Afatinib versus cisplatin plus gemcitabine for firstline treatment

Afatinib versus cisplatin plus gemcitabine for first-line treatment of Chinese patients with advanced non-small-cell lung cancer harboring EGFR mutations: subgroup analysis of the LUX-Lung 6 trial Yi-Long Wu, 1 Chong-Rui Xu, 1 Cheng-Ping Hu, 2 Jifeng Feng, 3 Shun Lu, 4 Yunchao Huang, 5 Wei Li, 6 Mei Hou, 7 Jian Hua Shi, 8 Angela Märten, 9 Jean Fan, 10 Barbara Peil, 11 Caicun Zhou 12 1 Guangdong Lung Cancer Institute, Guangdong General Hospital and Guangdong Academy of Medical Sciences, Guangzhou, China; 2 Department of Pulmonary Medicine, Xiangya Hospital, Central South University, Changsha, China; 3 Department of Internal Medicine, Jiangsu Provincial Tumor Hospital, Nanjing, Jiangsu, China; 4 Shanghai Lung Cancer Center, Shanghai Chest Hospital, Shanghai Jiao Tong University, Shanghai, China; 5 Yunnan Tumor Hospital (The Third Affiliated Hospital of Kunming Medical University), Kunming, Yunnan Province, China; 6 Cancer. Center, First Hospital of Jilin University, Changchun, China; 7 West China Hospital, Sichuan University, Chengdu, Sichuan, China; 8 Lin Yi Tumor Hospital, Linyi, Shandong Province, China; 9 Boehringer Ingelheim Gmb. H, Ingelheim, Germany; 10 Boehringer Ingelheim Pharmaceuticals, Inc. , Ridgefield, CT, USA; 11 Boehringer Ingelheim Pharma Gmb. H & Co. KG, Ingelheim, Germany; 12 Shanghai Pulmonary Hospital, Yangpu District, Shanghai, China

