Advancing Monoclonal Antibody Production with Improved Purification Technology

Advancing Monoclonal Antibody Production with Improved Purification Technology Nian Rui Qingdao Institute of Bioenergy and Bioprocess Technology, Chinese Academy of Sciences 4 th International Conference and Expo on Bioprocess & Biosystems Houston, USA, October 20 -21, 2016

Outline 1. Chromatin-directed cell culture clarification 2. Improved protein A chromatography Jun 2012 -Sep 2015 2

Introduction The standard model of protein A is simple. Ig. G binds. Contaminants do not. Load, wash, elute pure Ig. G. But in the real world, protein A does not elute pure Ig. G. Pg 3

Introduction Hypotheses to explain excess contamination include nonspecific interactions between contaminants and: the protein A ligand, the base matrix of the chromatography media, and with Ig. G. Other hypotheses have been suggested to explain how protein A chromatography promotes aggregation – or doesn’t. But all of them overlook two rogue elements in the equation. The first is Chromatin. The second is protein A itself. Pg 4

Starting from a clean slate… Chromatin from dead cells persists in cell culture harvests as heteroaggregates. Nucleosomal arrays provide their structural core. Individual nucleosomes contain 147 bp of DNA wrapped 1. 65 times around a core octamer of histones ((H 2 A, H 2 B, H 3, H 4)2). DNA and histones each contribute about 100 k. Da. Nucleosomes are linked in linear arrays by 20 -80 bp linker DNA segments associated with histone H 1. Histones are strongly hydrophobic with p. I ranges from 11 -12; DNA about 2. 6. Image modified from scbt. sastra. edu Pg 5
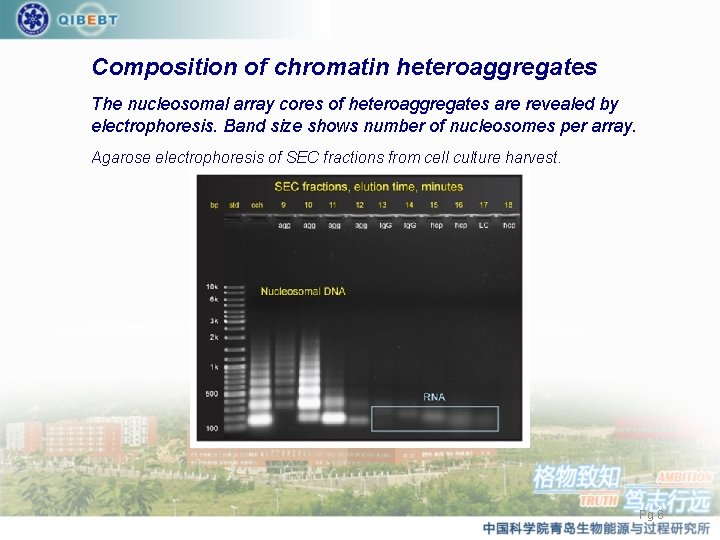
