Aerobic Metabolism of carbohydrate The three stages of

Aerobic Metabolism of carbohydrate

The three stages of respiration a. Stage I All the fuel molecules are oxidized to acetyl-Co. A. b. Stage II The acetyl-Co. A is completely oxidized into CO 2, electrons were collected by NAD and FAD via the citric acid cycle. c. Stage III Passage of electrons through the electron transport system to yield ATP from oxidative phosphorylation.
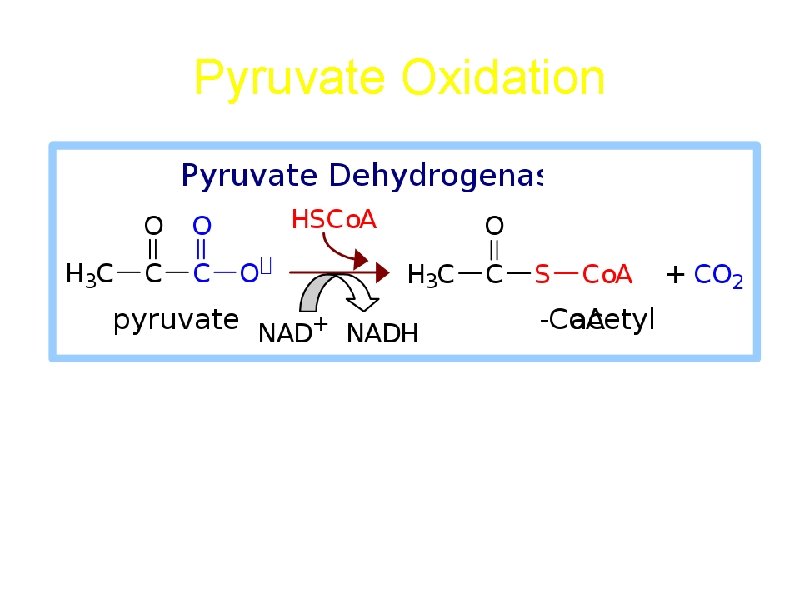
Pyruvate Oxidation Pyruvate + Co. A + NAD+ acetyl. Co. A + CO 2 + NADH + H+

Pyruvate Dehydrogenase is a large complex: pyruvate dehydrogenase (E 1), dihydrolipoyl transacetylase (E 2), dihydrolipoyl dehydrogenase (E 3) Requires 5 coenzymes: TPP, Lipoic Acid, Coenzyme A, FAD, NAD+
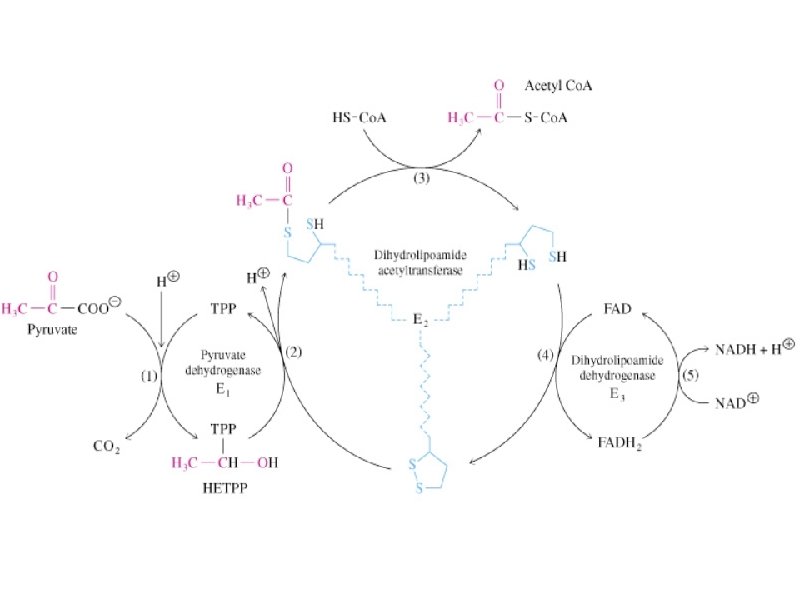
Reactions of the PDH complex

Regulation of Pyruvate Dehydrogenase 1). Product inhibition by NADH & acetyl Co. A 2). Covalent modification

Citric Acid Cycle a. Hans Krebs proposed the “citric acid cycle” for the complete oxidation of pyruvate in animal tissues in 1937 (1953 Nobel Prize laureate). b. The tricaboxylic acid (TCA) Cycle, Krebs Cycle

