Advances in Molecular Neurobiology Neural Stem Cells and
Advances in Molecular Neurobiology: Neural Stem Cells and Neuroregenerative Approaches Week 5 Adult neurogenic zones and adult neurogenesis

Adult neurogenesis The adult brain is a plastic place. To ensure that the mature nervous system's control of behaviour is flexible in the face of a varying environment, morphological and physiological changes are possible at many levels, including that of the entire cell. Adult neurogenesis is the process of generating new neurons which integrate into existing circuits after fetal and early postnatal development has ceased. In two areas of the adult brain — the olfactory bulb and the dentate gyrus — new neurons are generated throughout life and form an integral part of the normal functional circuitry. In addition there is a high level of adult neurogenesis in the olfactory epithelium (considered part of the peripheral nervous system) where olfactory receptor neurons are constantly replaced. This process is not fixed, but highly modulated, revealing a plastic mechanism by which the brain's performance can be optimized for a given environment. 1) http: //www. scholarpedia. org/article/Adult_neurogenesis 2) Lledo et al, 2006; Nat Rev Neurosci
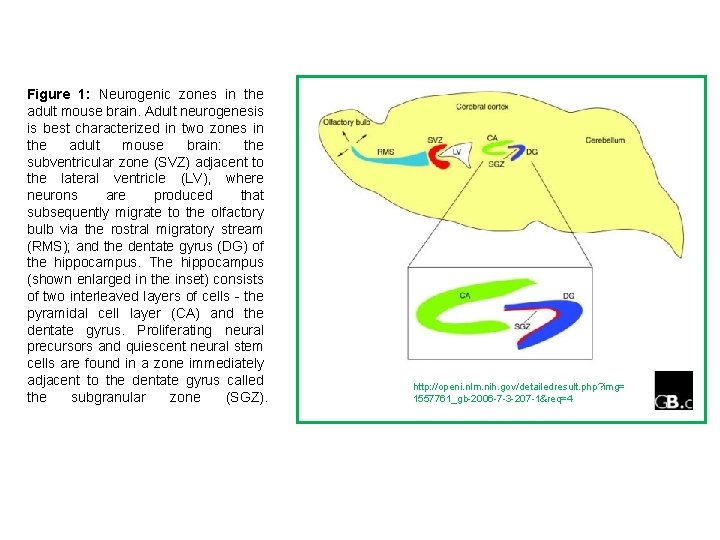
Figure 1: Neurogenic zones in the adult mouse brain. Adult neurogenesis is best characterized in two zones in the adult mouse brain: the subventricular zone (SVZ) adjacent to the lateral ventricle (LV), where neurons are produced that subsequently migrate to the olfactory bulb via the rostral migratory stream (RMS); and the dentate gyrus (DG) of the hippocampus. The hippocampus (shown enlarged in the inset) consists of two interleaved layers of cells - the pyramidal cell layer (CA) and the dentate gyrus. Proliferating neural precursors and quiescent neural stem cells are found in a zone immediately adjacent to the dentate gyrus called the subgranular zone (SGZ). http: //openi. nlm. nih. gov/detailedresult. php? img= 1557761_gb-2006 -7 -3 -207 -1&req=4
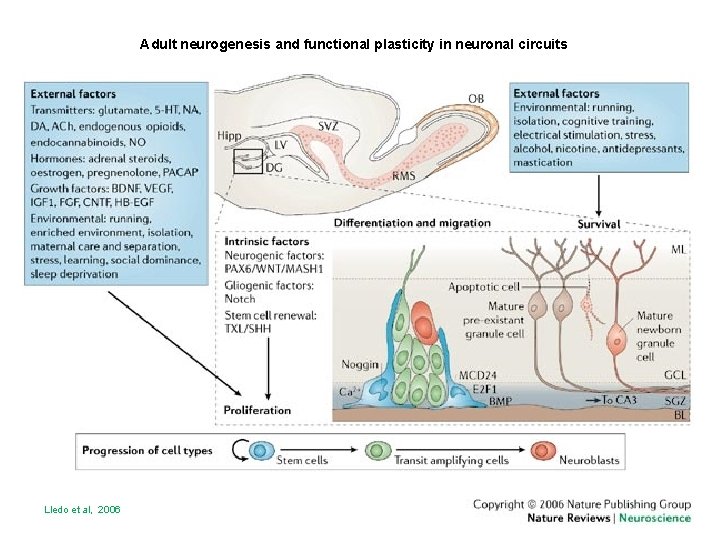
Adult neurogenesis and functional plasticity in neuronal circuits Lledo et al, 2006
