Aim How does temperature affect the solubility of

Aim: How does temperature affect the solubility of solids, liquids, and gases? Do Now: 1. Increasing temperature of a solution ________the solubility of a solid. 2. Increasing temperature of a solution ________ the solubility of a gas. 3. Increasing pressure on a solution ________ the solubility of a gas.

TEMPERATURE • Refer to Table G of your Reference Tables for Chemistry • The solubility of most solids increases with an increase in temperature • The solubility of gases decreases with an increase in temperature
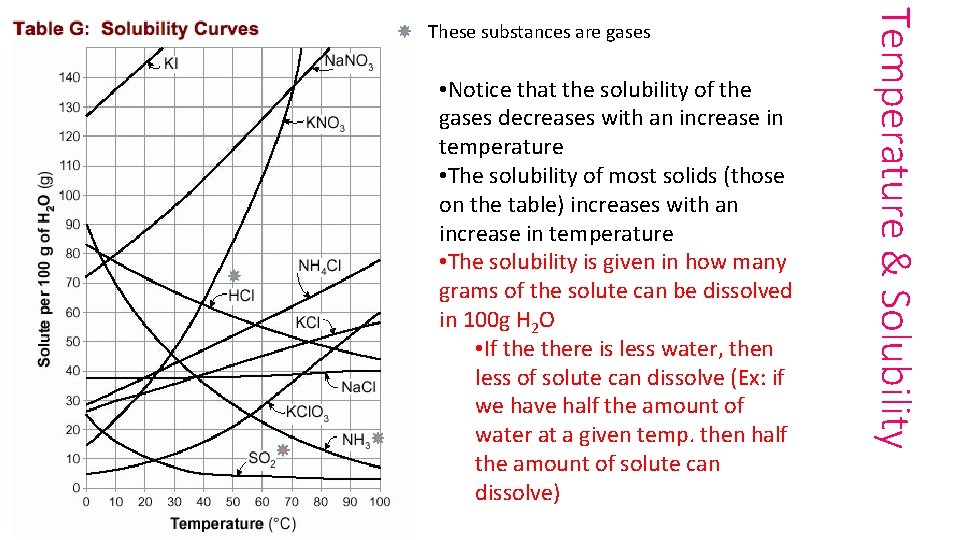
• Notice that the solubility of the gases decreases with an increase in temperature • The solubility of most solids (those on the table) increases with an increase in temperature • The solubility is given in how many grams of the solute can be dissolved in 100 g H 2 O • If there is less water, then less of solute can dissolve (Ex: if we have half the amount of water at a given temp. then half the amount of solute can dissolve) Temperature & Solubility These substances are gases
