Adventitious Agent Testing of Novel Cell Substrates for

Adventitious Agent Testing of Novel Cell Substrates for Vaccine Manufacture Arifa S. Khan, Ph. D. Division of Viral Products Office of Vaccines Research and Review CBER, FDA November 16, 2005 2/1/2022 1

Topics covered in the presentation: § Cell substrates used in U. S. licensed vaccines and current recommended testing for adventitious agents § Risks of viral contamination in vaccines § Special safety concerns and challenges for testing novel cell substrates, especially tumorigenic cells § Regulation of therapeutic products made in tumorigenic cell substrates § Testing recommendations to evaluate the safety of novel/tumorigenic cell substrates such as MDCK cells 2/1/2022 2

Types of Cell Substrates Used in Current U. S. Licensed Viral Vaccines § Primary Cells or Tissues: used without passage in tissue culture § Diploid Cells: cells with a finite lifespan and passage in tissue culture § Continuous Cell Lines: immortal, neoplastic cells with unrestricted passage in tissue culture -Non-tumorigenic 2/1/2022 3
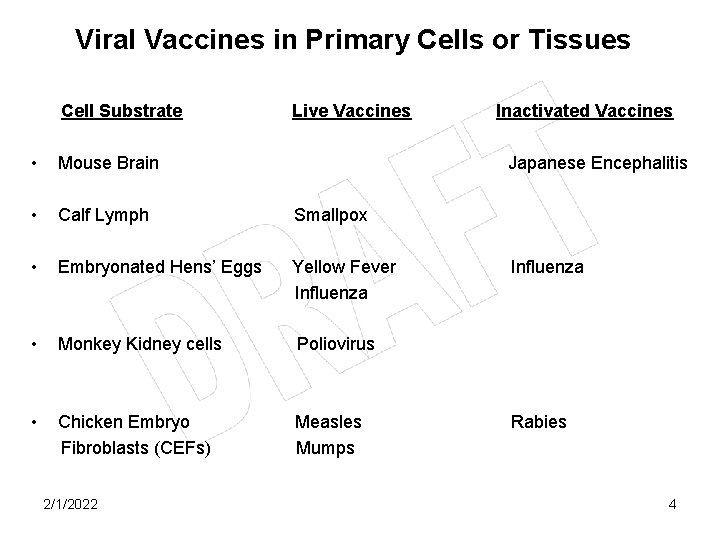
Viral Vaccines in Primary Cells or Tissues Cell Substrate Live Vaccines • Mouse Brain • Calf Lymph Smallpox • Embryonated Hens’ Eggs Yellow Fever Influenza • Monkey Kidney cells Poliovirus • Chicken Embryo Fibroblasts (CEFs) Measles Mumps 2/1/2022 Inactivated Vaccines Japanese Encephalitis Influenza Rabies 4

Viral Vaccines in Diploid Cells Cell Substrate Monkey • FRh. L-2 Human • WI-38 • MRC-5 2/1/2022 Live Vaccines Rotavirus Inactivated Vaccines Rabies Rubella Adenovirus Varicella Poliovirus Hepatitis A Rabies 5

Viral Vaccines in Continuous Cell Lines Cell Substrate Monkey • Vero 2/1/2022 Live Vaccines Poliovirus (Europe) Inactivated Vaccines Poliovirus 6

Current Adventitious Agent Testing 1: Detection of Non-viral Agents § Bacterial and Fungal Sterility: 21 CFR 610. 12 § Mycoplasma (& Spiroplasma): -agar and broth media culture method (21 CFR 610. 30) -indicator cell culture procedure -PCR § Mycobacterial Testing: -culture method [pre-1997 CFR 650. 13(b)(2)] -guinea pig test -PCR 1 References 2/1/2022 listed in the last two slides of presentation 7

