Agrobacterium tumefaciens pathogen and useful tool Agrobacterium tumefaciens

Agrobacterium tumefaciens – pathogen and useful tool Agrobacterium tumefaciens is a plant pathogen that induces tumors on about 60% of dicotyledonous angiosperms and gymnosperms Cherry Its tumor-inducing property also makes it a valuable tool for introducing genes into plants for research and agricultural purposes Casimiro, I. , Marchant, A. , Bhalerao, R. P. , Beeckman, T. , Dhooge, S. , Swarup, R. , Graham, N. , Inzé, D. , Sandberg, G. , Casero, P. J. and Bennett, M. (2001). Auxin Transport Promotes Arabidopsis Lateral Root Initiation. Plant Cell. 13: 843 -852. Herb Pilcher © 2014 American Society of Plant Biologists

Crown gall disease The first written record of crown gall disease, on grape, dates from 1853 Fridiano Cavara (1897) found that a bacterium causes crown gall in grape Crown gall induces growths at wound sites and severely limits crop yields and growth vigor Edward L. Barnard, Florida Department of Agriculture and Consumer Services, Bugwood. org; Mike Ellis, Ohio State University; University of Georgia Plant Pathology Archive, University of Georgia, Bugwood. org; Wikimedia commons © 2014 American Society of Plant Biologists

is caused by a “A 1907: plant. Crown tumor gall of bacterial origin” bacterium 1907 - Erwin Smith and C. O. Townsend isolated a bacterium from galls on daisy. When inoculated onto other plants, galls were produced gall Smith, E. F. and Townsend, C. O. (1907). A plant-tumor of bacterial origin. Science. 25: 671 -673. © 2014 American Society of Plant Biologists

Unusual compounds called opines are found in many crown galls The type of opine is determined by the bacterium, not the plant Octopineutilizing strain Nopalineutilizing strain Octopine Questions raised: • What are these compounds? • Do they cause the tumors? • How and why do the bacteria cause the plants to make opines? Nopaline 1960 s – 1970 s, numerous studies © 2014 American Society of Plant Biologists