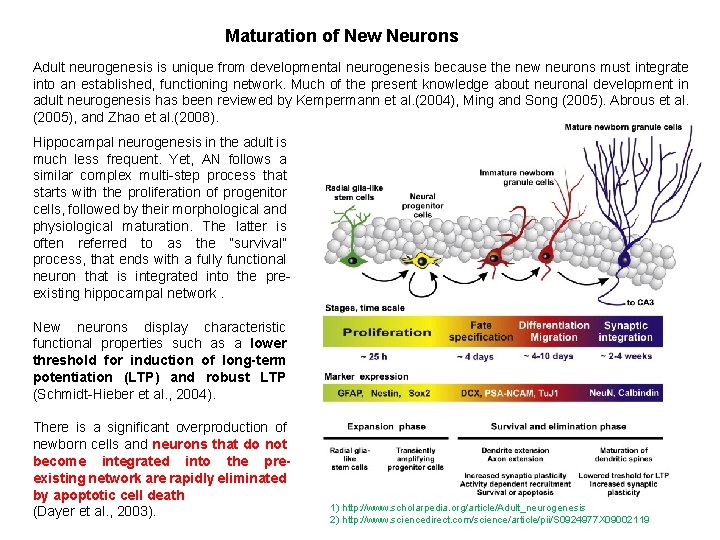
Maturation of New Neurons Adult neurogenesis is unique from developmental neurogenesis because the new neurons must integrate into an established, functioning network. Much of the present knowledge about neuronal development in adult neurogenesis has been reviewed by Kempermann et al. (2004), Ming and Song (2005). Abrous et al. (2005), and Zhao et al. (2008). Hippocampal neurogenesis in the adult is much less frequent. Yet, AN follows a similar complex multi-step process that starts with the proliferation of progenitor cells, followed by their morphological and physiological maturation. The latter is often referred to as the “survival” process, that ends with a fully functional neuron that is integrated into the preexisting hippocampal network. New neurons display characteristic functional properties such as a lower threshold for induction of long-term potentiation (LTP) and robust LTP (Schmidt-Hieber et al. , 2004). There is a significant overproduction of newborn cells and neurons that do not become integrated into the preexisting network are rapidly eliminated by apoptotic cell death (Dayer et al. , 2003). 1) http: //www. scholarpedia. org/article/Adult_neurogenesis 2) http: //www. sciencedirect. com/science/article/pii/S 0924977 X 09002119
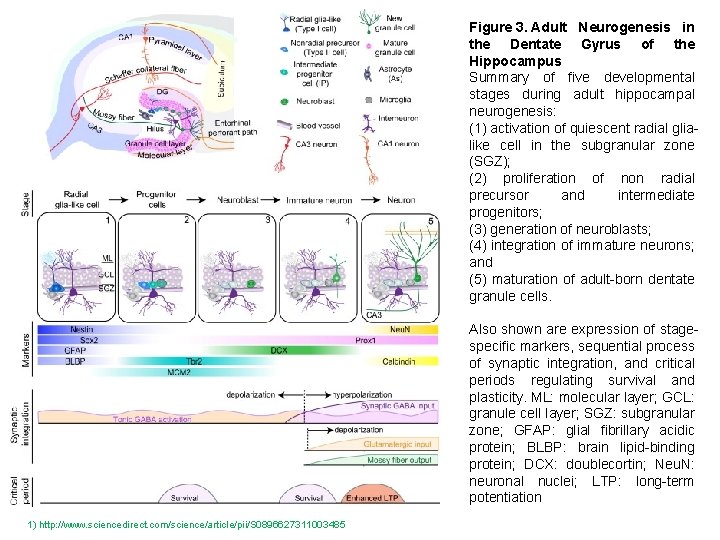
Figure 3. Adult Neurogenesis in the Dentate Gyrus of the Hippocampus Summary of five developmental stages during adult hippocampal neurogenesis: (1) activation of quiescent radial glialike cell in the subgranular zone (SGZ); (2) proliferation of non radial precursor and intermediate progenitors; (3) generation of neuroblasts; (4) integration of immature neurons; and (5) maturation of adult-born dentate granule cells. Also shown are expression of stagespecific markers, sequential process of synaptic integration, and critical periods regulating survival and plasticity. ML: molecular layer; GCL: granule cell layer; SGZ: subgranular zone; GFAP: glial fibrillary acidic protein; BLBP: brain lipid-binding protein; DCX: doublecortin; Neu. N: neuronal nuclei; LTP: long-term potentiation 1) http: //www. sciencedirect. com/science/article/pii/S 0896627311003485
