AIDS 2016 AIDSconference 1 Higher mortality in HIVHBV

#AIDS 2016 | @AIDS_conference 1

Higher mortality in HIV-HBV coinfected persons with elevated HBV replication in the Temprano Trial ANRS 12 136 and 12 240 G. Menan Kouamé 1, R. Moh 1, 4, A. Boyd 5, A. Badje 1, 2, D. Gabillard 2, E. Ouattara 2, JB Ntakpe 1, A. Emième 3, S. Maylin 6, S. Eholié 1, 4, K. Lacombe 5, X. Anglaret 1, 2, C. Danel 1, 2 for the ANRS 12 136 Temprano trial group 1 : Programme PAC-CI, ANRS research site, Abidjan, Côte d’Ivoire 2 : INSERM U 897, Université de Bordeaux, France 3 : Ce. DRe. S, CHU Treichville, Abidjan, Côte d’Ivoire 4 : Service des Maladies Infectieuses et Tropicale, CHU de Treichville, Abidjan, Côte d’Ivoire 5 : INSERM, UMR S 1136, Institut Pierre Louis d’Epidémiologie et de Santé Publique, Paris, France 6 : Laboratoire de Virologie, Hôpital Saint-Louis, AP-HP, Paris, France #AIDS 2016 | @AIDS_conference 2