Citric Acid Cycle a. The common pathway leading to complete oxidation of carbohydrates, fatty acids, and amino acids to CO 2. b. Some ATP is produced, More NADH is made , NADH goes on to make more ATP in electron transport and oxidative phosphorylation c. A pathway providing many precursors for biosynthesis
citric acid cycle overview

Individual reaction
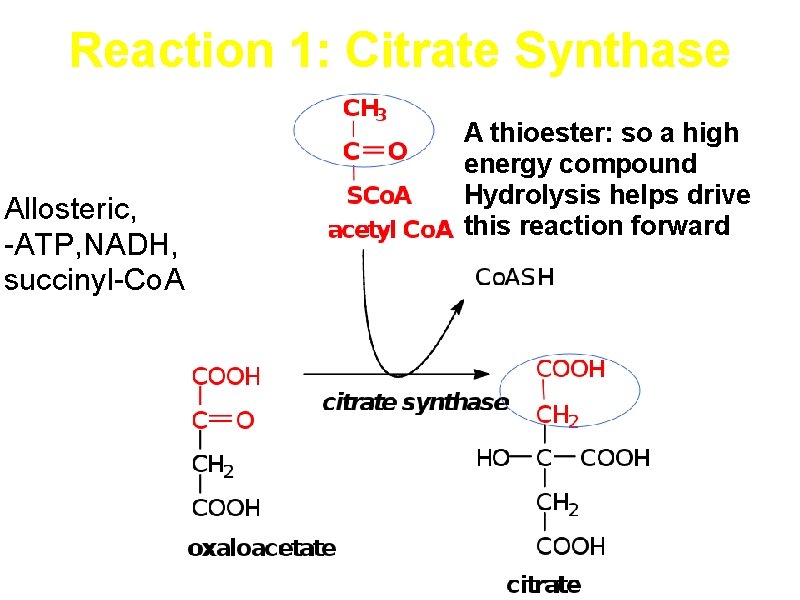
Reaction 1: Citrate Synthase Allosteric, -ATP, NADH, succinyl-Co. A A thioester: so a high energy compound Hydrolysis helps drive this reaction forward