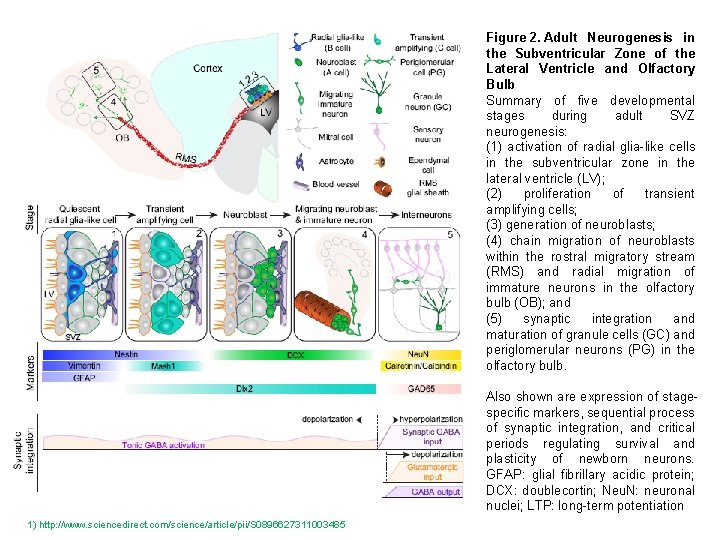
Figure 2. Adult Neurogenesis in the Subventricular Zone of the Lateral Ventricle and Olfactory Bulb Summary of five developmental stages during adult SVZ neurogenesis: (1) activation of radial glia-like cells in the subventricular zone in the lateral ventricle (LV); (2) proliferation of transient amplifying cells; (3) generation of neuroblasts; (4) chain migration of neuroblasts within the rostral migratory stream (RMS) and radial migration of immature neurons in the olfactory bulb (OB); and (5) synaptic integration and maturation of granule cells (GC) and periglomerular neurons (PG) in the olfactory bulb. Also shown are expression of stagespecific markers, sequential process of synaptic integration, and critical periods regulating survival and plasticity of newborn neurons. GFAP: glial fibrillary acidic protein; DCX: doublecortin; Neu. N: neuronal nuclei; LTP: long-term potentiation 1) http: //www. sciencedirect. com/science/article/pii/S 0896627311003485
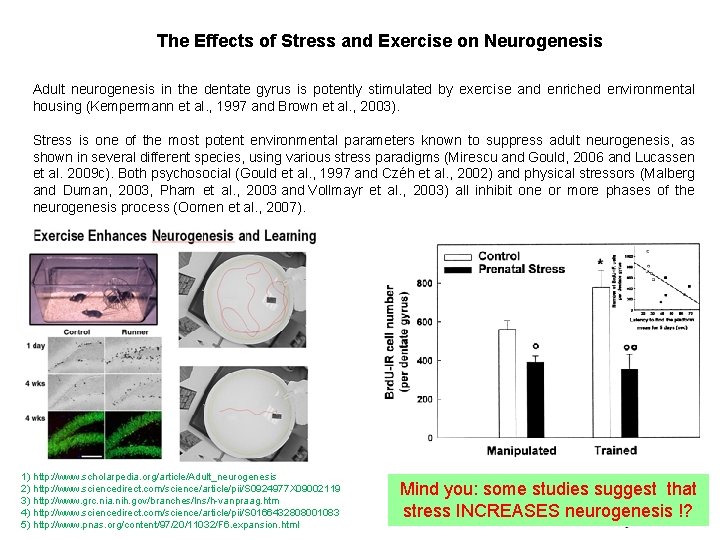
The Effects of Stress and Exercise on Neurogenesis Adult neurogenesis in the dentate gyrus is potently stimulated by exercise and enriched environmental housing (Kempermann et al. , 1997 and Brown et al. , 2003). Stress is one of the most potent environmental parameters known to suppress adult neurogenesis, as shown in several different species, using various stress paradigms (Mirescu and Gould, 2006 and Lucassen et al. 2009 c). Both psychosocial (Gould et al. , 1997 and Czéh et al. , 2002) and physical stressors (Malberg and Duman, 2003, Pham et al. , 2003 and Vollmayr et al. , 2003) all inhibit one or more phases of the neurogenesis process (Oomen et al. , 2007). 1) http: //www. scholarpedia. org/article/Adult_neurogenesis 2) http: //www. sciencedirect. com/science/article/pii/S 0924977 X 09002119 3) http: //www. grc. nia. nih. gov/branches/lns/h-vanpraag. htm 4) http: //www. sciencedirect. com/science/article/pii/S 0166432808001083 5) http: //www. pnas. org/content/97/20/11032/F 6. expansion. html Mind you: some studies suggest that stress INCREASES neurogenesis !?