Agrobacterium-induced galls do not require bacterial persistence Gall tissues without any bacteria can persist indefinitely in culture, in contrast with most other pathogen-induced neoplastic growths that require the presence of the pathogen Braun made fundamental discoveries about how Agrobacterium transforms plant cells Armin C. Braun 1911 - 1986 White, P. R. and Braun, A. C. (1941). Crown gall production by bacteria-free tumor tissues. Science. 94: 239 -241; Photo from Wood, H. N. , and Kelman, A. (1987) Phytopathology 77: 991. © 2014 American Society of Plant Biologists
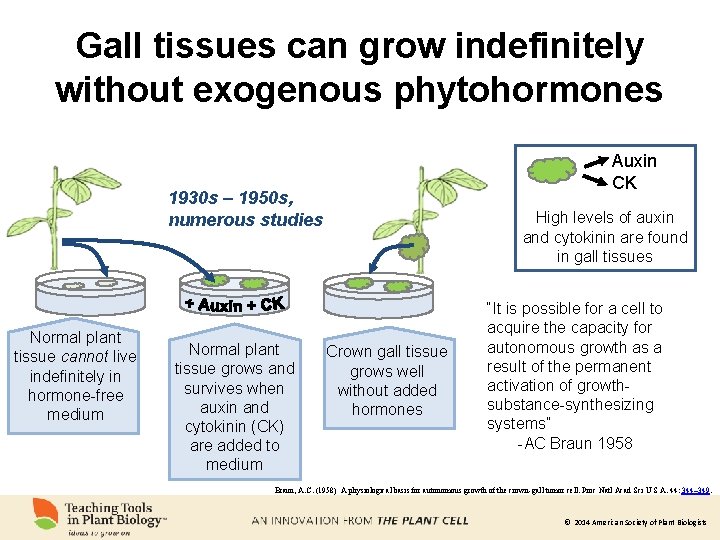
Gall tissues can grow indefinitely without exogenous phytohormones Auxin CK 1930 s – 1950 s, numerous studies Normal plant tissue cannot live indefinitely in hormone-free medium Normal plant tissue grows and survives when auxin and cytokinin (CK) are added to medium High levels of auxin and cytokinin are found in gall tissues Crown gall tissue grows well without added hormones “It is possible for a cell to acquire the capacity for autonomous growth as a result of the permanent activation of growthsubstance-synthesizing systems” -AC Braun 1958 Braun, A. C. (1958) A physiological basis for autonomous growth of the crown-gall tumor cell. Proc Natl Acad Sci U S A. 44: 344– 349. © 2014 American Society of Plant Biologists
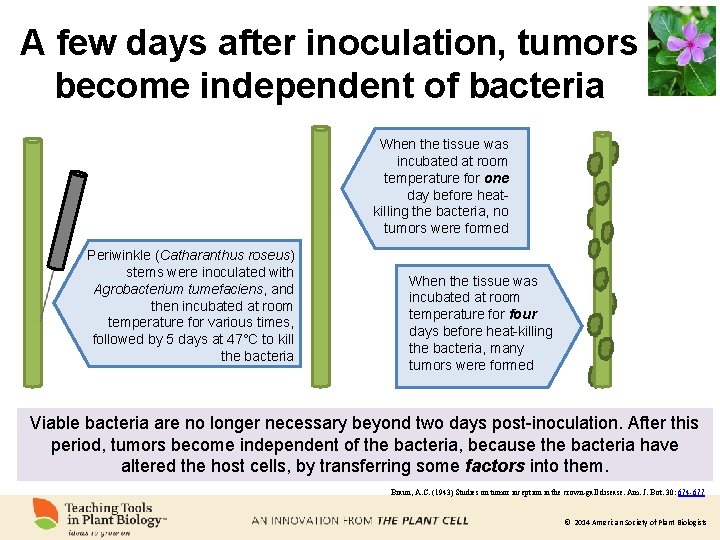
A few days after inoculation, tumors become independent of bacteria When the tissue was incubated at room temperature for one day before heatkilling the bacteria, no tumors were formed Periwinkle (Catharanthus roseus) stems were inoculated with Agrobacterium tumefaciens, and then incubated at room temperature for various