After today you will be able to Explain

After today, you will be able to… • Explain what ionization energy is • Describe the ionization energy trend from the Periodic Table • Explain why it requires more energy to remove each subsequent electron after the first

Unit 4: The Periodic Trends with Ionization Energy (I. E. )

Ionization Energy (I. E. ) • The amount of energy required to remove an electron from a gaseous atom. Example: Li Li+1 + 1 e • Depends on: 1. Distance between electron and nucleus 2. Nuclear charge (# of protons)

Group trend: I. E. decreases as you go down a group due to a greater # of energy levels Example: Ionization energy for elements in the same group: Lithium vs. Cesium e- More difficult to remove e- = e- closer to nucleus = higher I. E. e- Easier to remove e- = efurther away from nucleus = lower I. E.

Period trend: I. E. increases as you move across a period due to an increased nuclear charge Electrons are held more tightly because of more protons therefore more I. E. is required to remove an electron
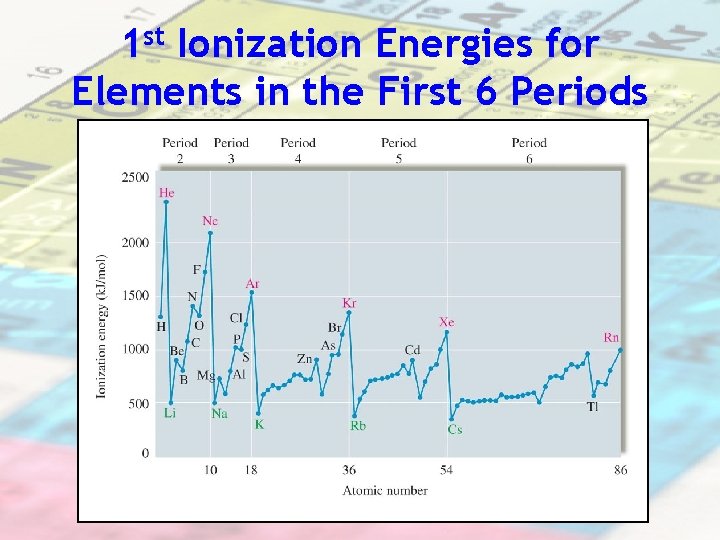
1 st Ionization Energies for Elements in the First 6 Periods

It always requires more energy to remove a 2 nd or 3 rd electron because they are more attracted to a positive ion than to a neutral atom.

1 st I. E. - Energy required to remove the 1 st electron. 2 nd I. E. - Energy required to remove the 2 nd electron. 3 rd I. E. - Energy required to remove the 3 rd electron. st 1 I. E. < nd 2 I. E. < rd 3 I. E.

1 st Ionization Energy 2 nd Ionization Energy 495. 9 k. J 4, 560 k. J Magnesium 738. 1 k. J 1, 450 k. J Sodium 3 rd Ionization Energy 7, 730 k. J There is a very large increase of ionization energy whenever an electron is removed from an atom/ion that is isoelectronic with a noble gas.
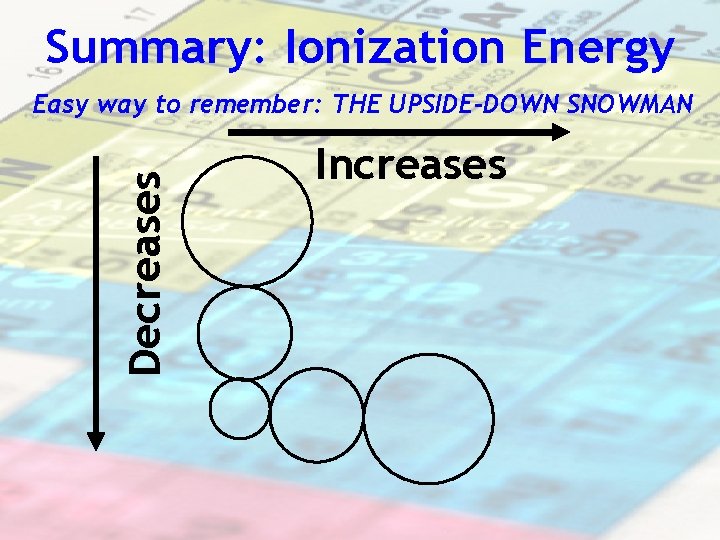
Summary: Ionization Energy Decreases Easy way to remember: THE UPSIDE-DOWN SNOWMAN Increases

Do now: Complete and Turn in “Exit Ticket” Then: Begin WS 3 (Homework)
- Slides: 11