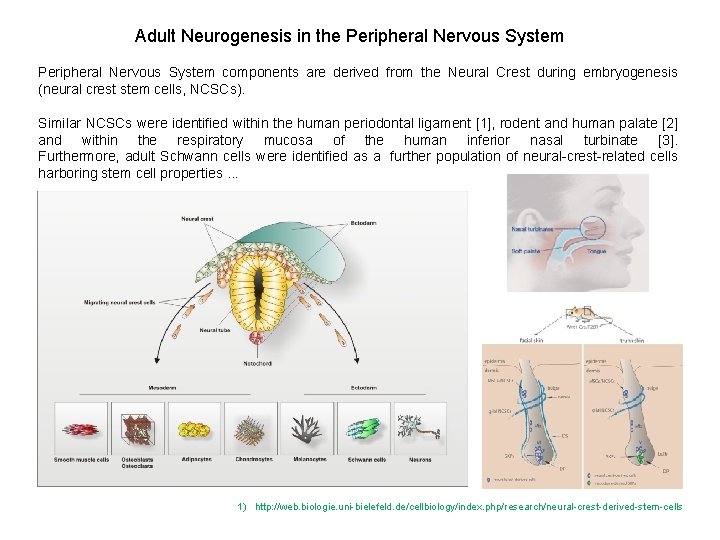
Adult Neurogenesis in the Peripheral Nervous System components are derived from the Neural Crest during embryogenesis (neural crest stem cells, NCSCs). Similar NCSCs were identified within the human periodontal ligament [1], rodent and human palate [2] and within the respiratory mucosa of the human inferior nasal turbinate [3]. Furthermore, adult Schwann cells were identified as a further population of neural-crest-related cells harboring stem cell properties. . . 1) http: //web. biologie. uni-bielefeld. de/cellbiology/index. php/research/neural-crest-derived-stem-cells
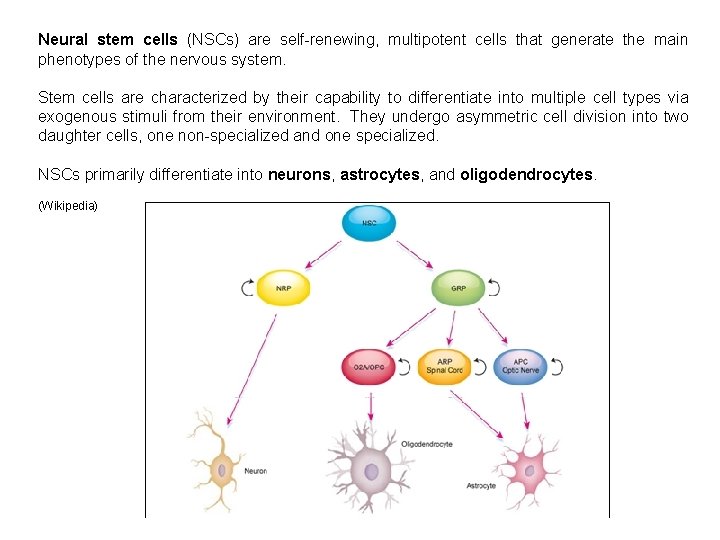
Neural stem cells (NSCs) are self-renewing, multipotent cells that generate the main phenotypes of the nervous system. Stem cells are characterized by their capability to differentiate into multiple cell types via exogenous stimuli from their environment. They undergo asymmetric cell division into two daughter cells, one non-specialized and one specialized. NSCs primarily differentiate into neurons, astrocytes, and oligodendrocytes. (Wikipedia)

Cell culture isolation, maintenance, environment
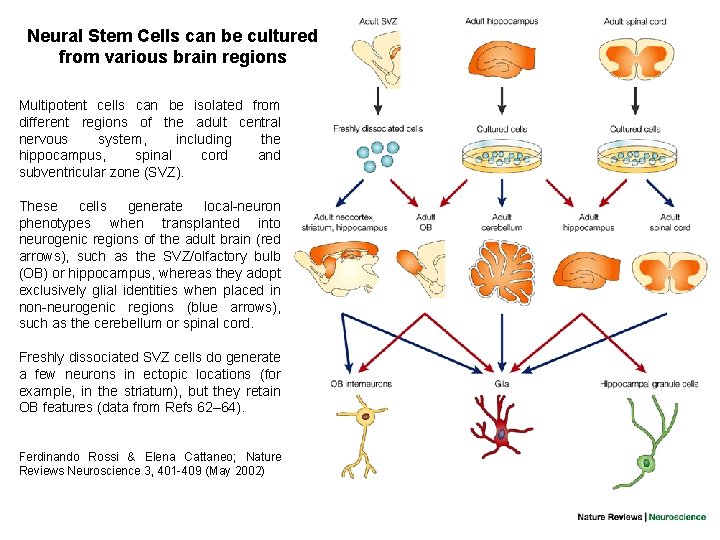
Neural Stem Cells can be cultured from various brain regions Multipotent cells can be isolated from different regions of the adult central nervous system, including the hippocampus, spinal cord and subventricular zone (SVZ). These cells generate local-neuron phenotypes when transplanted into neurogenic regions of the adult brain (red arrows), such as the SVZ/olfactory bulb (OB) or hippocampus, whereas they adopt exclusively glial identities when placed in non-neurogenic regions (blue arrows), such as the cerebellum or spinal cord. Freshly dissociated SVZ cells do generate a few neurons in ectopic locations (for example, in the striatum), but they retain OB features (data from Refs 62– 64). Ferdinando Rossi & Elena Cattaneo; Nature Reviews Neuroscience 3, 401 -409 (May 2002)