times, followed by 5 days at 47°C to kill the bacteria When the tissue was incubated at room temperature for four days before heat-killing the bacteria, many tumors were formed Viable bacteria are no longer necessary beyond two days post-inoculation. After this period, tumors become independent of the bacteria, because the bacteria have altered the host cells, by transferring some factors into them. Braun, A. C. (1943) Studies on tumor inception in the crown-gall disease. Am. J. Bot. 30: 674 -677 © 2014 American Society of Plant Biologists
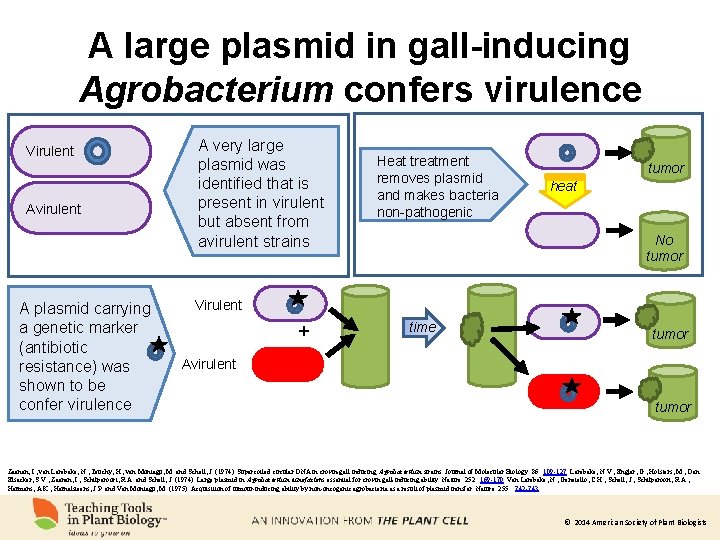
A large plasmid in gall-inducing Agrobacterium confers virulence Virulent Avirulent A plasmid carrying a genetic marker (antibiotic resistance) was shown to be confer virulence A very large plasmid was identified that is present in virulent but absent from avirulent strains Heat treatment removes plasmid and makes bacteria non-pathogenic tumor heat No tumor Virulent + time tumor Avirulent tumor Zaenen, I. , van Larebeke, N. , Teuchy, H. , van Montagu, M. and Schell, J. (1974). Supercoiled circular DNA in crown-gall inducing Agrobacterium strains. Journal of Molecular Biology. 86: 109 -127. Larebeke, N. V. , Engler, G. , Holsters, M. , Den Elsacker, S. V. , Zaenen, I. , Schilperoort, R. A. and Schell, J. (1974). Large plasmid in Agrobacterium tumefaciens essential for crown gall-inducing ability. Nature. 252: 169 -170. Van Larebeke, N. , Genetello, C. H. , Schell, J. , Schilperoort, R. A. , Hermans, A. K. , Hernalsteens, J. P. and Van Montagu, M. (1975). Acquisition of tumour-inducing ability by non-oncogenic agrobacteria as a result of plasmid transfer. Nature. 255: 742 -743. © 2014 American Society of Plant Biologists
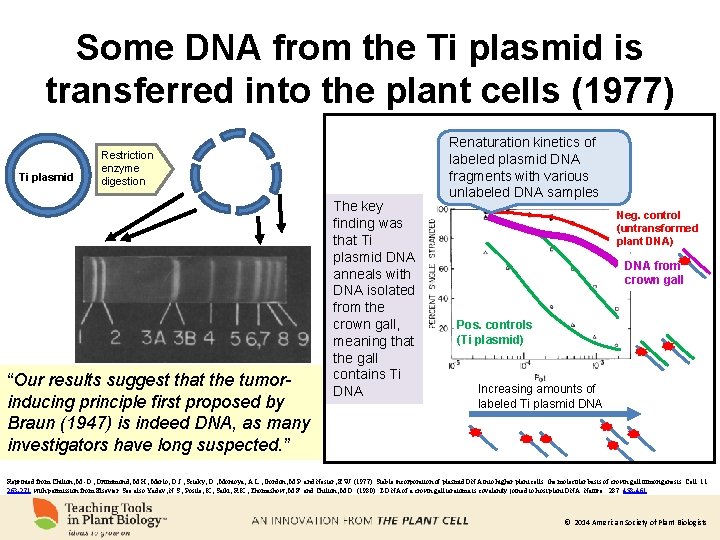