Composition of chromatin heteroaggregates The nucleosomal array cores of heteroaggregates are revealed by electrophoresis. Band size shows number of nucleosomes per array. Agarose electrophoresis of SEC fractions from cell culture harvest. Pg 6
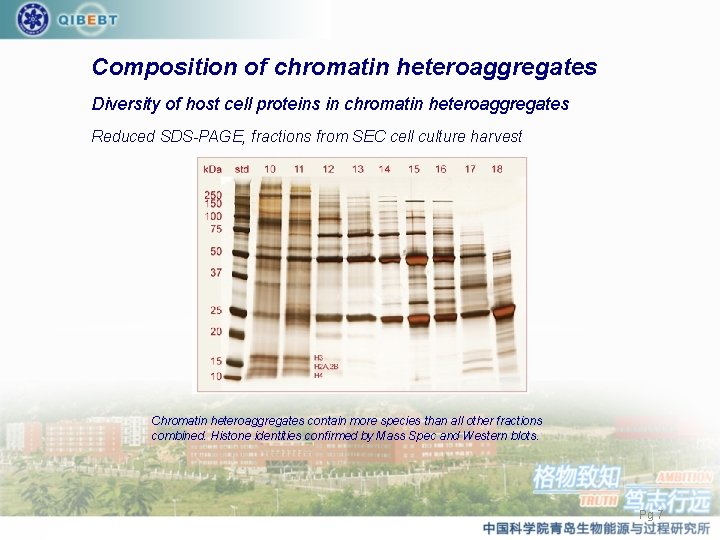
Composition of chromatin heteroaggregates Diversity of host cell proteins in chromatin heteroaggregates Reduced SDS-PAGE, fractions from SEC cell culture harvest Chromatin heteroaggregates contain more species than all other fractions combined. Histone identities confirmed by Mass Spec and Western blots. Pg 7
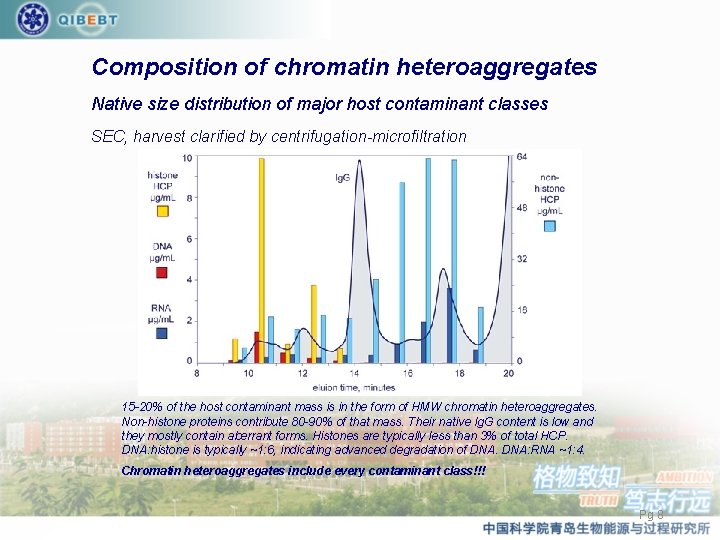
Composition of chromatin heteroaggregates Native size distribution of major host contaminant classes SEC, harvest clarified by centrifugation-microfiltration 15 -20% of the host contaminant mass is in the form of HMW chromatin heteroaggregates. Non-histone proteins contribute 80 -90% of that mass. Their native Ig. G content is low and they mostly contain aberrant forms. Histones are typically less than 3% of total HCP. DNA: histone is typically ~1: 6, indicating advanced degradation of DNA: RNA ~1: 4. Chromatin heteroaggregates include every contaminant class!!! Pg 8
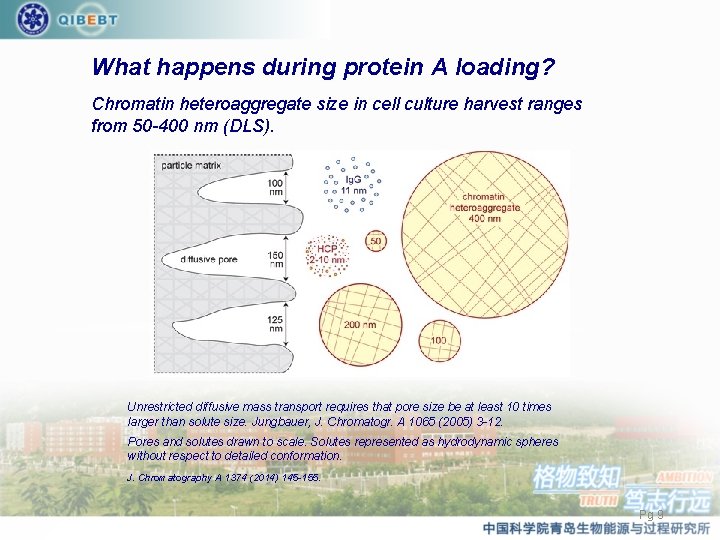
What happens during protein A loading? Chromatin heteroaggregate size in cell culture harvest ranges from 50 -400 nm (DLS). Unrestricted diffusive mass transport requires that pore size be at least 10 times larger than solute size. Jungbauer, J. Chromatogr. A 1065 (2005) 3 -12. Pores and solutes drawn to scale. Solutes represented as hydrodynamic spheres without respect to detailed conformation. J. Chromatography A 1374 (2014) 145 -155. Pg 9

