsaturated Solutions • Any point below a line represents an unsaturated solution. • In an unsaturated solution, the solvent contains less than the maximum amount of solute. • Example: At 90 o. C, 30 g of Na. Cl(s) in 100 g H 2 O(l) represent an unsaturated solution. 10 g of Na. Cl(s) have to be added to make the solution saturated. Explain.
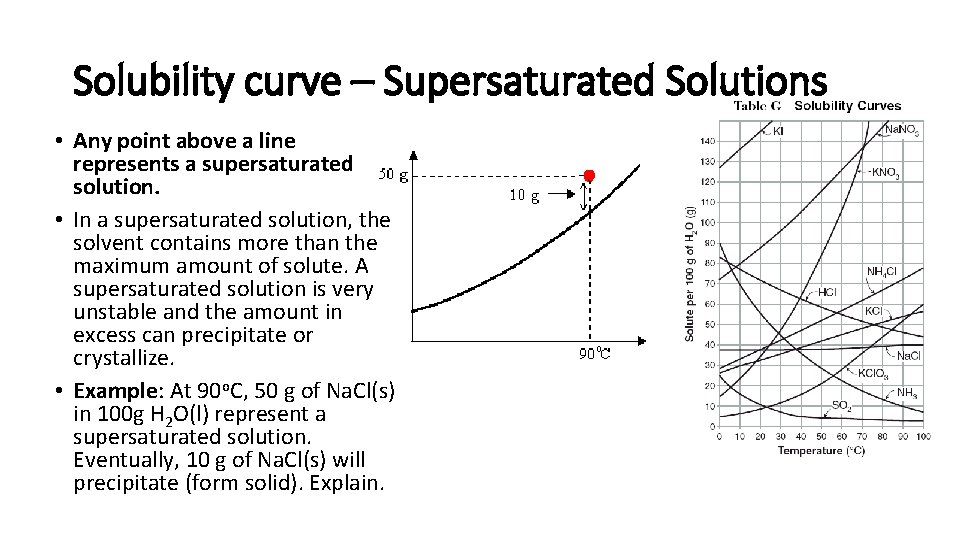
Solubility curve – Supersaturated Solutions • Any point above a line represents a supersaturated solution. • In a supersaturated solution, the solvent contains more than the maximum amount of solute. A supersaturated solution is very unstable and the amount in excess can precipitate or crystallize. • Example: At 90 o. C, 50 g of Na. Cl(s) in 100 g H 2 O(l) represent a supersaturated solution. Eventually, 10 g of Na. Cl(s) will precipitate (form solid). Explain.

How to read a solubility curve • To read a solubility curve, follow these steps 1. Determine what the question is asking. 1. Are you looking for temperature 2. Are you looking fro grams of solute that can be dissolved? 3. Are you deciding if a solution is saturated, unsaturated, or supersaturated? 2. Use the information given in the question to read graph. 3. Use the graph to answer the question.
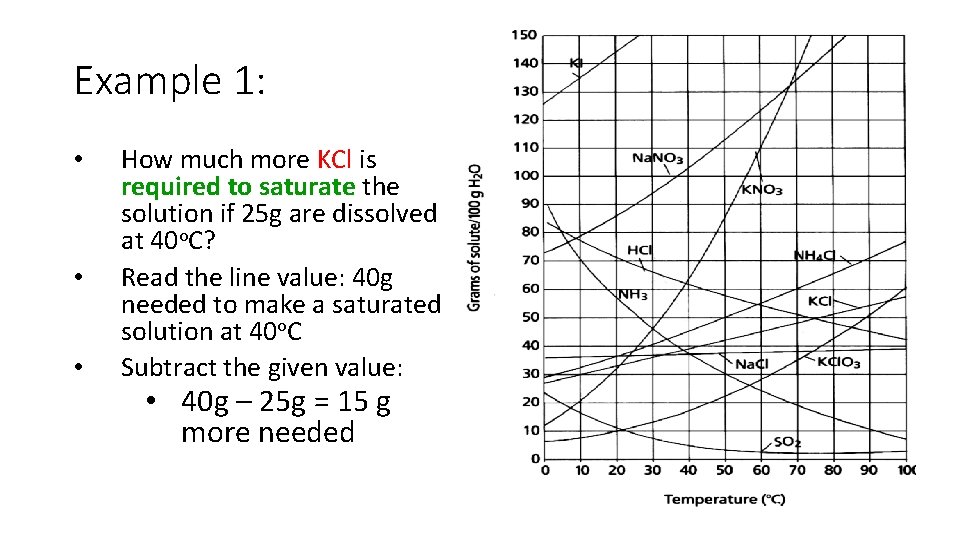
Example 1: • • • How much more KCl is required to saturate the solution if 25 g are dissolved at 40 o. C? Read the line value: 40 g needed to make a saturated solution at 40 o. C Subtract the given value: • 40 g – 25 g = 15 g more needed
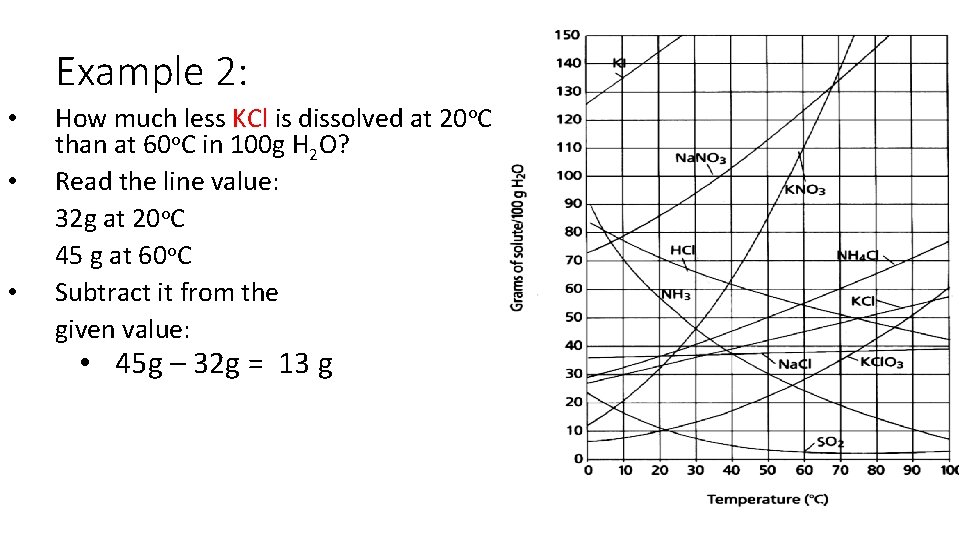