Current Adventitious Agent Testing 1: General Virus Detection IN VITRO ASSAYS: § § Direct examination of cells by transmission electron microscopy (TEM) Inoculation of 3 monolayer culture types and observation for cytopathic effect, hemagglutination or haemadsorption. Cells include: same species, human diploid cells, and monkey kidney cells: Human diploid cells: herpesviruses, adenoviruses, coronoviruses, reoviruses, alphaviruses, rubella, flaviviruses, rabies, enteroviruses, certain hepatitis A viruses, poliovirues, coxsackie B virus, echovirus, rhinoviruses, orthomyxoviruses, paramyxoviruses and simian CMV. Monkey kidney cells: same as human cells (except herpesviruses, coronoviruses and hepatitis A) PLUS herpes simplex virus, poxviruses, polyomaviruses, rotaviruses, vesiculoviruses, filoviruses, influenza viruses, bunyaviruses, arenaviruses, rhinoviruses, 2/1/2022 8 adenoviruses (some strains) and simian herpes B virus.

Current Adventitious Agent Testing 1: General Virus Detection IN VIVO ASSAYS: § Adult mice: LCMV, rhabdoviruses (including rabies) togaviruses (LDV) § Suckling mice: Coxsackie A viruses, Coxsackie B viruses, arboviruses, herpes simplex type 1 and 2, rhabdoviruses (including rabies), togaviruses (LDV), Junin, Herpes B. § Embryonated hens’ eggs: Herpes simplex type 1 and 2, rhabdoviruses (including rabies), herpes B, Mumps, influenza, parainfluenza (types 1, 2, and 3), vaccinia 2/1/2022 9

Current Adventitious Agent Testing 1: Detection of Known Viruses Based Upon Source Materials § Raw materials: Sera -bovine viruses; equine viruses Trypsin -porcine viruses; bovine viruses Detection per 9 CFR 113. 47 and 9 CFR 113. 53 § Cell substrate or passage history: Rodent -retroviruses, MAP/RAP/HAP, LCMV Simian -retroviruses, polyomaviruses Human -retroviruses, hepatitis viruses, enteroviruses, papillomaviruses, polyomaviruses, adenoviruses, herpesviruses Detection by Cell Culture assays, TEM, PERT or PCR assays 2/1/2022 10

Comprehensive testing regimens for detection of adventitious agents in vaccine cell substrates are designed to minimize the risk of virus contamination in vaccines and assure product safety. However, albeit rare, unexpected virus contamination has occurred in vaccine history. 2/1/2022 11

History of Contamination in Viral Vaccines 2 § Incomplete inactivation of virus ú ú § Early rabies vaccine Fortaleza incident (rabies) Cutter incident (polio) FMDV vaccine Viruses present in reagents ú Yellow fever vaccine contaminated with Hepatitis B virus present in sera added as stabilizer 2/1/2022 12

History of Contamination of Viral Vaccines 3, 4 § Naturally-occurring viruses in cell substrate (due to species of origin) Vaccines Cell Substrate Virus Poliovirus vaccines (1955 -1963) primary rhesus monkey kidney cells SV 40 Adenovirus vaccine primary rhesus monkey kidney cells SV 40 Yellow fever vaccine (early 1940 s) non-SPF eggs ALV 2/1/2022 13

Safety Concerns Related to Retroviruses 5 § Retrovirus infection results in stable integration of viral sequences into the host DNA, which persist for the life of the host. § Retrovirus-induced diseases can occur in animals and in humans after a prolonged incubation period. X Adverse Events Due to Retroviruses in Biologics 6, 7: - HIV-1 in Factor VIII [1980 s] Immune Dysfunction AIDS in Hemophiliacs - Murine Retrovirus Vector [2002] Insertional Mutagenesis T-cell Leukemias in X-SCID therapy 2/1/2022 14

Lessons from the Past § There is need for continued vigilance in adventitious virus testing of vaccines and other biological products: ú New testing strategies for detection of unknown adventitious viruses in novel cell substrates. ú Sensitive assays for detection of unknown, oncogenic viruses in tumorigenic cell substrates. 2/1/2022 15

