Aerogels Invented by S S Kistler Stanford U


Aerogels Invented by S. S. Kistler (Stanford U. ) in 1931 lengthy process, first major breakthrough: supercritical drying of wet gels retaining volume of the gel “Forgotten” for almost 30 years JPL Website, Stardust Program Very lightweight glass-like materials, but extremely fragile At best: • 1. 5 mg/cc, Guinness World Records • 99. 8% porosity • 1000 times less dense than glass • about 40 times better thermal insulators than the best fiberglass “Re-invented” in the 1960’s in France second major breakthrough: sol-gel process cutting Kistler’s method from weeks to hours

Current and Projected Use for Aerogels have been considered for: - thermal insulation (architectural, automotive industrial applications); - acoustic insulation (buildings, automobiles, aircraft); - dielectrics (for fast electronics); - supports for catalysts; and, - hosts of functional guests for chemical, electronic and optical applications. Silica aerogels have been actually used: - as Cerenkov radiation detectors - aboard spacecraft: o as collectors of cosmic particles (Stardust Program) o for thermal insulation (e. g. , Sojourner Rover - 1997) Commercialization has been slow, because silica aerogels are: - fragile; - hygroscopic; and, - require supercritical fluid (SCF) extraction

Crosslinking aerogels — Microscopically, nanocast conformal polymer coating on the silica nanoparticles thicker necks increase strength of material conformal polymer coating Micropores are blocked Leventis, et al, Nano Letters, 2002
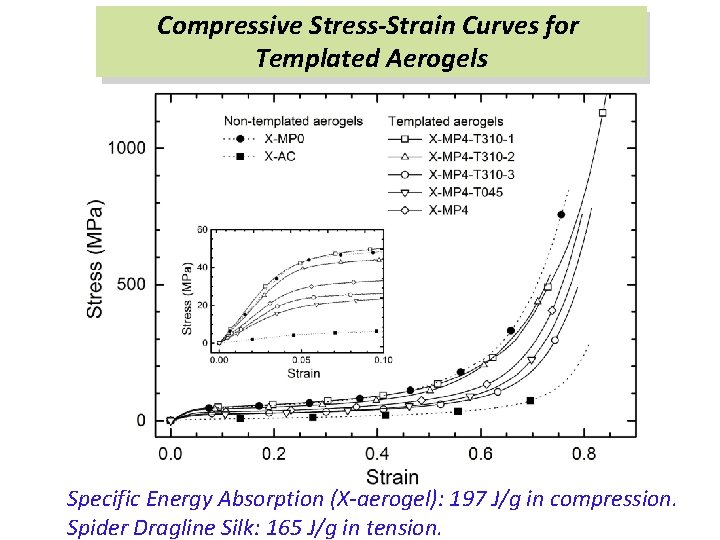
Compressive Stress-Strain Curves for Templated Aerogels Specific Energy Absorption (X-aerogel): 197 J/g in compression. Spider Dragline Silk: 165 J/g in tension.
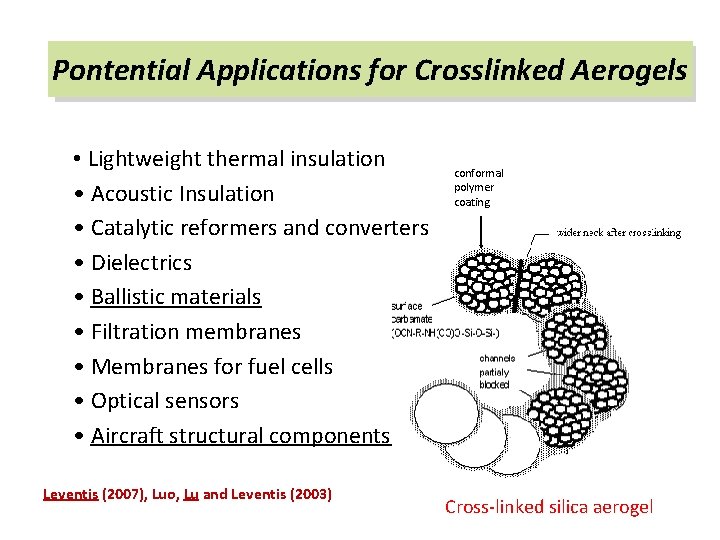
Pontential Applications for Crosslinked Aerogels • Lightweight thermal insulation • Acoustic Insulation • Catalytic reformers and converters • Dielectrics • Ballistic materials • Filtration membranes • Membranes for fuel cells • Optical sensors • Aircraft structural components Leventis (2007), Luo, Lu and Leventis (2003) conformal polymer coating Cross-linked silica aerogel
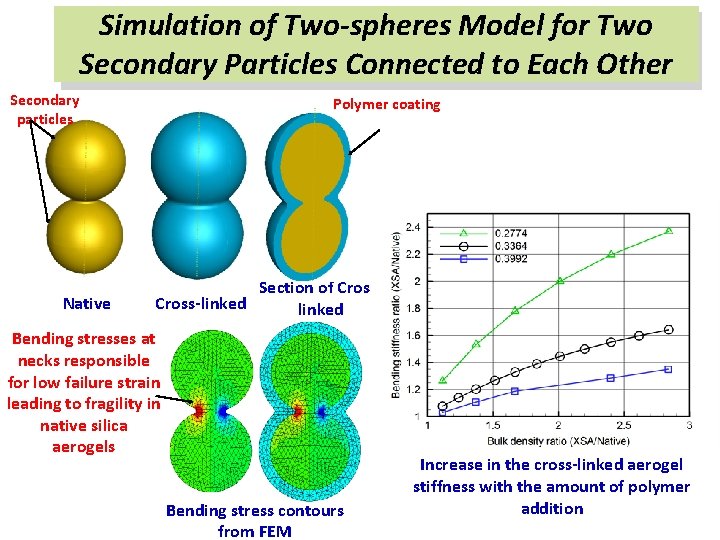
Simulation of Two-spheres Model for Two Secondary Particles Connected to Each Other Secondary particles Native Polymer coating Section of Cross-linked Bending stresses at necks responsible for low failure strain leading to fragility in native silica aerogels Bending stress contours from FEM Increase in the cross-linked aerogel stiffness with the amount of polymer addition

SEM of Crosslinked Templated Aerogels (X-MP 4 -T 045)
