Aim How does temperature affect the solubility of

Aim: How does temperature affect the solubility of solids, liquids, and gases? Do Now: 1. Increasing temperature of a solution ________the solubility of a solid. 2. Increasing temperature of a solution ________ the solubility of a gas. 3. Increasing pressure on a solution ________ the solubility of a gas.

TEMPERATURE • Refer to Table G of your Reference Tables for Chemistry • The solubility of most solids increases with an increase in temperature • The solubility of gases decreases with an increase in temperature
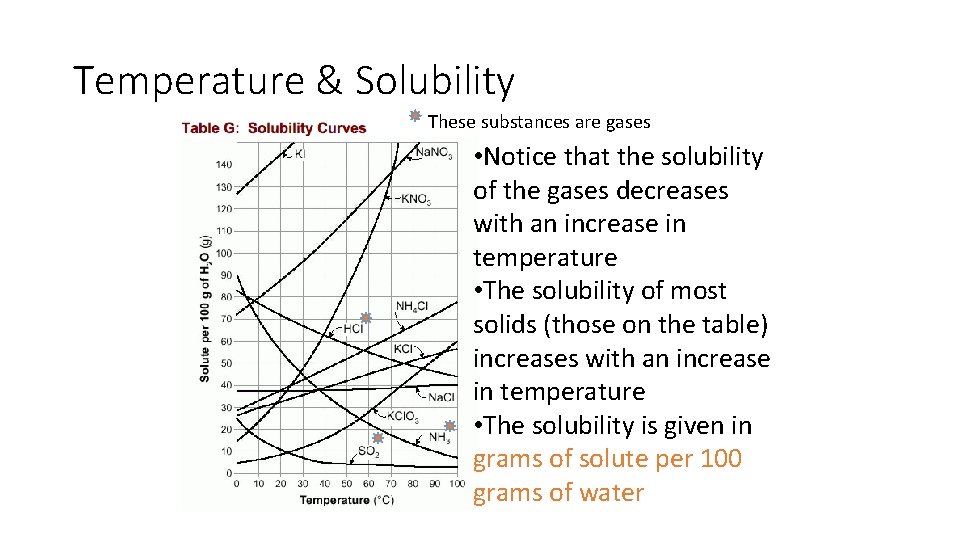
Temperature & Solubility These substances are gases • Notice that the solubility of the gases decreases with an increase in temperature • The solubility of most solids (those on the table) increases with an increase in temperature • The solubility is given in grams of solute per 100 grams of water

Question Which compound’s solubility decreases most rapidly as the temperature changes from 10 o. C to 70 o. C? a. NH 4 Cl b. NH 3 c. HCl d. KCl
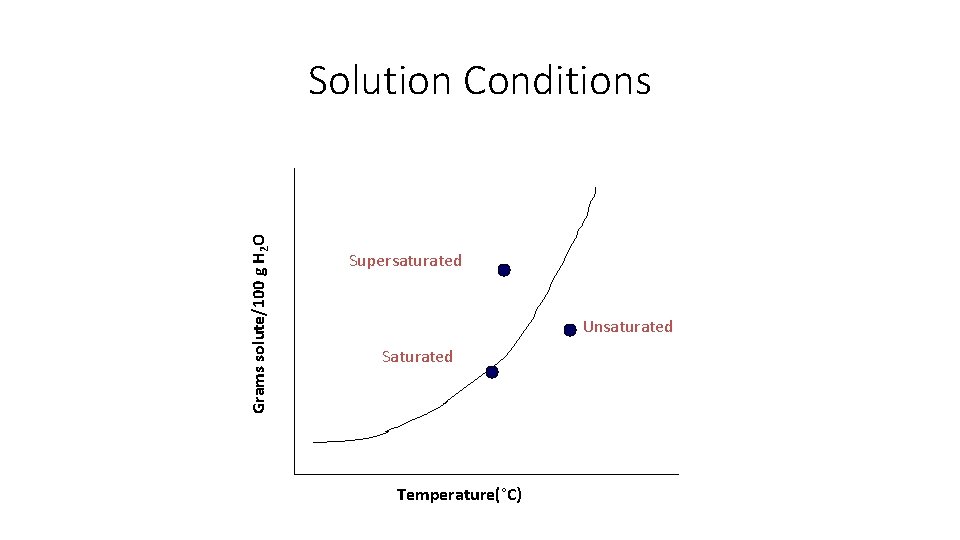
Grams solute/100 g H 2 O Solution Conditions Supersaturated Unsaturated Saturated Temperature(°C)

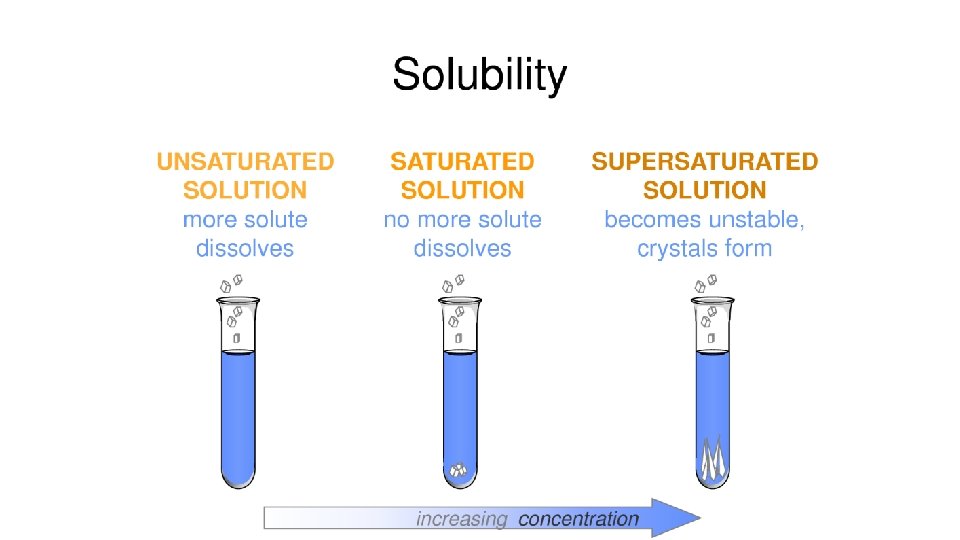
Solution Types • Unsaturated - the solution can dissolve more solute in the solvent at the specified temperature.

Solution Types • Saturated - the solution is holding as much solute as it can hold at the given temperature ( the rate of solution equals the rate of dissolution).

Solution Types • Supersaturated - the solution is holding more solute than it normally can hold at the specified temperature. These solutions are unstable and will seek to reach saturation when disturbed and the excess solute will precipitate out. • Example: rock candy • http: //chemed. chem. purdue. edu/demos/main_pages/15. 2. html
Question • How many grams of Na. NO 3 would be needed to saturate 200 grams of H 2 O at 40 o. C?
- Slides: 10