Concerns Regarding Novel Cell Substrates 8 -13, 19 § Insufficient information regarding health of the tissue donor § Lack of sufficient knowledge of viruses in donor species § Lack of sufficient documentation regarding cell passage history or exposure to biological materials: ú propagation in different labs • He. La cell contamination: Hep 2, D 96, FLA ú biological reagents used (sera, trypsin, others) • BVDV in RK-13 cells • Bovine polyoma virus in FRh. K cells ú other cell lines or viruses grown at same time • Retrovirus (type D) in He. La cells • SMRV in Namalwa cells ú exposure to mice in facility • MVM in CHO-K 1 and BHK-21 cells 2/1/2022 16

Concerns Regarding Tumorigenic Cell Substrates § Rapid cell growth might result in: ú ú § increased susceptibility for virus replication provide broader host range to different viruses activation of endogenous virus activation of latent virus Viruses may be involved in tumor phenotype ú Human papillomaviruses: • HPV-18 in He. La cells • HPV-16 in Si. Ha cells ú Human herpesviruses: • HHV-8 in BCP-1 • EBV in Raji cells • EBV in Namalwa cells ú Endogenous retroviruses in CHO and BHK cells 2/1/2022 17

FDA Experience with Tumorigenic Cells and Viruses in Therapeutic Products § Namalwa cells for interferon ú EBV genome naturally present due to derivation from Burkitt’s lymphoma ú SMRV contaminant due to lab passage § CHO cells for many products including tissue plasminogen activator, recombinant Factor VIII, recombinant hepatitis B vaccine § BHK cells for recombinant Factor VIIa ú Endogenous retroviruses produced spontaneously from cells ú Minute virus of mice due to contamination in fermenters 12 § 293 HEK cells for Drotrecogin alfa (activated) ú Adenovirus 5 DNA sequences present (partial) 2/1/2022 18

FDA Experience with Tumorigenic Cells and Viruses in Therapeutic Products § Murine Hybridoma cells (myeloma, NS 0 and SP 2/0) for monoclonal antibodies ú Naturally-occurring • murine viruses (LCMV, MVM, Sendai virus, EDIM, LDH) • murine retroviruses ú Due to raw materials • BVDV • Porcine parvovirus 2/1/2022 19

Tumor Cells Used in Inactivated Vaccines § He. La Cells (inadvertent use in 195914) ú Experimental Adenovirus Vaccines in 6 adult volunteers ú Inactivated poliovirus vaccine in 144 people § BHK-21 cells for JEV vaccine (China 15) § BHK-21 cells for veterinary vaccines 1960 s – 1980 s (FMDV and rabies 16) 2/1/2022 20

Regulation of Tumorigenic Cells in Biologics Accepted for human use based upon: § Advantages of using the cell line outweighs its tumorigenicity concerns § Development of sensitive assays for specific agents of concerns ú 324 K cell-based infectivity assay for MVM ú PERT assay for retroviruses § Viral clearance studies ú extensive cell bank and product characterization (different production stages; lot-to-lot testing) ú incorporation of orthogonal virus inactivation/removal steps in product manufacture 2/1/2022 21

Influence of Viral Clearance Steps in Vaccine Manufacture 17 § Evaluation and characterization of virus clearance (removal and/or inactivation procedures) from unprocessed bulk assures that the product is free of virus contamination by: ú evaluating processes for their ability to clear viruses that are known to be present in the cell substrate ú estimating the robustness of the process by characterizing the clearance of “model” viruses 2/1/2022 22

Viral Clearance Studies: Selection of Model Viruses 17 § Process evaluation of viral clearance using “relevant” or specific “model” viruses ú § selected based upon what is present or potential contaminant e. g. murine retroviruses for rodent cell substrates Process characterization of viral clearance using nonspecific model viruses (generally assessed using ate least 3 different viruses with differing characteristics) ú viruses that display a significant resistant to physical and/or chemical treatments ú viruses that display a wide range of physical and biololgoical properties: • • • 2/1/2022 small, nonenveloped viruses e. g. Polyomavirus medium-to-large enveloped DNA virus e. g. Herpesvirus medium-to-large enveloped RNA virus e. g. Retrovirus 23

