AI 444040 Study DCV SOF RBV for genotypes
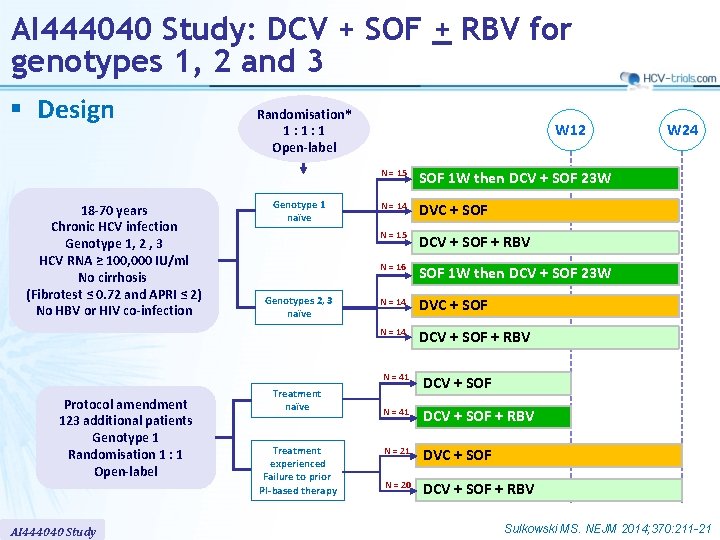
AI 444040 Study: DCV + SOF + RBV for genotypes 1, 2 and 3 § Design 18 -70 years Chronic HCV infection Genotype 1, 2 , 3 HCV RNA ≥ 100, 000 IU/ml No cirrhosis (Fibrotest ≤ 0. 72 and APRI ≤ 2) No HBV or HIV co-infection Protocol amendment 123 additional patients Genotype 1 Randomisation 1 : 1 Open-label AI 444040 Study Randomisation* 1: 1: 1 Open-label Genotype 1 naïve Genotypes 2, 3 naïve Treatment experienced Failure to prior PI-based therapy W 12 N = 15 SOF 1 W then DCV + SOF 23 W N = 14 DVC + SOF N = 15 DCV + SOF + RBV N = 16 SOF 1 W then DCV + SOF 23 W N = 14 DVC + SOF N = 14 DCV + SOF + RBV N = 41 DCV + SOF + RBV N = 21 DVC + SOF N = 20 DCV + SOF + RBV W 24 Sulkowski MS. NEJM 2014; 370: 211 -21
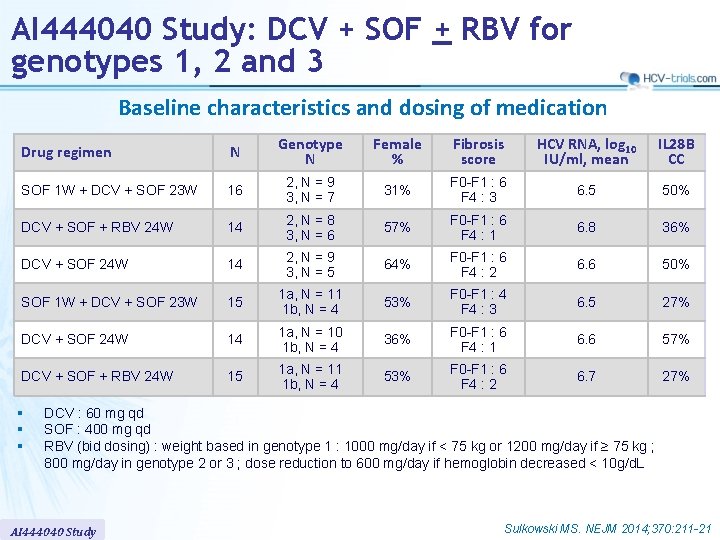
AI 444040 Study: DCV + SOF + RBV for genotypes 1, 2 and 3 Baseline characteristics and dosing of medication Drug regimen N Genotype N Female % Fibrosis score HCV RNA, log 10 IU/ml, mean IL 28 B CC SOF 1 W + DCV + SOF 23 W 16 2, N = 9 3, N = 7 31% F 0 -F 1 : 6 F 4 : 3 6. 5 50% DCV + SOF + RBV 24 W 14 2, N = 8 3, N = 6 57% F 0 -F 1 : 6 F 4 : 1 6. 8 36% DCV + SOF 24 W 14 2, N = 9 3, N = 5 64% F 0 -F 1 : 6 F 4 : 2 6. 6 50% SOF 1 W + DCV + SOF 23 W 15 1 a, N = 11 1 b, N = 4 53% F 0 -F 1 : 4 F 4 : 3 6. 5 27% DCV + SOF 24 W 14 1 a, N = 10 1 b, N = 4 36% F 0 -F 1 : 6 F 4 : 1 6. 6 57% DCV + SOF + RBV 24 W 15 1 a, N = 11 1 b, N = 4 53% F 0 -F 1 : 6 F 4 : 2 6. 7 27% § § § DCV : 60 mg qd SOF : 400 mg qd RBV (bid dosing) : weight based in genotype 1 : 1000 mg/day if < 75 kg or 1200 mg/day if ≥ 75 kg ; 800 mg/day in genotype 2 or 3 ; dose reduction to 600 mg/day if hemoglobin decreased < 10 g/d. L AI 444040 Study Sulkowski MS. NEJM 2014; 370: 211 -21
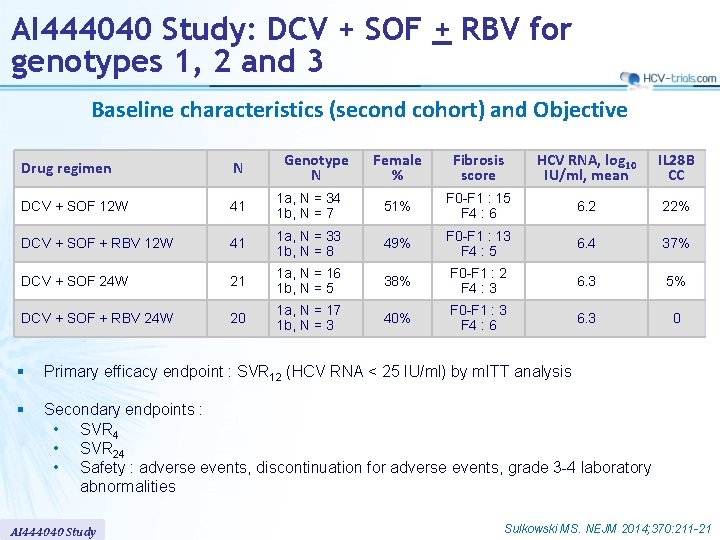