Fluoroacetate blocks the cycle a. Fluoroacetate is poisonous because it can convert to fluorocitrate which is an inhibitors of TCA cycle.
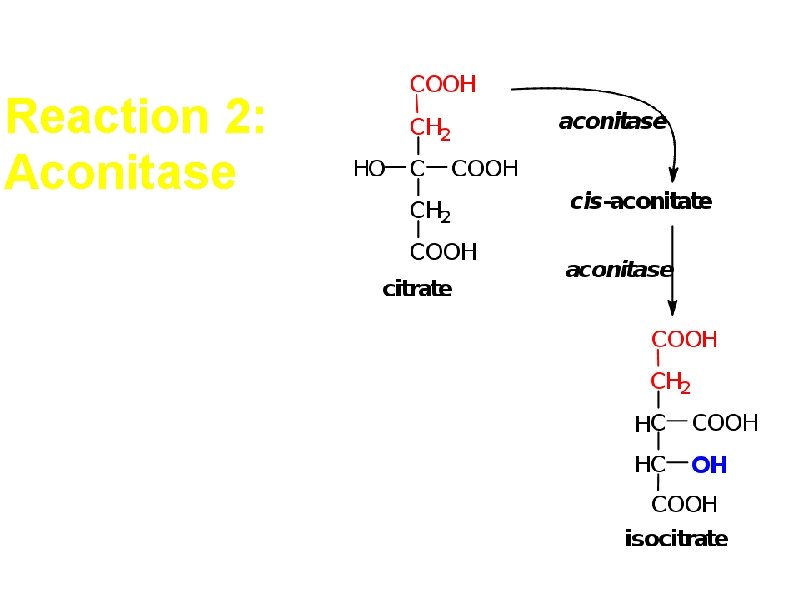
Reaction 2: Aconitase Dehydration followed by hydration
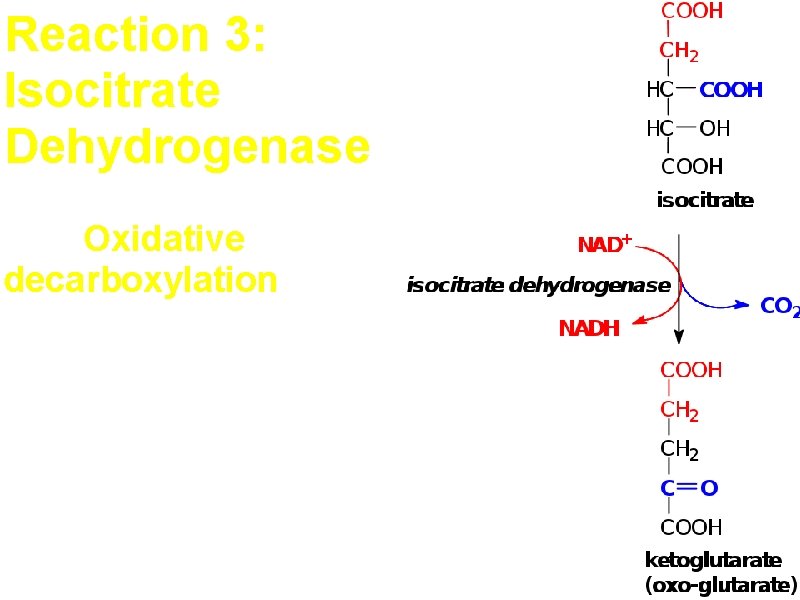
Reaction 3: Isocitrate Dehydrogenase First Oxidative decarboxylation Allosteric enzyme -ATP, NADH
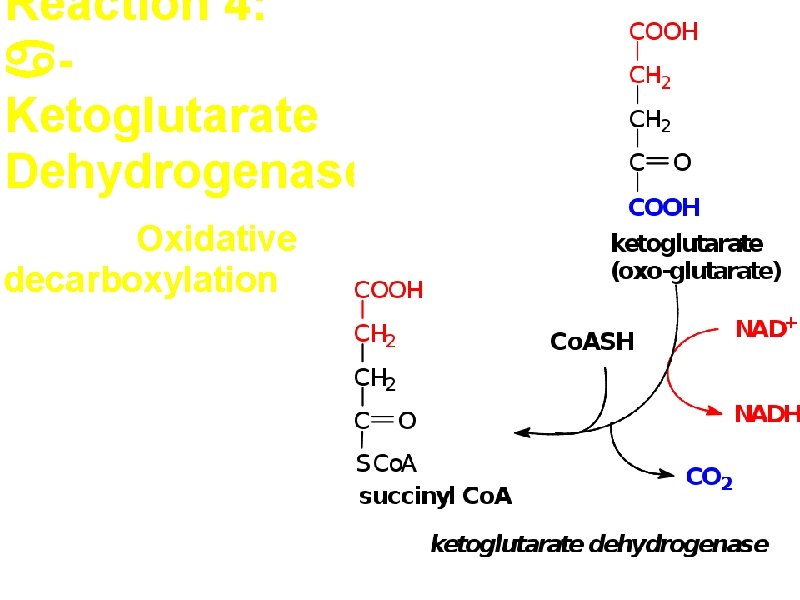
Reaction 4: Ketoglutarate Dehydrogenase Second Oxidative decarboxylation

Ketoglutarate dehydrogenase complex is very similar to the pyruvate degydrogenase complex. allosteric inhibitor: NADH, succinyl. Co. A, ATP a. Five coenzymes used – • TPP, Co. ASH, Lipoic acid, NAD+, FAD