Introduction • The reversible EGFR TKIs, erlotinib and gefitinib, and the irreversible ERBB family blocker, afatinib, are approved globally for the first-line treatment of advanced EGFR-mutation positive NSCLC 1– 4 – There are few data to guide treatment choice • In the Phase III LUX-Lung 3 (LL 3) and LL 6 studies, afatinib significantly improved PFS and several PROs versus chemotherapy 5, 6 • In a prespecified analysis of Del 19+ patients, afatinib also significantly prolonged OS versus chemotherapy 7 – Del 19 is the most common mutation found among Chinese patients with EGFR-mutation positive NSCLC (~50%)8 • Here, we report a post-hoc subgroup analysis of the efficacy and safety of afatinib versus chemotherapy in Chinese patients participating in LL 6 EGFR, epidermal growth factor receptor; PFS, progression-free survival; PROs, patient-reported outcomes; OS, overall survival; TKI, tyrosine kinase inhibitor 1. Novello S, et al. Ann Oncol 2016; 27: v 1–v 27; 2. Masters GA, et al. J Clin Oncol 2015; 33: 3488– 515; 3. Zhi X-Y, et al. Cancer 2015; 121: 3165– 81; 4. BI press release. http: //www. Boehringer. Ingelheim. com/press-release/afatinib-approved-lung-cancer-china. Accessed September 2017; 5. Sequist LV, et al. J Clin Oncol 2013; 31: 3327– 34; 6. Wu Y-L, et al. Lancet Oncol 2014; 15: 213– 22 7. Yang JC, et al. Lancet Oncol 2015; 16: 141– 51; 8. Shi Y, et al. PLo. S One 2015; 10: e 0143515
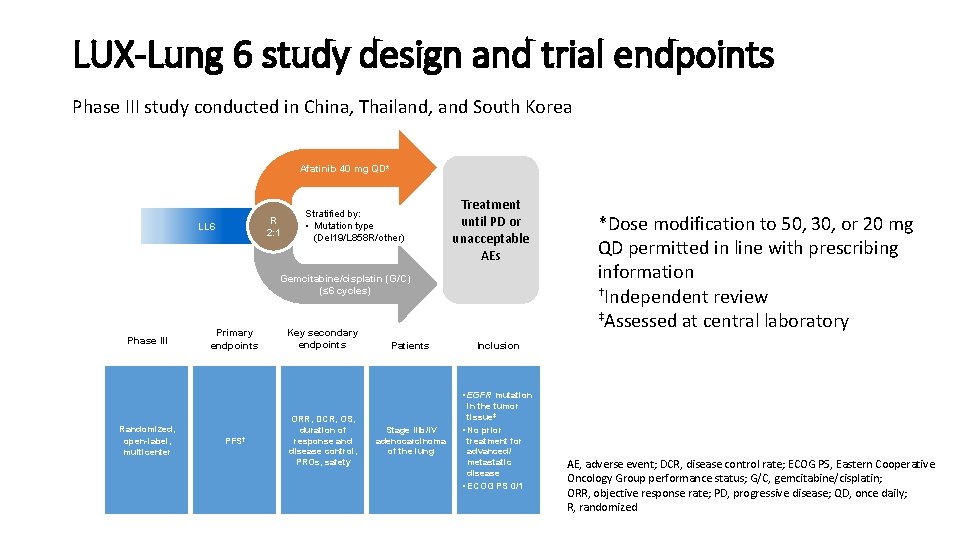
LUX-Lung 6 study design and trial endpoints Phase III study conducted in China, Thailand, and South Korea Afatinib 40 mg QD* R 2: 1 LL 6 Stratified by: • Mutation type (Del 19/L 858 R/other) Treatment until PD or unacceptable AEs Gemcitabine/cisplatin (G/C) (≤ 6 cycles) Phase III Primary endpoints Key secondary endpoints Patients *Dose modification to 50, 30, or 20 mg QD permitted in line with prescribing information †Independent review ‡Assessed at central laboratory Inclusion • EGFR mutation