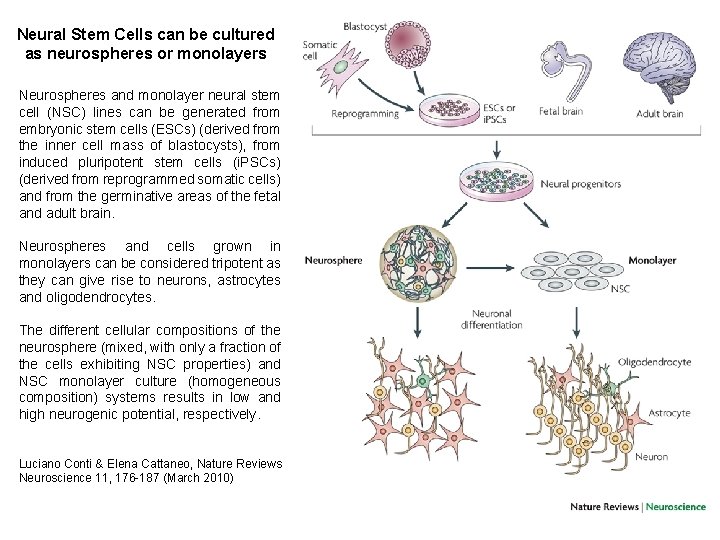
Neural Stem Cells can be cultured as neurospheres or monolayers Neurospheres and monolayer neural stem cell (NSC) lines can be generated from embryonic stem cells (ESCs) (derived from the inner cell mass of blastocysts), from induced pluripotent stem cells (i. PSCs) (derived from reprogrammed somatic cells) and from the germinative areas of the fetal and adult brain. Neurospheres and cells grown in monolayers can be considered tripotent as they can give rise to neurons, astrocytes and oligodendrocytes. The different cellular compositions of the neurosphere (mixed, with only a fraction of the cells exhibiting NSC properties) and NSC monolayer culture (homogeneous composition) systems results in low and high neurogenic potential, respectively. Luciano Conti & Elena Cattaneo, Nature Reviews Neuroscience 11, 176 -187 (March 2010)
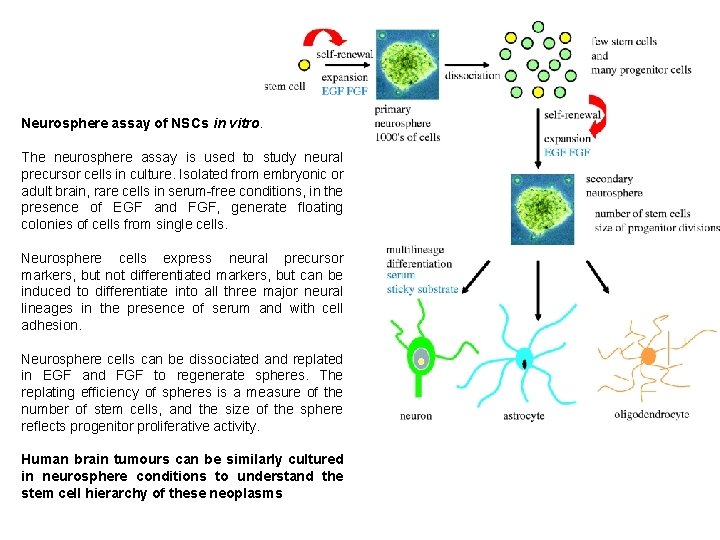
Neurosphere assay of NSCs in vitro. The neurosphere assay is used to study neural precursor cells in culture. Isolated from embryonic or adult brain, rare cells in serum-free conditions, in the presence of EGF and FGF, generate floating colonies of cells from single cells. Neurosphere cells express neural precursor markers, but not differentiated markers, but can be induced to differentiate into all three major neural lineages in the presence of serum and with cell adhesion. Neurosphere cells can be dissociated and replated in EGF and FGF to regenerate spheres. The replating efficiency of spheres is a measure of the number of stem cells, and the size of the sphere reflects progenitor proliferative activity. Human brain tumours can be similarly cultured in neurosphere conditions to understand the stem cell hierarchy of these neoplasms