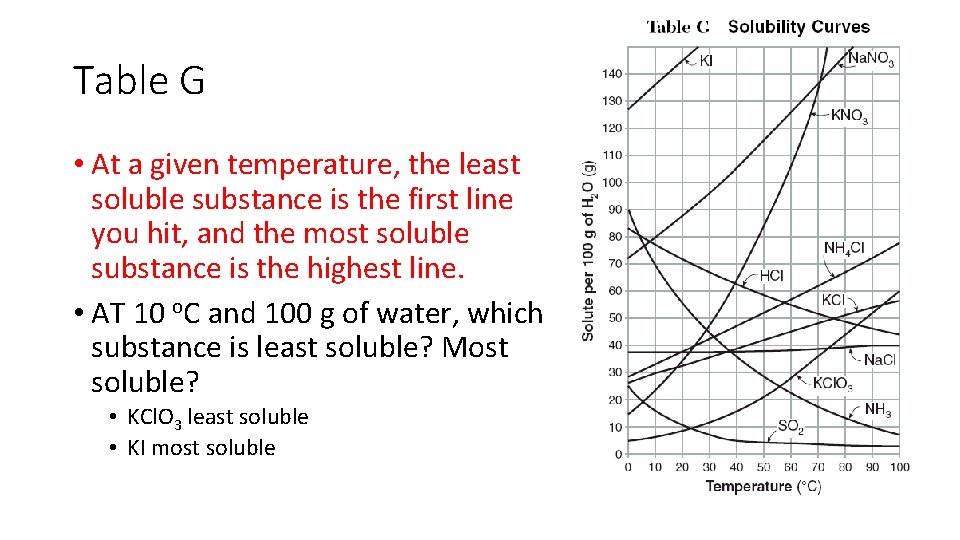
Table G • At a given temperature, the least soluble substance is the first line you hit, and the most soluble substance is the highest line. • AT 10 o. C and 100 g of water, which substance is least soluble? Most soluble? • KCl. O 3 least soluble • KI most soluble
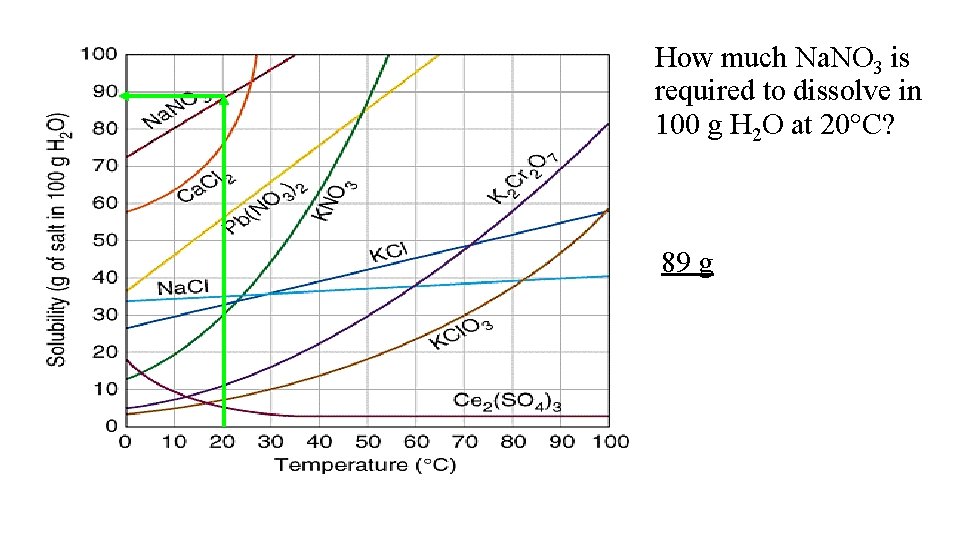
How much Na. NO 3 is required to dissolve in 100 g H 2 O at 20 C? 89 g
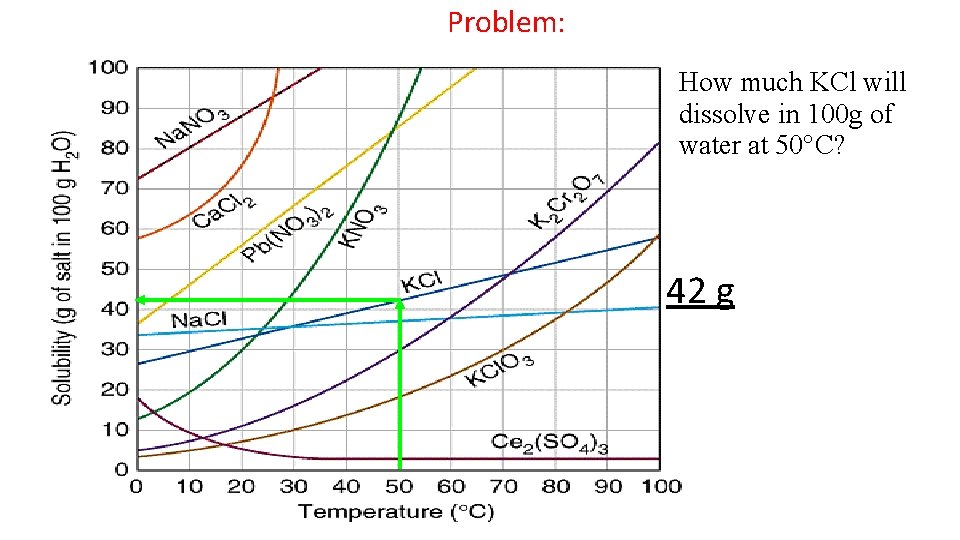
Problem: How much KCl will dissolve in 100 g of water at 50 C? 42 g
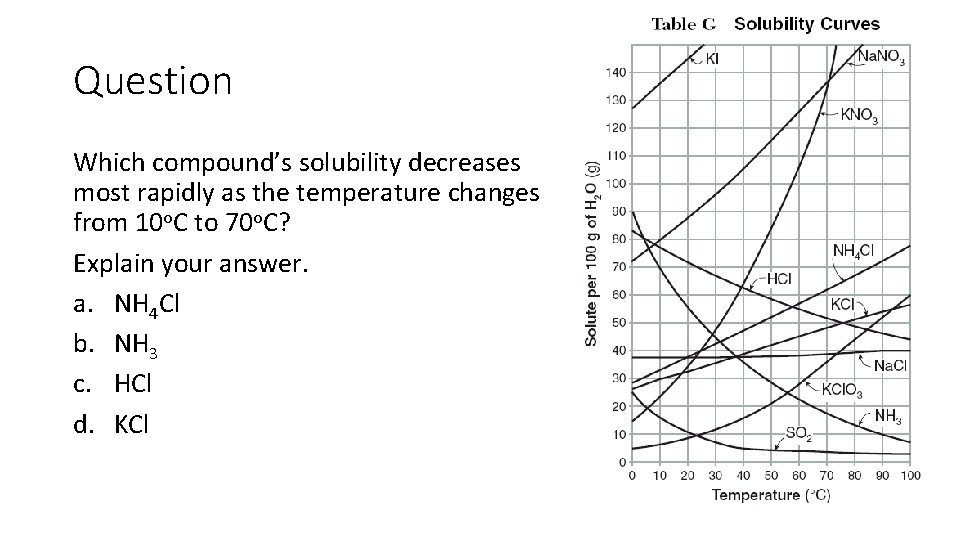
Question Which compound’s solubility decreases most rapidly as the temperature changes from 10 o. C to 70 o. C? Explain your answer. a. NH 4 Cl b. NH 3 c. HCl d. KCl

Solution Types • Unsaturated - the solution can dissolve more solute in the solvent at the specified temperature.

