AHCCCS Pharmacy and Therapeutics Committee 14 October 2020

AHCCCS Pharmacy and Therapeutics Committee 14 October 2020

Welcome and Introductions • Suzi Berman, RPh, Director of Pharmacy, AHCCCS o o o Meeting Minutes 19 May 2020 Review Vote 2

Magellan Class Reviews Classes for Review: Non-Supplemental Rebate Class Review Antimigraine Agents, Triptans Leukotriene Modifiers Phosphate Binders Sedative Hypnotics Antifungals, Oral Antifungals, Topical Beta Blockers BPH Treatments 3

Magellan Class Reviews Classes for Review: Non-Supplemental Rebate Class Review Calcium Channel Blockers Contraceptives, Oral Contraceptives, Other Bladder Relaxant Preparations Antiparkinsons Agents Lipotropics, Statins Lipotropics, Others Proton Pump Inhibitors Acne Agents, Topical 4

Magellan Drug Class Reviews Justin Johnson, Pharm. D, Magellan 5

Antimigraine Agents, Triptans 6

Antimigraine Agents, Triptans Class Overview – Agents in this class: • • • almotriptan malate - (almotriptan) eletriptan - (eletriptan; Relpax) frovatriptan - (frovatriptan; Frova) naratriptan - (naratriptan; Amerge) rizatriptan - (rizatriptan ODT & tablet; Maxalt, Maxalt MLT) sumatriptan - (sumatriptan kit, nasal, tablet & vial; Imitrex kit, nasal, tablet & vial; Onzetra Xsail, Sumavel Dose. Pro, Zembrace Sym. Touch) 7

Antimigraine Agents, Triptans Class Overview – Agents in this class: • sumatriptan/naproxen - (sumatriptan/naproxen; Treximet) • sumatriptan camphor/menthol • zolmitriptan - (zolmitriptan ODT, tablets; Zomig ODT, tablets) 8

Antimigraine Agents, Triptans • Migraine type headaches account of 10 to 20% of all headaches in adults, and have a high prevalence of over 39 million individuals in the US • During migraine diagnosis, regular tension-type headaches must be ruled out. • Key criteria for migraine diagnosis include an episodic headache lasting from 4 to 72 hours with at least two of the following symptoms: unilateral pain, throbbing, aggravation of pain upon moving, pain of moderate to severe intensity with nausea, vomiting, photophobia, or phonophobia • Non-opioid analgesics including acetaminophen, NSAIDs, and/or caffeine are considered first-line therapy for patients with mild to moderate migraine headaches 9

Antimigraine Agents, Triptans • Migraine-specific treatments such as triptans should be reserved for patients with moderate to severe migraine attacks • Currently, the triptans are considered the drugs of choice for treatment acute migraine attacks due to their efficacy profiles • Per The US Headache Consortium, which a multidisciplinary panel of several professional organizations, all of the triptans are considered effective for the acute migraine treatment and no significant differences exist among these agents in terms of safety and efficacy 10

Antimigraine Agents, Triptans • According to the American Academy of Neurology (AAN) and the American Headache Society (AHS), when it comes to pharmacologic treatment for episodic migraine prophylaxis in adults: o Drugs that are considered effective in migraine prevention include divalproex sodium, sodium valproate, topiramate metoprolol, propranolol, and timolol o Frovatriptan is established for short-term menstrually associated migraine (MAM) prevention o Drugs that are considered probably effective in migraine prevention include naratriptan, zolmitriptan, amitriptyline, venlafaxine, atenolol, and nadolol 11

Antimigraine Agents, Triptans • In addition to approval in adults, almotriptan, sumatriptan/naproxen (Treximet), and zolmitriptan nasal spray are FDA-approved for use in patients 12 to 17 years old, and rizatriptan is approved in patients 6 to 17 years old • Non-oral routes of administration are available for patients who experience nausea or vomiting with their migraine attacks, though nasal irritation can occur and unpleasant taste is common with nasal administration 12

Antimigraine Agents, Triptans Product Updates: • In April 2020, GSK has made a business decision to discontinue Imitrex 6 mg SDV. Generics will remain available. 13

Leukotriene Modifiers 14

Leukotriene Modifiers Class Overview – Agents in this class: • montelukast - (montelukast chewable tablet, granules & tablet; Singulair Chewable Tablet, Granules & Tablet) • zafirlukast - (zafirlukast; Accolate) • zileuton- (zileuton ER; Zyflo CR) 15

Leukotriene Modifiers • Zafirlukast, zileuton and zileuton ER are only approved for prophylaxis and chronic treatment of asthma, while montelukast is approved for asthma and allergic rhinitis, so it can be considered for patients with these two comorbidities but, it is not a first-line monotherapy treatment option for either condition • The NAEPP and the GINA guidelines both recommend inhaled corticosteroids as the mainstay of asthma treatment • Leukotriene modifiers are included as alternative or add-on treatment for certain patients • Per GINA guidelines, leukotriene modifiers are less effective than ICS, but may be appropriate for initial controller treatment for patients who are unable or unwilling to use ICS, intolerant to ICS, or who also suffer from allergic rhinitis 16

Leukotriene Modifiers • Leukotriene modifiers are also used as add-on therapy to reduce the ICS dose, and to potentially improve asthma management in those whose asthma is inadequately controlled on ICS • The use of leukotriene modifiers in acute asthma is not well established • The American Academy of Allergy, Asthma and Immunology (AAAAI, the American College of Allergy, Asthma, and Immunology (ACAAI, and the American Academy of Otolaryngology, Head and Neck Surgery recommend intranasal corticosteroids (INCS) as first line treatment for seasonal allergic rhinitis • Generally, montelukast is considered an alternative to first-line therapy with intranasal corticosteroids only in patients who suffer from both asthma and SAR 17

Leukotriene Modifiers • The International Consensus Statement on Allergy and Rhinology Allergic Rhinitis guidelines state that monotherapy with a leukotriene modifier may be appropriate in patients who are unable to use intranasal corticosteroids and oral antihistamines • Comparative trials of leukotriene modifiers are limited, and montelukast is the most widely used agent in this class due to its multiple indications, once daily dosing, and ease of administration due to multiple available dosage forms 18

Leukotriene Modifiers Product Updates: • In March 2020, the FDA has recommended a Boxed Warning be added to the prescribing information for montelukast (Singulair) due to the potential for serious behavior and mood-related changes, including the potential for suicidal ideation or behavior. • Product labeling already addressed the potential for these serious side effects; however the FDA has determined a stronger warning is warranted especially since there a number of alternative medications for the conditions for which montelukast is used. 19

Leukotriene Modifiers Product Updates: • The FDA has previously provided communications regarding the mental health side effects with this medication in 2008 and 2009, and although the current label includes a warning, prescribers/patients may not be aware of these safety concerns. • The FDA is recommending montelukast only be used for allergic rhinitis when other allergy medications are not tolerated or do not provide adequate symptom control. • In asthma patients, prescribers should evaluate the benefits versus risks of montelukast prior to prescribing. • In addition, the FDA is requiring a new patient Medication Guide providing details on these risks. 20

Phosphate Binders 21

Phosphate Binders Class Overview – Agents in the class: • calcium acetate - (calcium acetate capsule & tablet; Phoslyra) • ferric citrate - (Auryxia) • lanthanum carbonate - (lanthanum carbonate chewable tablet; Fosrenol Chewable Tablet, Fosrenol Powder Pack) • sevelamer carbonate - (sevelamer carbonate powder pack, tablet; Renvela Powder Pack, Renvela Tablet) • sevelamer HCL - (Renagel) • sucroferric oxyhydroxide - (Velphoro) 22

Phosphate Binders • • Chronic kidney disease (CKD) is another high prevalence medical condition affecting around 30 million people in the United States When Kidney function worsens, phosphorus clearance also decreases, resulting in hyperphosphatemia Patients with hyperphosphatemia usually in end stage renal disease become at high risk for: o renal bone disease o soft and vascular tissue calcification o cardiovascular disease o And increased mortality The National Kidney Foundation updated their guidelines in 2017 under The Kidney Disease: Improving Global Outcomes (KDIGO) foundation 23

Phosphate Binders • Hyperphosphatemia Treatment and management include reducing dietary phosphorus, using phosphate binding therapy, and removing phosphorus by dialysis • Studies have demonstrated that hyperphosphatemia control is crucial in preventing and delaying renal osteo-dystrophy and soft tissue calcifications • All phosphate binders are considered effective in reducing serum phosphate levels, and treatment guidelines do not strongly prefer one agent in this class over another for adults • Selection of an appropriate phosphate binder should be based on various clinical parameters, rather than phosphorus lowering properties alone, and, as such, treatment should be individualized accordingly 24

Phosphate Binders Product Updates: • None 25

Sedative Hypnotics 26

Sedative Hypnotics Class Overview - Benzodiazepine Agents: estazolam - (estazolam) flurazepam - (flurazepam) quazepam – (quazepam; Doral) temazepam - (temazepam, temazepam 7. 5 mg & 22. 5 mg; Restoril) • triazolam - (triazolam; Halcion) • • 27

Sedative Hypnotics Class Overview - Non-Benzodiazepine Agents: • • doxepin - (Silenor) eszopiclone - (eszopiclone; Lunesta) lemorexant - Dayvigo ramelteon - (ramelteon; Rozerem) suvorexant - (Belsomra) tasimelteon - (Hetlioz) zaleplon - (zaleplon; Sonata) zolpidem - (zolpidem, zolpidem SL, zolpidem ER; Ambien, Ambien CR, Edluar, Intermezzo, Zolpimist) 28

Sedative Hypnotics • Insomnia consists of variety of symptoms including difficulty falling asleep and staying asleep, or non-refreshing sleep along with daytime dysfunction or distress • Non-pharmacological interventions should always be used first for insomnia • The updated 2017 American Academy of Sleep Medicine (AASM) guidelines recommend psychological and behavioral strategies, as well as pharmacological interventions for insomnia • And they recommend initial behavioral interventions to include stimulus control, good sleep hygiene, and relaxation therapy, or a combination of treatments referred to as cognitive behavioral therapy for insomnia 29

Sedative Hypnotics • AASM guideline recommends that pharmacotherapy should be used to treat patients who failed to respond to CBT. They recommend: o zaleplon, triazolam, and ramelteon versus no treatment for sleep onset insomnia o suvorexant and doxepin over no treatment for sleep maintenance insomnia o eszopiclone, zolpidem, and temazepam for both sleep onset and sleep maintenance insomnia o Recommend against the use of trazodone or tiagabine for sleep onset or sleep maintenance insomnia in adults o Recommend against the use of over-the-counter medications or supplements (e. g. , diphenhydramine, tryptophan, melatonin) or herbal products (e. g. , valerian) as a treatment for sleep onset and sleep maintenance for chronic insomnia 30

Sedative Hypnotics • Current treatment guidelines for insomnia do not recommend one agent within this class over another, suggesting treatment be individualized • Choice of agent should be based on sleep pattern, treatment goals, past treatment response, patient preference, cost, availability of other treatments options, comorbid conditions, contraindications, potential interaction with concurrent medication, and side effects 31

Sedative Hypnotics • Hetlioz is only indicated for use in Non-24 -hour sleep-wake disorder (N 24 SWD or non-24), which is a chronic circadian rhythm condition that causes problems with the timing of sleep and sleep patterns in totally blind adults • With the exception of zolpidem SL, all agents should be administered immediately before going to bed or after the patient has gone to bed and experienced difficulty falling asleep • Zolpidem SL should be used for middle of the night awakenings when the patient still has more than 4 hours before waking up • In general, women require lower zolpidem doses due to differences in the clearance of this drug 32

Sedative Hypnotics • It is important to note that Doxepin, ramelteon, and tasimelteon are the only agents in this class that are not controlled substances and are not associated with abuse or physical dependence • All drugs in this class should be used at the lowest effective dose, and should be administered with caution in patients who exhibit signs and symptoms of depression • If insomnia does not resolve after 7 to 10 days of treatment, patients may need to be evaluated for other medical or psychological issues • Continuous use of these agents should be avoided, and patients should be encouraged to use these medications only as needed rather than regularly 33