Randomized, open-label, multicenter PFS† ORR, DCR, OS, duration of response and disease control, PROs, safety Stage IIIb/IV adenocarcinoma of the lung in the tumor tissue‡ • No prior treatment for advanced/ metastatic disease • ECOG PS 0/1 AE, adverse event; DCR, disease control rate; ECOG PS, Eastern Cooperative Oncology Group performance status; G/C, gemcitabine/cisplatin; ORR, objective response rate; PD, progressive disease; QD, once daily; R, randomized

Baseline demographics/characteristics were comparable between the Chinese subgroup and the overall LL 6 population Characteristic Sex, n (%) Female/ Male Median age, years (range) Race/ethnicity, * n (%) Southeast Asian South Korean Chinese ECOG PS, n (%) 0/ 1 Smoking status, n (%) Never smoked Other current or ex-smoker <15 pack-years and stopped >1 year ago Adenocarcinoma stage, n (%) IIIB IV Number of metastatic sites, n (%) 0 1 2 ≥ 3 EGFR mutation, n (%) Common mutations Del 19 L 858 R Uncommon mutations‡ Overall LL 6 population (N=364) Chinese patients (N=327) Afatinib (n=242) G/C (n=122) Afatinib (n=217) G/C (n=110) 155 (64. 0)/ 87 (36. 0) 58 (29– 79) 14 (5. 8) 11 (4. 5) 217 (89. 7) 83 (68. 0)/ 39 (32. 0) 58 (27– 76) 10 (8. 2) 2 (1. 6) 110 (90. 2) 136 (62. 7)/ 81 (37. 3) 58 (30– 78) 0 0 217 (100) 76 (69. 1)/ 34 (30. 9) 58 (27– 75) 0 0 110 (100) 48 (19. 8)/ 194 (80. 2) 181 (74. 8) 53 (21. 9) 8 (3. 3) 41 (33. 6)/ 81 (66. 4) 99 (81. 1) 19 (15. 6) 4 (3. 3) 44 (20. 3)/ 173 (79. 7) 159 (73. 3) 50 (23. 0) 8 (3. 7) 40 (36. 4)/ 70 (63. 6) 92 (83. 6) 15 (13. 6) 3 (2. 7) 16 (6. 6) 226 (93. 4) 5 (2. 1) 72 (29. 8) 91 (37. 6) 74 (30. 6) 216 (89. 3) 124 (51. 2) 92 (38. 0)† 26 (10. 7) 6 (4. 9) 116 (95. 1) 1 (0. 8) 52 (42. 