Solution Types • Saturated - the solution is holding as much solute as it can hold at the given temperature ( the rate of solution equals the rate of dissolution).
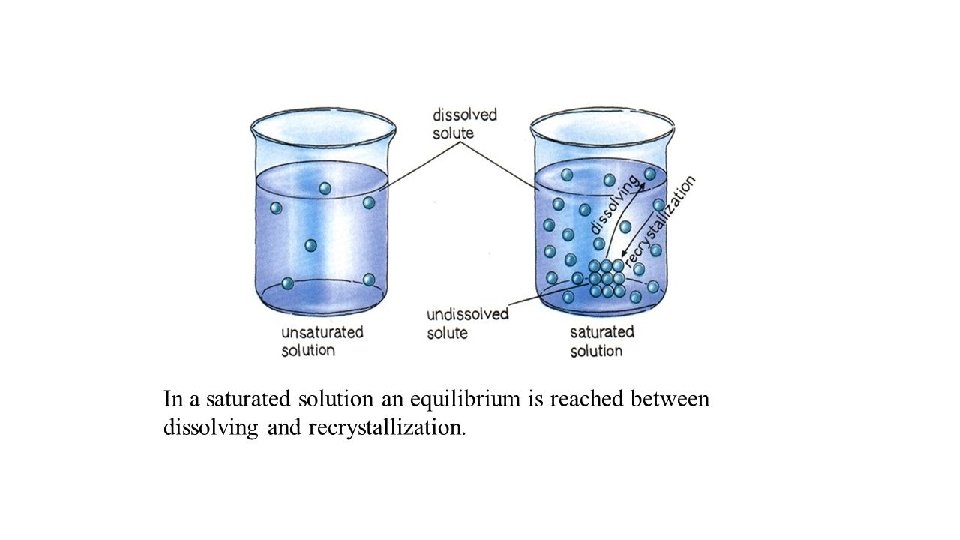

Solution Types • Supersaturated - the solution is holding more solute than it normally can hold at the specified temperature. These solutions are unstable and will seek to reach saturation when disturbed and the excess solute will precipitate out. • Example: rock candy • http: //chemed. chem. purdue. edu/demos/main_pages/15. 2. html
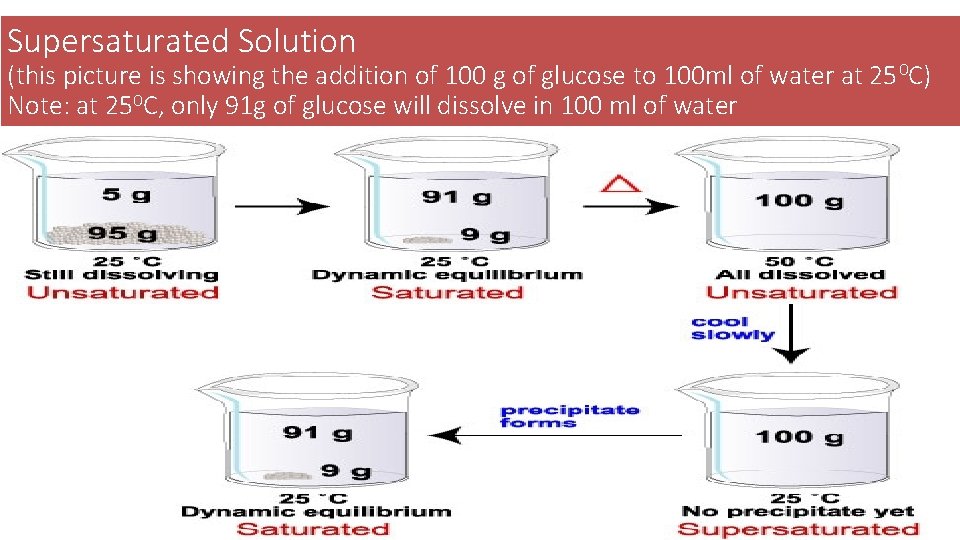