What happens during protein A loading? Chromatin heteroaggregates bind to protein A. Chromatin heteroaggregates obstruct access of Ig. G to particle pores and depress dynamic binding capacity by up to 20%. Experimental data indicate that protein A binding of chromatin heteroaggregates is mediated through multipoint attachments to surface-accessible histone elements. Alkaline hydrophobic histones are a good multimodal match for the acidic hydrophobic surface of protein A. Pg 10
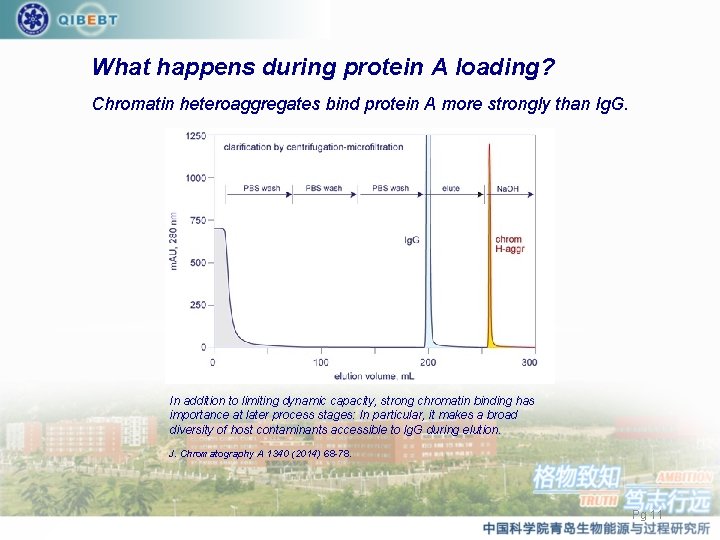
What happens during protein A loading? Chromatin heteroaggregates bind protein A more strongly than Ig. G. In addition to limiting dynamic capacity, strong chromatin binding has importance at later process stages: In particular, it makes a broad diversity of host contaminants accessible to Ig. G during elution. J. Chromatography A 1340 (2014) 68 -78. Pg 11
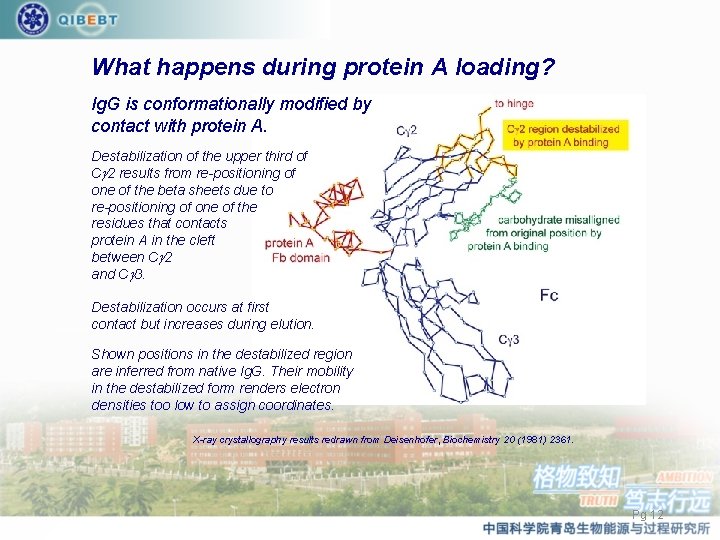
What happens during protein A loading? Ig. G is conformationally modified by contact with protein A. Destabilization of the upper third of Cg 2 results from re-positioning of one of the beta sheets due to re-positioning of one of the residues that contacts protein A in the cleft between Cg 2 and Cg 3. Destabilization occurs at first contact but increases during elution. Shown positions in the destabilized region are inferred from native Ig. G. Their mobility in the destabilized form renders electron densities too low to assign coordinates. X-ray crystallography results redrawn from Deisenhofer, Biochemistry 20 (1981) 2361. Pg 12
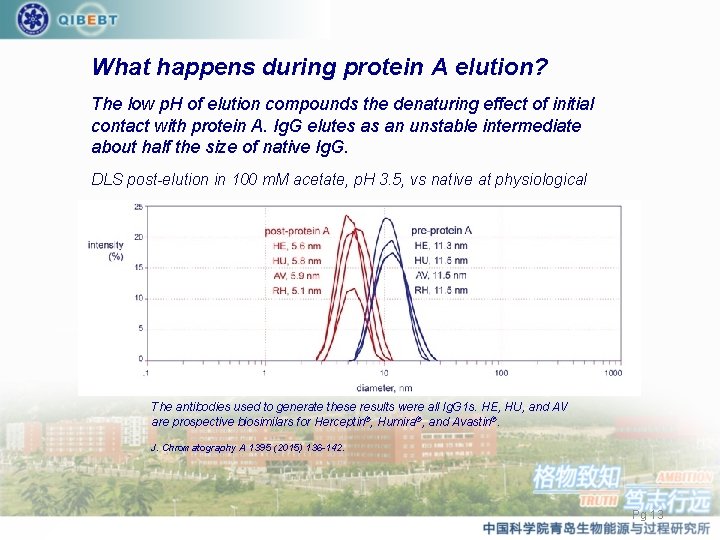