6) 36 (29. 5) 33 (27. 0) 108 (88. 5) 62 (50. 8) 46 (37. 7) 14 (11. 5) 13 (6. 0) 204 (94. 0) 3 (1. 4) 65 (30. 0) 84 (38. 7) 65 (30. 0) 193 (88. 9) 111 (51. 2) 82 (37. 8)† 24 (11. 1) 6 (5. 5) 104 (94. 5) 1 (0. 9) 48 (43. 6) 30 (27. 3) 31 (28. 2) 97 (88. 2) 55 (50. 0) 42 (38. 2) 13 (11. 8) • In LL 6, 327/364 (90%) patients randomized in LL 6 were from mainland China • The treated sets comprised 214 patients for afatinib and 103 for G/C • Median (range) of treatment duration was 389. 5 days (3– 1, 256) and 86 days (1– 154) with afatinib and G/C, respectively *Available categories within Asian race included Indian subcontinent Asian, Southeast Asian, Japanese, Korean, Taiwanese or Chinese, Asian – Other; †Including 4 patients with both L 858 R and Del 19 mutation. ‡Including T 790 M, exon 20 insertions, G 719 X, S 768 I, and L 861 Q, alone or as complex mutations in 2 or more exons.
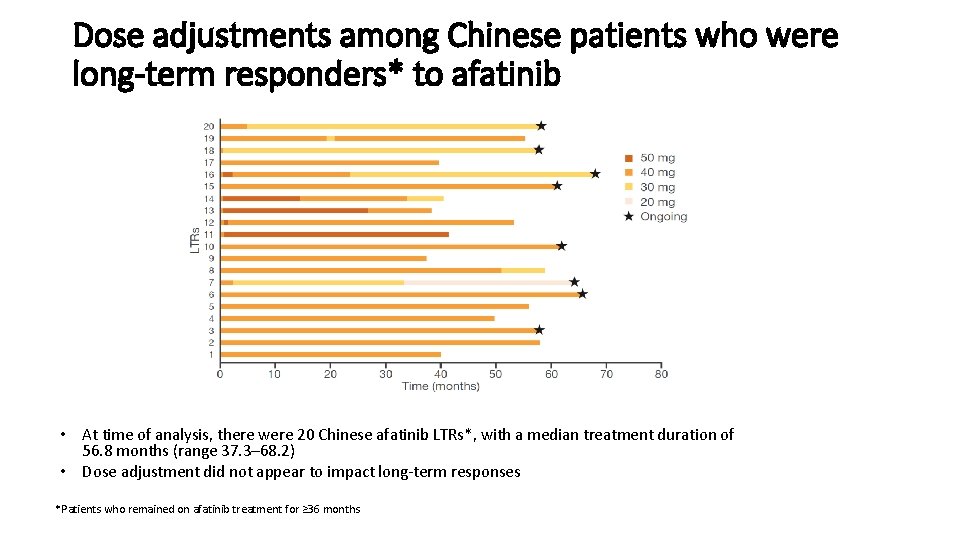
Dose adjustments among Chinese patients who were long-term responders* to afatinib • At time of analysis, there were 20 Chinese afatinib LTRs*, with a median treatment duration of 56. 8 months (range 37. 3– 68. 2) • Dose adjustment did not appear to impact long-term responses *Patients who remained on afatinib treatment for ≥ 36 months
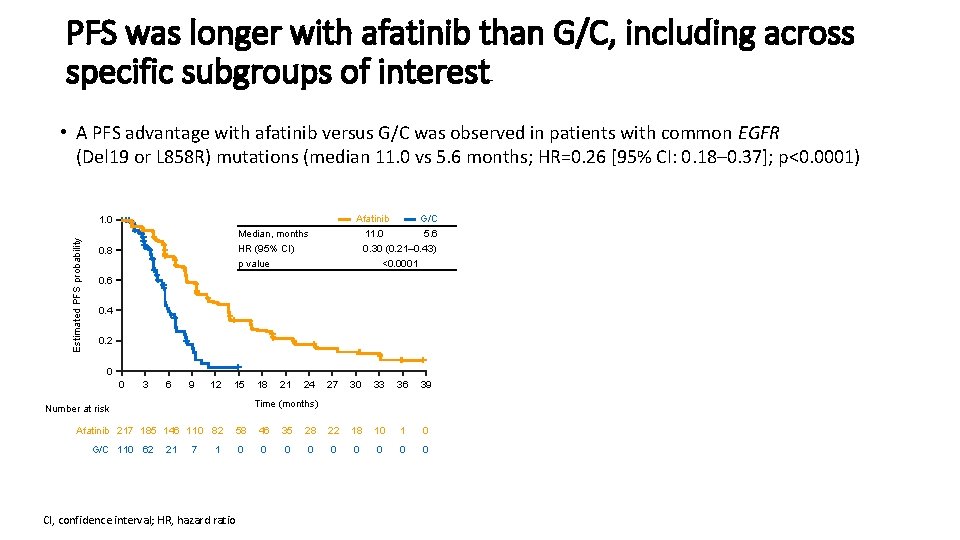
PFS was longer with afatinib than G/C, including across specific subgroups of interest • A PFS advantage with afatinib versus G/C was observed in patients with common EGFR (Del 19 or L 858 R) mutations (median 11. 0 vs 5. 6 months; HR=0. 26 [95% CI: 0. 18– 0. 37]; p<0. 0001) Afatinib G/C 11. 0 5. 6 0. 30 (0. 21– 0. 43) <0. 0001 Estimated PFS probability 1. 0 Median, months HR (95% CI) p value 0. 8 0. 6 0. 4 0. 2 0 0 3 6 9 12 15 21 24 27 30 33 36 39 Time (months) Number at risk Afatinib 217 185 146 110 82 G/C 110 62 18 21 7 58 46 35 28 22 18 10 1 0 0 0 0 0 1 CI, confidence interval; HR, hazard ratio