Background • Around 10% of HIV-infected adults are co-infected with hepatitis B virus (HBV) in West African countries. • HBV infection diagnosis is often ignored or delayed • The impact of HIV-HBV co-infection on mortality is largely unknown, especially among patients in West African countries with higher CD 4 cell counts. =>We aim to analyze the association between HBV replication and mortality during long-term follow-up in the Temprano trial ANRS 12136, a randomized controlled trial to assess the benefits and risks of early ART and 6 months IPT (Isoniazid Prophylaxis Therapy) on HIV infected adults in 9 centers, Abidjan Côte d’Ivoire 3 #AIDS 2016 | @AIDS_conference
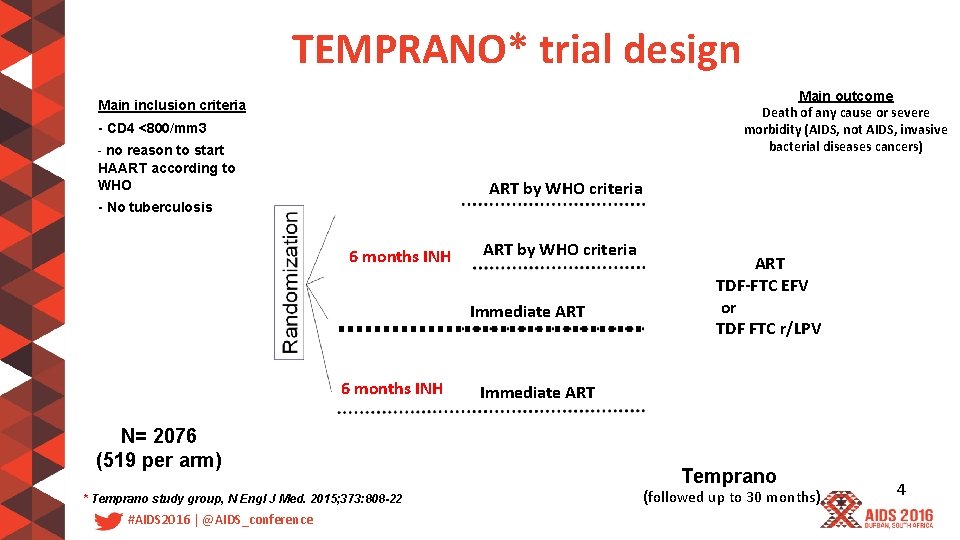
TEMPRANO* trial design Main outcome Death of any cause or severe morbidity (AIDS, not AIDS, invasive bacterial diseases cancers) Main inclusion criteria - CD 4 <800/mm 3 - no reason to start HAART according to WHO ART by WHO criteria - No tuberculosis 6 months INH ART by WHO criteria Immediate ART 6 months INH N= 2076 (519 per arm) * Temprano study group, N Engl J Med. 2015; 373: 808 -22 #AIDS 2016 | @AIDS_conference ART TDF-FTC EFV or TDF FTC r/LPV Immediate ART Temprano (followed up to 30 months) 4
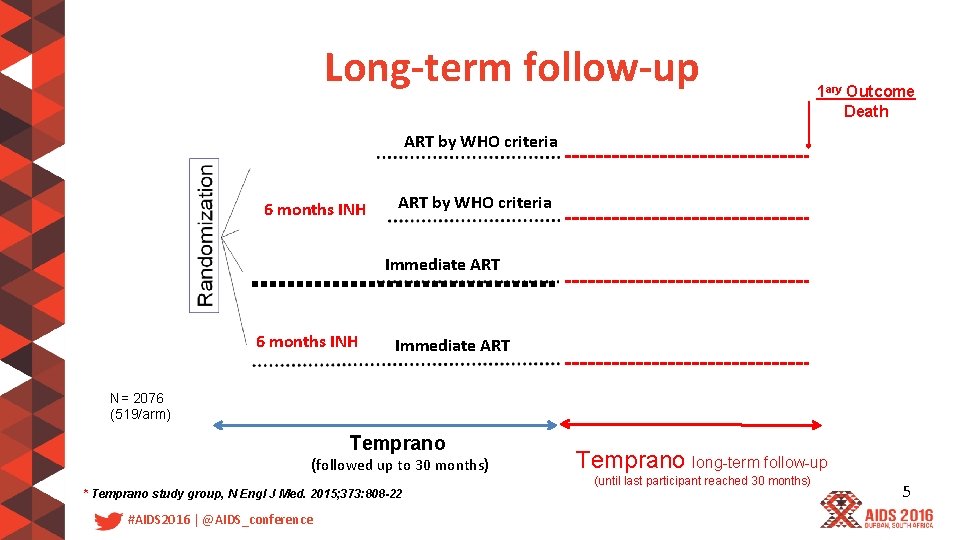
Long-term follow-up 1 ary Outcome Death ART by WHO criteria 6 months INH ART by WHO criteria Immediate ART 6 months INH Immediate ART N= 2076 (519/arm) Temprano (followed up to 30 months) * Temprano study group, N Engl J Med. 2015; 373: 808 -22 #AIDS 2016 | @AIDS_conference Temprano long-term follow-up (until last participant reached 30 months) 5