Sedative Hypnotics • In 2016, the FDA informed healthcare professionals that concurrent use of opioids and benzodiazepines or other CNS depressants may result in serious adverse reactions such as profound sedation, respiratory depression, coma, and death • Providers should limit prescribing opioids with benzodiazepines to patients in whom the treatment benefits clearly outweigh the risks 34

Sedative Hypnotics New Drug to Class: Dayvigo (lemorexant) 35

Sedative Hypnotics Treatment Updates: • Dayvigo (lemorexant), an orexin receptor antagonist, is indicated for the treatment of insomnia, characterized by difficulties with sleep onset and/or sleep maintenance, in adults. • Dayvigo is a DEA schedule IV product and is approved as 5 mg and 10 mg tablets. • The recommended dose is 5 mg taken immediately before going to bed with at least 7 hours remaining before the planned time of awakening. o The dose may be increased to a max of 10 mg based on clinical response and tolerability. 36

Sedative Hypnotics Treatment Updates: • Dayvigo is contraindicated in patient with narcolepsy. • Warnings include CNS depressant effects and daytime impairment, sleep paralysis and cataplexy-like symptoms, complex sleep behaviors, compromised respiratory function, worsening of depression/suicidal ideation, and need to evaluate co-morbid diagnoses if insomnia persists. • The most common adverse reaction was somnolence. 37

Sedative Hypnotics Treatment Updates: • The FDA approval of Dayvigo was based on two pivotal Phase III studies in a total of about 2, 000 adult patients with insomnia • SUNRISE 1 evaluated Dayvigo versus placebo for six-months. • SUNRISE 2 evaluated Dayvigo versus comparators for up to one month 38

Sedative Hypnotics Treatment Updates: • • SUNRISE 1 was a short-term (one month), randomized, double-blind, placebo- and active-controlled, multi-center, parallel-group clinical trial in adult female subjects age 55 and older and male subjects 65 years and older who met DSM-5 criteria for insomnia disorder. Patients were randomized to placebo (n=208), Dayvigo 5 mg (n=266) or 10 mg (n=269) or active comparator (n=263) once nightly. The primary efficacy endpoint was the mean change in latency to persistent sleep (LPS; defined as the number of minutes from lights off to the first 10 consecutive minutes of non-wakefulness) from baseline to end of treatment (day 29/30), as measured by overnight polysomnography (PSG) monitoring. Dayvigo 5 mg and 10 mg demonstrated statistically significant superiority on the primary efficacy measure, LPS, compared to placebo. Dayvigo 5 mg and 10 mg demonstrated statistically significant improvement in sleep efficiency (SE; defined as the proportion of time spent asleep during time in bed) and sleep onset and sleep maintenance (defined as the minutes of wake from the onset of persistent sleep until lights on) compared to placebo 39

Sedative Hypnotics Treatment Updates: • • SUNRISE 2 was a long-term (six month), randomized, double-blind, placebo-controlled, multicenter, trial in adult patients age 18 or older who met DSM-5 criteria for insomnia disorder. Patients were randomized to placebo (n=325), Dayvigo 5 mg (n=323), or Dayvigo 10 mg (n=323) once nightly. The primary efficacy endpoint was the mean change from baseline to end of treatment at six months for patient-reported (subjective) sleep onset latency (s. SOL), defined as the estimated minutes from the time that the subject attempted to sleep until falling asleep. Dayvigo 5 mg and 10 mg demonstrated statistically significant superiority on the primary efficacy measure, s. SOL, compared to placebo. Dayvigo 5 mg and 10 mg also showed statistically significant superiority in patient reported sleep efficiency and subjective sleep onset and sleep maintenance 40

Antifungals, Oral 41 41

Antifungals, Oral Class Overview – the indications for these products include, but are not limited to*: • Candidiasis fungal infections caused by candida • Cryptococcal infections • Tinea infections • Onychomycosis • Invasive aspergillosis *Not inclusive of all product indications, all products differ in indication 42

Antifungals, Oral Class Overview - Agents included in this class: • • • clotrimazole troche - (clotrimazole troche) fluconazole - (fluconazole; Diflucan) flucytosine - (flucytosine; Ancobon) griseofulvin suspension - (griseofulvin suspension) griseofulvin microsized - (griseofulvin microsized) griseofulvin ultramicrosized - (griseofulvin ultramicrosized) isavuconazonium - (Cresmba) itraconazole - (itraconazole; Onmel, Sporanox) itraconazole – (Tolsura) 43

Antifungals, Oral Class Overview - Agents in the class: • ketoconazole - (ketoconazole) • miconazole - (Oravig) • nystatin - (nystatin) • posaconazole - (Noxafil) • terbinafine - (terbinafine) • voriconazole - (voriconazole; Vfend) 44

Antifungals, Oral • Oral Antifungal agents have different spectrums of activity and are FDA-approved to treat a variety of infections in both the immunocompetent and the immunocompromised patients • As far as a direct comparison of the safety and efficacy of these drugs, only few trials have been conducted • Many of these drugs do carry boxed warnings related to adverse events and/or drug interactions 45

Antifungals, Oral • Fluconazole is considered an effective Candida treatment for different infections; it has excellent penetration into many tissues; and, unlike ketoconazole, it lacks concerns about p. H-dependent absorption • Fluconazole, itraconazole, ketoconazole, nystatin, and clotrimazole are all deemed effective treatments for oro-pharyngeal candidiasis • Voriconazole has been shown to have similar efficacy to fluconazole in the treatment of esophageal candidiasis; but it is also associated with more adverse effects than fluconazole • Posaconazole oral suspension carries an indication for oropharyngeal candidiasis treatment when refractory to itraconazole and/or fluconazole 46

Antifungals, Oral • Nystatin is used to treat intestinal candidiasis and may be used in infants and children • Cresemba, posaconazole, flucytosine, voriconazole, itraconazole, and fluconazole all carry indications for the treatment and/or prophylaxis of several serious fungal infections 47

Antifungals, Oral Product Updates: • None 48

Antifungals, Topical 49 49

Antifungals, Topical Class Overview - Product indications include* • Cutaneous Candidiasis • Tinea topical infections • Topical Onychomycosis • Seborrheic Dermatitis *Not inclusive of all product indications, all products differ in indication 50

Antifungals, Topical Agents include prescription and OTC products: • butenafine - (Mentax) • butenafine - (butenafine [OTC], Lotrimin Ultra [OTC]) • ciclopirox 0. 77% - (Ciclodan Cream, Kit; ciclopirox cream; Loprox Cream/Gel/Suspension) • ciclopirox 8% - (Ciclodan Solution, ciclopirox 8%, Penlac) • clotrimazole - (Alevazol [OTC], clotrimazole [OTC], Lotrimin AF [OTC]) • clotrimazole/betamethasone - (clotrimazole/betamethasone, Dermacin. Rx Therazole Pak, Lotrisone) • econazole cream - (econazole) • econazole foam - (Ecoza) 51

Antifungals, Topical Agents in this class: • efinaconazole - (Jublia) • ketoconazole - (Extina, ketoconazole, Nizoral A-D Shampoo, Nizoral Shampoo, Xolegel) • luliconazole - (Luzu) • miconazole - (Azolen [OTC], Desenex [OTC] , Fungoid [OTC], Lotrimin AF Spray, [OTC], miconazole [OTC], Zeasorb [OTC]) • miconazole/zinc oxide/white petrolatum - (Vusion) • naftifine - (naftifine, Naftin) • nystatin - (nystatin) • nystatin/triamcinolone - (nystatin/triamcinolone) 52

Antifungals, Topical Agents in the class: oxiconazole - (oxiconazole, Oxistat) sertaconazole - (Ertazco) sulconazole - (Exelderm) tavaborole - (Kerydin) terbinafine - (Lamisil [OTC], Lamisil AT [OTC], terbinafine [OTC]) tolnaftate - (Fungoid-D [OTC], Lamisil AF Defense [OTC], Tinactin [OTC], tolnaftate [OTC]) • undecylenic acid - (Hongo Cura, Sponix Anti-Fungal [OTC]) • undecylenic acid/zinc undecylenic - (Fungi-Nail [OTC], Hongo Cura [OTC]) • • • 53

Antifungals, Topical • Topical antifungal agents have different spectrums of activity and are indicated to treat a wide variety of infections • Topical antifungals are available in numerous formulations including creams, foams, gels, lotions, ointments, powders, etc. • When it comes to efficacy comparisons, limited data are available for the treatment of the topical fungal infections • An antifungal with a corticosteroid topical formulation may be considered when there is coexisting inflammation 54

Antifungals, Topical • In terms of seborrheic dermatitis, reliable data are lacking to compare efficacy. • Based on the limited efficacy data, choice of therapy may be based on factors such as prior treatments and the severity of the condition 55

Antifungals, Topical Product Updates: • None 56

Beta Blockers 57 57

Beta Blockers Class Overview - indications for beta blockers include*: • • Hypertension Heart Failure Angina pectoris Myocardial Infarction Cardiac Arrhythmias Migraine Prophylaxis Tremor Hypertrophic sub-aortic stenosis *Not inclusive of all product indications, all products differ in indication 58

Beta Blockers Class Overview – Single Agents in this class: • acebutolol - (acebutolol; Sectral) • atenolol - (atenolol; Tenormin) • betaxolol - (betaxolol) • bisoprolol - (bisoprolol) • carvedilol - (carvedilol; Coreg) • carvedilol extended-release - (carvedilol ER; Coreg CR) • labetalol - (labetalol) • metoprolol succinate ER - (metoprolol succinate ER; Toprol XL, Kapspargo Sprinkle) • metoprolol tartrate - (metoprolol tartrate; Lopressor) 59

Beta Blockers Class Overview – Single Agents in this class (cont. ): • nadolol - (nadolol; Corgard) • nebivolol - (Bystolic) • pindolol - (pindolol) • propranolol - (propranolol) • propranolol - (Hemangeol) • propranolol ER - (propranolol ER; Inderal XL, Innopran XL) • propranolol LA - (propranolol LA; Inderal LA) • sotalol - (sotalol; Betapace) 60

Beta Blockers Class Overview - Single Agents in this class (cont. ): • sotalol - (sotalol AF; Betapace AF) • sotalol - (Sotylize) • timolol - (timolol) Class Overview - Beta-Blocker/Diuretic Combinations • atenolol/chlorthalidone - (atenolol/chlorthalidone; Tenoretic) • bisoprolol/HCTZ - (bisoprolol/HCTZ; Ziac) • metoprolol succinate/HCTZ - (metoprolol succinate/HCTZ; Dutoprol) • metoprolol tartrate/HCTZ - (metoprolol tartrate/HCTZ) 61

Beta Blockers Class Overview - Beta-Blocker/Diuretic Combinations (cont. ) • nadolol/bendroflumethiazide - (nadolol/bendroflumethiazide; Corzide) • propranolol/HCTZ - (propranolol/HCTZ) 62

Beta Blockers • About 75 million adults in the U. S. have HTN, with the highest prevalence among African American men at 43% and women at 45. 7%. • It is estimated that HTN is controlled in only a little over half of patients • Hypertension is an independent risk factor for the development of CVD, and the more elevated the blood pressure, the higher the risk of MI, stroke, heart failure, and kidney disease. • For the most part, Beta-blockers are one of the classes suggested as first-line treatment in patients with coronary artery disease, post-MI, HF, and diabetes 63

Beta Blockers • It appears that beta-blockers have similar efficacy for the treatment of hypertension. Also, the role of beta-blockers in primary prevention for hypertension is questionable. • The JNC-8 guidelines do not recommend beta-blockers as initial treatment of hypertension due to a demonstrated higher rate of the primary composite outcome of CV death, MI, or stroke compared to use of an ARB with their use, a finding that was driven largely by an increase in stroke • Nevertheless, Beta-blockers do play an important role in preventing recurrent ischemia, life-threatening ventricular arrhythmias, reducing the incidence of sudden cardiac death, as well as improving survival in patients with prior MI 64

Beta Blockers • In addition, the 2007 ACC/AHA chronic stable angina guidelines recommend indefinite beta-blocker therapy for blood pressure control in patients with CAD, acute coronary syndrome (ACS), or left ventricular dysfunction (LVD), with or without heart failure symptoms • Beta-blockers have also been shown to reduce mortality in patients with chronic heart failure. Bisoprolol, carvedilol, and metoprolol succinate extended -release are evidence-based and have proven that when used with ACE inhibitor, they reduce symptoms of HF and improve clinical status and patients’ well-being plus reduce the risk of death AND the combined risk of death and hospitalization by 30 to 40%; it is also important to note that bisoprolol is not specifically indicated for HF 65

