Aim How to describe properties of matter Do

Aim: How to describe properties of matter Do Now: Come up with 3 tests to help distinguish a beaker of water from a beaker of salt water.

Define Matter • Matter is anything that has mass and takes up space • Physical and chemical properties are used to describe matter

Physical Property • Can be observed without changing the composition of the substance • Use 5 senses to make observations – color – texture – density – viscosity – change in state (melting point, boiling point, freezing point) – solubility – malleability – mass – volume

Conductivity • Material’s ability to allow heat or electricity to flow

Malleability • Ability of a material to be shaped or hammered without breaking
Freezing point • The temperature at which a liquid changes into a solid. Freezing point of water is 0 o. C

Boiling point • The temperature at which it a liquid changes into a gas • the boiling point of water is 100 o. C.

Melting point • The temperatures at which the solid changes into a liquid • The melting point of water is 0 o. C

Density • Mass/volume • Can be used to test the purity of a substance • Table S has density for elements

Chemical Properties – Describes the ability of a substance to undergo changes in composition – A new substance is produced – reactivity – toxicity – p. H – flammability – fermenting – oxidation

Flammability • Material’s ability to burn in the presence of oxygen

Reactivity • How readily a substance combines chemically with other substances
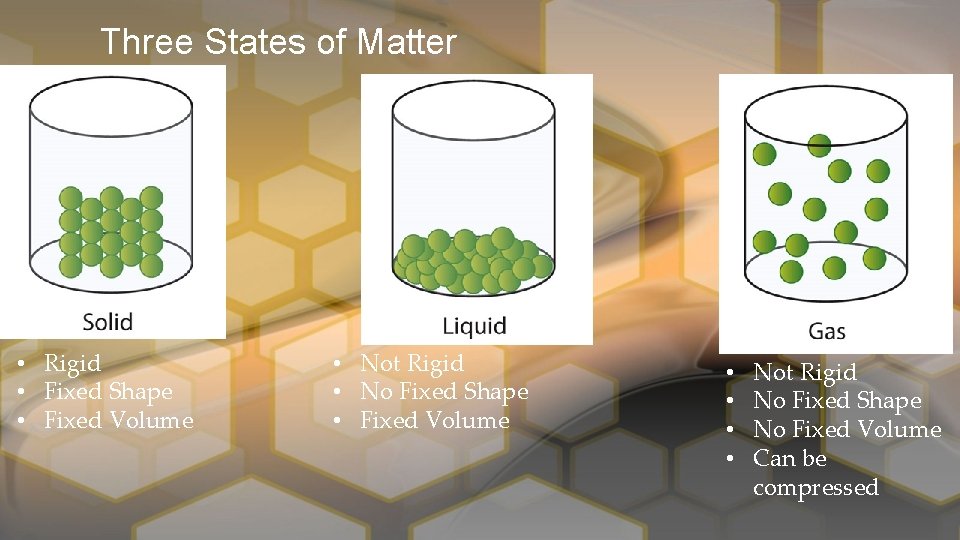
Three States of Matter • Rigid • Fixed Shape • Fixed Volume • Not Rigid • No Fixed Shape • Fixed Volume • • Not Rigid No Fixed Shape No Fixed Volume Can be compressed

Check Point ( 2 min) • Turn to the person next to you and discuss the following: • What are some differences between chemical and physical properties? Give examples and explain why.

Matter’s Changes • Physical Change – properties remain the same – changes the form of a substance without changing its identity – All changes in state! (freeze, melt, condense, evaporate, and sublimate) – Examples: – Boil, dissolve, break, split, crack, grind, cut, crush, and bend.

Matter’s Changes • Chemical Change – changes the identity of a substance – a new substance is produced – Examples: – rust, rot, burn, decompose, corrode, oxidize, ferment, and explode! – Evidence of a chemical change: – change in color or odor, formation of a gas, formation of a precipitate (solid), change in light or heat

Summary Questions 1. Which statement describes a chemical property of aluminum? a. Aluminum has a density of 2. 698 g/cm 3 at STP b. Aluminum reacts with sulfuric acid c. Aluminum conducts an electric current

Summary Questions 2. Which statement describes a chemical property of the element magnesium? a. Magnesium reacts with an acid b. Magnesium has a high boiling point c. Magnesium conducts electricity d. Magnesium is malleable

Summary Questions 3. Which process is a chemical change? a. Decomposition of water b. Melting of ice c. Subliming of ice d. Boiling of water

Summary Questions 4. Which set of procedures and observations indicates a chemical change? a. Large crystals are crushed and become a powder b. A solid is heated and turns into a liquid c. A cool, shiny metal is added to water in a beaker and rapid bubbling occurs d. Ethanol liquid is added to a an empty beaker and eventually disappears
- Slides: 20