Methods • Hepatitis B : – Hepatitis B surface Antigen at inclusion in the trial for all the patients (Mini Vidas®, Biomérieux) – for HBs. Ag-positive patients: • Hepatitis B « e » antigen and antibody (n=125; Elecsys Assay, Roche) • HBV DNA (PCR technique 1 ) • Analysis for this sub-study: Cox multivariate models adjusted on CD 4, HIV viral load, Isoniazid prophylaxis, ARVs and Gender – Main variable of interest: HBV DNA viral load, cut of choose by Youden index – Primary endpoint : All-cause mortality 1 Boyd A et al. , Antiviral Ther, 2015. #AIDS 2016 | @AIDS_conference 6
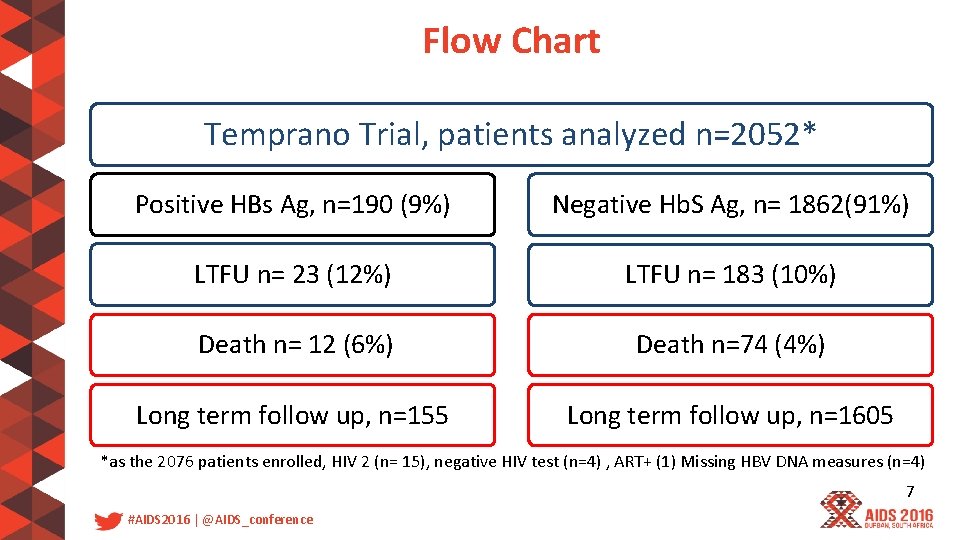
Flow Chart Temprano Trial, patients analyzed n=2052* Positive HBs Ag, n=190 (9%) Negative Hb. S Ag, n= 1862(91%) LTFU n= 23 (12%) LTFU n= 183 (10%) Death n= 12 (6%) Death n=74 (4%) Long term follow up, n=155 Long term follow up, n=1605 *as the 2076 patients enrolled, HIV 2 (n= 15), negative HIV test (n=4) , ART+ (1) Missing HBV DNA measures (n=4) 7 #AIDS 2016 | @AIDS_conference