What happens during protein A elution? The low p. H of elution compounds the denaturing effect of initial contact with protein A. Ig. G elutes as an unstable intermediate about half the size of native Ig. G. DLS post-elution in 100 m. M acetate, p. H 3. 5, vs native at physiological The antibodies used to generate these results were all Ig. G 1 s. HE, HU, and AV are prospective biosimilars for Herceptin®, Humira®, and Avastin®. J. Chromatography A 1395 (2015) 136 -142. Pg 13
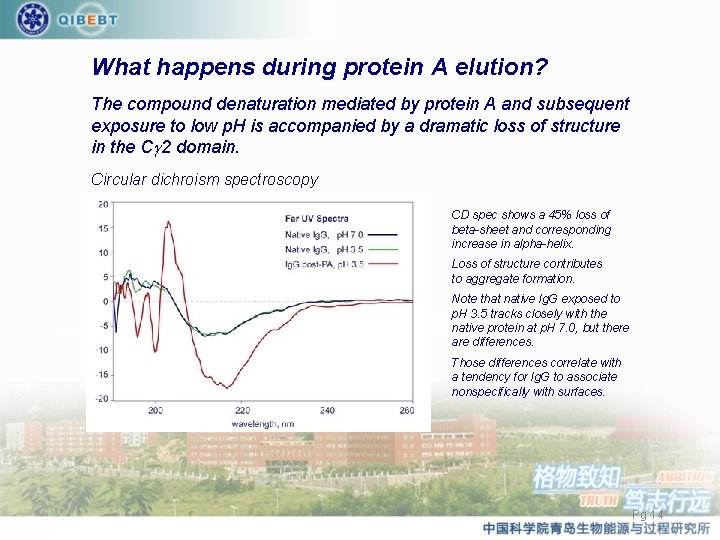
What happens during protein A elution? The compound denaturation mediated by protein A and subsequent exposure to low p. H is accompanied by a dramatic loss of structure in the Cg 2 domain. Circular dichroism spectroscopy CD spec shows a 45% loss of beta-sheet and corresponding increase in alpha-helix. Loss of structure contributes to aggregate formation. Note that native Ig. G exposed to p. H 3. 5 tracks closely with the native protein at p. H 7. 0, but there are differences. Those differences correlate with a tendency for Ig. G to associate nonspecifically with surfaces. Pg 14
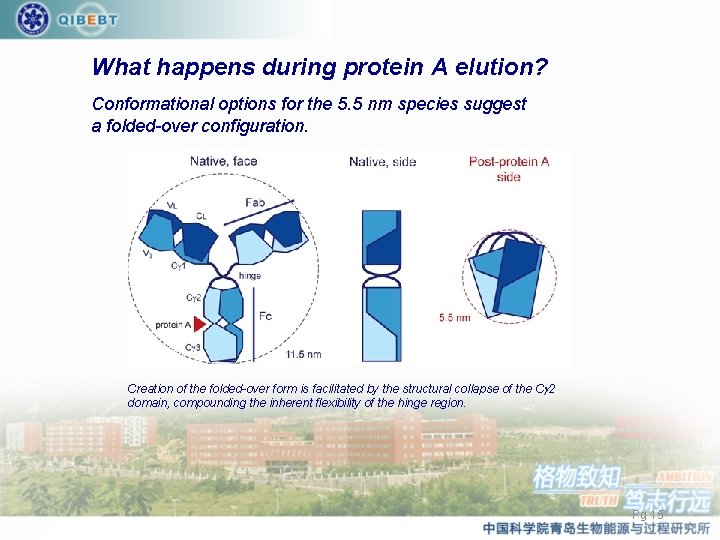
What happens during protein A elution? Conformational options for the 5. 5 nm species suggest a folded-over configuration. Creation of the folded-over form is facilitated by the structural collapse of the Cg 2 domain, compounding the inherent flexibility of the hinge region. Pg 15
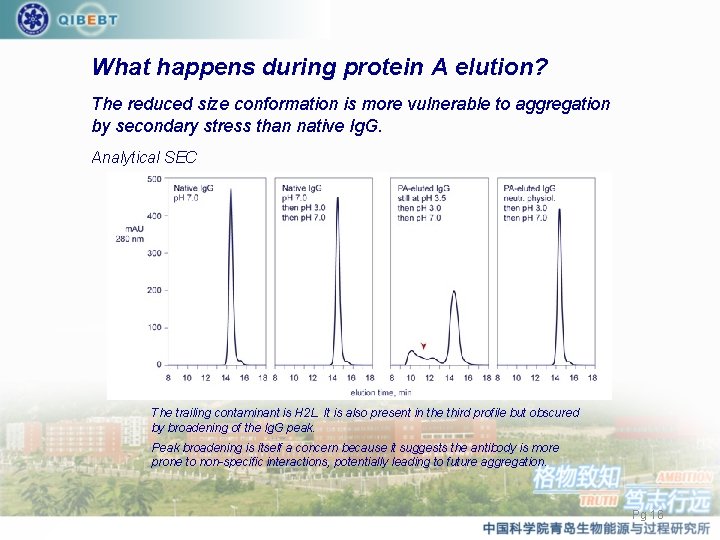
What happens during protein A elution? The reduced size conformation is more vulnerable to aggregation by secondary stress than native Ig. G. Analytical SEC The trailing contaminant is H 2 L. It is also present in the third profile but obscured by broadening of the Ig. G peak. Peak broadening is itself a concern because it suggests the antibody is more prone to non-specific interactions, potentially leading to future aggregation. Pg 16