Beta Blockers Product Updates: • None 66

BPH Treatments 67

BPH Treatments Class Overview: Alpha-Blockers • alfuzosin ER - (alfuzosin ER; Uroxatral) • doxazosin - (doxazosin; Cardura) • doxazosin ER - (Cardura XL) • silodosin - (silodosin; Rapaflo) • tamsulosin - (tamsulosin; Flomax) • terazosin - (terazosin) 68

BPH Treatments Class Overview: 5 -Alpha Reductase (5 AR) Inhibitors • dutasteride - (dutasteride; Avodart) • finasteride - (finasteride; Proscar) Class Overview: 5 -Alpha Reductase (5 AR) Inhibitor/Alpha Blocker Combinations • dutasteride/tamsulosin - (dutasteride/tamsulosin; Jalyn) Class Overview: • tadalafil - (tadalafil; Cialis) 69

BPH Treatments • BPH is one of the most common medical conditions in aging men, and it affects about 14 million men in the US • By age 60 years, approximately 50% of men demonstrate histo-pathologic BPH; and by 85 years of age, the prevalence becomes as high as 90% • Roles of drugs used for BPH include relieving LUTS, preventing complications, and, in some cases, can serve as an alternative to surgery • So, all products in this class are FDA approved for BPH but none are indicated for prostate cancer prevention • Many of the drugs in this class carry other non-BPH indications 70

BPH Treatments • Per the American Urological Association treatment guidelines, patients with mild BPH, as well as, patients with moderate or severe BPH who do not experience bothersome symptoms do not typically require pharmacotherapy • Alpha blockers are an appropriate treatment option for patients with moderate to severe BPH-related LUTS • According to the AUA, alfuzosin (Uroxatral), doxazosin (Cardura), tamsulosin (Flomax), and terazosin have comparable clinical efficacy; but when it comes to safety, selective alpha-blockers, such as alfuzosin (Uroxatral), tamsulosin (Flomax), and silodosin (Rapaflo), may have a decreased incidence of hypotension 71

BPH Treatments • Silodosin (Rapaflo) did not have any peer-reviewed studies published prior to the guideline update • The AUA states that the 5 AR inhibitors are appropriate and effective in treating men with LUTS associated with prostatic enlargement, but not for those with LUTS without prostatic enlargement • 5 AR inhibitors may also be used to prevent progression of BPH-related LUTS and to reduce the risk of urinary retention and future prostate-related surgery • Treatment with Combination therapy consisting of an alpha blocker and a 5 AR inhibitor is appropriate and effective for patients at highest risk for disease progression and for those who exhibit LUTS symptoms and have demonstrable prostate enlargement 72

BPH Treatments • The NIH-funded Medical Therapy of Prostatic Symptoms (MTOPS) and Comba. T studies indicated that combination therapy is likely to be more effective at inhibiting disease progression than monotherapy • 5 ARs are not to be administered to women or children, and women who are pregnant or may become pregnant should not handle dutasteride or finasteride 73

BPH Treatments Product Updates: • None 74

Calcium Channel Blockers 75

Calcium Channel Blockers Class Overview - indications for products in this class include*: • • Hypertension Angina Vasospastic Angina Ventricular Rate Control Unstable Angina Coronary Artery Disease Subarachnoid hemorrhage *Not inclusive of all product indications, all products differ in indication 76

Calcium Channel Blockers Class Overview - Dihydropyridines: • amlodipine - (amlodipine, Norvasc) • felodipine ER - (felodipine ER, Plendil) • isradipine - (isradipine) • nicardipine - (Cardene, nicardipine) • nicardipine SR - (Cardene SR) • nifedipine - (nifedipine, Procardia) • nifedipine ER, SA, SR - (Adalat CC; Afeditab CR; Nifediac CC; Nifedical XL; nifedipine ER, SA, SR; Procardia XL) • nimodipine - (nimodipine) 77

Calcium Channel Blockers Class Overview: Dihydropyridines (cont. ) • nimodipine solution - (Nymalize) • nisoldipine ER- (nisoldipine ER; Sular) Class Overview: Non-dihydropyridines • diltiazem - (diltiazem; Cardizem) • diltiazem ER - (diltiazem ER; Cardizem LA, Matzim LA) • diltiazem ER - (diltiazem ER; Cardizem CD, Cartia XT, Dilacor XR, Dilt CD, Taztia XT, Tiazac) 78

Calcium Channel Blockers Class Overview: Non-dihydropyridines (cont. ) • diltiazem ER - (Dilt XR, Diltia XT) • verapamil - (verapamil; Calan) • verapamil ER - (Covera-HS) • verapamil ER - (verapamil ER; Verelan PM) • verapamil SR - (verapamil SR; Calan SR, Isoptin SR, Verelan) 79

Calcium Channel Blockers • Calcium channel blockers (CCBs) are widely used in the treatment of hypertension and angina pectoris • JNC-8 lists thiazide diuretics, CCBs, ACE inhibitors, and ARBs as first-line therapy options for HTN in non-African American patients; for African Americans, only thiazide diuretics and CCBs are first line treatments • The benefits of CCBs have been clearly demonstrated for controlling angina and hypertension • For hypertension, no CCB has demonstrated an improved clinical outcome over another • Because of their potent peripheral vasodilating effects, dihydropyridines may cause a baroreceptor-mediated reflex increase in heart rate 80

Calcium Channel Blockers • Diltiazem decreases AV conduction and heart rate, while verapamil decreases heart rate, slows AV nodal conduction to the greatest extent of the CCBs and is useful for supraventricular tachyarrhythmias • Short-acting nifedipine has been associated with increased coronary mortality in patients with a history of MI and should not be used for hypertension • ALLHAT is one of the main studies which enrolled patients with hypertension and a known CAD risk factor. • It showed that chlorthalidone, amlodipine, and lisinopril had similar outcomes of combined fatal coronary heart disease (CHD) and nonfatal MI 81

Calcium Channel Blockers • Several large trials enrolling patients with hypertension have shown that CCBs have beneficial clinical effects on composite cardiovascular outcomes or individual clinical outcomes equivalent to the comparator antihypertensives, but not superior 82

Contraceptives, Oral 83

Contraceptives, Oral Class Overview - indications for products in this class include: • The prevention of pregnancy 84

Contraceptives, Oral Class Overview – Combined Pill: desogestrel/ethinyl estradiol APRI EMOQUETTE KARIVA VELIVET AZURETTE ENSKYCE MIRCETTE VIORELE BEKYREE ISIBLOOM PIMTREA VOLNEA CAZIANT JULEBER RECLIPSEN CYRED/CYRED EQ KALLIGA SIMLIYA drospirenone/ethinyl estradiol/levomefolate BEYAZ SAYFRAL TYDEMY estradiol valerate/dienogest NATAZIA 85
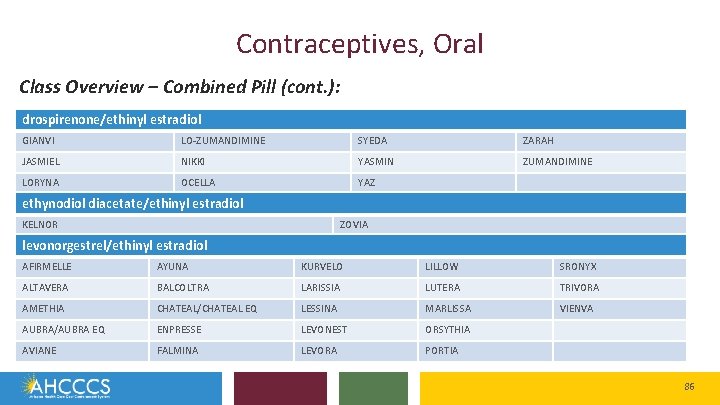
Contraceptives, Oral Class Overview – Combined Pill (cont. ): drospirenone/ethinyl estradiol GIANVI LO-ZUMANDIMINE SYEDA ZARAH JASMIEL NIKKI YASMIN ZUMANDIMINE LORYNA OCELLA YAZ ethynodiol diacetate/ethinyl estradiol KELNOR ZOVIA levonorgestrel/ethinyl estradiol AFIRMELLE AYUNA KURVELO LILLOW SRONYX ALTAVERA BALCOLTRA LARISSIA LUTERA TRIVORA AMETHIA CHATEAL/CHATEAL EQ LESSINA MARLISSA VIENVA AUBRA/AUBRA EQ ENPRESSE LEVONEST ORSYTHIA AVIANE FALMINA LEVORA PORTIA 86
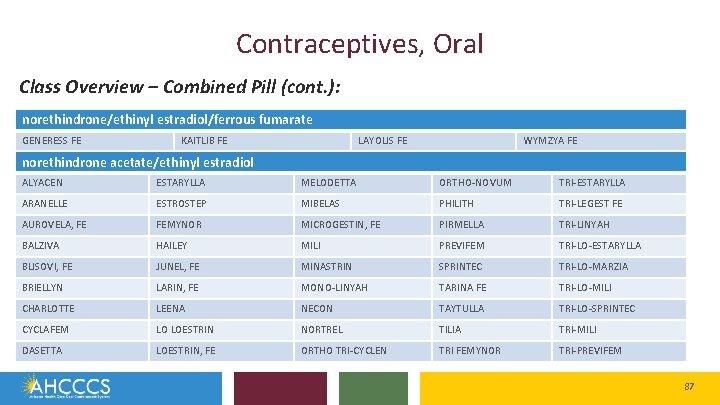
Contraceptives, Oral Class Overview – Combined Pill (cont. ): norethindrone/ethinyl estradiol/ferrous fumarate GENERESS FE KAITLIB FE LAYOLIS FE WYMZYA FE norethindrone acetate/ethinyl estradiol ALYACEN ESTARYLLA MELODETTA ORTHO-NOVUM TRI-ESTARYLLA ARANELLE ESTROSTEP MIBELAS PHILITH TRI-LEGEST FE AUROVELA, FE FEMYNOR MICROGESTIN, FE PIRMELLA TRI-LINYAH BALZIVA HAILEY MILI PREVIFEM TRI-LO-ESTARYLLA BLISOVI, FE JUNEL, FE MINASTRIN SPRINTEC TRI-LO-MARZIA BRIELLYN LARIN, FE MONO-LINYAH TARINA FE TRI-LO-MILI CHARLOTTE LEENA NECON TAYTULLA TRI-LO-SPRINTEC CYCLAFEM LO LOESTRIN NORTREL TILIA TRI-MILI DASETTA LOESTRIN, FE ORTHO TRI-CYCLEN TRI FEMYNOR TRI-PREVIFEM 87
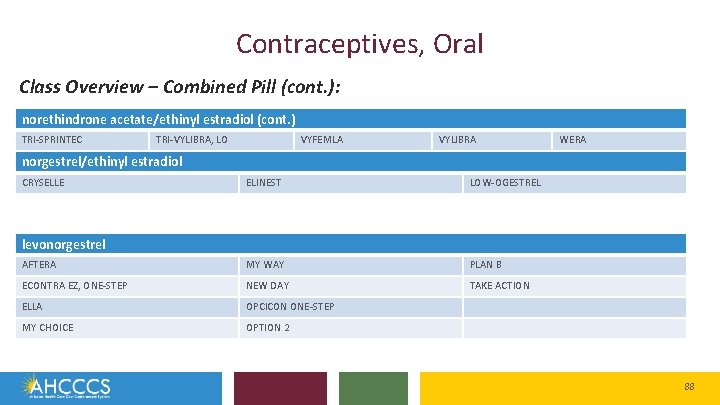
Contraceptives, Oral Class Overview – Combined Pill (cont. ): norethindrone acetate/ethinyl estradiol (cont. ) TRI-SPRINTEC TRI-VYLIBRA, LO VYFEMLA VYLIBRA WERA norgestrel/ethinyl estradiol CRYSELLE ELINEST LOW-OGESTREL levonorgestrel Class Overview – Emergency Contraceptive: AFTERA MY WAY PLAN B ECONTRA EZ, ONE-STEP NEW DAY TAKE ACTION ELLA OPCICON ONE-STEP MY CHOICE OPTION 2 88