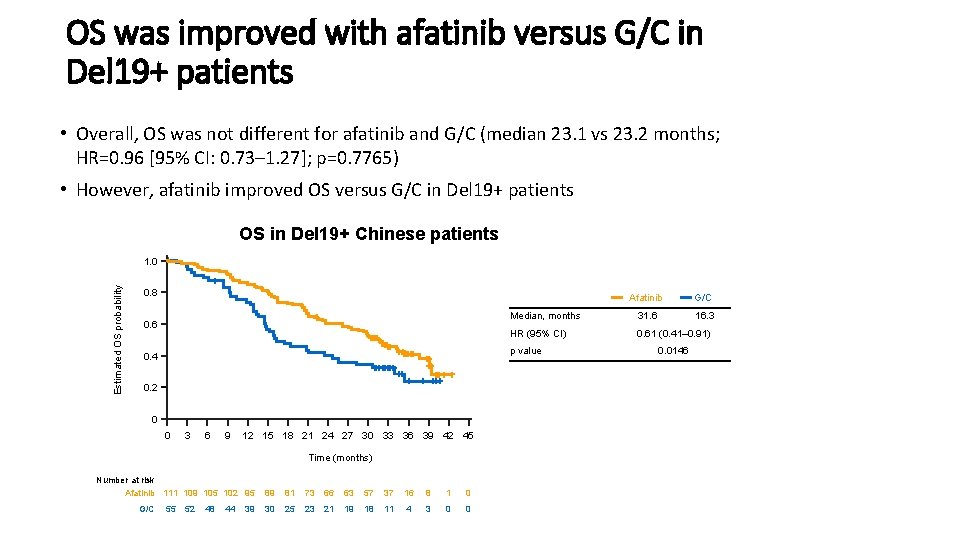
OS was improved with afatinib versus G/C in Del 19+ patients • Overall, OS was not different for afatinib and G/C (median 23. 1 vs 23. 2 months; HR=0. 96 [95% CI: 0. 73– 1. 27]; p=0. 7765) • However, afatinib improved OS versus G/C in Del 19+ patients OS in Del 19+ Chinese patients Estimated OS probability 1. 0 0. 8 0. 6 G/C Median, months 31. 6 16. 3 HR (95% CI) 0. 61 (0. 41– 0. 91) p value 0. 4 0. 2 0 0 3 6 9 12 15 18 21 24 27 30 33 36 39 42 45 Time (months) Number at risk Afatinib 111 109 105 102 95 G/C Afatinib 55 52 48 44 39 89 81 73 66 63 57 37 16 8 1 0 30 25 23 21 19 18 11 4 3 0 0 0. 0146
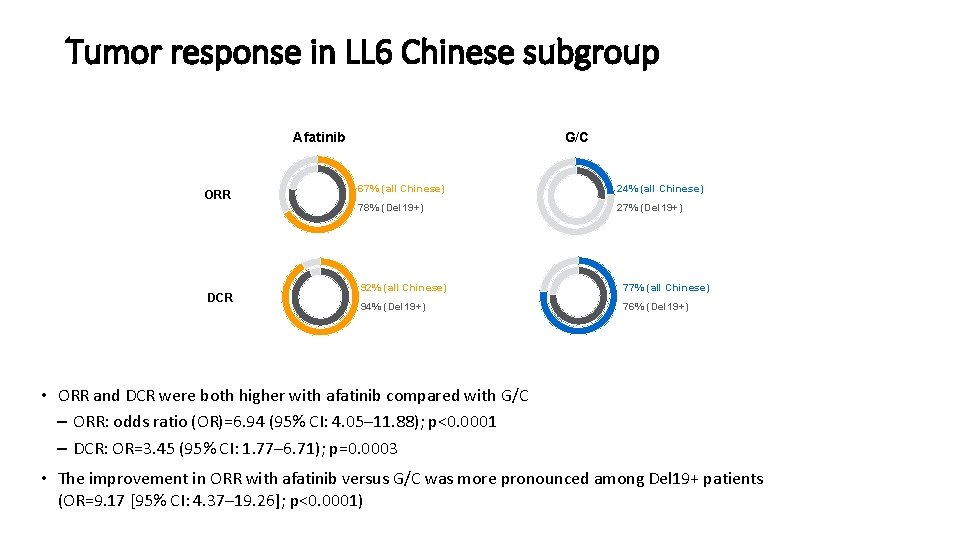
Tumor response in LL 6 Chinese subgroup Afatinib ORR DCR G/C 67% (all Chinese) 24% (all Chinese) 78% (Del 19+) 27% (Del 19+) 92% (all Chinese) 77% (all Chinese) 94% (Del 19+) 76% (Del 19+) • ORR and DCR were both higher with afatinib compared with G/C – ORR: odds ratio (OR)=6. 94 (95% CI: 4. 05– 11. 88); p<0. 0001 – DCR: OR=3. 45 (95% CI: 1. 77– 6. 71); p=0. 0003 • The improvement in ORR with afatinib versus G/C was more pronounced among Del 19+ patients (OR=9. 17 [95% CI: 4. 37– 19. 26]; p<0. 0001)
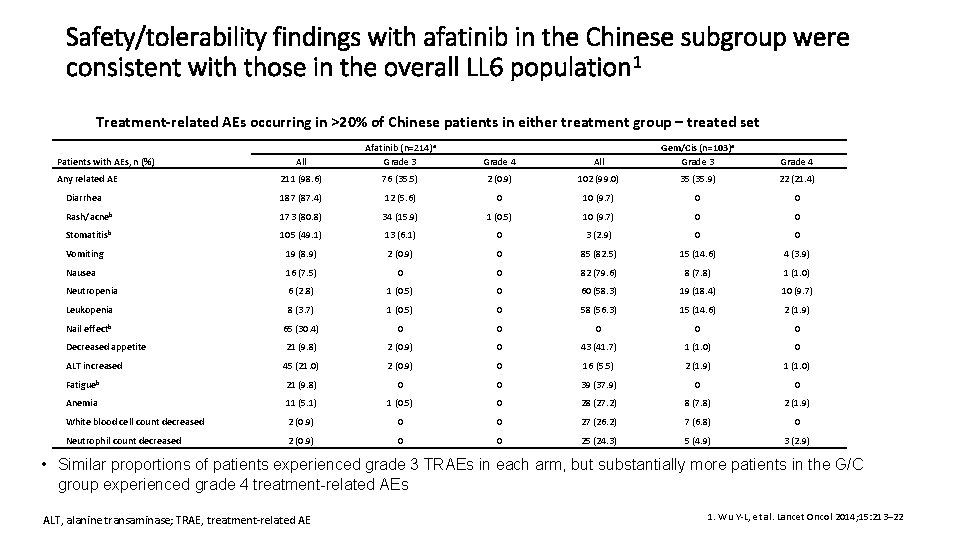