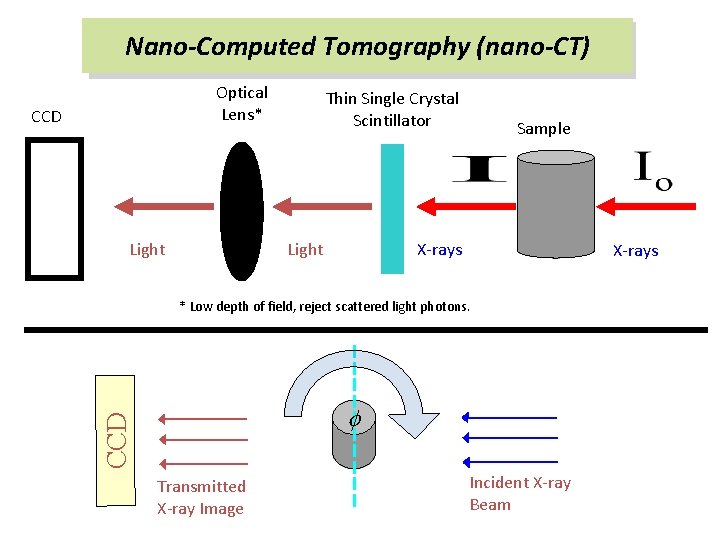
Nano-Computed Tomography (nano-CT) Optical Lens* CCD Light Thin Single Crystal Scintillator Light Sample X-rays * Low depth of field, reject scattered light photons. CCD f Transmitted X-ray Image Incident X-ray Beam
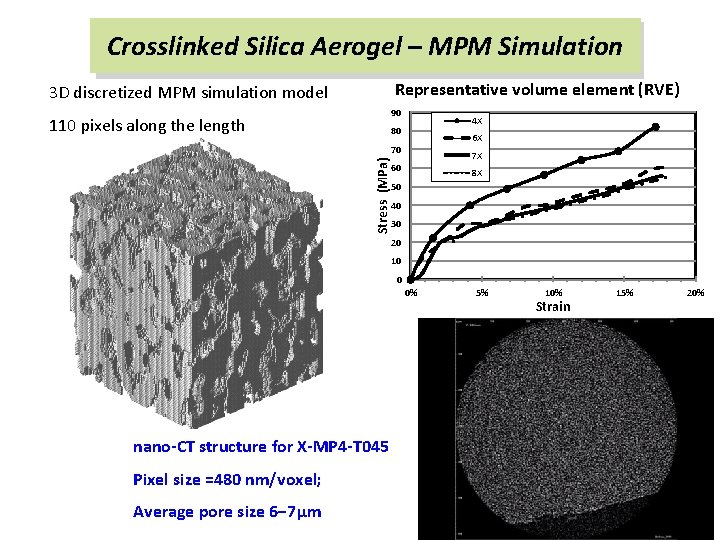
Crosslinked Silica Aerogel – MPM Simulation Representative volume element (RVE) 3 D discretized MPM simulation model 90 110 pixels along the length 4 X 80 6 X Stress (MPa) 70 7 X 60 8 X 50 40 30 20 10 0 0% nano-CT structure for X-MP 4 -T 045 Pixel size =480 nm/voxel; Average pore size 6− 7μm 5% 10% Strain 15% 20%

Crosslinked Silica Aerogel – MPM Simulation Flexural Modulus Estimation Weight ratio( Polymer: Silica)=70%: 30% Density of polymer = 1. 2 g/cc Density of silica = 2. 6 g/cc Modulus of polymer = 2 Gpa Modulus of silica =70 Gpa The volume ratio( Polymer: Silica) ≈5: 1 The dimensionless radius of the model R=6 Based on the bending equation for the composite material The silica-aerogel modulus is about 3. 889 Gpa

Simulation Results Ø Microstructural evolution under compression Microstructure deformation characteristic (comparison with rohacell foam) Ø Dynamic equilibrium Dynamic stress equilibrium; velocity loading history Ø Compressive stress-strain curve Typical silica-aerogel