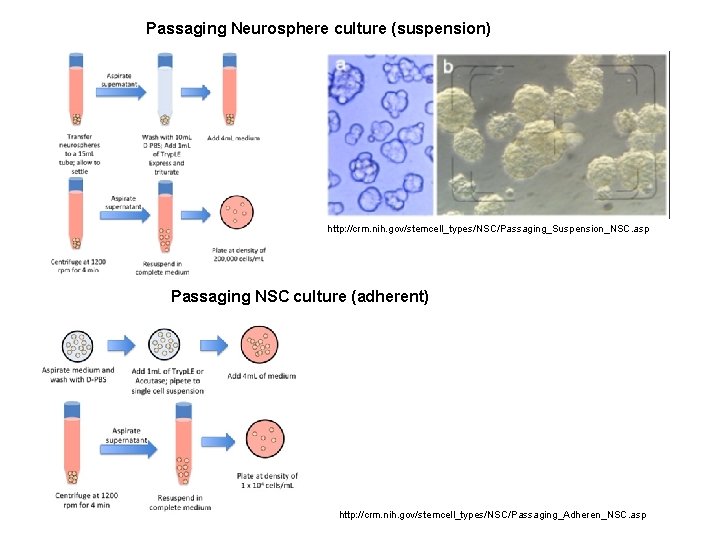
Passaging Neurosphere culture (suspension) http: //crm. nih. gov/stemcell_types/NSC/Passaging_Suspension_NSC. asp Passaging NSC culture (adherent) http: //crm. nih. gov/stemcell_types/NSC/Passaging_Adheren_NSC. asp
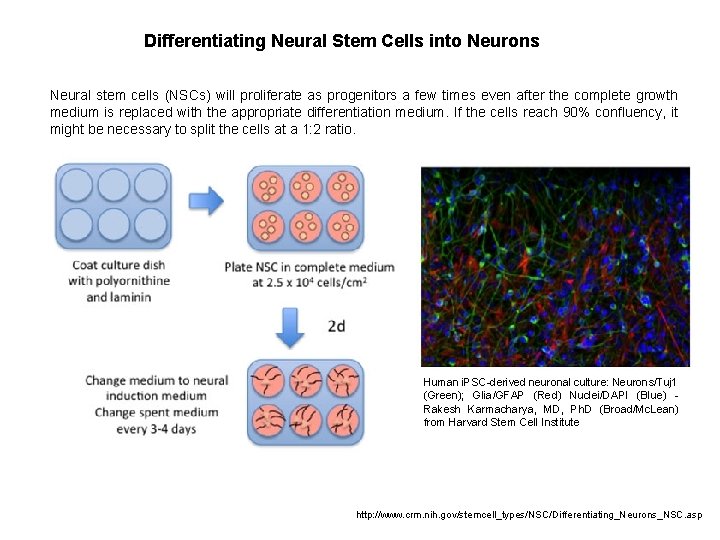
Differentiating Neural Stem Cells into Neurons Neural stem cells (NSCs) will proliferate as progenitors a few times even after the complete growth medium is replaced with the appropriate differentiation medium. If the cells reach 90% confluency, it might be necessary to split the cells at a 1: 2 ratio. Human i. PSC-derived neuronal culture: Neurons/Tuj 1 (Green); Glia/GFAP (Red) Nuclei/DAPI (Blue) - Rakesh Karmacharya, MD, Ph. D (Broad/Mc. Lean) from Harvard Stem Cell Institute http: //www. crm. nih. gov/stemcell_types/NSC/Differentiating_Neurons_NSC. asp
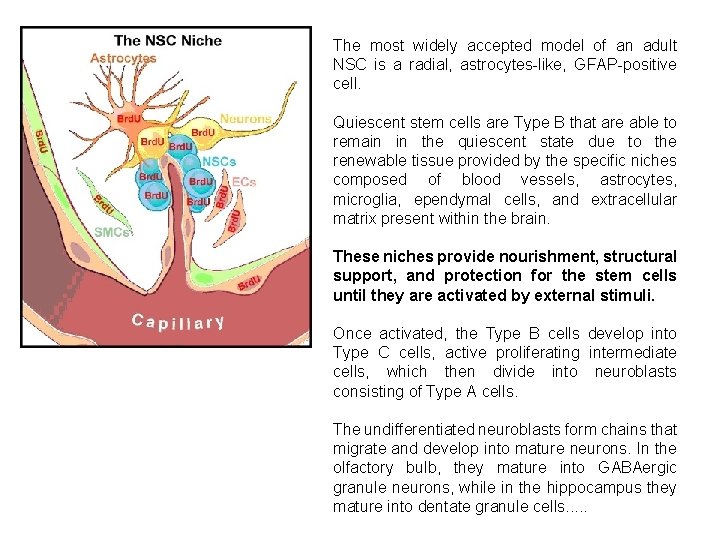
The most widely accepted model of an adult NSC is a radial, astrocytes-like, GFAP-positive cell. Quiescent stem cells are Type B that are able to remain in the quiescent state due to the renewable tissue provided by the specific niches composed of blood vessels, astrocytes, microglia, ependymal cells, and extracellular matrix present within the brain. These niches provide nourishment, structural support, and protection for the stem cells until they are activated by external stimuli. Once activated, the Type B cells develop into Type C cells, active proliferating intermediate cells, which then divide into neuroblasts consisting of Type A cells. The undifferentiated neuroblasts form chains that migrate and develop into mature neurons. In the olfactory bulb, they mature into GABAergic granule neurons, while in the hippocampus they mature into dentate granule cells. . .