Safety/tolerability findings with afatinib in the Chinese subgroup were consistent with those in the overall LL 6 population 1 Treatment-related AEs occurring in >20% of Chinese patients in either treatment group – treated set All Gem/Cis (n=103)a Grade 3 Grade 4 2 (0. 9) 102 (99. 0) 35 (35. 9) 22 (21. 4) 12 (5. 6) 0 10 (9. 7) 0 0 173 (80. 8) 34 (15. 9) 1 (0. 5) 10 (9. 7) 0 0 105 (49. 1) 13 (6. 1) 0 3 (2. 9) 0 0 Vomiting 19 (8. 9) 2 (0. 9) 0 85 (82. 5) 15 (14. 6) 4 (3. 9) Nausea 16 (7. 5) 0 0 82 (79. 6) 8 (7. 8) 1 (1. 0) Neutropenia 6 (2. 8) 1 (0. 5) 0 60 (58. 3) 19 (18. 4) 10 (9. 7) Leukopenia 8 (3. 7) 1 (0. 5) 0 58 (56. 3) 15 (14. 6) 2 (1. 9) Nail effectb 65 (30. 4) 0 0 0 Decreased appetite 21 (9. 8) 2 (0. 9) 0 43 (41. 7) 1 (1. 0) 0 ALT increased 45 (21. 0) 2 (0. 9) 0 16 (5. 5) 2 (1. 9) 1 (1. 0) Fatigueb 21 (9. 8) 0 0 39 (37. 9) 0 0 Anemia 11 (5. 1) 1 (0. 5) 0 28 (27. 2) 8 (7. 8) 2 (1. 9) White blood cell count decreased 2 (0. 9) 0 0 27 (26. 2) 7 (6. 8) 0 Neutrophil count decreased 2 (0. 9) 0 0 25 (24. 3) 5 (4. 9) 3 (2. 9) All Afatinib (n=214)a Grade 3 Grade 4 211 (98. 6) 76 (35. 5) Diarrhea 187 (87. 4) Rash/acneb Stomatitisb Patients with AEs, n (%) Any related AE • Similar proportions of patients experienced grade 3 TRAEs in each arm, but substantially more patients in the G/C group experienced grade 4 treatment-related AEs ALT, alanine transaminase; TRAE, treatment-related AE 1. Wu Y-L, et al. Lancet Oncol 2014; 15: 213– 22
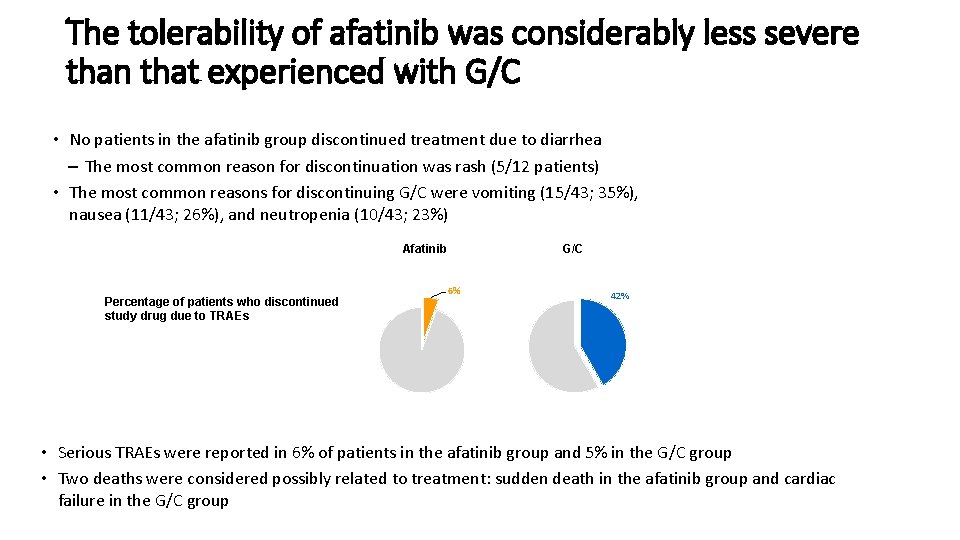
The tolerability of afatinib was considerably less severe than that experienced with G/C • No patients in the afatinib group discontinued treatment due to diarrhea – The most common reason for discontinuation was rash (5/12 patients) • The most common reasons for discontinuing G/C were vomiting (15/43; 35%), nausea (11/43; 26%), and neutropenia (10/43; 23%) Afatinib Percentage of patients who discontinued study drug due to TRAEs G/C 6% 42% • Serious TRAEs were reported in 6% of patients in the afatinib group and 5% in the G/C group • Two deaths were considered possibly related to treatment: sudden death in the afatinib group and cardiac failure in the G/C group