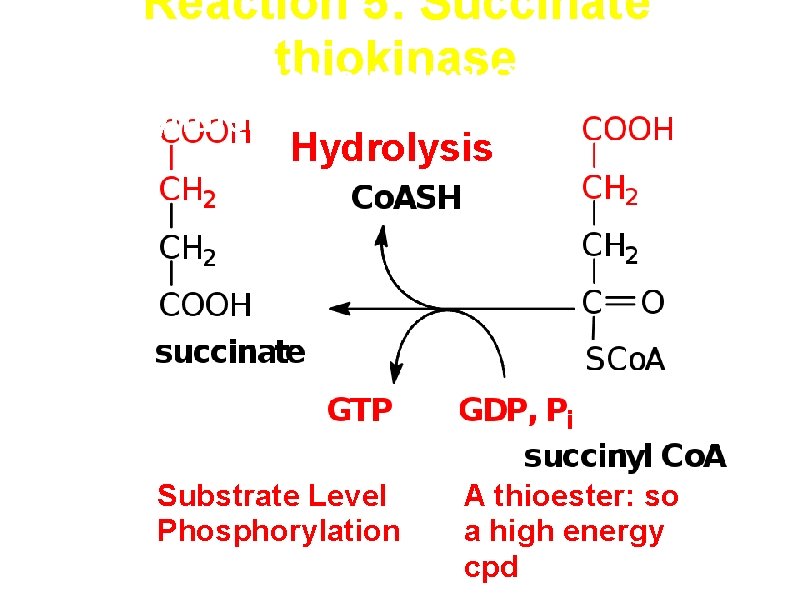
Reaction 5: Succinate thiokinase (also called succinyl Co. A synthetase) Hydrolysis Substrate Level Phosphorylation A thioester: so a high energy cpd
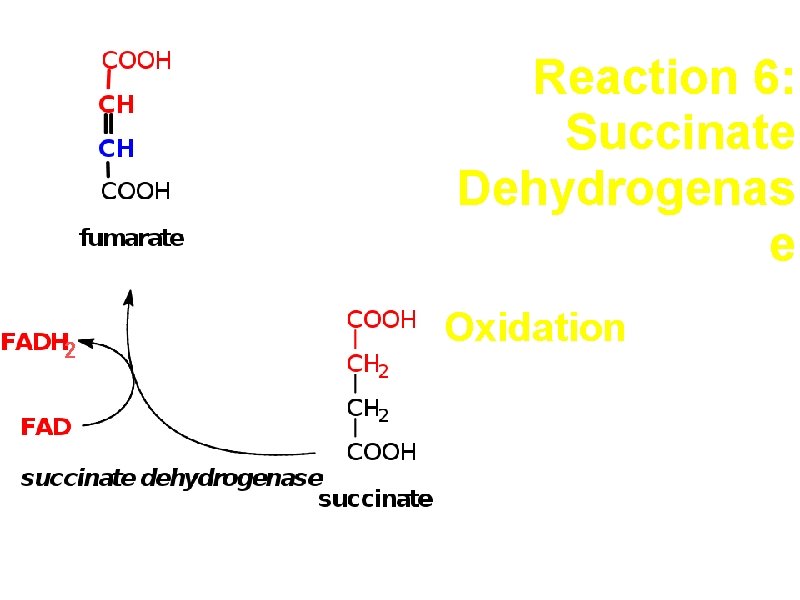
Reaction 6: Succinate Dehydrogenas e 2 Oxidation e- carrier is FAD