Contraceptives, Oral Class Overview – Extended Continuous Use Combined Pill: levonorgestrel/ethinyl estradiol AMETHYST DAYSEE JOLESSA RIVELSA AMETHIA LO FAYOSIM LOJAIMIESS SEASONIQUE ASHLYNA INTROVALE LOSEASONIQUE SETLAKIN CAMRESE, LO JAIMIESS QUARTETTE SIMPESSE drospirenone Class Overview – Progestin Only: SLYND norethindrone CAMILA HEATHER LYZA SHAROBEL DEBLITANE INCASSIA NORA-BE TULANA ERRIN JENCYCLA NORLYDA 89

Contraceptives, Oral • This is a first-time review of this class • Hormonal oral contraceptives (OCs) are available in various dosage forms for prevention of pregnancy and are available as either a combination of estrogen and progestin (combination OCs) or progestin alone. • Products differ in the specific hormones they contain and how these hormones are dosed throughout the cycle (hormone phases) resulting in several product options. • The various hormone combinations and phases in which they are dosed create products that produce different cycle lengths and physiological effects 90

Contraceptives, Oral • OC’s work primarily by suppression of ovulation • Progestin prevents the luteinizing hormone (LH) surge required for the release of the ovum o Secondarily, progestin thickens cervical mucus and decreases tubal motility (creating a more difficult passage for sperm), and thins the endometrium (resulting in tissue that is less receptive to implantation) • Estrogen stabilizes the endometrium providing for an acceptable cycle control and bleeding profile, inhibits the release of follicle-stimulating hormone (FSH) from the pituitary which inhibits the development of a dominant follicle and thus potentiates inhibition of the LH surge 91

Contraceptives, Oral • The majority of OCs contain the synthetic estrogen ethinyl estradiol, and the dose varies across products from 20 mcg/day to 50 mcg/day. o Estradiol valerate is a prodrug of estradiol, utilized in Natazia • There are currently 9 different progestins contained in OCs and is the primary differentiator among different OCs • OCs are commonly referred to as first through fourth generation progestins based on when they were introduced into the market o o 1 st generation agents include norethindrone, norethindrone acetate, and ethynodiol diacetate 2 nd generation progestins include norgestrel and its active isomer, levonorgestrel 3 rd generation agents are norgestimate and desogestrel 4 th generation agents are drospirenone and dienogest 92

Contraceptives, Oral • Progestins vary in their progestational, estrogenic, antiestrogenic, androgenic activity. o o The 1 st generation progestins are generally well tolerated but are associated with spotting and breakthrough bleeding. The 2 nd generation drugs are more potent progestins with longer half-lives, and have more androgenic activity compared to the first generation drugs and may be associated with more androgenic side effects, such as hirsutism, acne, or dyslipidemia. Norgestrel is the most androgenic of the progestins. The 3 rd generation drugs are similar to the 2 nd generation drugs in their progestational activity but have less androgenic activity; adverse effects, such as acne, may occur less frequently. Norgestimate and desogestrel have the least androgenic activity of the progestins; however, they have been associated with a slightly higher risk of thrombosis than earlier agents. The 4 th generation, dienogest, the most recently introduced progestin, is antiandrogenic 93

Contraceptives, Oral • Traditional OCs are administered daily for 21 days, followed by a hormone-free week during which menstruation occurs • Extended cycle products (e. g. , 91 day cycle) delay or completely eliminate the break in hormone use and may be desirable to women who wish to avoid menstruation • Progestin-only OCs, also known as the “mini pill”, contain lower doses of the progestin norethindrone or, the newer product, drospirenone • The Centers for Disease Control and Prevention (CDC) continue to recommend combination oral contraceptives and progestin-only oral contraceptives as effective methods of contraception 94

Contraceptives, Oral • The use of OCs is associated with increased risks of serious conditions, including myocardial infarction (MI), thromboembolism, stroke, hepatic tumors, and gallbladder disease • While the risk of serious morbidity or mortality is very small in healthy women, underlying risk factors, such as obesity, diabetes, hypertension, and hyperlipidemia can increase morbidity and mortality. • OCs are contraindicated in women with a prior thromboembolic event or stroke, hepatic disease, cerebral vascular or coronary artery disease, undiagnosed abnormal uterine bleeding, uncontrolled hypertension, migraine headaches, particularly with focal neurologic symptoms, diabetes, a history of estrogen-dependent tumor, such as breast cancer or endometrium cancer, or major surgery with prolonged immobilization 95

Contraceptives, Oral Product Updates: • None 96

Contraceptives, Other 97

Contraceptives, Other Class Overview - indications for products in this class include: • The prevention of pregnancy 98

Contraceptives, Other Class Overview – Implant: Drug Availability FDA Classification etonorgestrel Implanon, Nexlpanon Implantable Rod Drug Availability FDA Classification medroxyprogesterone acetate vial, syringe; Depo-Provera syringe, vial Vaginal Contraceptive Ring Class Overview – Injection: Drug Availability Class Overview – Intrauterine Device (IUD): etonogestrel/ethinyl estradiol ring; Eluryng, Nuvaring Segesterone acetate/ethinyl estradiol Annovera FDA Classification Vaginal Contraceptive Ring 99
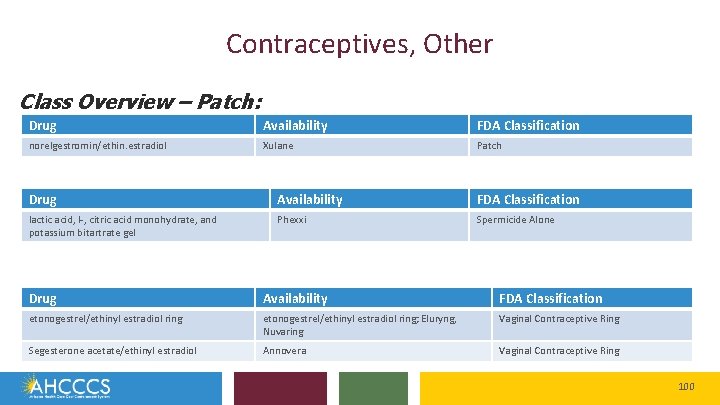
Contraceptives, Other Class Overview – Patch: Drug Availability FDA Classification norelgestromin/ethin. estradiol Xulane Patch Drug Availability Class Overview – Spermicide: lactic acid, l-, citric acid monohydrate, and potassium bitartrate gel Phexxi FDA Classification Spermicide Alone Drug Availability FDA Classification etonogestrel/ethinyl estradiol ring; Eluryng, Nuvaring Vaginal Contraceptive Ring Segesterone acetate/ethinyl estradiol Annovera Vaginal Contraceptive Ring Class Overview – Vaginal Ring: 100

Contraceptives, Other • This is a first-time review of this class • Hormonal contraceptives are available in various dosage forms for prevention of pregnancy and are available as either a combination of estrogen and progestin (combination OCs) or progestin alone. • Selection of the most appropriate product for a patient may depend on the desired phases of hormones, cycle length, associated product risks, side effect profile, and tolerability. • The American College of Obstetricians and Gynecologists (ACOG) published guidance that states long-acting reversible contraceptives (LARC) are safe and have higher rates of efficacy, continuation, and satisfaction compared with short-acting contraceptives 101

Contraceptives, Other • Implantable Rod: o o o Nexplanon is a progestin containing implant placed subdermally for up to 3 years Previously, Implanon was also available however the manufacturer discontinued this product Insertion and removal complications include pain, paresthesias, bleeding, hematoma, scarring, infection, or migration to vasculature, including pulmonary vessels, may occur Surgical interventions may be necessary to remove implants The most common adverse reactions were change in menstrual bleeding pattern, headache, vaginitis, weight increase, acne, breast pain, abdominal pain, and pharyngitis Nexplanon may become less effective in overweight women over time, especially in the presence of other factors that could decrease etonogestrel concentrations 102

Contraceptives, Other • Injection: o o medroxyprogesterone acetate is a derivative of progesterone and can be dosed every 12 to 14 weeks Contraindications include active thrombophlebitis, or current or history of thromboembolic disorders, or cerebral vascular disease, Known/suspected/ or past malignancy of the breast, significant liver disease, Known hypersensitivity, and undiagnosed vaginal bleeding Warnings include loss of bone mineral density, arterial and venous thromboembolic disorders, cancer risks (e. g. , breast, cervical), ectopic pregnancy, anaphylaxis, fluid retention, weight gain, delayed return of ovulation or fertility, depression, injection site reactions, bleeding irregularities, risk of hyperglycemia in patients with diabetes, and jaundice and elevated transaminase The most frequently reported adverse reactions were dysfunctional uterine bleeding (e. g. , irregular, increased, decreased, or spotting), headache, increased weight, amenorrhea, and injection site reactions (e. g. , pain/tenderness, nodule/lump, persistent atrophy/indentation/dimpling or lipodystrophy) 103

Contraceptives, Other • IUD: o o Intrauterine Devices (IUD’s) are a very small T-shaped device inserted into the uterus that are used to prevent pregnancy (the T-shape helps prevent sperm from reaching the egg) There are 2 types of IUD’s available § § o Copper IUD (Paragard): IUDs that use copper to prevent pregnancy (no hormones); copper is toxic to sperm Hormonal IUD (Kyleena, Mirena, Liletta, Skyla): IUDs that uses the hormone progestin to prevent pregnancy; progestin thickens the mucus in the cervix to trap and block sperm, it can also prevent eggs from leaving the ovaries The durations of insertion vary for the different IUD products § § § Paragard: 10 – 12 years Kyleena: 5 years Liletta: 6 years Mirena: 5 – 7 years Skyla: 3 years 104

Contraceptives, Other • IUD: o Contraindications for IUD’s include use as post-coital contraception (emergency contraception), congenital or acquired uterine anomaly (including fibroids, that distorts the uterine cavity), acute pelvic inflammatory disease (PID) or a history of PID unless there has been a subsequent intrauterine pregnancy, postpartum endometritis or infected abortion in the past 3 months, known or suspected uterine or cervical neoplasia, known or suspected breast cancer or other progestinsensitive cancer, uterine bleeding of unknown etiology, untreated acute cervicitis or vaginitis, (including bacterial vaginosis or other lower genital tract infections until infection is controlled), acute liver disease or liver tumor (benign or malignant), conditions associated with increased susceptibility to pelvic infections, previously inserted intrauterine device (IUD) that has not been removed, and hypersensitivity to any component of this product 105

Contraceptives, Other • IUD: o o o Warning for IUD’s include risk of ectopic pregnancy, risk of intrauterine pregnancy, sepsis, pelvic infection, perforation, expulsion, ovarian cysts, bleeding pattern alterations, and breast cancer Adverse reactions include vulvovaginitis, ovarian cysts, abdominal pain/pelvic pain, headache/migraine, acne/seborrhea, dysmenorrhea/uterine spasm, breast pain/breast discomfort, and increased bleeding Product labeling for IUD’s contain clinical considerations for removal including coagulopathies, migraine/severe headache, marked increase in blood pressure, severe arterial disease such as stroke/myocardial infarction 106

Contraceptives, Other • Patch: o o The transdermal patch (Xulane) is an estrogen/progestin combination hormonal contraceptive (CHC) indicated for the prevention of pregnancy in women with a BMI < 30 kg/m 2 for whom a transdermal delivery system is an appropriate method of contraception The patch uses a 28 -day cycle where a patch is placed weekly for 3 weeks, followed by 1 week with no patch § o A new patch should be placed on the same day every week; only 1 patch should be worn at a time Contraindications include use in patients with high risk of arterial or venous thromboembolic events, BMI ≥ 30 kg/m 2, breast cancer or other estrogen- or progestin-sensitive cancer, liver tumors or liver disease, undiagnosed abnormal uterine bleeding, pregnancy, co-administration with hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir (with or without dasabuvir) 107

Contraceptives, Other • Patch: o Warnings include vascular risks (thrombotic events), liver disease, high blood pressure, carbohydrate and lipid metabolic effects (dyslipidemia), headache, and uterine bleeding o The most frequent adverse reactions reported were breast symptoms, nausea/vomiting, headache, application site disorder, abdominal pain, dysmenorrhea, vaginal bleeding and menstrual disorders, and mood/affect and anxiety disorders 108