Supersaturated Solution (this picture is showing the addition of 100 g of glucose to 100 ml of water at 25 0 C) Note: at 250 C, only 91 g of glucose will dissolve in 100 ml of water
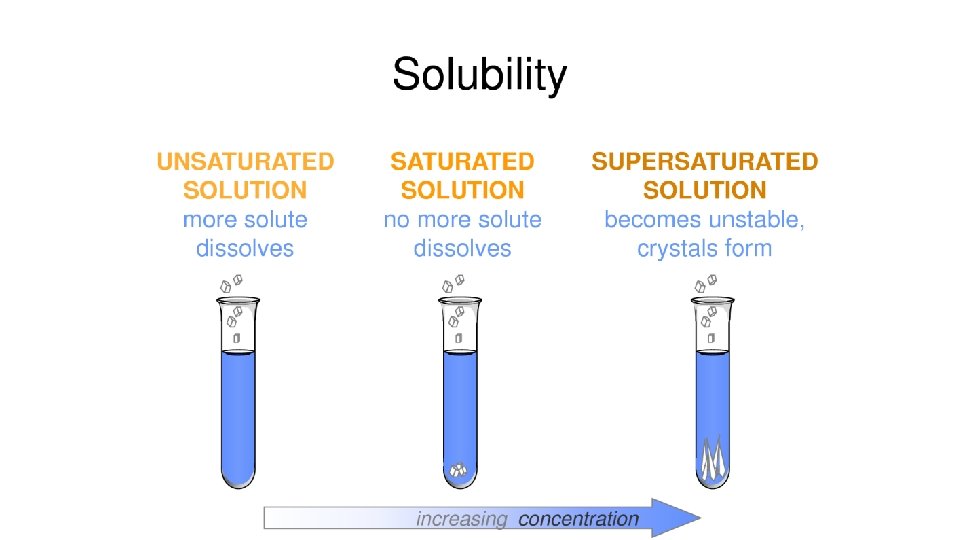
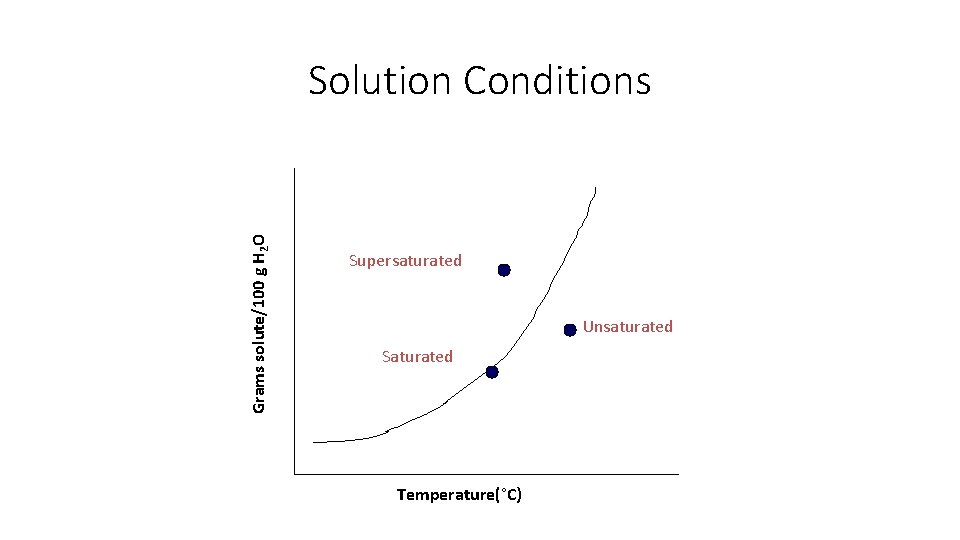
Grams solute/100 g H 2 O Solution Conditions Supersaturated Unsaturated Saturated Temperature(°C)
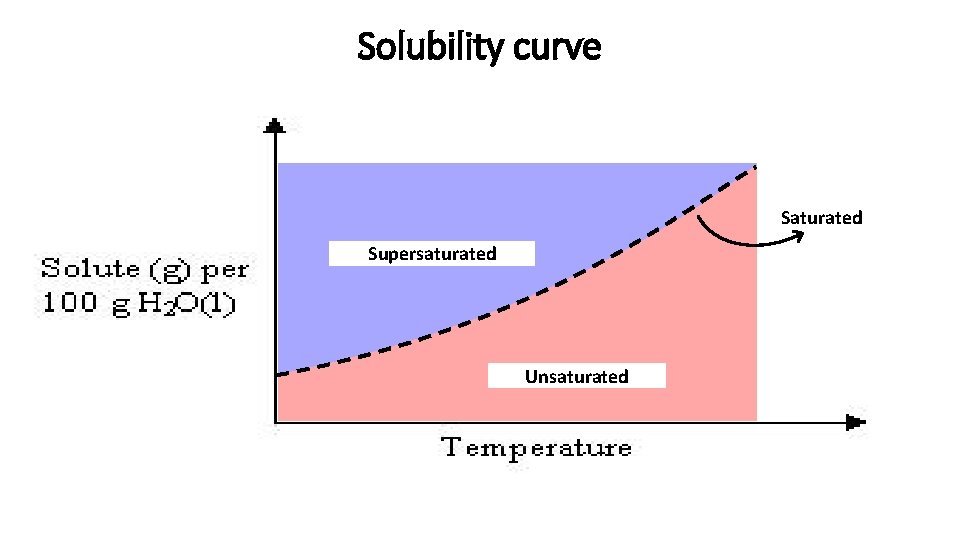
Solubility curve Saturated Supersaturated Unsaturated
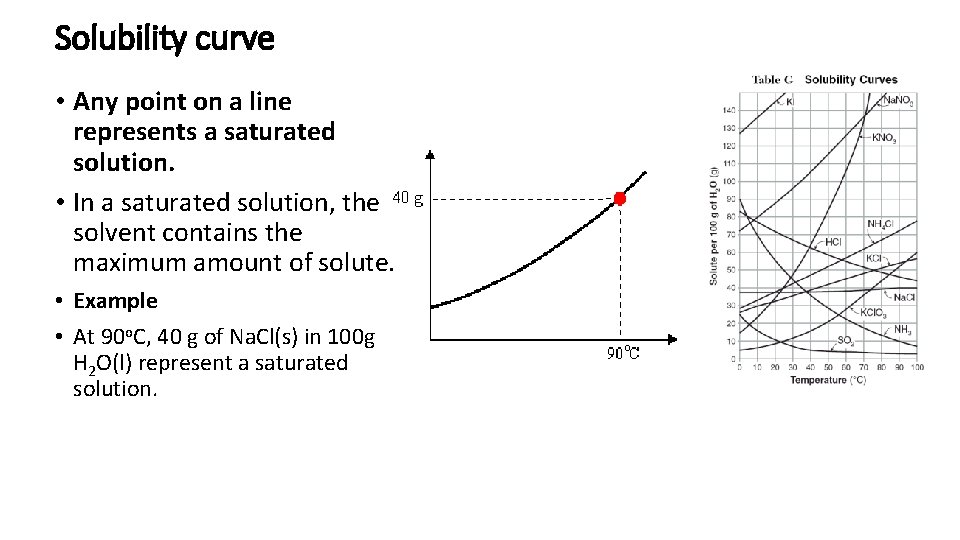