Succinate Dehydrogenase a. Part of electron transport chain in the inner membrane of mitochondria. b. Removal of H across a C-C bond is not sufficiently exergonic to reduce NAD+, but it does yield enough energy to reduce FAD. c. Malonate is a competitive inhibitor
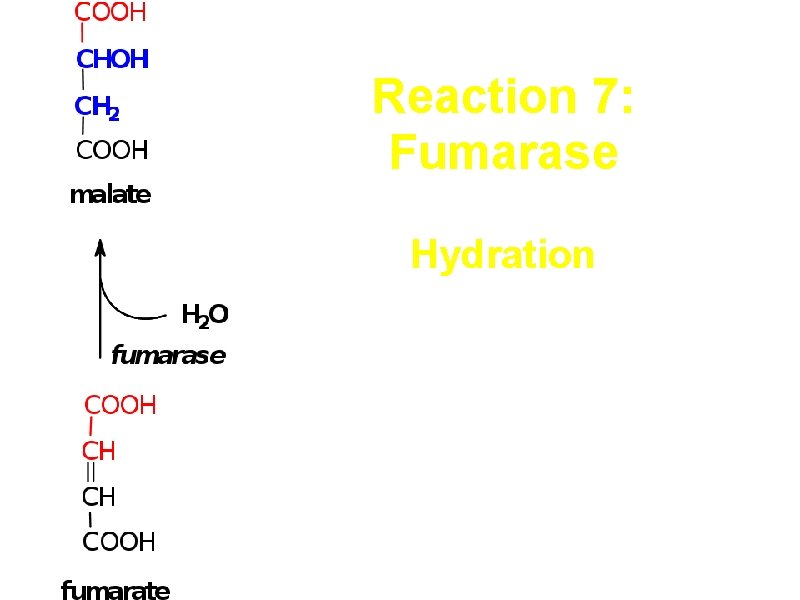
Reaction 7: Fumarase Hydration trans-addition of the elements of water across the double bond, forms L-malate
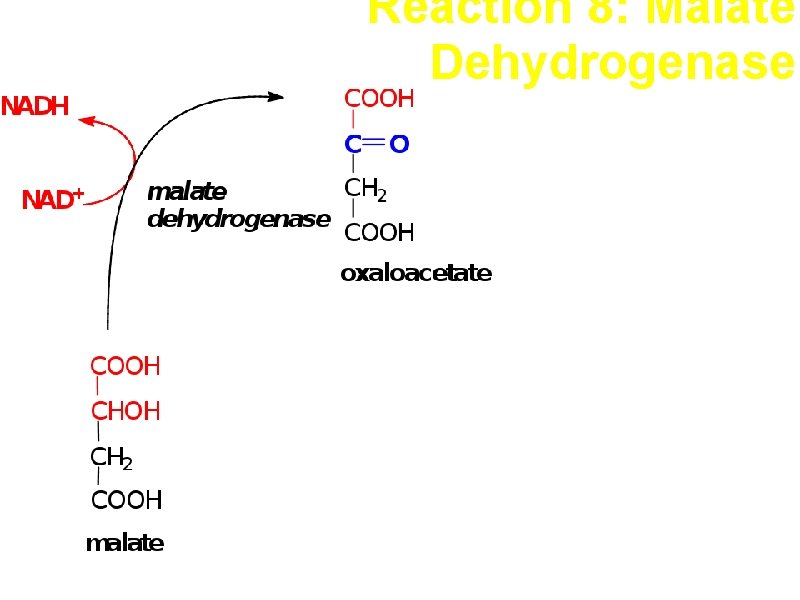
Reaction 8: Malate Dehydrogenase Oxidation This and the previous two reactions form a reaction triad +
TCA Cycle Summary 1 acetate through the cycle produces 2 CO 2, 1 GTP, 3 NADH, 1 FADH 2

Aerobic Nature of the Cycle NADH and FADH 2 must be reoxidized by the electron transport chain. Succinate Dehydrogenase is part of electron transport chain in the inner membrane of mitochondria.
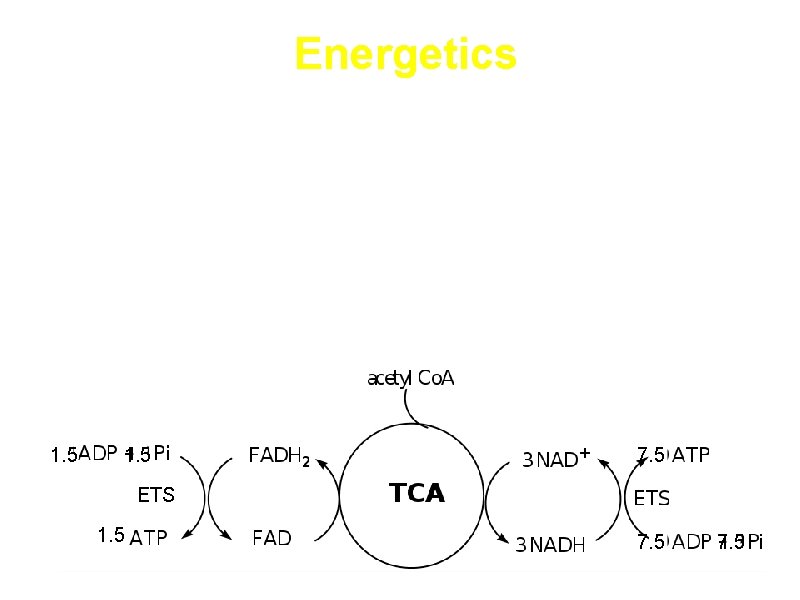
Energetics a. Energy is conserved in the reduced coenzymes NADH, FADH 2 and one GTP b. NADH, FADH 2 can be oxidized to produce ATP by oxidative phosphorylation 1. 5 7. 5 ETS 1. 5 7. 5
ATP generated by the cycle 3 NADH 3 NAD+ ETS 3*2. 5=7. 5 ATP FADH 2 FAD ETS 1. 5 ATP Substrate level phosphorylation 1 GTP 10 ATP Equivalents Total