Baseline characteristics according to HBs status Characteristics HBs. Ag serology p-value Positive Négative N (%) 190 (9%) 1862 (91%) Male sex, N(%) 57 (29%) 385 (20%) 0, 004 CD 4, cells/mm 3 , median 456 465 0, 70 HIV viral load Log 10/ml, median 4. 8 4. 6 0, 08 Early ART, N (%) 103 (53%) 930 (50%) 0, 36 IPT, N (%) 102 (53%) 928 (50%) 0, 42 TGP > N , UI/ml, N (%) 29 (15%) 129 (7%) <0, 01 Ag Hbe Positive 17 (14%) - - HBV DNA, copies/ml, Médian (IQR) HBV DNA , copies/ml #AIDS 2016 | @AIDS_conference 523 (<12 -11420) < 7000 139 (73 %) - - > 7000 51 (27%) - - 8
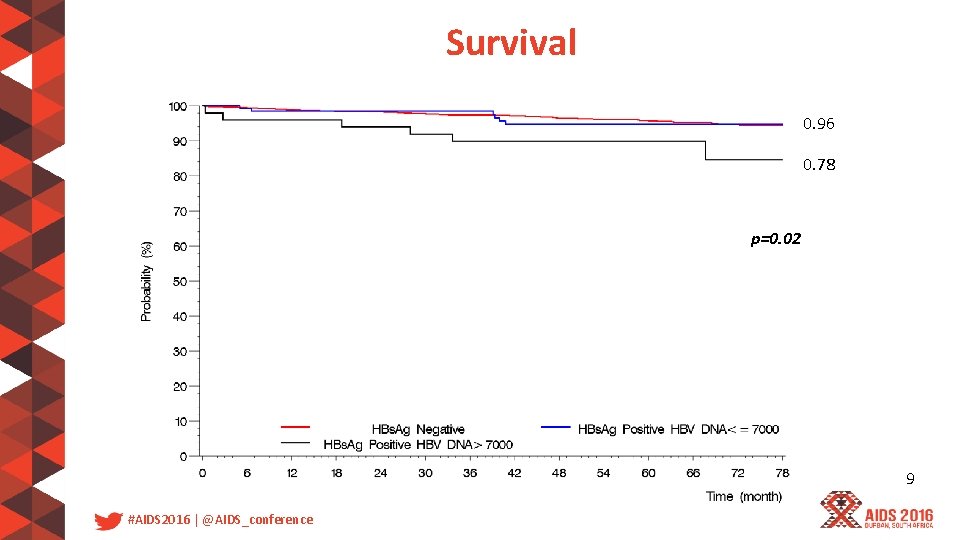
Survival 0. 96 0. 78 p=0. 02 9 #AIDS 2016 | @AIDS_conference
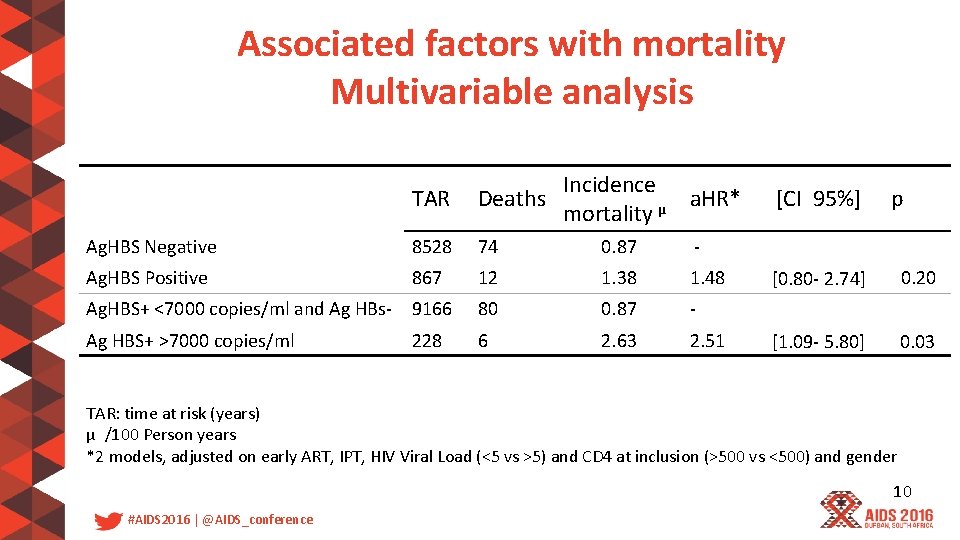
Associated factors with mortality Multivariable analysis TAR Incidence Deaths a. HR* µ mortality Ag. HBS Negative 8528 74 0. 87 - Ag. HBS Positive 867 12 1. 38 1. 48 Ag. HBS+ <7000 copies/ml and Ag HBs- 9166 80 0. 87 - Ag HBS+ >7000 copies/ml 6 2. 63 2. 51 228 [CI 95%] p [0. 80 - 2. 74] 0. 20 [1. 09 - 5. 80] 0. 03 TAR: time at risk (years) µ /100 Person years *2 models, adjusted on early ART, IPT, HIV Viral Load (<5 vs >5) and CD 4 at inclusion (>500 vs <500) and gender 10 #AIDS 2016 | @AIDS_conference

Discussion • There is a 2. 5 -times higher mortality in patients with HBV DNA greater than 7000 copies/ml enrolled in the Temprano trial • Early ART needs to be provided to all HIV infected-patients and especially to coinfected patients with high HBV replication 11 #AIDS 2016 | @AIDS_conference

THANKS All individuals who agreed to be included and followed in the Temprano trial and all the teams of CEPREF, CMSDS, CIRBA, La Pierre Angulaire, Hôpital Nord d’Abobo, Centre El Rapha, FSU Anonkoua Koute, USAC, SMIT. Programme PAC-CI Site ANRS de Côte d’Ivoire #AIDS 2016 | @AIDS_conference 12

FULLY FUND THE GLOBAL FUND 13 #AIDS 2016 | @AIDS_conference
- Slides: 13