Solubility curve • Any point on a line represents a saturated solution. • In a saturated solution, the solvent contains the maximum amount of solute. • Example • At 90 o. C, 40 g of Na. Cl(s) in 100 g H 2 O(l) represent a saturated solution.
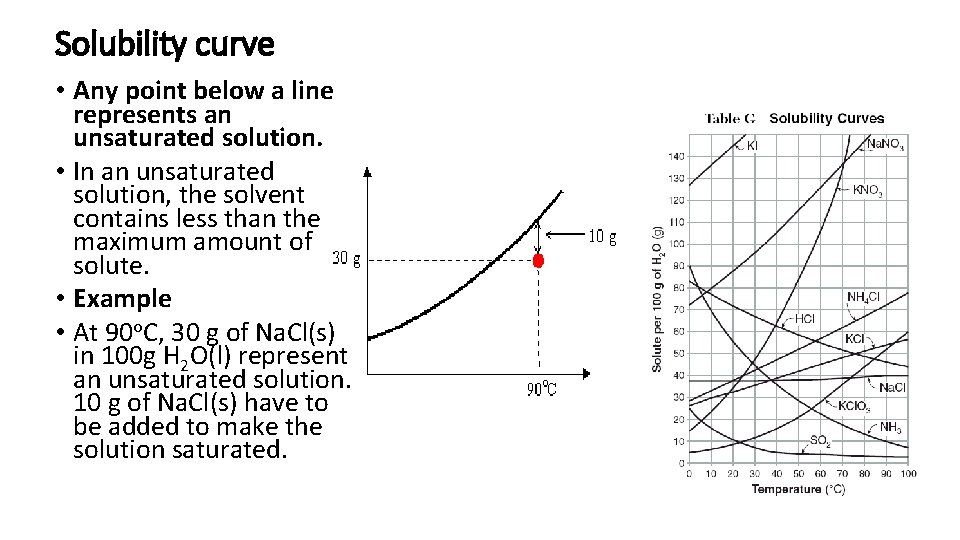
Solubility curve • Any point below a line represents an unsaturated solution. • In an unsaturated solution, the solvent contains less than the maximum amount of solute. • Example • At 90 o. C, 30 g of Na. Cl(s) in 100 g H 2 O(l) represent an unsaturated solution. 10 g of Na. Cl(s) have to be added to make the solution saturated.