AI 444040 Study: DCV + SOF + RBV for genotypes 1, 2 and 3 Baseline characteristics (second cohort) and Objective Female % Fibrosis score HCV RNA, log 10 IU/ml, mean IL 28 B CC 1 a, N = 34 1 b, N = 7 51% F 0 -F 1 : 15 F 4 : 6 6. 2 22% 41 1 a, N = 33 1 b, N = 8 49% F 0 -F 1 : 13 F 4 : 5 6. 4 37% DCV + SOF 24 W 21 1 a, N = 16 1 b, N = 5 38% F 0 -F 1 : 2 F 4 : 3 6. 3 5% DCV + SOF + RBV 24 W 20 1 a, N = 17 1 b, N = 3 40% F 0 -F 1 : 3 F 4 : 6 6. 3 0 Drug regimen N DCV + SOF 12 W 41 DCV + SOF + RBV 12 W Genotype N § Primary efficacy endpoint : SVR 12 (HCV RNA < 25 IU/ml) by m. ITT analysis § Secondary endpoints : • SVR 4 • SVR 24 • Safety : adverse events, discontinuation for adverse events, grade 3 -4 laboratory abnormalities AI 444040 Study Sulkowski MS. NEJM 2014; 370: 211 -21
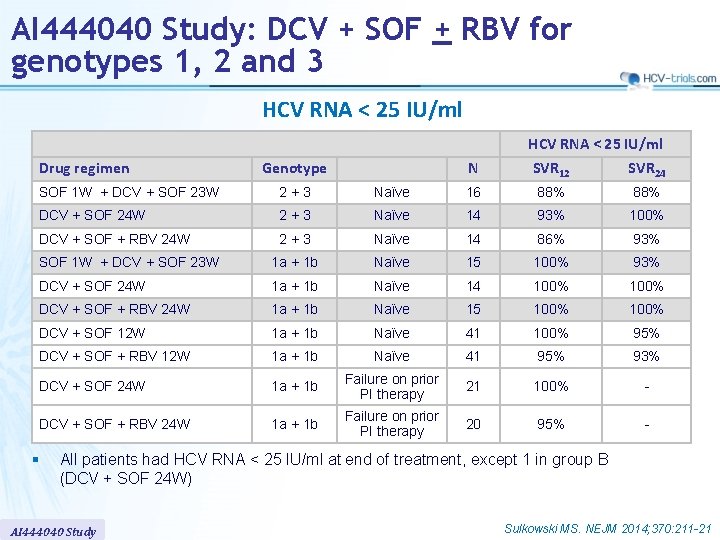
AI 444040 Study: DCV + SOF + RBV for genotypes 1, 2 and 3 HCV RNA < 25 IU/ml Drug regimen Genotype N HCV RNA < 25 IU/ml SVR 12 SVR 24 SOF 1 W + DCV + SOF 23 W 2+3 Naïve 16 88% DCV + SOF 24 W 2+3 Naïve 14 93% 100% DCV + SOF + RBV 24 W 2+3 Naïve 14 86% 93% SOF 1 W + DCV + SOF 23 W 1 a + 1 b Naïve 15 100% 93% DCV + SOF 24 W 1 a + 1 b Naïve 14 100% DCV + SOF + RBV 24 W 1 a + 1 b Naïve 15 100% DCV + SOF 12 W 1 a + 1 b Naïve 41 100% 95% DCV + SOF + RBV 12 W 1 a + 1 b Naïve 41 95% 93% DCV + SOF 24 W 1 a + 1 b Failure on prior PI therapy 21 100% - DCV + SOF + RBV 24 W 1 a + 1 b Failure on prior PI therapy 20 95% - § All patients had HCV RNA < 25 IU/ml at end of treatment, except 1 in group B (DCV + SOF 24 W) AI 444040 Study Sulkowski MS. NEJM 2014; 370: 211 -21

AI 444040 Study: DCV + SOF + RBV for genotypes 1, 2 and 3 § Virologic relapse post-treatment : 1 patient with genotype 3 who did not received RBV – NS 5 A A 30 K polymorphism (DCV resistance) at baseline and failure § Resistance testing (sequencing) – NS 5 A polymorphisms associated with loss of susceptibility to DCV in vitro detected at baseline in 32 patients : 8% in genotype 1, 61% in genotype 