Contraceptives, Other • Spermicide: o o Spermicide is a type of contraception that either kills sperm or stops it from moving There are many over the counter products that contain the active ingredient nanoxynol-9. The prescription product, Phexxi gel, contains lactic acid, citric acid, and potassium bitartrate which lowers the vaginal p. H and reduces sperm motility One pre-filled dose applicator should be administered vaginally immediately before vaginal intercourse (up to one hour) 109

• Spermicide: o o Contraceptives, Other There are no boxed warning or contraindications for Phexxi Warnings include cystitis/pyelonephritis (should not be used in women with a history of recurrent urinary tract infection or urinary abnormalities) The most common adverse reactions were vulvovaginal burning sensation, vulvovaginal pruritus, vulvovaginal mycotic infection, urinary tract infection, vulvovaginal discomfort, bacterial vaginosis, vaginal discharge, genital discomfort, dysuria, and vulvovaginal pain Phexxi may be used: § § § during any part of the menstrual cycle with other forms of contraception (hormonal contraceptives, condoms, diaphragms) with products for vaginal infections (miconazole, metronidazole) 110

• Vaginal Rings: o o o Contraceptives, Other Vaginal rings contain an estrogen/progestin combination in a non-biodegradable ring One ring is inserted into the vagina and remains in place continually for 3 weeks, then removed for 1 week Patients should regularly check for the presence of vaginal rings as they can be accidentally expelled (i. e. , while removing a tampon, during intercourse, or with straining during a bowel movement) If the vaginal ring is accidentally expelled and is left outside of the vagina for < 3 hours, contraceptive efficacy is not reduced, and it can be rinsed and reinserted If a vaginal ring is lost, a new vaginal ring should be inserted and the regimen should be continued without alteration 111

• Vaginal Rings: o o o Contraceptives, Other Contraindications include use in patients with high risk of arterial or venous thromboembolic events, breast cancer or other estrogen- or progestin-sensitive cancer, liver tumors or liver disease, undiagnosed abnormal uterine bleeding, hypersensitivity to any of the components, pregnancy, co -administration with hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir (with or without dasabuvir) Warnings include vascular risks (thrombotic events), toxic shock syndrome, liver disease, high blood pressure, carbohydrate and lipid metabolic effects (dyslipidemia), headache, and uterine bleeding The most common adverse reactions were vaginitis, headache (including migraine), mood changes (e. g. , depression, mood swings, mood altered, depressed mood, affect lability), device-related events (e. g. , expulsion/discomfort/foreign body sensation), nausea/vomiting, vaginal discharge, increased weight, vaginal discomfort, breast pain/discomfort/tenderness, dysmenorrhea, abdominal pain, acne, and decreased libido 112

Contraceptives, Other Product Updates: • None 113

Bladder Relaxant Preparations 114

Bladder Relaxant Preparations Class Overview – Agents in the class: • darifenacin - (darifenacin; Enablex) • fesoterodine ER - (Toviaz) • mirabegron ER - (Myrbetriq) • oxybutynin – (oxybutynin; Ditropan XL, Gelnique [gel], Oxytrol [patch]) • solifenacin - (solifenacin; Vesicare) • tolterodine ER - (tolterodine ER; Detrol, Detrol LA) • trospium – (trospium, trospium ER) 115

Bladder Relaxant Preparations • • This is a first-time review of this class Overactive bladder (OAB) is a chronic and debilitating syndrome that is characterized by: o o • The overall prevalence of OAB occurs equally in men (16%) and women (16. 9%); however, more women suffer from OAB with incontinence. o • urinary urgency with or without urge incontinence usually in combination with urinary frequency (8 or more voiding episodes per 24 hours) and nocturia (awakening 1 or more times per night to void) The prevalence of OAB is almost 20% in those older than 60 years of age Many conditions are associated with the symptoms of OAB including: o lower urinary tract conditions (e. g. , urinary tract infection, obstruction), neurological conditions (e. g. , stroke, Alzheimer’s disease), systemic conditions (e. g. , heart failure, vascular insufficiency), functional and behavioral conditions (e. g. , impaired mobility), and use of various medications (e. g. , diuretics, narcotics). 116

Bladder Relaxant Preparations • OAB and urinary incontinence (UI) are associated with skin infections and irritations and, in the elderly, an increased risk of falls and fractures. • The symptoms of OAB are usually associated with overactivity of the detrusor muscle as it contracts spastically, sometimes without a known cause. • This results in sustained high bladder pressure and urgency or urge incontinence depending on the sphincter response. • The management of OAB includes both pharmacological and nonpharmacological interventions (e. g. , bladder training, pelvic floor muscle exercises). • Antimuscarinic agents that relax the detrusor muscle or prevent a bladder contraction are effective for OAB. 117

Bladder Relaxant Preparations • Per the American Urological Association (AUA) treatment guidelines, the following is recommended: o o o Behavioral therapy (e. g. , bladder training, bladder control strategies, pelvic floor muscle training, and fluid management) as first-line therapy. Pharmacological therapy may be combined with behavioral therapy as first-line treatment as well. Oral antimuscarinics (darifenacin, fesoterodine, oxybutynin, solifenacin, tolterodine, trospium) or beta-3 adrenergic receptor agonists should be offered as second-line therapy. There may be consideration of combination anti-muscarinic and beta-3 adrenergic receptor agonist in patients refractory to monotherapy with either mechanism alone. Surgery is reserved for patients with severe refractory OAB symptoms or who are not candidates for oral therapy 118

Bladder Relaxant Preparations • Silodosin (Rapaflo) did not have any peer-reviewed studies published prior to the guideline update. • The AUA states that the 5 AR inhibitors are appropriate and effective in treating men with LUTS associated with prostatic enlargement, but not for those with LUTS without prostatic enlargement • 5 AR inhibitors may also be used to prevent progression of BPH-related LUTS and to reduce the risk of urinary retention and future prostate-related surgery • Treatment with Combination therapy consisting of an alpha blocker and a 5 AR inhibitor is appropriate and effective for patients at highest risk for disease progression and for those who exhibit LUTS symptoms and have demonstrable prostate enlargement 119

Bladder Relaxant Preparations • The NIH-funded Medical Therapy of Prostatic Symptoms (MTOPS) and Comba. T studies indicated that combination therapy is likely to be more effective at inhibiting disease progression than monotherapy • 5 ARs are not to be administered to women or children, and women who are pregnant or may become pregnant should not handle dutasteride or finasteride 120

Bladder Relaxant Preparations Product Updates: • None 121

Antiparkinson’s Agents 122

Antiparkinson’s Agents cont’d Class Overview – Anticholinergics: • benztropine^ trihexyphenidyl^ Class Overview - Dopa decarboxylase Inhibitor: • carbidopa* - (carbidopa; Lodosyn) Class Overview - Dopamine precursor: • levodopa* - (Inbrija; levodopa) Class Overview – Dopamine precursor/dopa decarboxylase inhibitor combinations: • levodopa/carbidopa (levodopa/carbidopa tablet, ODT; Duopa, Rytary, Sinemet CR) * indicates the agent is approved for adjunctive therapy ^ indicates the agent has an additional indication for drug induced extrapyramidal symptoms (EPS) 123

Antiparkinson’s Agents cont’d Class Overview – MAO-B inhibitors: • rasagiline - (Azilect; rasagiline) • safinamide* - (Xadago) • selegiline* – (selegiline, seligiline ODT; Zelapar) Class Overview – Dopamine agonists: • bromocriptine* - (bromocriptine; Parlodel) • pramipexole+ - (Mirapex, Mirapex ER; pramipexole, pramipexole ER) • ropinirole+ - (ropinirole, ropinirole ER; Requip, Requip XL) • rotigotine+ - (Neupro) * indicates the agent is approved for adjunctive therapy + indicates the agent is approved for Restless Leg Syndrome 124

Antiparkinson’s Agents Class Overview – COMT inhibitors: o o entacapone* - (Comtan; entacapone) tolcapone* - (Tasmar; tolcapone) Class Overview – Dopamine precursor/dopa decarboxylase inhibitor/ COMT inhibitor: o levodopa/carbidopa/entacapone (levodopa/carbidopa/entacapone; Stalevo) 125

Antiparkinson’s Agents Class Overview – N-Methyl-D-aspartate (NMDA) receptor type: • amantadine^ • amantadine ER - (Gocovri*; Osmolex ER^) Class Overview – Adenosine receptor antagonist: • istradefylline* (Nourianz) * indicates the agent is approved for adjunctive therapy ^ indicates the agent has an additional indication for drug induced extrapyramidal symptoms (EPS) 126

Antiparkinson’s Agents • This is a first-time review of this class • Parkinson’s disease (PD) is a progressive, neurodegenerative disorder with cardinal motor features of tremor, bradykinesia, and rigidity • This disease affects approximately 1% of individuals older than 60 years and the incidence increases significantly with age • The term “parkinsonism” describes the motor syndrome of bradykinesia, rigidity, tremor, and balance and gait disturbances • Secondary parkinsonism, which has a different etiology and pathology than PD, is the main clinical manifestation of multiple disorders including brain tumors near the basal ganglia, cerebral atherosclerosis, head trauma, and progressive supranuclear palsy and can also be caused by toxins and drugs (antipsychotics agents) 127

Antiparkinson’s Agents • Parkinson’s disease and secondary parkinsonism are characterized by striatal dopamine deficiency. • In PD, the degeneration of dopamine-containing neurons in the substantia nigra leads to the formation of Lewy bodies (intracellular neuronal inclusion bodies). • While Lewy bodies are not present in secondary parkinsonism, the nigral striatal pathway may be impaired and nigral cell loss or loss of striatal cellular elements may occur. 128

• Antiparkinson’s Agents Anticholinergics were the first medications indicated for the treatment of PD. o o o • Anticholinergics, such as benztropine and trihexyphenidyl, improve motor symptoms in some patients with PD, especially younger patients with resting tremor as a predominant symptom. Today, they are used primarily as adjuncts to levodopa treatment and as treatments for tremor symptoms. These drugs often cause side effects in the elderly and are contraindicated in patients with glaucoma, benign prostatic hypertrophy, and dementia. Levodopa replaces dopamine in the brain which represented a major breakthrough in the treatment of PD (exogenous dopamine does not cross the blood-brain barrier). o o o The combination of levodopa with carbidopa (a peripheral dopa decarboxylase inhibitor that does not cross the blood-brain barrier) led to an increase in the amount of levodopa available to the brain for conversion to dopamine and a reduction in the incidence of nausea and vomiting Long-term treatment with levodopa is complicated by the development of motor fluctuations, dyskinesias, and neuropsychiatric complications Patients may experience a “wearing-off” effect characterized by a shorter duration of benefit from each levodopa dose, causing parkinsonian symptoms to re-emerge 129

• Antiparkinson’s Agents Monoamine oxidase B (MAO-B) is an enzyme predominantly located in the brain that breaks down several chemicals, but primarily dopamine. o o • MAO-B is abundant in the striatum and involved in dopamine metabolism, theory is that MAO-B inhibition will increase the quantity of dopamine available and result in the reduction of some of the motor symptoms seen with PD. rasagiline (generic, Azilect) and selegiline (generic, Zelapar), highly selective inhibitors of MAO-B, have been shown to cause a slight improvement in motor performance upon initiation of therapy and to delay the development of disability that requires the addition of levodopa Dopamine agonists including bromocriptine (Parlodel), pramipexole (Mirapex, Mirapex ER), ropinirole (Requip, Requip XL), and rotigotine (Neupro) are used in early PD o o These agents have a levodopa-sparing effect and can reduce the frequency of “off” time. Monotherapy with dopamine agonists has been shown to reduce the subsequent dyskinesias and other motor complications in comparison to levodopa, however, there is the potential for orthostatic hypotension and neuropsychiatric adverse effects, such as confusion and hallucinations 130

Antiparkinson’s Agents • Catechol-O-methyltransferase (COMT) inhibitors, entacapone (Comtan), and tolcapone (Tasmar) reduces the end-of-dose failure (“wearing off”) of levodopa therapy that causes motor complications o o • By reducing the peripheral metabolism of levodopa, COMT inhibitors allow for the use of lower doses of levodopa and both agents are approved as adjuncts to levodopa therapy. Some experts recommend the initiation of a COMT inhibitor at the onset of levodopa therapy to reduce the risk of developing motor complications An adenosine A 2 a antagonist, istradefylline (Nourianz) is approved as adjunctive therapy to levodopa/carbidopa to decrease “off” episodes and is a once daily oral option o o The exact mechanism of adenosine A 2 a antagonists in PD is not known, but it is not dopaminergic. istradefylline may be neuroprotective and prevent dyskinesia when used with levodopa 131