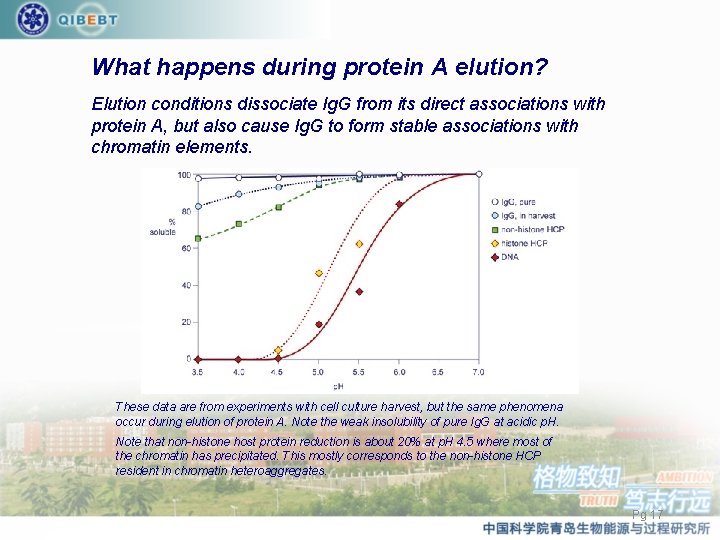
What happens during protein A elution? Elution conditions dissociate Ig. G from its direct associations with protein A, but also cause Ig. G to form stable associations with chromatin elements. These data are from experiments with cell culture harvest, but the same phenomena occur during elution of protein A. Note the weak insolubility of pure Ig. G at acidic p. H. Note that non-histone host protein reduction is about 20% at p. H 4. 5 where most of the chromatin has precipitated. This mostly corresponds to the non-histone HCP resident in chromatin heteroaggregates. Pg 17
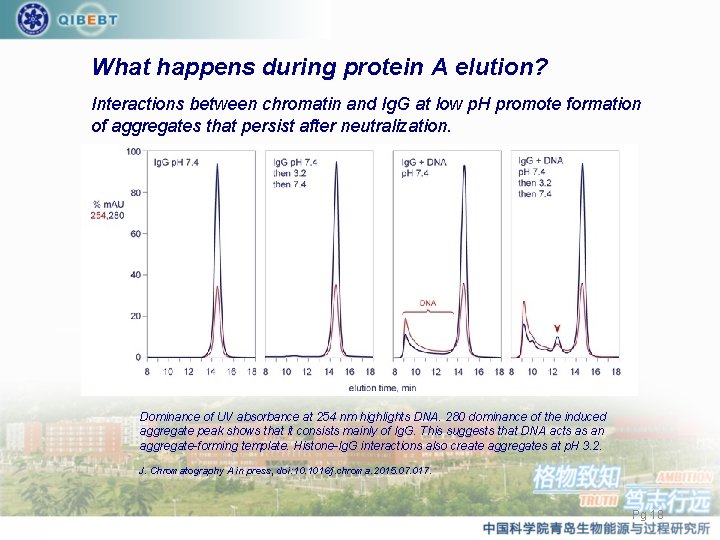
What happens during protein A elution? Interactions between chromatin and Ig. G at low p. H promote formation of aggregates that persist after neutralization. Dominance of UV absorbance at 254 nm highlights DNA. 280 dominance of the induced aggregate peak shows that it consists mainly of Ig. G. This suggests that DNA acts as an aggregate-forming template. Histone-Ig. G interactions also create aggregates at p. H 3. 2. J. Chromatography A in press, doi: 10. 1016/j. chroma. 2015. 07. 017. Pg 18

What happens during protein A elution? Elution conditions also cause subsets of contaminants to dissociate from chromatin elements that remain bound to protein A. Eluted Ig. G can bind to either leached chromatin elements, or to chromatin elements that remain bound to protein A. Ig. G bound to leached chromatin forms aggregates. Ig. G bound to protein A-bound chromatin is lost from elution. Typically ~5%. Leached chromatin in the elution fraction is the main cause of turbidity. Pg 19