Some DNA from the Ti plasmid is transferred into the plant cells (1977) Ti plasmid Restriction enzyme digestion “Our results suggest that the tumorinducing principle first proposed by Braun (1947) is indeed DNA, as many investigators have long suspected. ” The key finding was that Ti plasmid DNA anneals with DNA isolated from the crown gall, meaning that the gall contains Ti DNA Renaturation kinetics of labeled plasmid DNA fragments with various unlabeled DNA samples Neg. control (untransformed plant DNA) DNA from crown gall Pos. controls (Ti plasmid) Increasing amounts of labeled Ti plasmid DNA Reprinted from Chilton, M. -D. , Drummond, M. H. , Merlo, D. J. , Sciaky, D. , Montoya, A. L. , Gordon, M. P. and Nester, E. W. (1977). Stable incorporation of plasmid DNA into higher plant cells: the molecular basis of crown gall tumorigenesis. Cell. 11: 263 -271. with permission from Elsevier. See also Yadav, N. S. , Postle, K. , Saiki, R. K. , Thomashow, M. F. and Chilton, M. D. (1980). T-DNA of a crown gall teratoma is covalently joined to host plant DNA. Nature. 287: 458 -461. © 2014 American Society of Plant Biologists
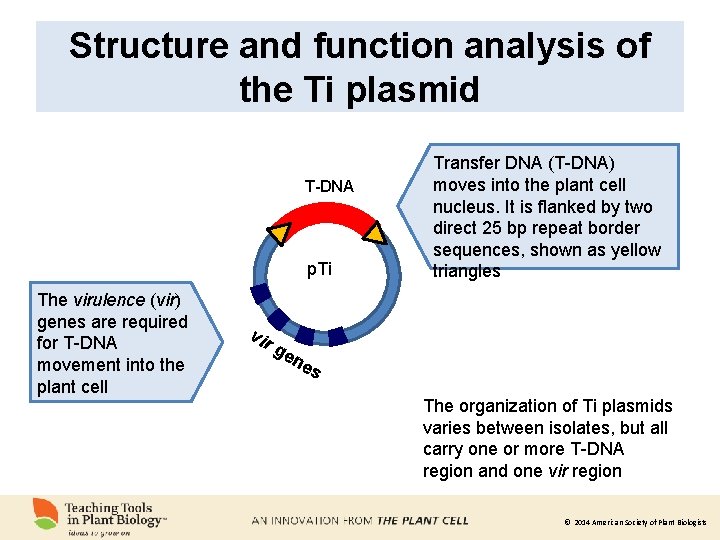
Structure and function analysis of the Ti plasmid T-DNA p. Ti The virulence (vir) genes are required for T-DNA movement into the plant cell vir ge Transfer DNA (T-DNA) moves into the plant cell nucleus. It is flanked by two direct 25 bp repeat border sequences, shown as yellow triangles ne s The organization of Ti plasmids varies between isolates, but all carry one or more T-DNA region and one vir region © 2014 American Society of Plant Biologists
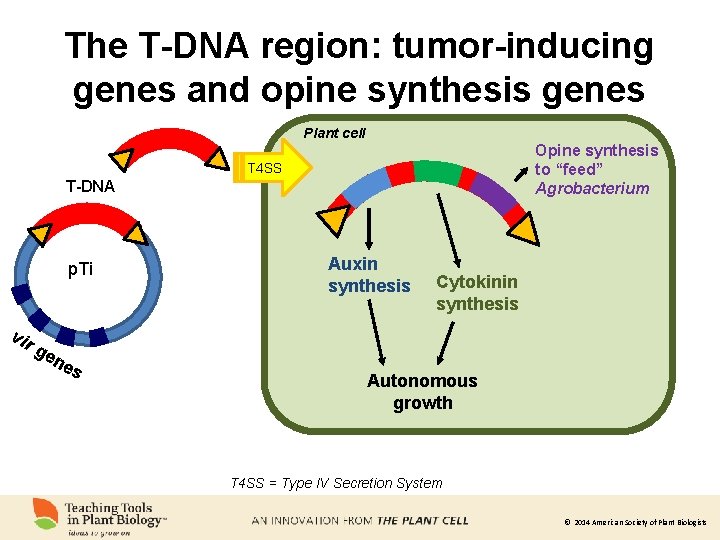
The T-DNA region: tumor-inducing genes and opine synthesis genes Plant cell Opine synthesis to “feed” Agrobacterium T 4 SS T-DNA p. Ti vir ge ne s Auxin synthesis Cytokinin synthesis Autonomous growth T 4 SS = Type IV Secretion System © 2014 American Society of Plant Biologists