material stress-strain relation Ø Effect of porosity on the material properties Gibson & Ashby beam structure analog for the honeycomb structure material; response for the different porosity silicaaerogel models
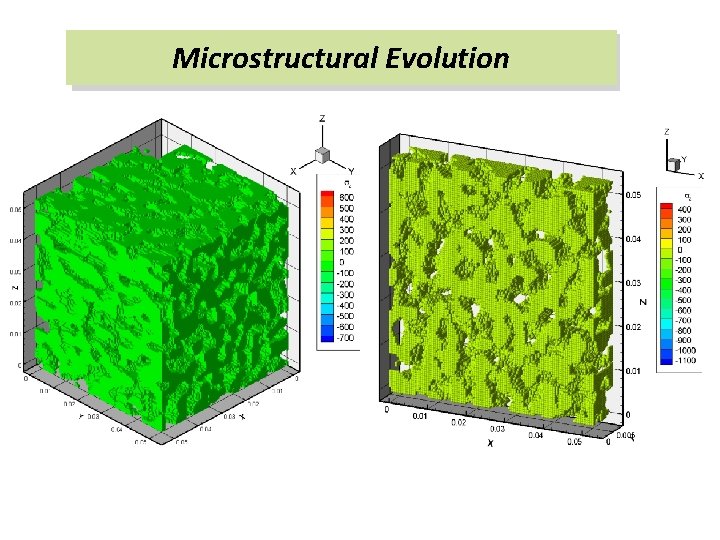
Microstructural Evolution
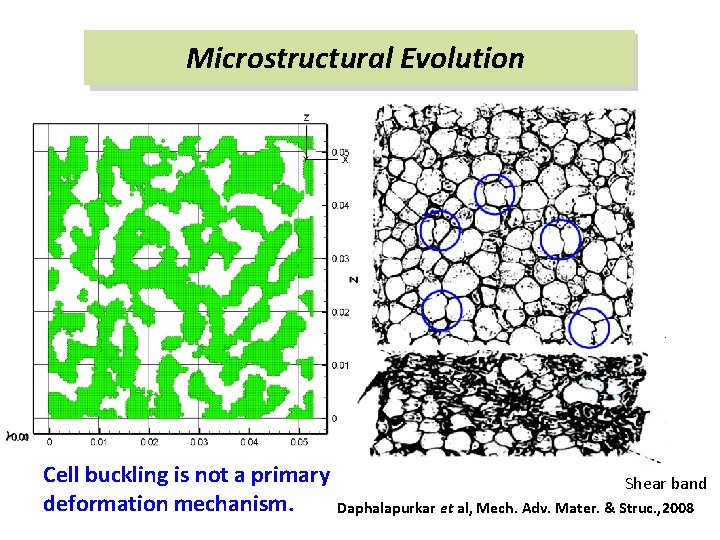
Microstructural Evolution Cell buckling is not a primary deformation mechanism. Shear band Daphalapurkar et al, Mech. Adv. Mater. & Struc. , 2008
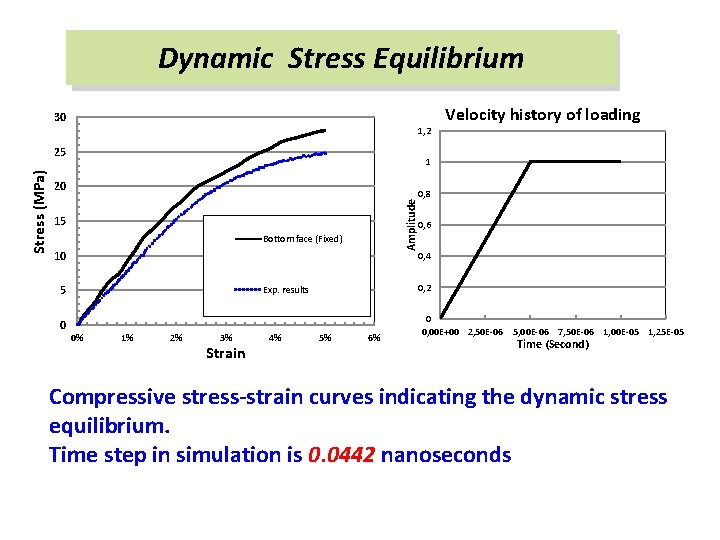
Dynamic Stress Equilibrium 30 1, 2 1 20 Amplitude Stress (MPa) 25 15 Bottom face (Fixed) 10 5 0 Velocity history of loading 1% 2% 3% Strain 4% 0, 6 0, 4 0, 2 Exp. results 0% 0, 8 5% 6% 0 0, 00 E+00 2, 50 E-06 5, 00 E-06 7, 50 E-06 1, 00 E-05 1, 25 E-05 Time (Second) Compressive stress-strain curves indicating the dynamic stress equilibrium. Time step in simulation is 0. 0442 nanoseconds
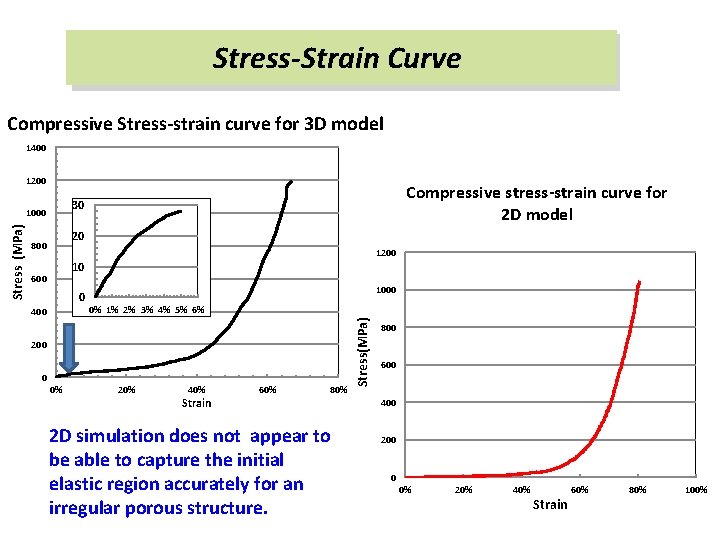
Stress-Strain Curve Compressive Stress-strain curve for 3 D model 1400 1200 1000 20 800 1200 10 600 0 400 1000 0% 1% 2% 3% 4% 5% 6% 200 0 0% 20% 40% Strain 60% 80% 2 D simulation does not appear to be able to capture the initial elastic region accurately for an irregular porous structure. Stress(MPa) Stress (MPa) Compressive stress-strain curve for 2 D model 30 800 600 400 200 0 0% 20% 40% Strain 60% 80% 100%
