Agenda 1 Book scavenger hunt 2 Review Chemical

Agenda 1. Book scavenger hunt 2. Review Chemical Reactions 3. Properties of Water Notes 4. p. H scale practice 5. Pass back outlines

d n a r e t a W f o Properties Life

e c a f r u S d n a n o i s e h d A & n o i Cohes Tension cohesion- hydrogen bonds hold or link substances together
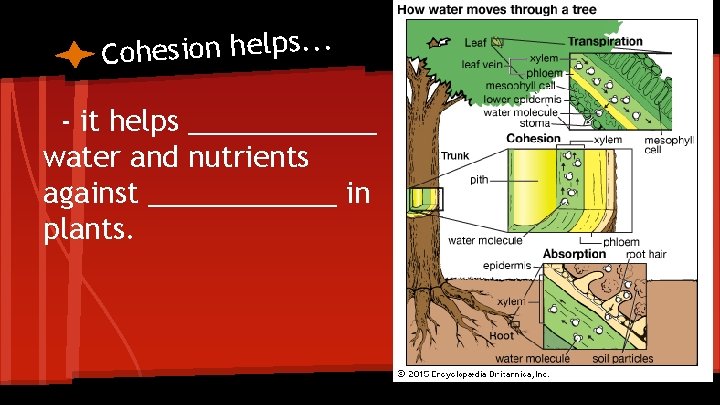
. . . s p l e h n o i s e h o C - it helps ______ water and nutrients against ______ in plants.

Adhesion: the clinging of one substance to another ●the clinging/adhesion of water by hydrogen bonds to the molecules of the cell wall helps go against the downward pull of _______.

n o i s n e T e c a f Sur This is the measure of how difficult it is to stretch or break the surface of a liquid. *it’s kind of like water is coated with an invisible film …. that’s why spiders can walk across water!

: s e i t r e p o r P l a Therm t a e h c i f i c e p S High This is the amount of heat that must be absorbed or lost for 1 g of a substance to change it’s temp 1 degree Celsius. Water has high specific heat (think of specific heat as the measure of the resistance of a substance to changing its temperature when it absorbs or releases heat. )
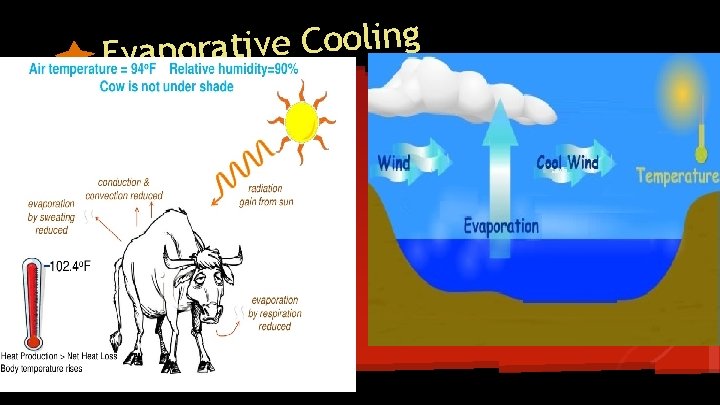
g n i l o o C e v i t a Evapor

p m e t s l o r t n o c Water

Density Which is less dense? The cork or rock? W Water is LESS dense as a solid than as a liquid! Few things are like this. ICE FLOATS ON WATER!!! Water expands when it solidifies. Most other things contract and become denser when they solidify.

. . . e s u a c e b t n a t r o p m i s i y t i s Den If all ice sank…. all ponds, lakes, and oceans would freeze completely solid, making life impossible.

t n e v l o S l a s r e v i Un Due to its polarity, water is an excellent and universal solvent. MANY compounds are water soluble and will dissolve when water surrounds the molecules, forming hydrogen bonds with them

? r e t a w n i s e v l What disso ionic and polar things are the best, but not everything needs to be ionic in order to dissolve. Sugar is non-ionic, but polar, so it dissolves (water soluble).


- Slides: 15