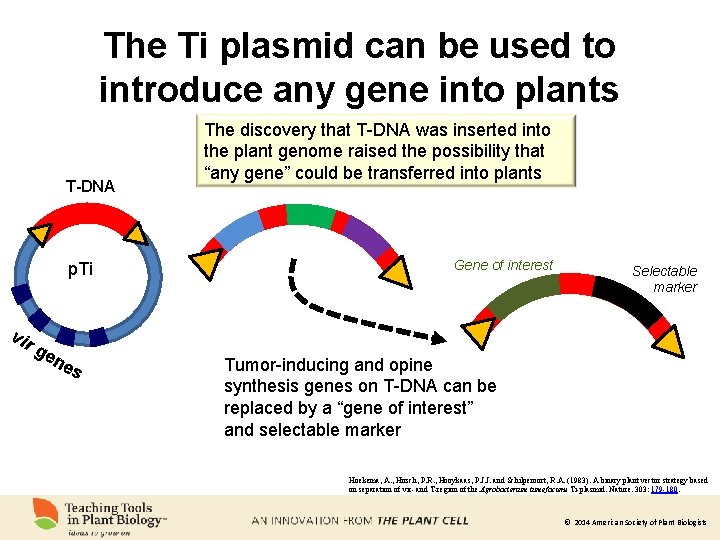
The Ti plasmid can be used to introduce any gene into plants T-DNA p. Ti vir ge ne s The discovery that T-DNA was inserted into the plant genome raised the possibility that “any gene” could be transferred into plants Gene of interest Selectable marker Tumor-inducing and opine synthesis genes on T-DNA can be replaced by a “gene of interest” and selectable marker Hoekema, A. , Hirsch, P. R. , Hooykaas, P. J. J. and Schilperoort, R. A. (1983). A binary plant vector strategy based on separation of vir- and T-region of the Agrobacterium tumefaciens Ti-plasmid. Nature. 303: 179 -180. © 2014 American Society of Plant Biologists
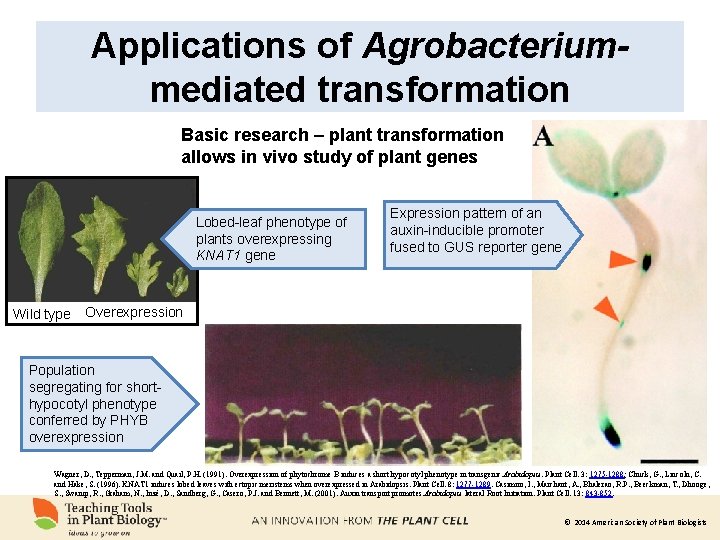
Applications of Agrobacteriummediated transformation Basic research – plant transformation allows in vivo study of plant genes Lobed-leaf phenotype of plants overexpressing KNAT 1 gene Wild type Expression pattern of an auxin-inducible promoter fused to GUS reporter gene Overexpression Population segregating for shorthypocotyl phenotype conferred by PHYB overexpression Wagner, D. , Tepperman, J. M. and Quail, P. H. (1991). Overexpression of phytochrome B induces a short hypocotyl phenotype in transgenic Arabidopsis. Plant Cell. 3: 1275 -1288; Chuck, G. , Lincoln, C. and Hake, S. (1996). KNAT 1 induces lobed leaves with ectopic meristems when overexpressed in Arabidopsis. Plant Cell. 8: 1277 -1289. Casimiro, I. , Marchant, A. , Bhalerao, R. P. , Beeckman, T. , Dhooge, S. , Swarup, R. , Graham, N. , Inzé, D. , Sandberg, G. , Casero, P. J. and Bennett, M. (2001). Auxin transport promotes Arabidopsis lateral Root Initiation. Plant Cell. 13: 843 -852. © 2014 American Society of Plant Biologists
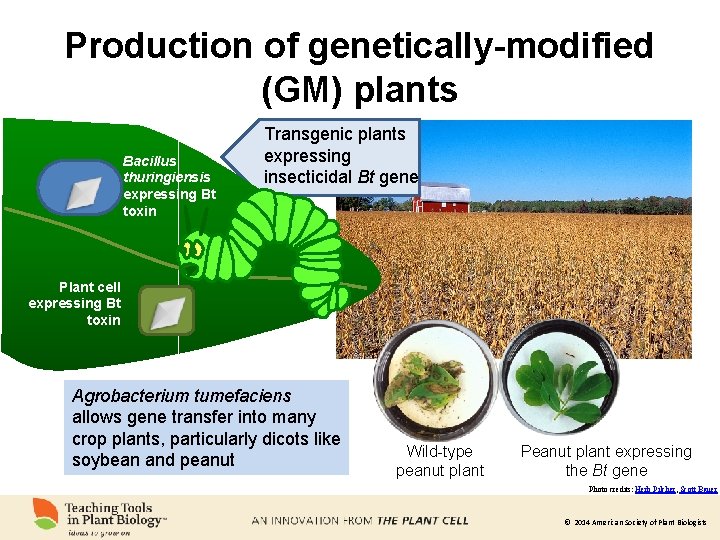