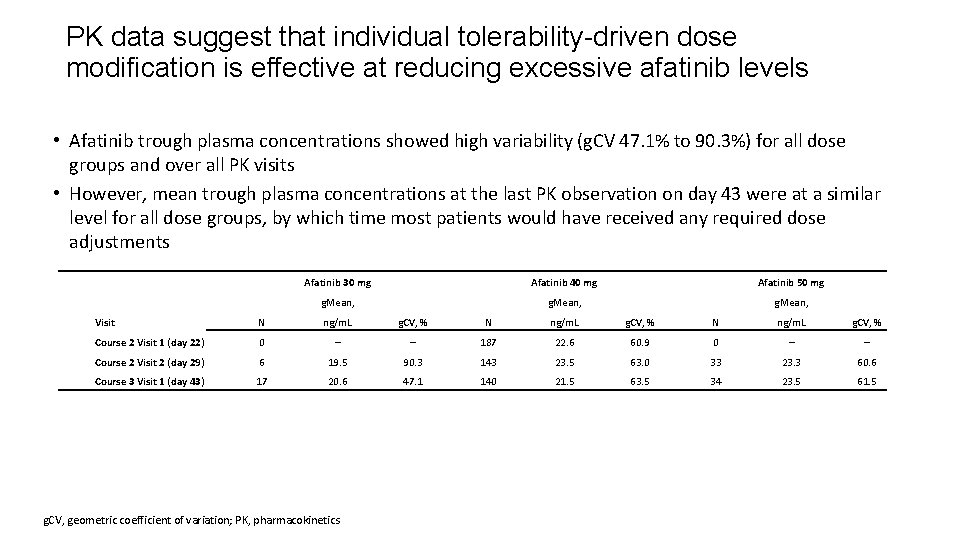
PK data suggest that individual tolerability-driven dose modification is effective at reducing excessive afatinib levels • Afatinib trough plasma concentrations showed high variability (g. CV 47. 1% to 90. 3%) for all dose groups and over all PK visits • However, mean trough plasma concentrations at the last PK observation on day 43 were at a similar level for all dose groups, by which time most patients would have received any required dose adjustments Afatinib 30 mg Afatinib 40 mg Afatinib 50 mg g. Mean, Visit N ng/m. L g. CV, % Course 2 Visit 1 (day 22) 0 – – 187 22. 6 60. 9 0 – – Course 2 Visit 2 (day 29) 6 19. 5 90. 3 143 23. 5 63. 0 33 23. 3 60. 6 Course 3 Visit 1 (day 43) 17 20. 6 47. 1 140 21. 5 63. 5 34 23. 5 61. 5 g. CV, geometric coefficient of variation; PK, pharmacokinetics
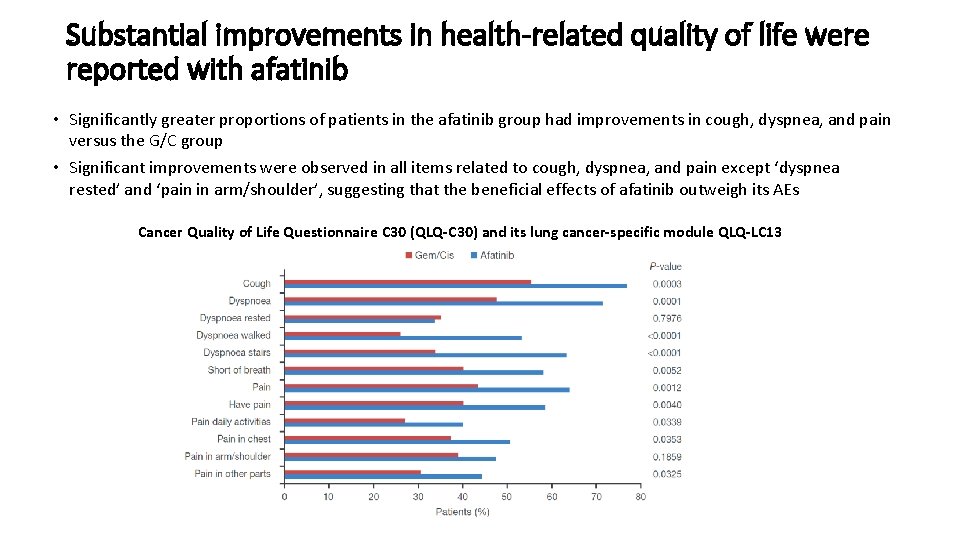
Substantial improvements in health-related quality of life were reported with afatinib • Significantly greater proportions of patients in the afatinib group had improvements in cough, dyspnea, and pain versus the G/C group • Significant improvements were observed in all items related to cough, dyspnea, and pain except ‘dyspnea rested’ and ‘pain in arm/shoulder’, suggesting that the beneficial effects of afatinib outweigh its AEs Cancer Quality of Life Questionnaire C 30 (QLQ-C 30) and its lung cancer-specific module QLQ-LC 13