Antiparkinson’s Agents • Despite advances in treatments over the years, there is no cure for PD. • Symptomatic therapy can provide benefit for quite some time, but the continued, however slow, progression of PD eventually results in significant disability. • Patients may not require treatment in the early stages of PD if symptoms do not cause functional impairment. • As the disease progresses, however, therapy becomes more complex, requiring dosage adjustments, incorporation of multiple medications, and the use of rescue treatments. • It is generally recommended that medication regimens be kept as simple as possible since the risk of adverse effects is generally lower when fewer agents are used at higher doses than when multiple drugs are used at lower doses 132

Antiparkinson’s Agents • An evidence-based review updated in 2018 by the Movement Disorder Society ranked the efficacy of the various treatments based on placebo-controlled trials of patients with PD between 2004 and 2016. o o o In the review, oral levodopa/carbidopa, the MAO-B inhibitors, and the dopamine agonists are all rated as efficacious for symptomatic monotherapy in patients with PD; exceptions to this are bromocriptine and ropinirole ER which are considered likely efficacious. The anticholinergics, as well as amantadine, are rated as likely efficacious as monotherapy. As symptomatic adjunct therapy to levodopa, the nonergot dopamine agonists (rotigotine, tolcapone, and rasagiline) are considered efficacious; anticholinergics, amantadine, and bromocriptine are likely efficacious. There is insufficient evidence to rate selegiline. Entacapone and safinamide are noted to be non-efficacious as an adjunct therapy to levodopa. 133

Antiparkinson’s Agents • An evidence-based review updated in 2018 by the Movement Disorder Society ranked the efficacy of the various treatments based on placebocontrolled trials of patients with PD between 2004 and 2016 (cont. ) o o For the prevention/delay of dyskinesia, pramipexole and ropinirole are efficacious; bromocriptine is likely efficacious. Efficacious treatments for motor fluctuations include COMT inhibitors, levodopa/carbidopa (standard and extended-release formulations), rasagiline, and dopamine agonists; the exception to this is bromocriptine, which is likely efficacious. Amantadine is rated efficacious for the treatment of dyskinesia. Duodenal administration of levodopa/carbidopa was also likely efficacious for treatment of motor fluctuations and dyskinesia. 134

Antiparkinson’s Agents Product Updates: • None 135

Lipotropics, Statins 136

Lipotropics, Statins Class Overview - indications for products in this class include*: • • • Heterozygous familial hypercholesterolemia Homozygous familial hypercholesterolemia Mixed dyslipidemia Primary hypercholesterolemia Primary prevention of coronary events in patients with CVD Secondary prevention of coronary events in patients with CHD *Not inclusive of all product indications, all products differ in indication 137

Lipotropics, Statins Class Overview - Statins: • atorvastatin – (atorvastatin; Lipitor) • fluvastatin ER – (fluvastatin ER; Lescol XL) • Lovastatin • lovastatin ER – (lovastatin ER; Altoprev) • pitavastatin – (Livalo, Zypitamag) • pravastatin – (pravastatin; Pravachol) • Rosuvastatin – (rosuvastatin; Crestor, Ezallor Sprinkle) • Simvastatin – (simvastatin; Flolipid, Zocor) 138

Lipotropics, Statins Class Overview – Statin Combinations: • amlodipine/atorvastatin – (amlodipine/atorvastatin; Caduet) • ezetimibe/simvastatin – (ezetimibe/simvastatin; Vytorin) 139

Lipotropics, Statins • This is a first-time review of this therapeutic class • Clinical studies clearly show that ASCVD events are reduced by using the maximum tolerated statin intensity in 1 of 4 predefined groups shown to benefit from statin therapy. o o Adults with clinical ASCVD Patients with primary elevations of LDL-C ≥ 190 mg/d. L Patients who are 40 to 75 years of age with diabetes Patients 40 to 75 years of age with an estimated 10 -year ASCVD risk estimated by Pooled Cohort Equations (PCE) • ASCVD prevention benefit of statin therapy may be less clear in selected individuals who do not fall into 1 of the 4 benefit groups 140

Lipotropics, Statins • The ACC/AHA addresses the use of statins in cholesterol management. • ACC/AHA classifies the intensity of statin therapy based on the average expected LDL-C response to a specific statin and dose. • Generally, high-intensity statin therapy on average lowers LDL-C by ≥ 50%, moderate-intensity therapy lowers LDL-C by 30% to < 50%, and lower-intensity statin therapy lowers LDL-C by < 30%. o o o High-intensity statin therapy: atorvastatin 40 mg and 80 mg, rosuvastatin 20 mg and 40 mg, Moderate-intensity therapy: atorvastatin 10 mg and 20 mg, rosuvastatin 5 mg and 10 mg, simvastatin 20 mg to 40 mg, pravastatin 40 mg and 80 mg, lovastatin 40 mg, fluvastatin 80 mg, and pitavastatin 2 mg to 4 mg. All remaining lower dosages are classified as lower-intensity therapy 141

Lipotropics, Statins • For primary prevention of ASCVD in patients ages 40 to 75 years with LDL-C 70 to 189 mg/d. L, lifestyle management alone is advised in those with low ASCVD risk (10 -year risk < 5%). • For patients with borderline risk (10 -year risk of 5% to < 7. 5%), moderateintensity statin therapy may be added if risk enhancers are present. • Moderate-intensity statin therapy is recommended in patients with intermediate risk (10 -year risk ≥ 7. 5% to < 20%) and as initial therapy in diabetic patients (regardless of ASCVD risk). • High-intensity statin therapy is recommended in high-risk (10 -year risk ≥ 20%) patients and diabetic adults with multiple risk factors. 142

Lipotropics, Statins • Multiple other organizations recommend the use of statins in the management of hypercholesterolemia including: o o o The American Association of Clinical Endocrinologists (AACE) and the American College of Endocrinology (ACE) The American Diabetes Association (ADA) The National Heart, Lung and Blood Institute (NHLBI) The National Lipid Association (NLA) The US Preventive Services Task Force (USPSTF) American Academy of Pediatrics (AAP) endorsed the 2012 guidelines by the NHLBI on cardiovascular health and risk reduction in children and adolescents which outlines age appropriate lipid screening in the pediatric population 143

Lipotropics, Statins Product Updates: • None 144

Lipotropics, Other 145

Lipotropics, Other Class Overview - indications for products in this class include*: • Primary hypercholesterolemia • To reduce elevated LDL-C, total-cholesterol (Total-C), triglycerides (TG), and Apo B, and to increase HDL-C • Severe Hypertriglyceridemia *Not inclusive of all product indications, all products differ in indication 146

Lipotropics, Other Class Overview: adenosine triphosphate-citrate lyase (ACL) inhibitor & combinations • bempedoic acid - (Nexletol) • bempedoic acid/ezetimibe - (Nexlizet) Class Overview: Apolipoprotein B Synthesis Inhibitor • lomitapide - (Juxtapid) Class Overview: Bile Acid Sequestrants • cholestyramine - (cholestyramine; Questran, Questran Light) • colesevelam – (colesevelam; Welchol) • colestipol – (colestipol; Colestid) 147

Lipotropics, Other Class Overview: Cholesterol Absorption Inhibitor • ezetimibe (ezetimibe; Zetia) Class Overview: Fibric Acids • fenofibrate (fenofibrate; Antara, Fenoglide, Fibricor, Lipofen, Tricor, Triglide, Trilipix) Class Overview: Fibric Acids • fenofibrate – (fenofibrate; Antara, Fenoglide, Lipofen, Tricor, Triglide) • fenofibric acid – (fenofibric acid; Fibricor, Trilipix) • gemfibrozil – (gemfibrozil; Lopid) Class Overview: Niacin • niacin - (niacin, niacin ER; Niaspan, Niacor) 148

Lipotropics, Other Class Overview: Omega-3 Fatty Acids • icosapent ethyl - (Vascepa) • omega-3 -acid ethyl esters – (omega-3 -acid ethyl esters; Lovaza) Class Overview: Proprotein Convertase Subtilisin/Kexin Type 9 (PCSK 9) Inhibitors • alirocumab - (Praluent) • evolocumab - (Repatha) 149
Lipotropics, Other • This is a first-time review of this class • Many clinical trials have demonstrated that a high serum concentration of lowdensity lipoprotein cholesterol (LDL-C) and low levels of high-density lipoprotein cholesterol (HDL-C) are major risk factors for coronary heart disease (CHD) • The National Health and Nutrition Examination Survey (NHANES) reported that in 2015 to 2016 approximately 12. 4% of adults had high total cholesterol (≥ 240 mg/d. L) and 18. 4% had low HDL-C (< 40 mg/d. L); the incidence being higher in women (13. 7%) compared to men (11. 2%) 150

Lipotropics, Other • These non-statin lipotropic agents have various mechanisms of action: o o o Apolipoprotein B (apo-B) is a structural protein of very low-density lipoproteins (VLDL) and low-density lipoproteins (LDL). Microsomal triglyceride transfer protein (MTP) transfers triglycerides onto apo B during the production of VLDL, a precursor to LDL. Lomitapide (Juxtapid) directly binds and inhibits MTP, preventing the synthesis of apo-B-containing proteins in enterocytes and hepatocytes. This results in decreased synthesis of VLDL and, thereby, reduced plasma LDL-C levels. Bile acids emulsify the dietary fat thus facilitating absorption. The bile acid sequestrants, cholestyramine, colestipol (Colestid), and colesevelam (Welchol), bind bile acids in the intestine to form an insoluble complex which is excreted in the feces, thereby interrupting enterohepatic circulation. 151

Lipotropics, Other • These non-statin lipotropic agents have various mechanisms of action: o o Ezetimibe (Zetia) inhibits cholesterol absorption along the brush border of the small intestine. This leads to a decrease in the delivery of intestinal cholesterol to the liver. The effects of the fibric acids, fenofibrate (Antara, Fenoglide, Lipofen, Tricor, Triglide), fenofibric acid (Fibricor, Trilipix; the active metabolite of fenofibrate), and gemfibrozil (Lopid), have been explained by the activation of peroxisome proliferator activated receptor alpha (PPARα) which increases lipolysis and elimination of TG from plasma Niacin (nicotinic acid) inhibits lipolysis in adipocytes and possibly inhibits hepatic TG production resulting in a reduction in the synthesis of VLDL that is available for conversion to LDL-C The mechanism of action of omega-3 -acid ethyl esters is not completely understood. It is thought that the omega-3 -acid ethyl esters may reduce the synthesis of TG by the liver. 152

Lipotropics, Other • These non-statin lipotropic agents have various mechanisms of action: o PCSK 9 binds to low density lipoprotein receptors (LDLR) at the surface of hepatocytes and, thereby, targets internalized LDLR for lysosomal degradation. By inhibiting the binding of PCSK 9 to LDLR, the PCSK 9 inhibitors, alirocumab (Praluent) and evolocumab (Repatha), increase the number of LDLR available to clear LDL particles 153

Lipotropics, Other • Many non-statin therapies do not provide adequate ASCVD risk reduction benefits compared to their potential for adverse effects in the routine prevention of ASCVD. • AIM-HIGH study showed the additional reduction in non-HDL-C (as well as apolipoprotein B [Apo B], lipoprotein (a) [Lp(a)], and triglycerides) levels with niacin therapy did not further reduce ASCVD risk in those treated to LDL-C levels of 40 to 80 mg/d. L. • ACCORD trial reported that, in diabetic patients with and without clinical CV disease, the addition of a fenofibrate to simvastatin therapy did not reduce the risk for CV events compared with simvastatin alone. 154
Lipotropics, Other • Studies to date have not demonstrated an overall benefit of fibrates for reduction of CV events or total mortality; although post-hoc subgroup analyses have reported a decrease in composite CV events with the use of fibrates in patients with moderate hypertriglyceridemia. • Notably, the REDUCE-IT trial revealed that icosapent ethyl (Vascepa) reduces CV event risk by 25% 155