ATP generated by complete oxidation of Glucose glycolysis a. 2 ATP(Substrate-level phosphorylation) 2 b. 2 NADH ( oxphos) 3 -5 2 Pyruvate a. oxidative decarboxylation 2 NADH ( oxphos) 5 2 Acetyl Co. A TCA cycle 20 6 NADH 2 FADH 2 2 GTP total 30 -32 ATP CO 2 5 -7 ATP 25 ATP

The Fate of Carbon in TCA
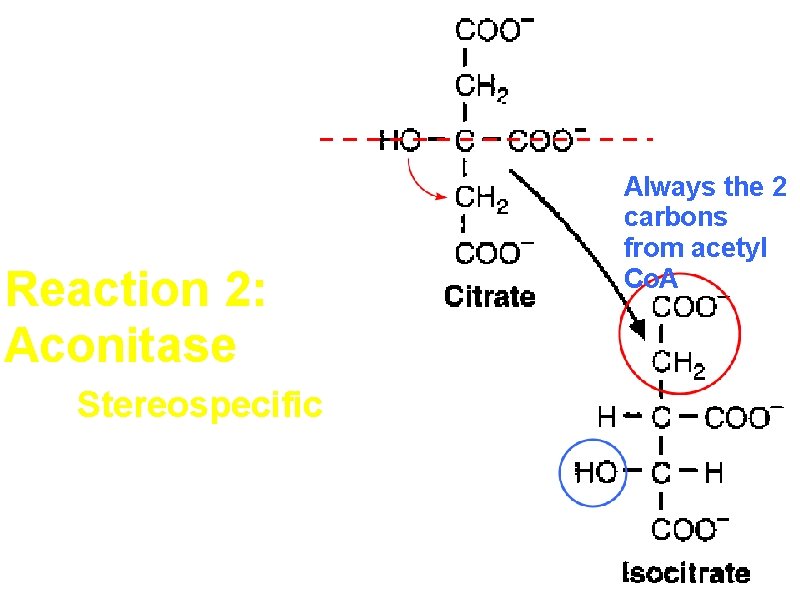
Reaction 2: Aconitase Stereospecific A symmetrical compound is converted to a chiral compound Always the 2 carbons from acetyl Co. A
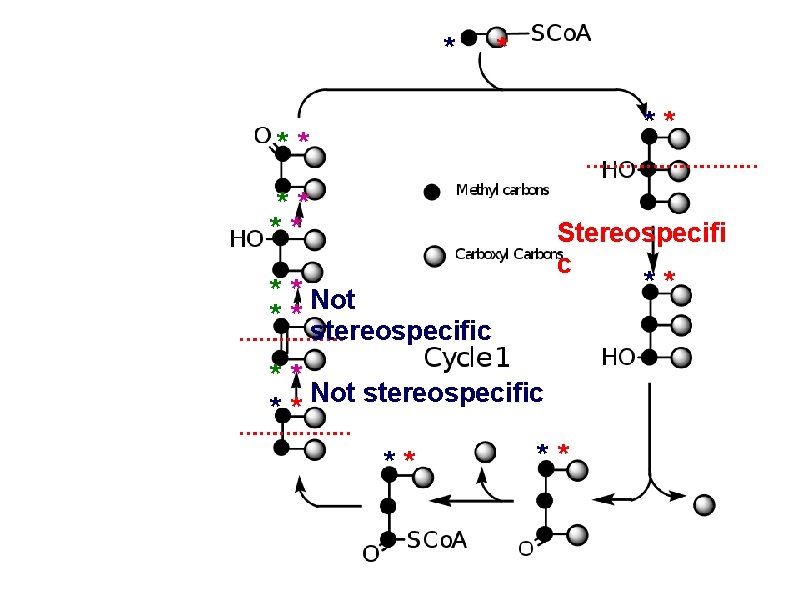
How much radioactivity has been lost after one turn? * * ** ** Zero Stereospecifi c ** * * Not * * stereospecific ** Not stereospecific ** ** **
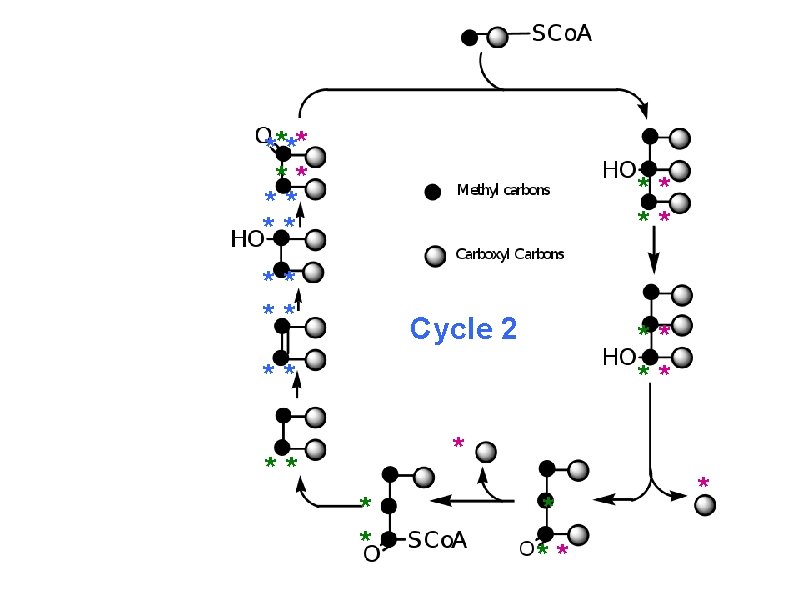
How much radioactivity has been lost after 2 turns? 50% **** ** Cycle 2 ** ** ** * * * ** *