Key findings and conclusions • Afatinib improved PFS and ORR versus G/C in Chinese patients • An improvement in OS was also observed versus G/C in Chinese patients with an EGFR Del 19 mutation • The improvement in efficacy with afatinib versus G/C was reflected in a delayed time to deterioration of PROs for the symptoms most important to patients with NSCLC: cough, dyspnea, and pain • There were no unexpected safety findings with afatinib in Chinese patients, and discontinuation due to TRAEs was uncommon • Afatinib should therefore be considered as a first-line treatment option for Chinese patients with EGFR-mutation positive NSCLC

References 1. 2. 3. 4. 5. 6. 7. 8. Novello S, et al. Ann Oncol 2016; 27: v 1–v 27 Masters GA, et al. J Clin Oncol 2015; 33: 3488– 515 Zhi X-Y, et al. Cancer 2015; 121: 3165– 81 BI press release. http: //www. Boehringer-Ingelheim. com/press-release/afatinib-approved-lung-cancer-china. Accessed September 2017 Sequist LV, et al. J Clin Oncol 2013; 31: 3327– 34 Wu Y-L, et al. Lancet Oncol 2014; 15: 213– 22 Yang JC, et al. Lancet Oncol 2015; 16: 141– 51 Shi Y, et al. PLo. S One 2015; 10: e 0143515 Acknowledgments This study was funded by Boehringer Ingelheim. The authors were fully responsible for all content and editorial decisions, were involved at all stages of manuscript development and have approved the final version. Medical writing assistance, supported financially by Boehringer Ingelheim, was provided by Lynn Pritchard of Geo. Med, an Ashfield company, part of UDG Healthcare plc, during the development of the manuscript and accompanying slide set These materials are for personal use only and may not be reproduced without written permission of the authors and the appropriate copyright permissions
- Slides: 14