Production of genetically-modified (GM) plants Bacillus thuringiensis expressing Bt toxin Transgenic plants expressing insecticidal Bt gene Plant cell expressing Bt toxin Agrobacterium tumefaciens allows gene transfer into many crop plants, particularly dicots like soybean and peanut Wild-type peanut plant Peanut plant expressing the Bt gene Photo credits: Herb Pilcher, Scott Bauer © 2014 American Society of Plant Biologists
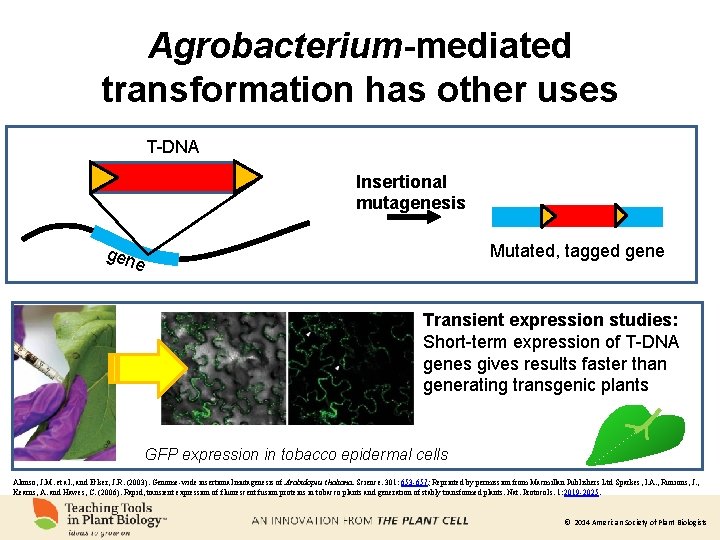
Agrobacterium-mediated transformation has other uses T-DNA Insertional mutagenesis Mutated, tagged gene gen e Transient expression studies: Short-term expression of T-DNA genes gives results faster than generating transgenic plants GFP expression in tobacco epidermal cells Alonso, J. M. et al. , and Ecker, J. R. (2003). Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science. 301: 653 -657; Reprinted by permission from Macmillan Publishers Ltd Sparkes, I. A. , Runions, J. , Kearns, A. and Hawes, C. (2006). Rapid, transient expression of fluorescent fusion proteins in tobacco plants and generation of stably transformed plants. Nat. Protocols. 1: 2019 -2025. © 2014 American Society of Plant Biologists
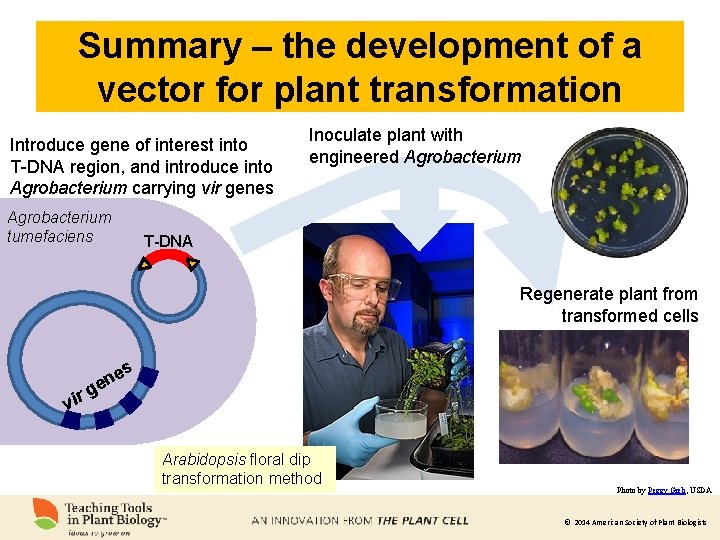
Summary – the development of a vector for plant transformation Introduce gene of interest into T-DNA region, and introduce into Agrobacterium carrying vir genes Agrobacterium tumefaciens Inoculate plant with engineered Agrobacterium T-DNA Regenerate plant from transformed cells g vir es n e Arabidopsis floral dip transformation method Photo by Peggy Greb, USDA © 2014 American Society of Plant Biologists
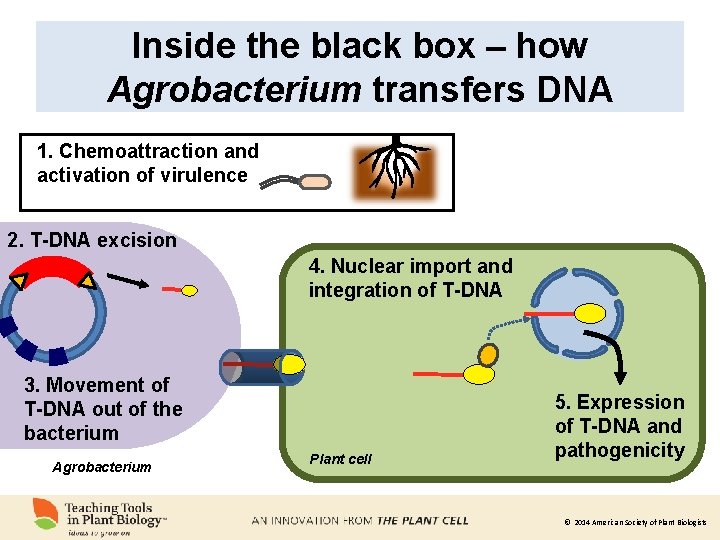
Inside the black box – how Agrobacterium transfers DNA 1. Chemoattraction and activation of virulence 2. T-DNA excision 4. Nuclear import and integration of T-DNA 3. Movement of T-DNA out of the bacterium Agrobacterium Plant cell 5. Expression of T-DNA and pathogenicity © 2014 American Society of Plant Biologists