a. Carboxyl C of acetate turns to CO 2 only in the second turn of the cycle b. Methyl C of acetate survives two cycles completely, but half of what's left exits the cycle on each turn after that.

Regulation of the TCA Cycle Again, 3 irreversible reactions are the key sites a. Citrate synthase - regulated by availability of substrates - acetyl-Co. A and oxaloacetate, citrate is a competitive inhibitor; Allosteric: - NADH , ATP, succinyl-Co. A a. Isocitrate dehydrogenase – NADH, ATP inhibit, ADP and NAD+ Ca++ activate • -Ketoglutarate dehydrogenase - NADH and succinyl-Co. A inhibit, AMP Ca++activate
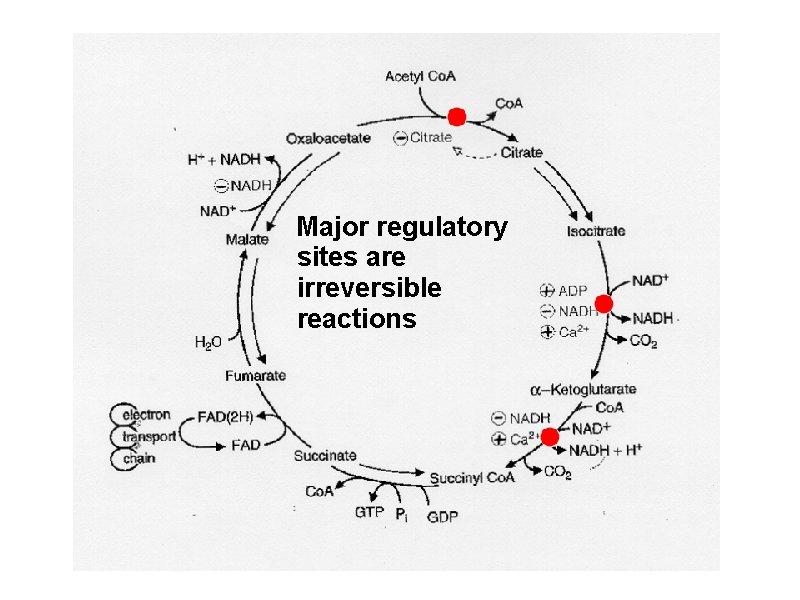
Major regulatory sites are irreversible reactions