Example 2: • • • How much less KCl is dissolved at 20 o. C than at 60 o. C in 100 g H 2 O? Read the line value: 32 g at 20 o. C 45 g at 60 o. C Subtract it from the given value: • 45 g – 32 g = 13 g
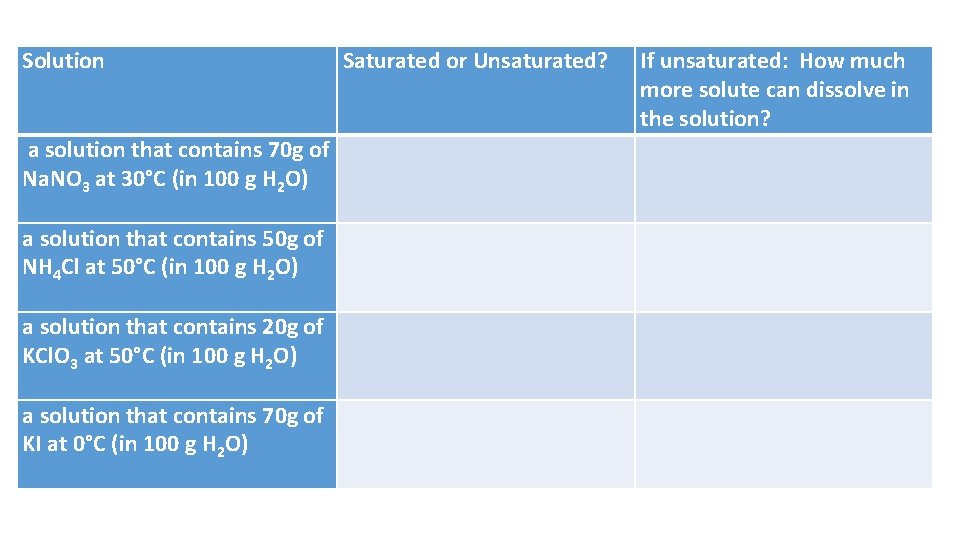
Solution Saturated or Unsaturated? If unsaturated: How much more solute can dissolve in the solution? a solution that contains 70 g of Na. NO 3 at 30°C (in 100 g H 2 O) a solution that contains 50 g of NH 4 Cl at 50°C (in 100 g H 2 O) a solution that contains 20 g of KCl. O 3 at 50°C (in 100 g H 2 O) a solution that contains 70 g of KI at 0°C (in 100 g H 2 O)

Homework. Explain how you got your answer. 1. How many grams of KNO 3 are dissolved in 100 g of H 2 O at 60 o. C? Explain. 2. How many grams of Na. NO 3 would be needed to saturate 200 grams of H 2 O at 40 o. C? Explain. 3. Which solutes vary inversely with temperature? Explain. 4. If 55 grams of potassium chlorate are mixed with 100 grams of water at 55 o. C, how much will not dissolve? Explain. 5. If 125 grams of potassium iodide are mixed with 100 grams of water at 10 o. C, how much more must be added to saturate the solution? 6. How many grams of potassium chlorate are needed to make a saturated solution in 50 grams of water at 35 o. C? Explain. 7. If you had a saturated KCl solution at 70 o. C and cooled it down to 50 o. C, how much of the solute would precipitate? Explain. 8. How many grams of Na. NO 3 can dissolve in 50 g of H 2 O at 10 o. C? Explain. 9. If you had 40 g of NH 3 in 100 g of water at 10 o. C would the solution be saturated, unsaturated or supersatured? Explain.
- Slides: 21