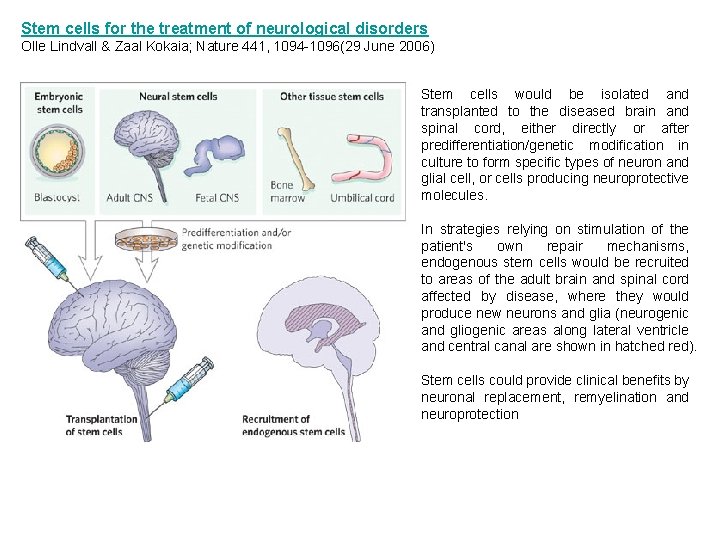
Stem cells for the treatment of neurological disorders Olle Lindvall & Zaal Kokaia; Nature 441, 1094 -1096(29 June 2006) Stem cells would be isolated and transplanted to the diseased brain and spinal cord, either directly or after predifferentiation/genetic modification in culture to form specific types of neuron and glial cell, or cells producing neuroprotective molecules. In strategies relying on stimulation of the patient's own repair mechanisms, endogenous stem cells would be recruited to areas of the adult brain and spinal cord affected by disease, where they would produce new neurons and glia (neurogenic and gliogenic areas along lateral ventricle and central canal are shown in hatched red). Stem cells could provide clinical benefits by neuronal replacement, remyelination and neuroprotection
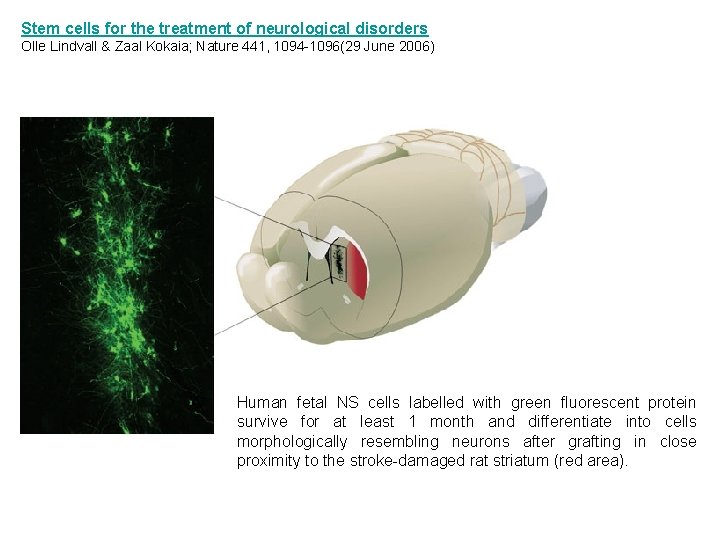
Stem cells for the treatment of neurological disorders Olle Lindvall & Zaal Kokaia; Nature 441, 1094 -1096(29 June 2006) Human fetal NS cells labelled with green fluorescent protein survive for at least 1 month and differentiate into cells morphologically resembling neurons after grafting in close proximity to the stroke-damaged rat striatum (red area).
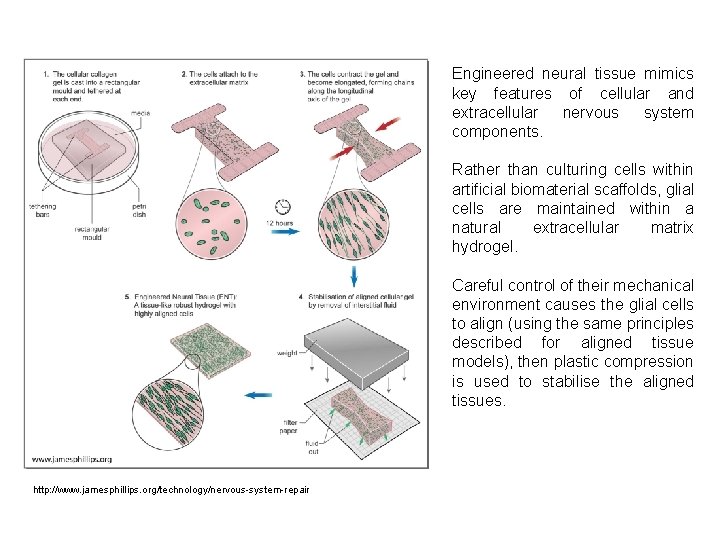
Engineered neural tissue mimics key features of cellular and extracellular nervous system components. Rather than culturing cells within artificial biomaterial scaffolds, glial cells are maintained within a natural extracellular matrix hydrogel. Careful control of their mechanical environment causes the glial cells to align (using the same principles described for aligned tissue models), then plastic compression is used to stabilise the aligned tissues. http: //www. jamesphillips. org/technology/nervous-system-repair
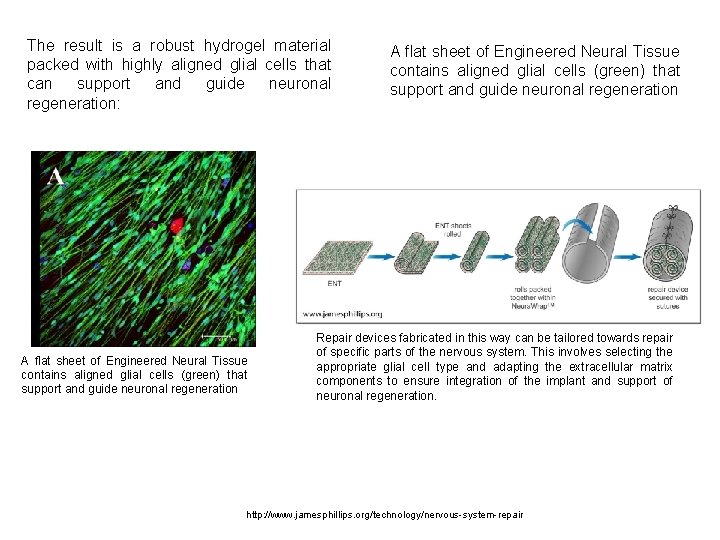