Anaplerotic reactions a. Anaplerotic (filling up) reactions replenish citric acid cycle intermediates b. Amphibolic Nature of TCA Cycle means it both Anabolic and Catabolic. TCA cycle provides several of Intermediates for Biosynthesis
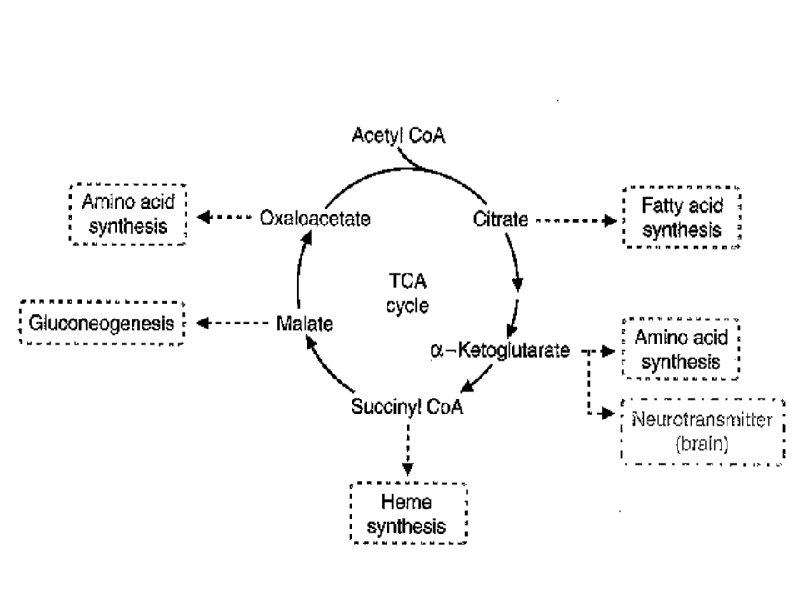

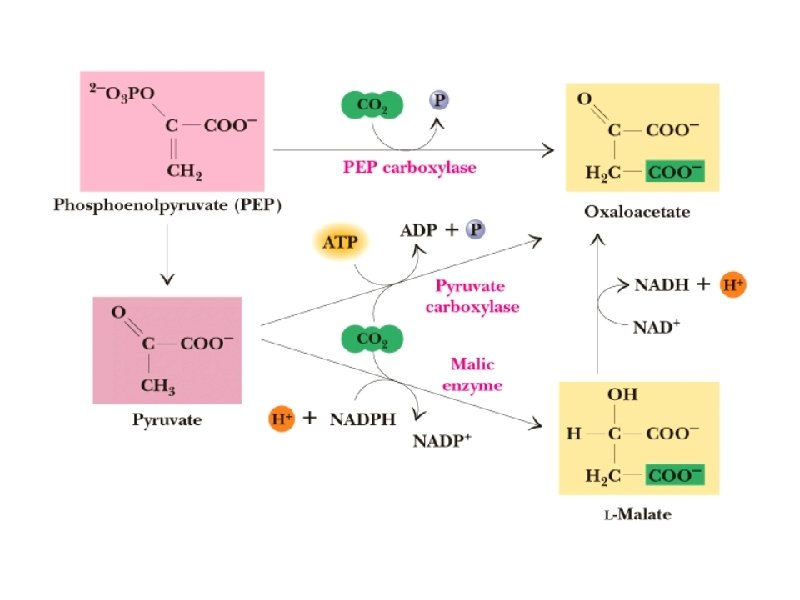
Anaplerotic reactions a. PEP carboxylase - converts PEP to oxaloacetate , Anaplerotic reaction in plants and bacteria b. Pyruvate carboxylase - converts pyruvate to oxaloacetate, a major anaplerotic reaction in mammalian tissues c. Malic enzyme converts pyruvate into malate

The Glyoxylate Cycle An Anabolic Variant of the Citric Acid Cycle for plants and bacteria a. Acetate-based growth - net synthesis of carbohydrates and other intermediates from acetate - is not possible with TCA • Glyoxylate cycle offers a solution for plants and some bacteria and algae • The CO 2 -evolving steps are bypassed an extra acetate is utilized • Isocitrate lyase and malate synthase are the short-circuiting enzymes

Acetyl-Co. A Citrate Oxaloacetate Isocitrate Malate sythase Acetyl-Co. A Glyoxalate Isocitrate lyase Succinate



Home work a. Starting with pyruvate labelled with 13 C on the keto-carbon, where will this isotopic label end up in oxaloacetate after one round of the citric acid cycle?

Briefly describe a. The reaction catalyzed by the pyruvate dehydrogenase complex in carbohydrate metabolism: • One advantage of a multienzyme complex • explain how pyruvate dehydrogenase is regulated. (Graphs of enzyme activity would be cool)! • What effect would increasing expression of the PDH kinase gene have on carbohydrate metabolism?

a. If you add pyruvate labeled in the carboxylate group to actively metabolizing eukaryotic cells under aerobic conditions, under what conditions will that labeled carbon enter the citric acid cycle?

a. What is the primary regulatory site in glycolysis? Explain the role of energy charge in controlling the enzyme. b. Name other sites for regulation in glycolysis.
- Slides: 46