Lipotropics, Other • • • Non-statin agents can be considered when maximally tolerated statin therapy does not lower LDL-C sufficiently per the American College of Cardiology/American Heart Association (ACC/AHA) recommendations. The Endocrine Society recommends drug therapy (along with lifestyle changes) to reduce the risk of pancreatitis in patients with severe and very severe hypertriglyceridemia; a fibrate is considered first-line treatment. The National Lipid Association (NLA) 2017 guidance determined that PSCK 9 inhibitors will provide reasonable value in the following 3 groups of patients on maximally tolerated statin therapy: o o o (1) extremely high-risk (≥ 40% 10 -year ASCVD risk) patients with LDL-C ≥ 70 mg/d. L, including patients with extensive or active ASCVD burden and those with less extensive ASCVD plus extremely high-risk cardiometabolic risk factors; (2) very high-risk (≥ 30% to 39% 10 -year ASCVD risk) patients with LDL-C ≥ 100 mg/d. L; and (3) high-risk (< 30% 10 -year ASCVD risk) patients with LDL-C ≥ 130 mg/d. L, including patients with He. FH or severe hypercholesterolemia ≥ 220 mg/d. L 156

Lipotropics, Other New Drug to Class: Nexlitol/Nexlizet (bempedoic acid; bempedoic acid/ezetimibe) 157

Lipotropics, Other Treatment Updates: • Bempedoic acid (Nexletol) is an adenosine triphosphate-citrate lyase (ACL) inhibitor and bempedoic acid/ezetimibe (Nexlizet) contains an ACL inhibitor and a cholesterol absorption inhibitor. • Both agents are indicated as an adjunct to diet and maximally tolerated statin therapy for the treatment of adults with heterozygous familial hypercholesterolemia (He. FH) or established atherosclerotic cardiovascular disease (ASCVD) who require additional lowering of low-density lipoprotein-cholesterol (LDL-C) • The recommended dosage of bempedoic acid is 180 mg orally once daily. • The recommended dosage of bempedoic acid/ezetimibe is 1 tablet (180 mg/10 mg) orally once daily 158

Lipotropics, Other Treatment Updates: • Bempedoic acid/ezetimibe (nexlizet) is contraindicated in patients with known hypersensitivity to ezetimibe. o Anaphylaxis, angioedema, rash and urticaria have been reported with ezetimibe. • Warnings include increased blood uric acid level, and increased risk of tendon rupture. • The most common adverse reactions were upper respiratory tract infection, muscle spasms, hyperuricemia, back pain, abdominal pain/discomfort, bronchitis, pain in extremity, anemia, and elevated liver enzymes. 159

Lipotropics, Other Treatment Updates: • The CLEAR trial program includes four phase 3, double-blind, randomized clinical trials evaluating efficacy and safety of bempedoic acid. • The CLEAR Harmony 52 -week trial enrolled adults with ASCVD, He. FH, or both. o o Baseline LDL-C levels were ≥ 70 mg/d. L while on maximally tolerated stain therapy with or without additional lipid-lowering therapies. Significant differences in changes from baseline compared to placebo were also seen in non-high-density lipoprotein cholesterol (HDL-C), total cholesterol (TC), apolipoprotein B (apo-B), and high-sensitivity C-reactive protein (hs. CRP) at week 12. 160

Lipotropics, Other Treatment Updates: • The CLEAR Wisdom 52 -week trial included adults with ASCVD, He. FH, or both. o o Baseline LDL-C levels were ≥ 70 mg/d. L while on maximally tolerated statin therapy. At week 12, the difference in mean percent change in LDL-C from baseline between bempedoic acid and placebo was -17. 4% (p<0. 001). • The CLEAR Serenity trial randomized 345 patients with hypercholesterolemia and a history of intolerance to at least 2 statins. o o Baseline LDL-C levels were ≥ 130 mg/d. L for primary prevention patients and ≥ 100 mg/d. L for patients with He. FH while on maximally tolerated statin therapy. At week 12, bempedoic acid significantly reduced LDL-C from baseline. Significant reductions from baseline compared to placebo were also seen with non-HDL-C, TC, apo-B, and hs. CRP (high sensitivity C-reactive protein). 161

Lipotropics, Other Treatment Updates: • The CLEAR Tranquility trial enrolled adults with a history of statin intolerance and an LDL-C ≥ 100 mg/d. L while on stable lipid-lowering therapy. o o The change in LDL-C from baseline to week 12 was significantly greater with bempedoic acid compare to placebo (difference from placebo, -28. 5% [95% CI, -34. 4% to -22. 5%; p<0. 001). Significant reductions in LDL-C with bempedoic acid compared to placebo were also observed with non-HDL-C, TC, apo-B, and hs. CRP. 162

Proton Pump Inhibitors 163

Proton Pump Inhibitors Class Overview - indications for products in this class include*: • • Gastric ulcers GERD H. pylori eradication (as part of combination therapy) Maintenance/treatment of Erosive Esophagitis Maintenance/treatment of Duodenal Ulcer Pathological hypersecretory conditions Pyrosis (Heartburn) *Not inclusive of all product indications, all products differ in indication 164

Proton Pump Inhibitors Class Overview – PPI’s: • dexlansoprazole - (Dexilant) • esomeprazole - (esomeprazole; Esomep-EZS, Nexium 24 HR) • lansoprazole - (lansoprazole; Prevacid, Prevacid 24 -HR) • omeprazole - (omeprazole; Prilosec, Prilosec OTC) • omeprazole/sodium bicarbonate - (omeprazole/sodium bicarbonate; Zegerid) • pantoprazole - (pantoprazole; Protonix) • rabeprazole - (rabeprazole; Aciphex) 165

Proton Pump Inhibitors • This is a first-time review of this therapeutic class • All PPIs are substituted benzimidazole derivatives that reduce gastric acid secretion by specifically inhibiting the proton pump (H+/K+ATPase) at the secretory surface of the gastric parietal cell. • Proton pump inhibitors (PPIs) demonstrate gastric acid suppression superior to histamine-2 receptor antagonists (H 2 RAs). • PPIs achieve a more rapid and sustained increase in gastric p. H and are not associated with the rapid tachyphylaxis seen with H 2 RAs, thereby, offering improved treatment of various acid-peptic disorders, including gastroesophageal reflux disease (GERD), peptic ulcer disease (PUD), and nonsteroidal anti-inflammatory drug (NSAID)-induced gastropathy 166

Proton Pump Inhibitors • The American Gastroenterological Association (AGA) and the American College of Gastroenterology (ACG) recommend PPIs as first-line therapy for the treatment of severe GERD-related symptoms or erosive esophagitis (EE). o PPIs provide the most rapid symptomatic relief and heal esophagitis in the highest percentage of patients. • PPIs are used in conjunction with various antimicrobials for the eradication of Helicobacter pylori, the most common cause of PUD. Antisecretory therapy with either H 2 RAs or PPIs accelerates ulcer healing and provides rapid symptomatic improvement. 167

Proton Pump Inhibitors • All PPIs have the potential to cause p. H-dependent interactions with drugs from a various classes including anticonvulsants, antifungals, HIV medications, etc. • Duration of PPI treatment depends on the indication and can range from 10 days (H. pylori) to 4 weeks (Symptomatic Nonerosive GERD) to ongoing treatment (Maintenance of erosive esophagitis). • It is recommended to avoid administration of PPIs for longer than medically indicated. 168

Proton Pump Inhibitors Product/Guideline Updates: • The American Gastroenterological Association (AGA) and the Joint Task Force on Allergy-Immunology Practice Parameters (JTF) has issued a clinical practice guideline on the management of eosinophilic esophagitis (Eo. E) recommending proton pump inhibition (over no treatment) for patients with symptomatic esophageal eosinophilia 169