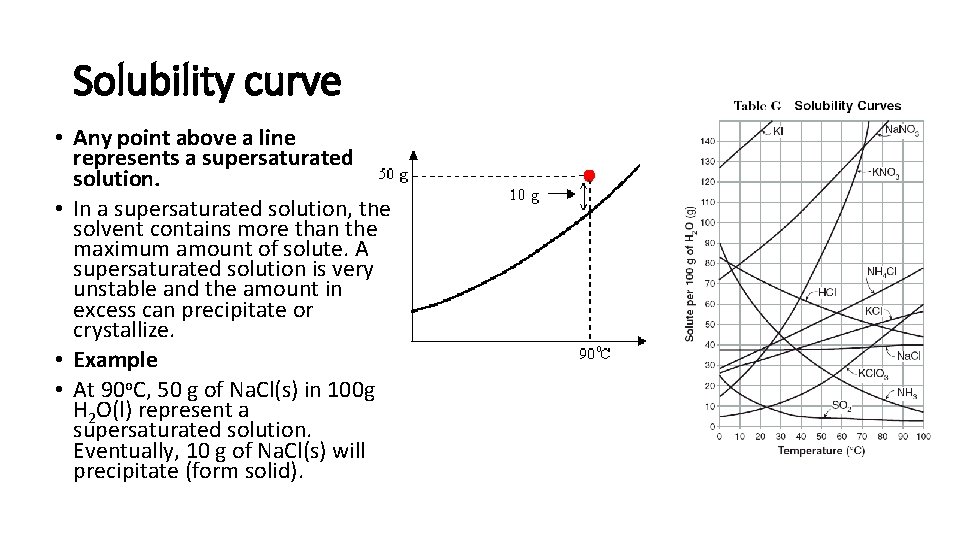
Solubility curve • Any point above a line represents a supersaturated solution. • In a supersaturated solution, the solvent contains more than the maximum amount of solute. A supersaturated solution is very unstable and the amount in excess can precipitate or crystallize. • Example • At 90 o. C, 50 g of Na. Cl(s) in 100 g H 2 O(l) represent a supersaturated solution. Eventually, 10 g of Na. Cl(s) will precipitate (form solid).
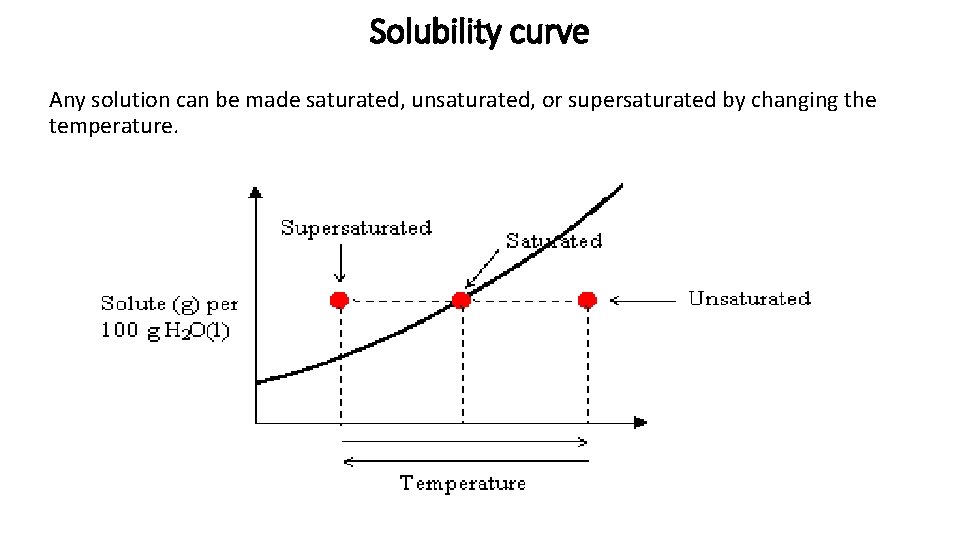
Solubility curve Any solution can be made saturated, unsaturated, or supersaturated by changing the temperature.