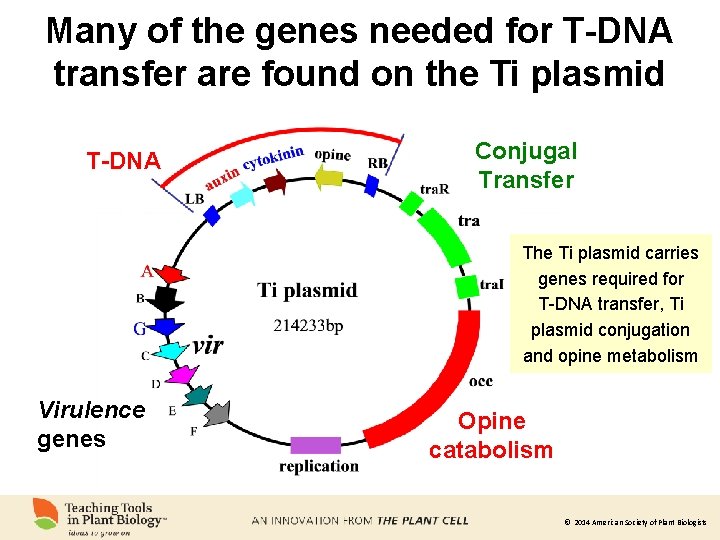
Many of the genes needed for T-DNA transfer are found on the Ti plasmid T-DNA Conjugal Transfer The Ti plasmid carries genes required for T-DNA transfer, Ti plasmid conjugation and opine metabolism Virulence genes Opine catabolism © 2014 American Society of Plant Biologists
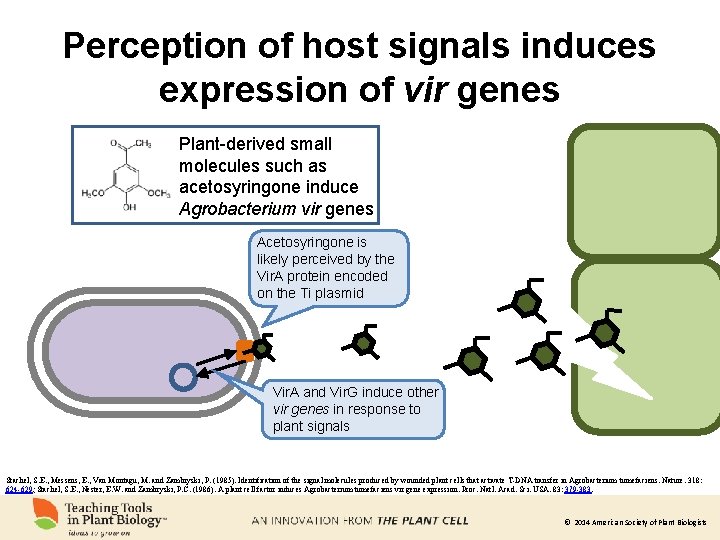
Perception of host signals induces expression of vir genes Plant-derived small molecules such as acetosyringone induce Agrobacterium vir genes Acetosyringone is likely perceived by the Vir. A protein encoded on the Ti plasmid Vir. A and Vir. G induce other vir genes in response to plant signals Stachel, S. E. , Messens, E. , Van Montagu, M. and Zambryski, P. (1985). Identification of the signal molecules produced by wounded plant cells that activate T-DNA transfer in Agrobacterium tumefaciens. Nature. 318: 624 -629; Stachel, S. E. , Nester, E. W. and Zambryski, P. C. (1986). A plant cell factor induces Agrobacterium tumefaciens vir gene expression. Proc. Natl. Acad. Sci. USA. 83: 379 -383. © 2014 American Society of Plant Biologists
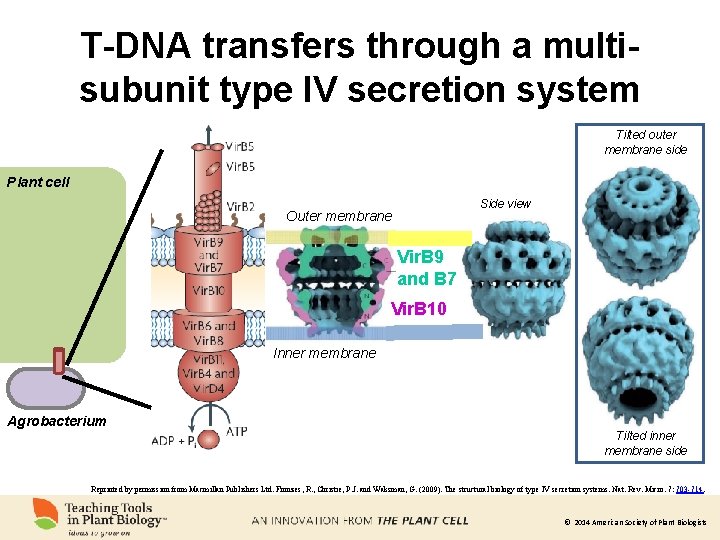
T-DNA transfers through a multisubunit type IV secretion system Tilted outer membrane side Plant cell Side view Outer membrane Vir. B 9 and B 7 Vir. B 10 Inner membrane Agrobacterium Tilted inner membrane side Reprinted by permission from Macmillan Publishers Ltd. Fronzes, R. , Christie, P. J. and Waksman, G. (2009). The structural biology of type IV secretion systems. Nat. Rev. Micro. 7: 703 -714. © 2014 American Society of Plant Biologists
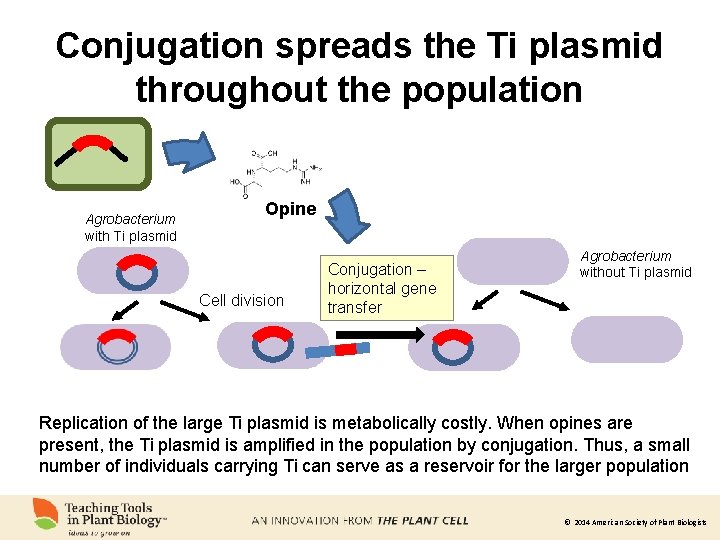