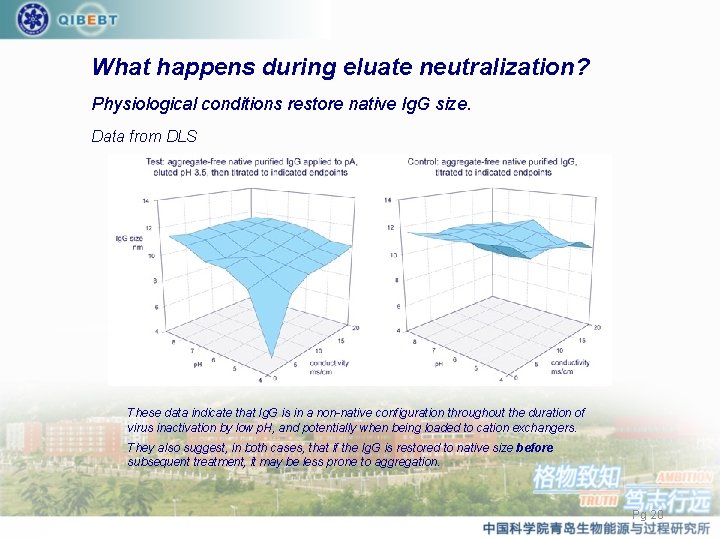
What happens during eluate neutralization? Physiological conditions restore native Ig. G size. Data from DLS These data indicate that Ig. G is in a non-native configuration throughout the duration of virus inactivation by low p. H, and potentially when being loaded to cation exchangers. They also suggest, in both cases, that if the Ig. G is restored to native size before subsequent treatment, it may be less prone to aggregation. Pg 20

What happens during eluate neutralization? Neutralization has two important additional effects: Native Ig. G becomes dissociated from chromatin heteroaggregates. Chromatin elements meanwhile re-associate into very large particles that cause the high turbidity commonly observed among neutralized eluates. This is what enables contaminant and turbidity reduction by microfiltration, and why it does not reduce Ig. G recovery. Pg 21
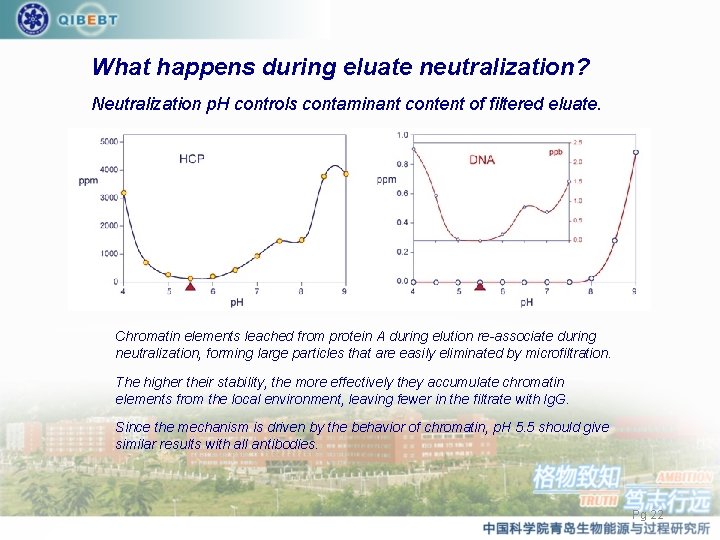
What happens during eluate neutralization? Neutralization p. H controls contaminant content of filtered eluate. Chromatin elements leached from protein A during elution re-associate during neutralization, forming large particles that are easily eliminated by microfiltration. The higher their stability, the more effectively they accumulate chromatin elements from the local environment, leaving fewer in the filtrate with Ig. G. Since the mechanism is driven by the behavior of chromatin, p. H 5. 5 should give similar results with all antibodies. Pg 22
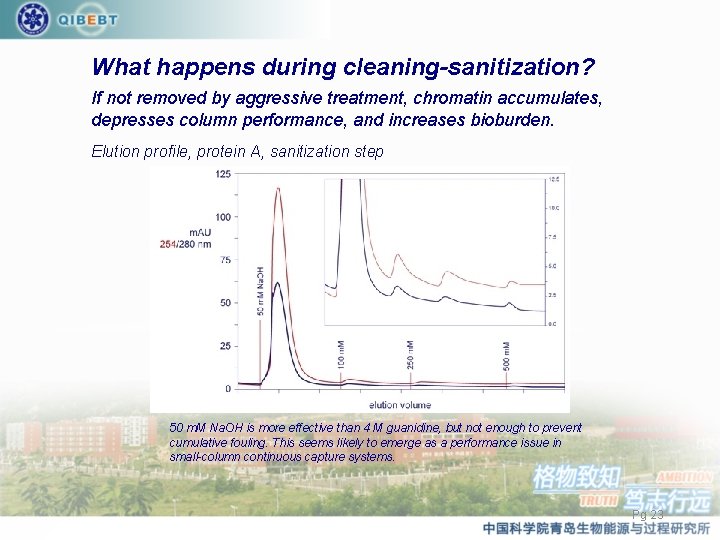