Acne Agents, Topical 170

Acne Agents, Topical Class Overview – indication for these products include*: • The topical treatment of acne vulgaris *Tazorac 0. 1% gel also indicated for the topical treatment of plaque psoriasis 171
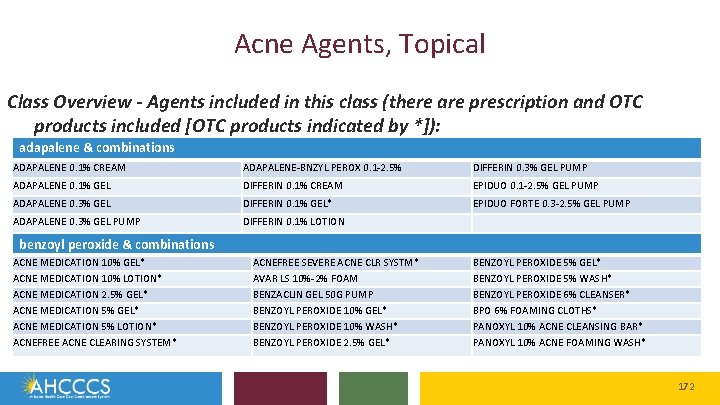
Acne Agents, Topical Class Overview - Agents included in this class (there are prescription and OTC products included [OTC products indicated by *]): adapalene & combinations ADAPALENE 0. 1% CREAM ADAPALENE-BNZYL PEROX 0. 1 -2. 5% DIFFERIN 0. 3% GEL PUMP ADAPALENE 0. 1% GEL DIFFERIN 0. 1% CREAM EPIDUO 0. 1 -2. 5% GEL PUMP ADAPALENE 0. 3% GEL DIFFERIN 0. 1% GEL* EPIDUO FORTE 0. 3 -2. 5% GEL PUMP ADAPALENE 0. 3% GEL PUMP DIFFERIN 0. 1% LOTION benzoyl peroxide & combinations ACNE MEDICATION 10% GEL* ACNE MEDICATION 10% LOTION* ACNE MEDICATION 2. 5% GEL* ACNE MEDICATION 5% LOTION* ACNEFREE ACNE CLEARING SYSTEM* ACNEFREE SEVERE ACNE CLR SYSTM* AVAR LS 10%-2% FOAM BENZACLIN GEL 50 G PUMP BENZOYL PEROXIDE 10% GEL* BENZOYL PEROXIDE 10% WASH* BENZOYL PEROXIDE 2. 5% GEL* BENZOYL PEROXIDE 5% WASH* BENZOYL PEROXIDE 6% CLEANSER* BPO 6% FOAMING CLOTHS* PANOXYL 10% ACNE CLEANSING BAR* PANOXYL 10% ACNE FOAMING WASH* 172
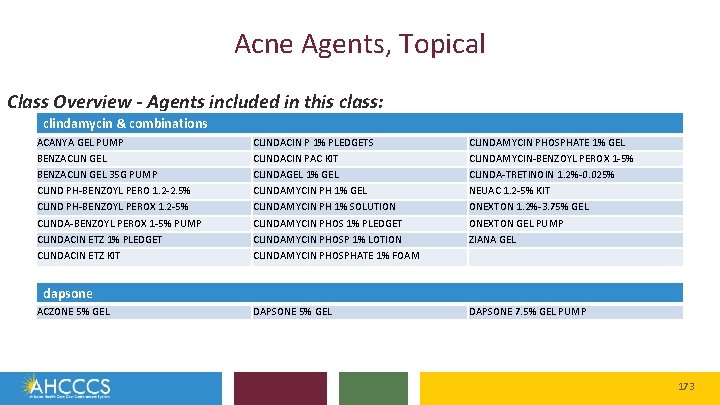
Acne Agents, Topical Class Overview - Agents included in this class: clindamycin & combinations ACANYA GEL PUMP BENZACLIN GEL 35 G PUMP CLIND PH-BENZOYL PERO 1. 2 -2. 5% CLIND PH-BENZOYL PEROX 1. 2 -5% CLINDA-BENZOYL PEROX 1 -5% PUMP CLINDACIN ETZ 1% PLEDGET CLINDACIN ETZ KIT CLINDACIN P 1% PLEDGETS CLINDACIN PAC KIT CLINDAGEL 1% GEL CLINDAMYCIN PH 1% SOLUTION CLINDAMYCIN PHOS 1% PLEDGET CLINDAMYCIN PHOSP 1% LOTION CLINDAMYCIN PHOSPHATE 1% FOAM CLINDAMYCIN PHOSPHATE 1% GEL CLINDAMYCIN-BENZOYL PEROX 1 -5% CLINDA-TRETINOIN 1. 2%-0. 025% NEUAC 1. 2 -5% KIT ONEXTON 1. 2%-3. 75% GEL ONEXTON GEL PUMP ZIANA GEL DAPSONE 5% GEL DAPSONE 7. 5% GEL PUMP dapsone ACZONE 5% GEL 173
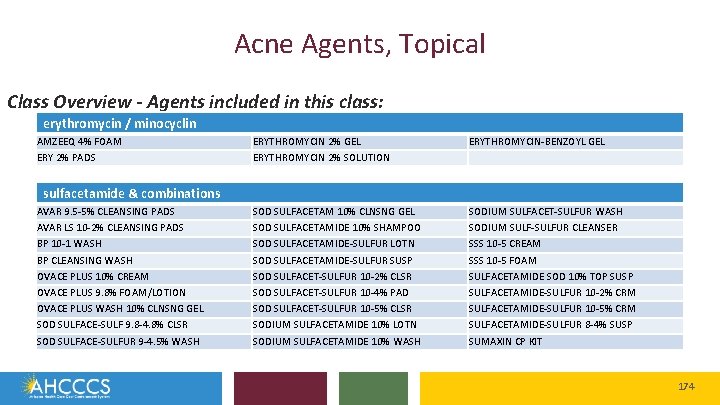
Acne Agents, Topical Class Overview - Agents included in this class: erythromycin / minocyclin AMZEEQ 4% FOAM ERY 2% PADS ERYTHROMYCIN 2% GEL ERYTHROMYCIN 2% SOLUTION ERYTHROMYCIN-BENZOYL GEL SOD SULFACETAM 10% CLNSNG GEL SOD SULFACETAMIDE 10% SHAMPOO SOD SULFACETAMIDE-SULFUR LOTN SOD SULFACETAMIDE-SULFUR SUSP SOD SULFACET-SULFUR 10 -2% CLSR SOD SULFACET-SULFUR 10 -4% PAD SOD SULFACET-SULFUR 10 -5% CLSR SODIUM SULFACETAMIDE 10% LOTN SODIUM SULFACETAMIDE 10% WASH SODIUM SULFACET-SULFUR WASH SODIUM SULF-SULFUR CLEANSER SSS 10 -5 CREAM SSS 10 -5 FOAM SULFACETAMIDE SOD 10% TOP SUSP SULFACETAMIDE-SULFUR 10 -2% CRM SULFACETAMIDE-SULFUR 10 -5% CRM SULFACETAMIDE-SULFUR 8 -4% SUSP SUMAXIN CP KIT sulfacetamide & combinations AVAR 9. 5 -5% CLEANSING PADS AVAR LS 10 -2% CLEANSING PADS BP 10 -1 WASH BP CLEANSING WASH OVACE PLUS 10% CREAM OVACE PLUS 9. 8% FOAM/LOTION OVACE PLUS WASH 10% CLNSNG GEL SOD SULFACE-SULF 9. 8 -4. 8% CLSR SOD SULFACE-SULFUR 9 -4. 5% WASH 174
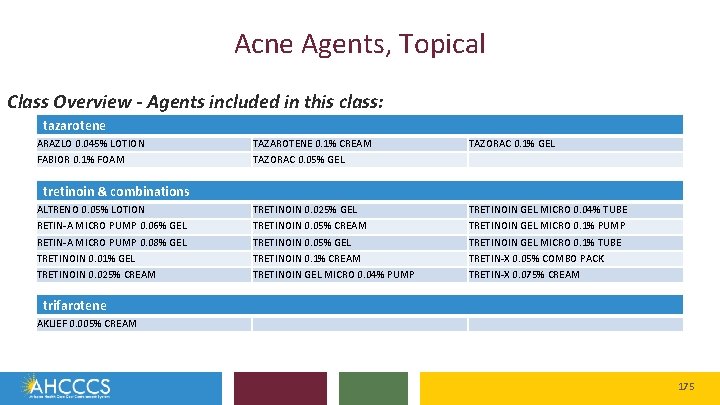
Acne Agents, Topical Class Overview - Agents included in this class: tazarotene ARAZLO 0. 045% LOTION FABIOR 0. 1% FOAM TAZAROTENE 0. 1% CREAM TAZORAC 0. 05% GEL TAZORAC 0. 1% GEL TRETINOIN 0. 025% GEL TRETINOIN 0. 05% CREAM TRETINOIN 0. 05% GEL TRETINOIN 0. 1% CREAM TRETINOIN GEL MICRO 0. 04% PUMP TRETINOIN GEL MICRO 0. 04% TUBE TRETINOIN GEL MICRO 0. 1% PUMP TRETINOIN GEL MICRO 0. 1% TUBE TRETIN-X 0. 05% COMBO PACK TRETIN-X 0. 075% CREAM tretinoin & combinations ALTRENO 0. 05% LOTION RETIN-A MICRO PUMP 0. 06% GEL RETIN-A MICRO PUMP 0. 08% GEL TRETINOIN 0. 01% GEL TRETINOIN 0. 025% CREAM trifarotene AKLIEF 0. 005% CREAM 175

Acne Agents, Topical • This is a first-time review of this class • Acne vulgaris is the most common cutaneous condition in the United States. • It is a disorder that affects primarily teenagers and young adults, but it can sometimes persist beyond young adulthood. • Small cysts called comedones form in hair follicles due to blockage of the pore from accumulated sebum and keratinous material. • Bacteria, most often Propionibacterium acnes, release free fatty acids from sebum within the comedones causing inflammation to form a cyst. • This results in rupture of the cyst wall and subsequent inflammatory reaction due to extrusion of oily and keratinous debris from the cyst. 176

Acne Agents, Topical • The goals of treatment include resolution of lesions, scar prevention, and reduction of psychological morbidities. • This is achieved by decreasing sebaceous gland activity, bacterial population, and inflammation. • The available products work by different mechanisms to attack the causative events. • Typically, retinoids, such as tretinoin, adapalene, tazarotene, and trifarotene are used to inhibit comedone formation, while an antibiotic, such as clindamycin, or erythromycin suppresses P. acnes. 177

Acne Agents, Topical • Benzoyl peroxide has bactericidal, keratolytic, and comedolytic activity and has been useful as a single agent and in combination with antibiotics or retinoids in decreasing the number of lesions in mild to moderate acne. • Trifarotene (Aklief) is an agonist of retinoic acid receptors (RAR), with particular activity at the gamma subtype of RAR. Stimulation of RAR results in modulation of target genes which are associated with various processes, including cell differentiation and mediation of inflammation. • Combination therapy is useful to limit growing resistance to antibacterial therapy, as well as, enhance the efficacy of antibiotics by improving penetration into the lesions. 178

Acne Agents, Topical • According to the 2018 update to the consensus guidelines developed by the Global Alliance to Improve Outcomes in Acne: o o o Retinoids should form the cornerstone of acne therapy. Retinoid products are generally similar in efficacy, and their efficacy improves with higher concentrations. Early treatment with retinoids can minimize the risk of post inflammatory hyperpigmentation (PIH), by inhibiting tyrosinase and by blocking pigment transfer from melanocytes to keratinocytes. Topical retinoid monotherapy may be sufficient in some cases. Benzoyl peroxide or an oral antibiotic may be added as needed. For most patients with inflammatory acne, comedonal acne, or both, a topical retinoid plus benzoyl peroxide is first-line therapy. Topical antibiotics should not be used as monotherapy due to rapid development of resistance 179

Acne Agents, Topical • 2018 update to the consensus guidelines developed by the Global Alliance to Improve Outcomes in Acne (cont. ): o Benzoyl peroxide is a potent bactericidal agent and is the preferred topical antimicrobial to support good antimicrobial stewardship. o Topical retinoids, with or without benzoyl peroxide, is the treatment choice for maintenance therapy. o Studies have shown that maintenance therapy with a topical retinoid achieved sustained reductions in micro comedones, resulting in fewer active acne lesions. o Efficacy in maintenance therapy has also been demonstrated for adapalene, adapalene/benzoyl peroxide, tazarotene, and azelaic acid. o Trifarotene was FDA approved in 2019 and use of this agent is not addressed in the guidelines discussed above 180

New Drug Reviews Justin Johnson, Pharm. D. 181

New Products • Oriahnn (elagolix/estradiol/norethindrone acetate) 182

Oriahnn (elagolix/estradiol/norethindrone acetate; elagolix) • Oriahnn is indicated for the management of heavy menstrual bleeding associated with uterine leiomyomas (fibroids) in premenopausal women o Due to the risk of continued bone loss (which may not be reversible), limit use to 24 months o Elagolix is a gonadotropin-releasing hormone (Gn. RH) receptor antagonist, estradiol is an estrogen, and norethindrone acetate is a progestin • Oriahnn is taken once in the morning and once in the evening, at approximately the same time each day, with or without food, for up to 24 months 183

• • Oriahnn (elagolix/estradiol/norethindrone acetate; elagolix) Boxed warning – thromboembolic disorders and vascular events Contraindications include: o o o o known hypersensitivity osteoporosis liver impairment or disease high risk arterial, venous thrombotic, or thromboembolic disorders (e. g. , female smokers > 35 years of age, uncontrolled hypertension, headache with focal neurological symptoms or migraines with aura and > 35 years) pregnancy history of or current hormonally-sensitive malignancies (e. g. , breast cancer) undiagnosed abnormal uterine bleeding use with organic anion transporting polypeptide (OATP)1 B 1 inhibitors known or expected to increase elagolix exposure to a clinically significant extent 184

• Oriahnn (elagolix/estradiol/norethindrone acetate; elagolix) Warnings include: o bone loss o suicidal ideation or behavior/mood disorders o hepatic impairment/transaminase elevations o increased blood pressure o menstrual bleeding changes/decreased ability to recognize pregnancy o allergic reactions secondary to inactive components (e. g. , FD&C Yellow No 5 [tartrazine]), o gallbladder disease/cholestatic jaundice history o alopecia, carbohydrate and lipid metabolism effects (e. g. , increased glucose, changed in lipid levels) o laboratory result effects (e. g. , select thyroid or corticosteroid levels) 185

Oriahnn (elagolix/estradiol/norethindrone acetate; elagolix) • Common Adverse Effects (occurred ≥ 5% and higher than placebo) o o hot flush headache fatigue metrorrhagia 186

• Oriahnn (elagolix/estradiol/norethindrone acetate; elagolix) Drug Interactions include: o CYP 3 A substrates (e. g. , midazolam) o CYP 2 C 19 substrates (e. g. , omeprazole) o P-gp substrates (e. g. , digoxin) o rosuvastatin o elagolix is a substrate of CYP 3 A, P-gp, and OATP 1 B 1; estradiol and norethindrone acetate are CYP 3 A substrates o The following agents may impact the effects of Oriahnn: § Strong CYP 3 A inducers – decreased Oriahnn efficacy § Strong CYP 3 A inhibitors – increased Oriahnn exposure; avoid use § OATP 1 B 1 inhibitors – increased Oriahnn exposure; contraindicated 187

Oriahnn (elagolix/estradiol/norethindrone acetate; elagolix) • • There are no comparative trials available The FDA approval of Oriahnn was based on two, randomized Phase 3 uterine fibroid clinical trials, ELARIS UF-I and ELARIS UF-II Oriahnn achieved the primary endpoint of clinically meaningful reduction in bleeding (defined as the proportion of women who achieved both at least a 50% reduction in menstrual blood loss at final month of treatment and a total menstrual blood loss amount of less than 80 ml), compared with placebo in final month of study for patients o 7/10 women no longer experiencing heavy menstrual bleeding versus 1/10 women on placebo. Oriahnn also reduced heavy menstrual bleeding due to uterine fibroids by 50% within the first month of use. 188

Executive Session 189

Public Therapeutic Class Votes 190

Biosimilar Update Justin Johnson, Pharm. D, Magellan 191

BIOSIMILAR UPDATE • Herzuma (trastuzumab-pkrb) and Ontruzant (trastuzumab-dttb) are biosimilars for Herceptin, launched on 3/16/2020 and 4/15/2020, respectively. • Both products are approved for the same indications as Herceptin • Herzuma and Ontruzant are more costly than Herceptin • Herceptin will remain the preferred agent for AHCCCS and it’s Contractors. 192

P&T Meeting Dates • 2020 Meeting Dates: o January 26, 2021 o May 19, 2021 193
- Slides: 193