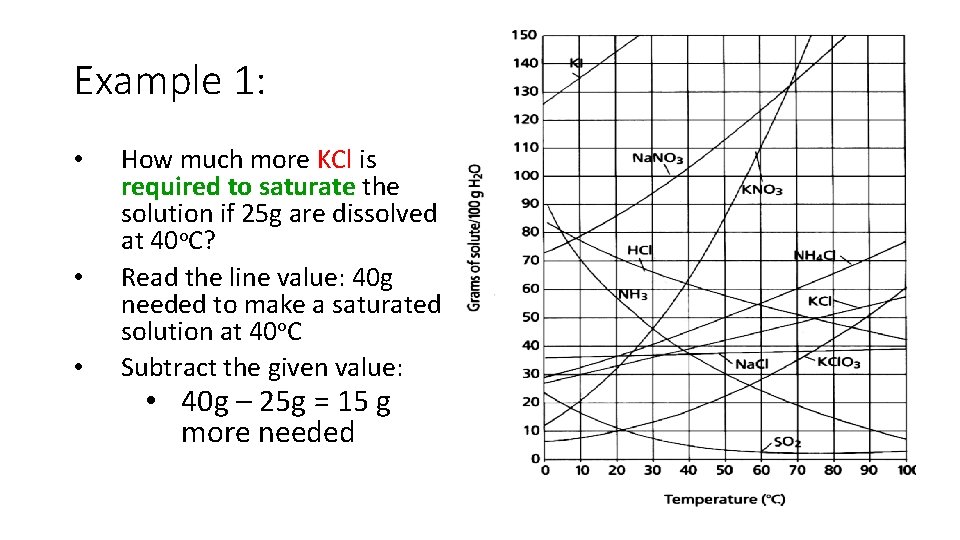
Example 1: • • • How much more KCl is required to saturate the solution if 25 g are dissolved at 40 o. C? Read the line value: 40 g needed to make a saturated solution at 40 o. C Subtract the given value: • 40 g – 25 g = 15 g more needed
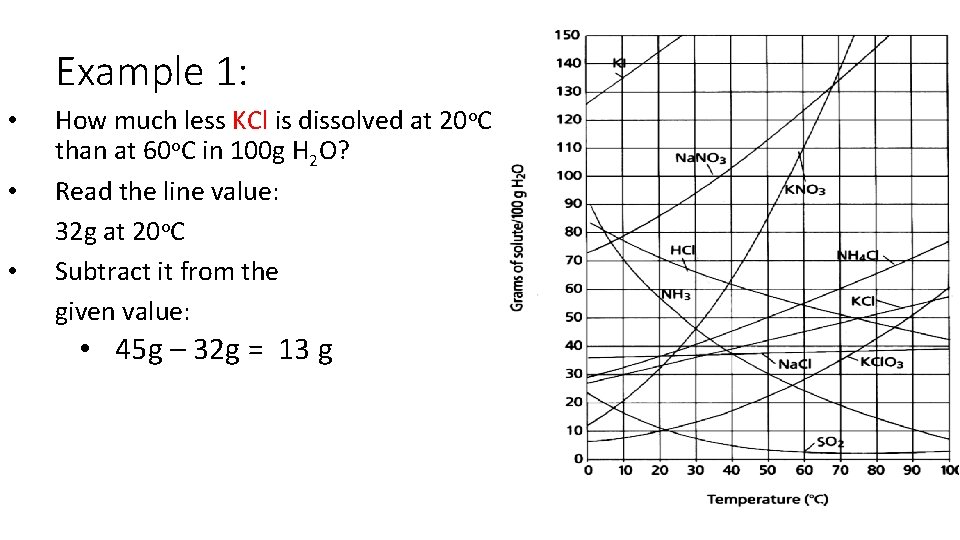
Example 1: • • • How much less KCl is dissolved at 20 o. C than at 60 o. C in 100 g H 2 O? Read the line value: 32 g at 20 o. C Subtract it from the given value: • 45 g – 32 g = 13 g
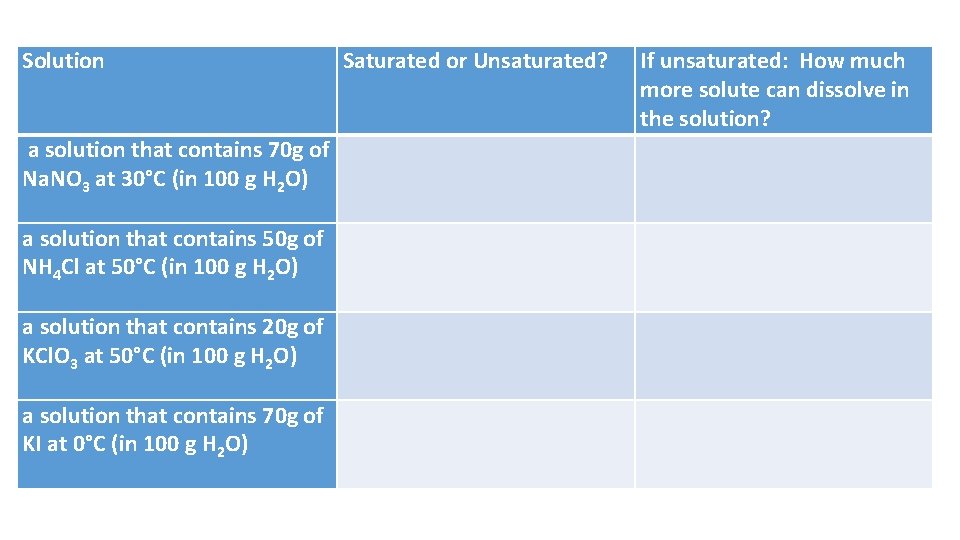
Solution Saturated or Unsaturated? If unsaturated: How much more solute can dissolve in the solution? a solution that contains 70 g of Na. NO 3 at 30°C (in 100 g H 2 O) a solution that contains 50 g of NH 4 Cl at 50°C (in 100 g H 2 O) a solution that contains 20 g of KCl. O 3 at 50°C (in 100 g H 2 O) a solution that contains 70 g of KI at 0°C (in 100 g H 2 O)
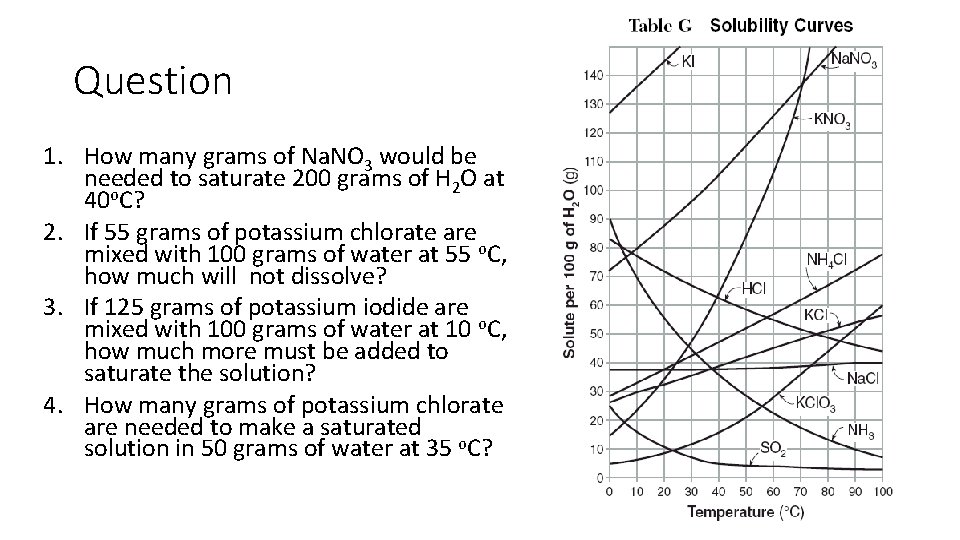
Question 1. How many grams of Na. NO 3 would be needed to saturate 200 grams of H 2 O at 40 o. C? 2. If 55 grams of potassium chlorate are mixed with 100 grams of water at 55 o. C, how much will not dissolve? 3. If 125 grams of potassium iodide are mixed with 100 grams of water at 10 o. C, how much more must be added to saturate the solution? 4. How many grams of potassium chlorate are needed to make a saturated solution in 50 grams of water at 35 o. C?

Answers 1. 210 g - In 100 g of H 2 O and 40 o. C, 105 g of Na. NO 3. Therefore, in twice the amount of water, twice the amount of solute can dissolve 2. 30 g - At 55 o. C , 25 g of KCl. O 3 dissolves in 100 g of H 2 O. The remaining 30 g out of the 55 g does not dissolve. 3. 10 g - At 10 o. C , 135 grams of KI in 100 g of H 2 O are needed to make a saturated solution. Therefore, 10 g more of KI are needed. 4. 7 g - From the table we can find how many grams of KCl. O 3 are needed in 100 g of H 2 O at 35 o. C to make a saturated solutions; about 14 g. - In half the amount of water, only 7 g of KI is needed.
- Slides: 24