2, 28% in genotype 3 – Most frequent mutations : Q 30 H (genotype 1 a), L 31 M (genotype 1 b and 2), Y 93 H (genotype 3) – Except the patient with relapse, all other patients with preexisting DCV resistant variants had a SVR – No mutation (S 282 T) to SOF at baseline or in the patient with breakthrough AI 444040 Study Sulkowski MS. NEJM 2014; 370: 211 -21

AI 444040 Study: DCV + SOF + RBV for genotypes 1, 2 and 3 § Adverse events – Most common : fatigue, headache, nausea (≥ 25% in any group) – Grade 3 -4 adverse events : 7 (3. 3%) – Discontinuation of treatment for adverse events : 2 (both achieved SVR) • DCV-SOF 24 W : 1 cerebrovascular accident • DCV-SOF + RBV 24 W : 1 fibromyalgia exacerbation – Serious adverse events : 10 (4. 7%) – Most common grade 3 -4 laboratory abnormalities : low phosphorus and elevation of glucose levels – Hemoglobin level more reduced in groups with RBV • Reduction of RBV dose in 5 patients because of anemia AI 444040 Study Sulkowski MS. NEJM 2014; 370: 211 -21

AI 444040 Study: DCV + SOF + RBV for genotypes 1, 2 and 3 § Summary – DCV + SOF was assessed in untreated patients and patients in whom previous treatment with telaprevir or boceprevir had failed – Most patients had a SVR, • including 98% of patients with genotype 1 infection, regardless of viral subtype or failure of prior treatment with PI, • and 91% of naïve patients infected with genotype 2 or 3 – The most common adverse event was fatigue, which was reported in approximately one third of patients – Virologic breakthrough and relapse were rare and were not observed in any of the patients infected with HCV genotype 1 or 2, despite preexisting DCV-resistant variants in 14% – In genotype 3, 1 relapse in a patient with baseline DCV-resistant variant – No additional benefit of RBV addition but greater decrease in hemoglobin AI 444040 Study Sulkowski MS. NEJM 2014; 370: 211 -21
- Slides: 7