Viral Clearance Studies: Quantitation of Virus Reduction Factor 17 § The virus reduction factor (Ri) of an individual purification or inactivation step is defined as the log 10 of the ratio of the virus load in the prepurification material and the virus load in the post-purification material which is ready for use in the next step of the process. 10 Ri = (v’) (10 a’) / (v”) (10 a”) (this formula takes into account both the titers and the volumes of the materials before and after the purification step). § The overall reduction factor for a complete production process is the sum logarithm of the reduction factors of the individual steps. It represents the logarithm of the ratio of the virus load at the beginning of the first process clearance step and at the end of the last process clearance steps. 2/1/2022 24

Viral Clearance Studies: Viral Safety Evaluation 17 § When expected or known viruses are present, the number of virus particles in the starting material can be estimated and a specific clearance value may be used to calculate a specific safety risk (e. g. for rodent cells a 6 log reduction of virus above the starting value is recommended) § In the case of unknown potential contaminants, the goal should be to provide sufficient virus clearance that can assure that the product is free of virus contamination. Determination of the virus clearance factor needs consideration of some of the limitations of the virus clearance studies § The summation of the reduction factors of multiple steps may overestimate the true potential for virus elimination. § Reduction factors are normally expressed on a logarithmic scale, which implies that, while residual virus infectivity will never be reduced to zero, it may be greatly reduced mathematically § The behavior of the tissue culture grown model viruses used in the virus clearance studies may be different from that of the native virus (in the case of unknown viruses, the model viruses are selected based upon best representation) 2/1/2022 25

General Approaches for Viral Safety of Biologics § Qualification of cell banks and raw materials ú Extensive testing of cell substrate and use of raw materials certified or tested to be free of detectable virus § Process Validation ú Design an efficient process • to avoid risk of contamination • eliminate or reduce potential virus load • inactivate potentially contaminating virus § In-Process Testing ú Develop a comprehensive testing plan to evaluate production lots for known and novel viruses 2/1/2022 26

Potential Agents of Concern in MDCK Cells Due to Naturally-Occurring Viruses Different Families of Dog Viruses are prevalent Worldwide: Virus family Paramyxoviridae Parvoviridae Adenoviridae Rhabdoviridae Coronaviridae Herpesviridae Papillomaviridae* Retroviridae* Disease Canine distemper Parainfluenzavirus 2 infection Parvovirus infection Infectious canine hepatitis Canine laryngotracheitis Rabies Canine coronovirus infection Hemorrhagic disease of pups Canine papillomatosis Canine leukemia (lentivirus) Lymphosarcomas (type C) Persistence + (sometimes) + + ? + + *Oncogenic Viruses 2/1/2022 27

Additional Agents of Potential Concern in MDCK Cells Due to Cell Susceptibility • • Influenza virus Vesicular stomatitis virus (Indiana) Vaccinia virus Coxsackie B 5 virus Vesicular exanthema of swine Reovirus type 2 and 3 (persistence) Bluetongue virus (persistence) LCMV (persistence) 2/1/2022 28