What happens during cleaning-sanitization? If not removed by aggressive treatment, chromatin accumulates, depresses column performance, and increases bioburden. Elution profile, protein A, sanitization step 50 m. M Na. OH is more effective than 4 M guanidine, but not enough to prevent cumulative fouling. This seems likely to emerge as a performance issue in small-column continuous capture systems. Pg 23
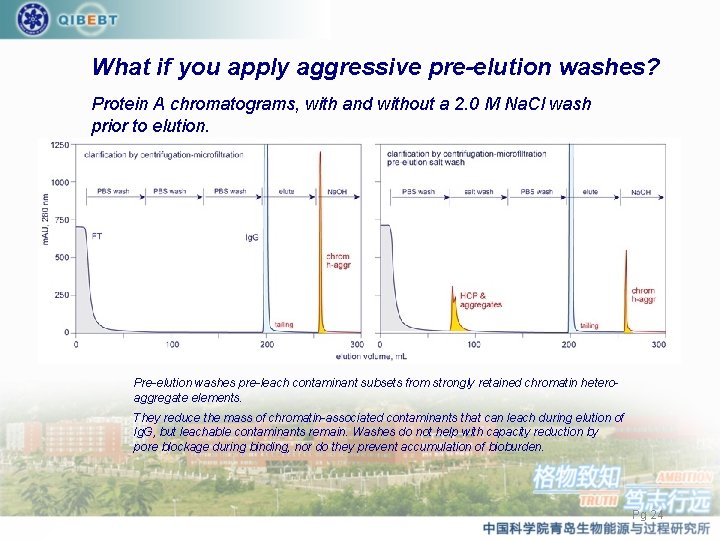
What if you apply aggressive pre-elution washes? Protein A chromatograms, with and without a 2. 0 M Na. Cl wash prior to elution. Pre-elution washes pre-leach contaminant subsets from strongly retained chromatin heteroaggregate elements. They reduce the mass of chromatin-associated contaminants that can leach during elution of Ig. G, but leachable contaminants remain. Washes do not help with capacity reduction by pore blockage during binding, nor do they prevent accumulation of bioburden. Pg 24
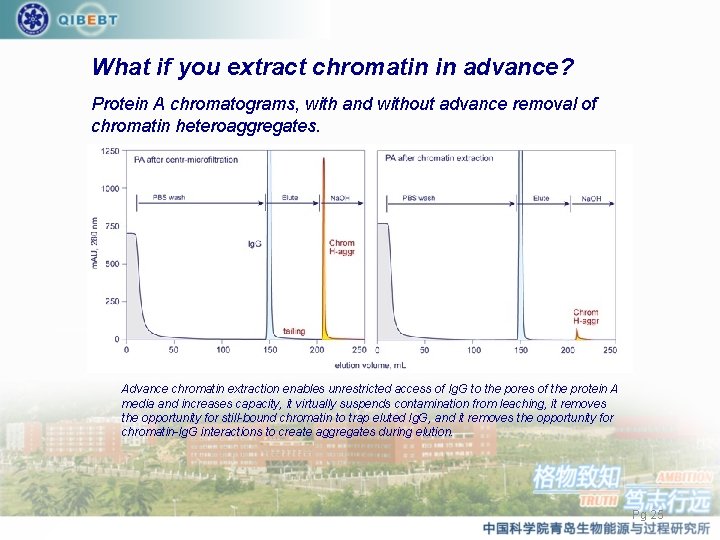
What if you extract chromatin in advance? Protein A chromatograms, with and without advance removal of chromatin heteroaggregates. Advance chromatin extraction enables unrestricted access of Ig. G to the pores of the protein A media and increases capacity, it virtually suspends contamination from leaching, it removes the opportunity for still-bound chromatin to trap eluted Ig. G, and it removes the opportunity for chromatin-Ig. G interactions to create aggregates during elution. Pg 25
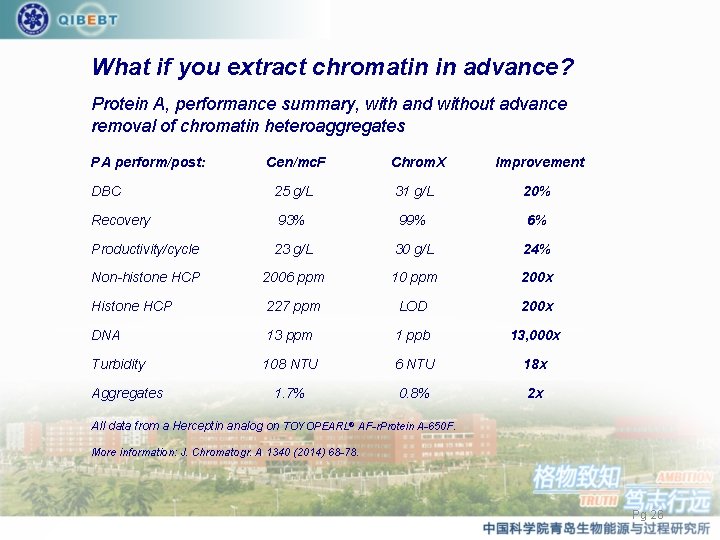
What if you extract chromatin in advance? Protein A, performance summary, with and without advance removal of chromatin heteroaggregates PA perform/post: Cen/mc. F Chrom. X Improvement DBC 25 g/L 31 g/L 20% Recovery 93% 99% 6% Productivity/cycle 23 g/L 30 g/L 24% Non-histone HCP 2006 ppm 10 ppm 200 x Histone HCP 227 ppm LOD 200 x DNA 13 ppm 1 ppb 13, 000 x Turbidity 108 NTU 6 NTU 18 x 1. 7% 0. 8% 2 x Aggregates All data from a Herceptin analog on TOYOPEARL® AF-r. Protein A-650 F. More information: J. Chromatogr. A 1340 (2014) 68 -78. Pg 26

Conclusions Ig. G purification by affinity chromatography with protein A is more complicated than the traditional model suggests. Protein A-induces a major conformational change in Ig. G that persists as long as elution conditions are maintained. This appears to be universal among Ig. G 1 antibodies, likely occurs among other subclasses, other species, and probably among Fc-fusion proteins. How it affects subsequent downstream operations remains to be investigated. Pg 27

Conclusions Chromatin heteroaggregates interfere with protein A purification in many important ways: They block Ig. G access to particle pores, reduce capacity. They induce aggregation under elution conditions. They inflate host contamination of the eluted Ig. G. They mediate on-column loss of Ig. G. They increase bioburden. Their leachates impair the ability of subsequent purification steps to remove host contaminants. Since chromatin is a universal element of cell culture harvests, these effects should be understood to be universal. Pg 28

Conclusions If you extract chromatin in advance, the protein A model becomes nearly as simple as everyone assumes it is. Protein A-mediated conformational change remains a potential concern, but chromatin-mediated effects are suspended. This enables better process economics, reproducibility, and product quality with a single polishing step than is otherwise possible with two polishing steps. Pg 29

Thank you! Pg 30
- Slides: 30