Conjugation spreads the Ti plasmid throughout the population Agrobacterium with Ti plasmid Opine Cell division Conjugation – horizontal gene transfer Agrobacterium without Ti plasmid Replication of the large Ti plasmid is metabolically costly. When opines are present, the Ti plasmid is amplified in the population by conjugation. Thus, a small number of individuals carrying Ti can serve as a reservoir for the larger population © 2014 American Society of Plant Biologists
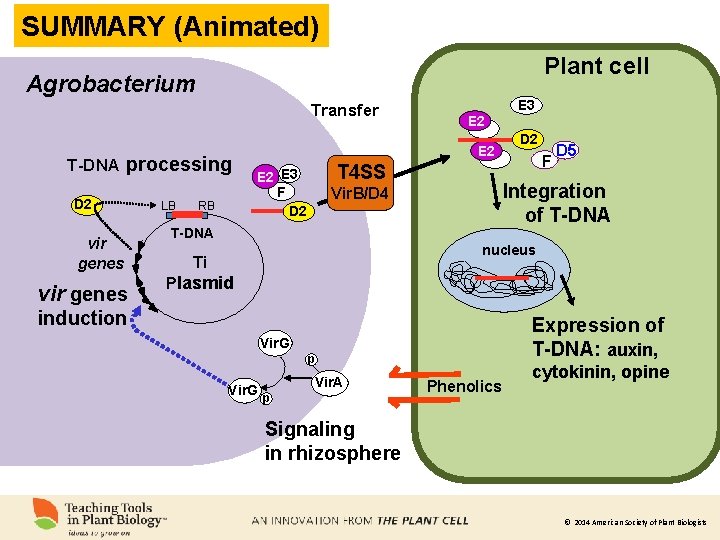
SUMMARY (Animated). Plant cell Agrobacterium Transfer T-DNA processing D 2 vir genes LB RB E 3 E 2 T 4 SS E 2 E 3 F D 2 F D 5 Integration of T-DNA Vir. B/D 4 T-DNA nucleus Ti Plasmid induction Expression of T-DNA: auxin, Vir. G p Vir. A Phenolics cytokinin, opine Signaling in rhizosphere © 2014 American Society of Plant Biologists

Agrobacterium rhizogenes: inducer of roots T-DNA vir The oncogenic genes on A. rhizogenes TDNA are not as well understood as those on the Ti plasmid ge ne s Infection by A. rhizogenes leads to production of a large root mass, rather than a tumor. Therefore, the large plasmid carried by A. rhizogenes is called a Root-inducing (Ri) plasmid p. Ri Mulberry infected with A. rhizogenes Image credits: William M. Brown Jr. , Bugwood. org; Reprinted from Dhakulkar, S. , Ganapathi, T. R. , Bhargava, S. and Bapat, V. A. (2005). Induction of hairy roots in Gmelina arborea Roxb. and production of verbascoside in hairy roots. Plant Sci. 169: 812 -818 with permission from Elsevier. © 2014 American Society of Plant Biologists

Conclusions Agrobacterium is an amazing organism, with a unique ability to transfer DNA into diverse host genomes, which has been exploited to facilitate research and breeding Agrobacterium research and its application went far beyond what Smith and Townsend could foresee when they found crown gall was caused by the bacterium in 1907 Edward L. Barnard, Florida Department of Agriculture and Consumer Services, Bugwood. org; Mike Ellis, Ohio State University; © 2014 American Society of Plant Biologists
- Slides: 24