Qualification of MDCK Cells § Current Established Assays (generally validated) ú PCR assays (specific and generic) ú Infectivity assays (in vitro, in vivo) ú Western blot, ELISA, IFA § Novel Assays with broad detection for unknown viruses of different families including non-oncogenic and oncogenic (not validated) ú Induction of latent and endogenous viruses by chemical treatment of cell substrate ú Assays for detection of oncogenic viruses by inoculation of cell lysate (and DNA) into newborn rodents (3 species) 2/1/2022 29
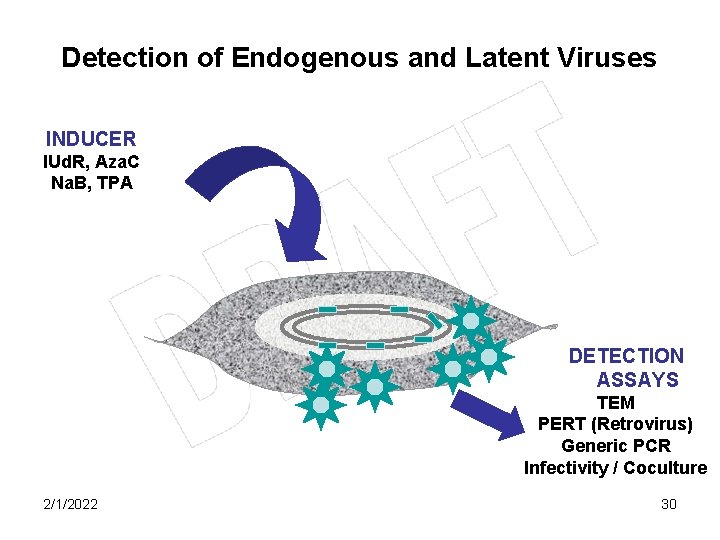
Detection of Endogenous and Latent Viruses INDUCER IUd. R, Aza. C Na. B, TPA DETECTION ASSAYS TEM PERT (Retrovirus) Generic PCR Infectivity / Coculture 2/1/2022 30

Chemical Inducers are Potent Virus Activators 18, 20 -23 § 5’-iodo-2’-deoxyuridine (IUd. R) and 5 -azacytidine (Aza. C) are known inducers of endogenous retroviruses from cells of different species including avian and mammalian (ranging from mouse to human). § 12 -O-tetradecanoly phorbol-13 -acetate (TPA) and sodium butyrate (Na. B) can induce various latent DNA viruses including herpesviruses and some retroviruses (HIV-1) 2/1/2022 31
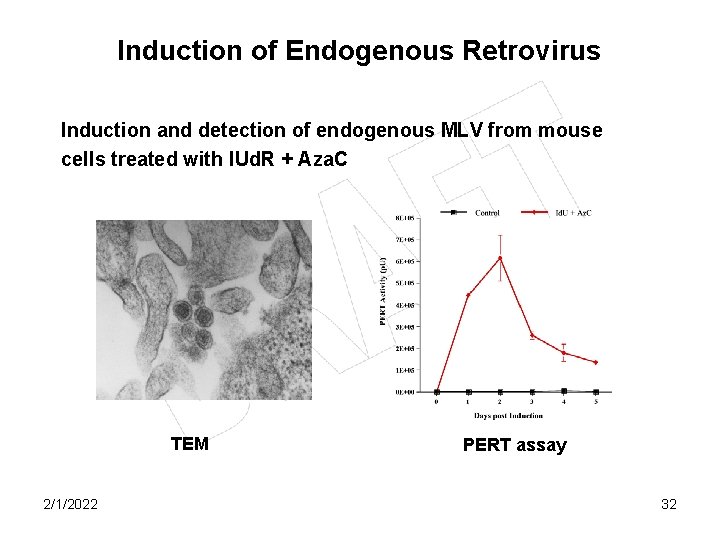
Induction of Endogenous Retrovirus Induction and detection of endogenous MLV from mouse cells treated with IUd. R + Aza. C TEM 2/1/2022 PERT assay 32
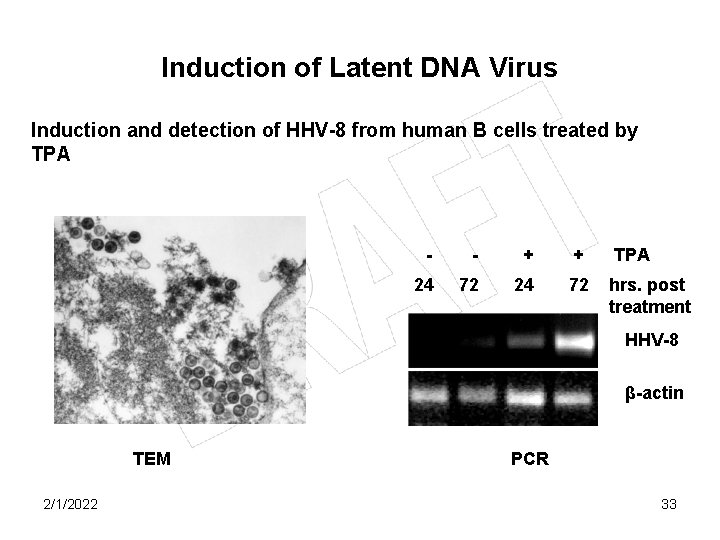
Induction of Latent DNA Virus Induction and detection of HHV-8 from human B cells treated by TPA - - + + TPA 24 72 hrs. post treatment HHV-8 β-actin TEM 2/1/2022 PCR 33