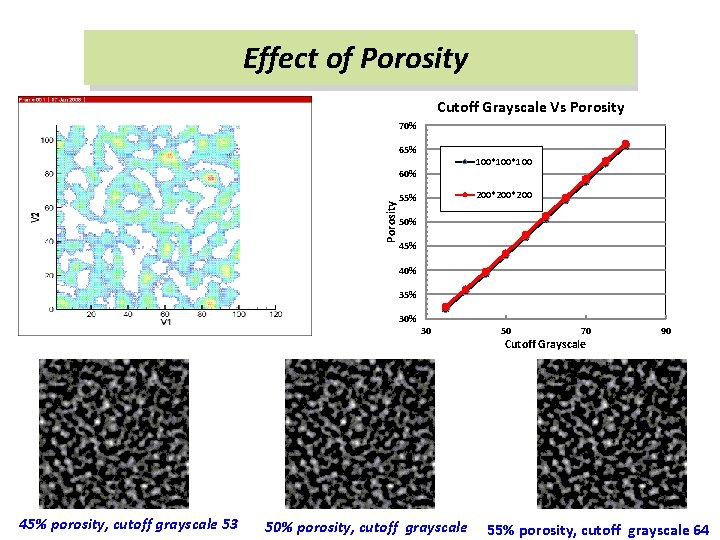
Effect of Porosity Cutoff Grayscale Vs Porosity 70% 65% 100*100 Porosity 60% 200*200 55% 50% 45% 40% 35% 30 45% porosity, cutoff grayscale 53 50% porosity, cutoff grayscale 50 70 Cutoff Grayscale 90 55% porosity, cutoff grayscale 64
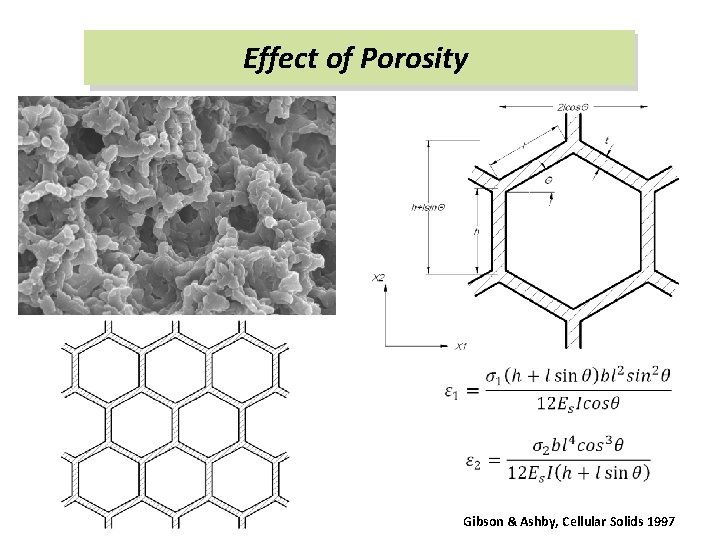
Effect of Porosity Gibson & Ashby, Cellular Solids 1997
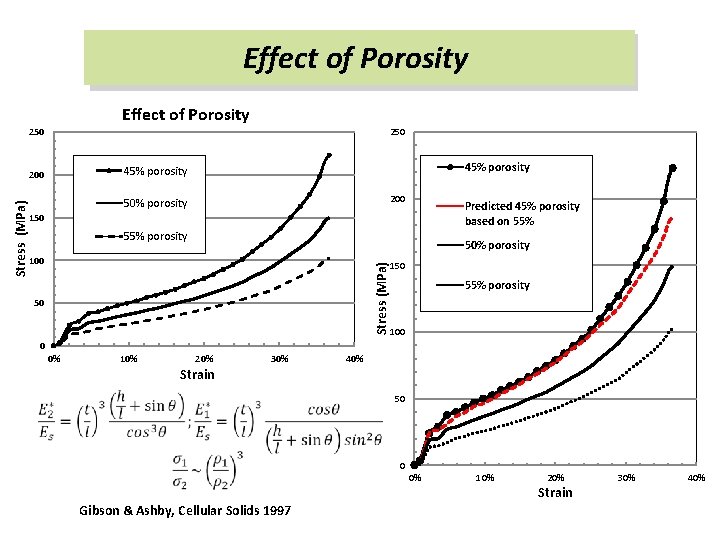
Effect of Porosity 250 200 50% porosity Predicted 45% porosity based on 55% 150 55% porosity 50% porosity 100 Stress (MPa) 45% porosity 200 50 150 55% porosity 100 0 0% 10% 20% 30% 40% Strain 50 0 0% Gibson & Ashby, Cellular Solids 1997 10% 20% Strain 30% 40%

Conclusion ØMPM simulation indicates the dynamic stress equilibrium condition has been reached. The stress-strain relation agrees with the experimental results in the elastic region and yielding region. ØThe simulation shows the potential to simulate the nanostructure property relationship of the crosslinked templated aerogels. ØThe simulation capture the elastic, compaction and densification behavior of the silica-aerogel. ØThe mechanical behavior of silica-aerogel follows a cubic power relation when the pores are not fully compacted. The relation does not hold when the pores are all closed.
- Slides: 20