In Vivo Assays for Oncogenic Viruses of concern: = Gamma-Herpesviruses, Polyomaviruses, Papillomaviruses, Adenoviruses, certain Poxviruses, Retroviruses § 3 species assay (cell lysates [and DNA] from 107 cells equivalent; <4 day-old animals): ú newborn hamster ú newborn nude mice ú newborn rats 5 months 2/1/2022 34

Detection of Oncogenic Viruses in Cell Lysate 24, 25 • 1911, Peyton Rous discovered the first avian retrovirus (Rous sarcoma virus) by injecting filtered extract into chickens • 1950 -1953, Gross discovered Gross leukemia virus and polyoma virus when he obtained tumors in newborn C 3 H mice that were inoculated with cell-free material from Ak mouse leukemia cells. • Extracts from a variety of different transplantable sarcomas and carcinomas injected into newborn mice ----- leukemias --- Murine Retroviruses: Graffi, 1955, Moloney, 1960, Friend 1957, and Rauscher 1962. • 1957 -1958, Bernice Eddy used extracts of primary rhesus monkey kidney cells to induce tumors in newborn hamsters (due to SV 40) 2/1/2022 35

In Vivo Detection of Oncogenic Viruses 26 NB Syrian Hamster NB Mice NB Rats Murine leukemia/sarcoma + + + Hamster Polyoma + - - Bovine Papilloma + + + Human Adeno 12, 18, 21, 31 Adeno 3, 7, 14, 16, 21 Adeno 9 Adeno: SA 7, CELO, canine + + - Papilloma 16, 18 polyoma (SV 40, JCV, BKV) + - - Herpes 1, 2 HHV-8 + - + - 2/1/2022 36

Extended Adventitious Agent Testing of Novel Cell Substrates (MDCK Cells for Inactivated Flu Vaccine) CELL BANK TESTING § Testing for viruses based upon species of origin § Testing for viruses that could potentially be present based upon susceptibility of cells § Testing for rodent viruses due to extensive and unknown passage history of cells in different laboratories § Testing for viruses due to raw materials used in history of cell propagation (equine viruses, bovine viruses, porcine viruses) § Testing for endogenous and latent viruses by chemical Induction with subsequent virus detection using broad and specific detection assays § Testing for potential unknown oncogenic viruses due to tumorigenicity of cells ú cell lysate and DNA testing in 3 rodent species 2/1/2022 37

Additional Safety Steps in Use of Novel Cell Substrates (MDCK Cells for Inactivated Flu Vaccine) § IN PROCESS TESTING ú Evaluation for presence of any potential viruses • virus seed • biological raw materials § VIRAL CLEARANCE STUDIES ú Evaluation of inactivation using different viruses ú Evaluation of virus removal during the manufacturing process ú Estimation of virus reduction using appropriate model viruses (spiking studies) 2/1/2022 38
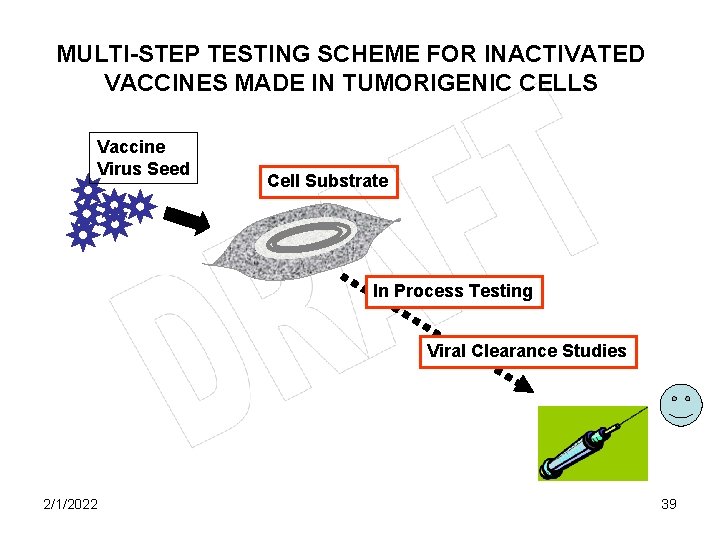
MULTI-STEP TESTING SCHEME FOR INACTIVATED VACCINES MADE IN TUMORIGENIC CELLS Vaccine Virus Seed Cell Substrate In Process Testing Viral Clearance Studies 2/1/2022 39
References 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. FDA Points to Consider in the Characterization of Cell Lines Used to Produce Biologicals (1993) F. Houraud, 1990. Dev. Biol. Standard. Vol. 75, p. 3 -7 J. S. Robertson, 2003. Dev. Biol. Vol. 113, p. 73 -77; B. H. Sweet and M. R. Hilleman, 1960. P. S. E. B. M. Vol 105. p. 420 -427 Ono, 1988. Dev. Biol. Stand. Vol. 70, p. 69 -81 C. J. M. Melief and J. Goudsmith 1986. Vox Sanguinis 50, 1 -11 A. Fischer et al. , 2004. NEJM 350, 913 -922 G. A. Erickson et al. , 1990. Dev. Biol. Stand. Vol. 75 p. 173 -175 R. L. Levings and S. J. Wessman, 1990. Dev. Biol. Stand. Vol. 75 p. 177 -181 R. J. Hay, 1990. Dev. Biol. Stand. Vol. 75 p. 193 -204 P. Minor. Develop. Biol. Standard. 1996. Vol. 88 p. 25 -29 R. L. Garnick, Develop. Biol. Standard. 1996. Vol. 88 p. 49 -56 Nicklas et al. , 1993, Lab. Animal Science L. Hayflick. 1988. Dev. Biol. Std. Vol. 70 p. 11 -26 G. Peiwei, D. Zhifen and W. Zhongquan. 1988. Dev. Biol. Std. Vol. 70 p. 223 -226 J. C. Petricciani 1988 Dev. Biol. Std. Vol. 70 p. 3 -10 2/1/2022 40

References (cont. ) 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. International Conference on Harmonisation: Q 5 A Viral Safety Evaluation of Biotechnology Products Derived from Cell Lines of Human or Animal Origin, 1998. AS Khan and JF Sears. 2001. Dev. Biol. Stand. Vol. 106. p. 387 -393 D. Onions. Dev. Biol. 2004 vol. 118. p. 155 -163 Weiss, Teich, Varmus, Coffin. 1984. Endogenous viruses in RNA Tumor Viruses, 2 nd ed. Vol. 1 pp. 111109 -1203. Bedigian et al. 1978. J. Virol. 27. 313 -319 Yu et al. 1999. J. Gen. Virol. 80. 83 -90 Khan et al. 2001. Virus Res. 79. 39 -45 L. Gross. 1970 Oncogenic Viruses 2 nd ed. Eddy et al 1961. Proc Soc Expt. Biol Med 107, 191 -197. Table submitted in Briefing Package “Summary of Oncogenicity of Tumor Virues in Laboratory Rodents and Other Species”. 2/1/2022 41
- Slides: 41