The result is a robust hydrogel material packed with highly aligned glial cells that can support and guide neuronal regeneration: A flat sheet of Engineered Neural Tissue contains aligned glial cells (green) that support and guide neuronal regeneration Repair devices fabricated in this way can be tailored towards repair of specific parts of the nervous system. This involves selecting the appropriate glial cell type and adapting the extracellular matrix components to ensure integration of the implant and support of neuronal regeneration. http: //www. jamesphillips. org/technology/nervous-system-repair
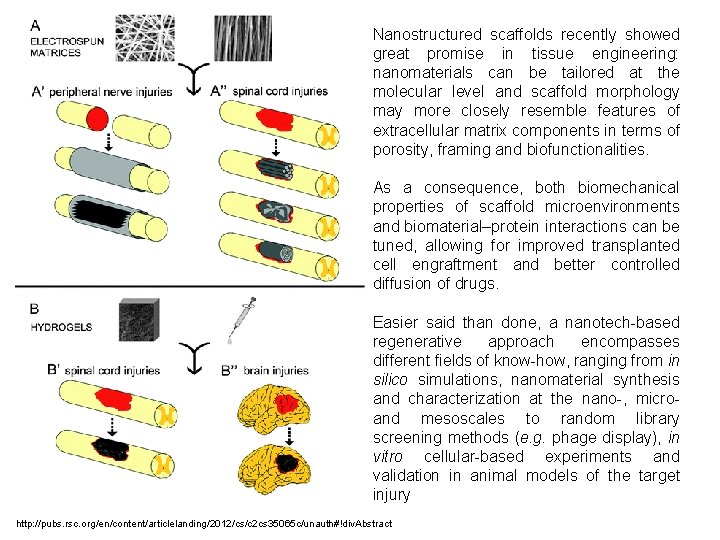
Nanostructured scaffolds recently showed great promise in tissue engineering: nanomaterials can be tailored at the molecular level and scaffold morphology may more closely resemble features of extracellular matrix components in terms of porosity, framing and biofunctionalities. As a consequence, both biomechanical properties of scaffold microenvironments and biomaterial–protein interactions can be tuned, allowing for improved transplanted cell engraftment and better controlled diffusion of drugs. Easier said than done, a nanotech-based regenerative approach encompasses different fields of know-how, ranging from in silico simulations, nanomaterial synthesis and characterization at the nano-, micro- and mesoscales to random library screening methods (e. g. phage display), in vitro cellular-based experiments and validation in animal models of the target injury http: //pubs. rsc. org/en/content/articlelanding/2012/cs/c 2 cs 35065 c/unauth#!div. Abstract
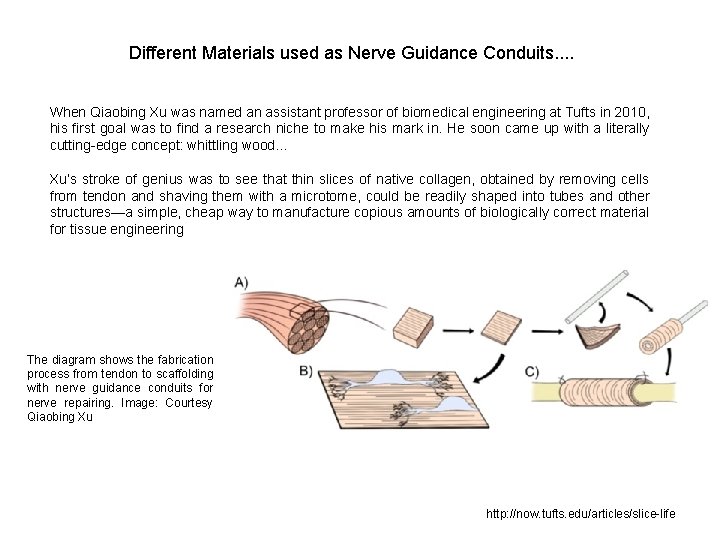
Different Materials used as Nerve Guidance Conduits. . When Qiaobing Xu was named an assistant professor of biomedical engineering at Tufts in 2010, his first goal was to find a research niche to make his mark in. He soon came up with a literally cutting-edge concept: whittling wood. . . Xu’s stroke of genius was to see that thin slices of native collagen, obtained by removing cells from tendon and shaving them with a microtome, could be readily shaped into tubes and other structures—a simple, cheap way to manufacture copious amounts of biologically correct material for tissue engineering The diagram shows the fabrication process from tendon to scaffolding with nerve guidance conduits for nerve repairing. Image: Courtesy Qiaobing Xu http: //now. tufts. edu/articles/slice-life
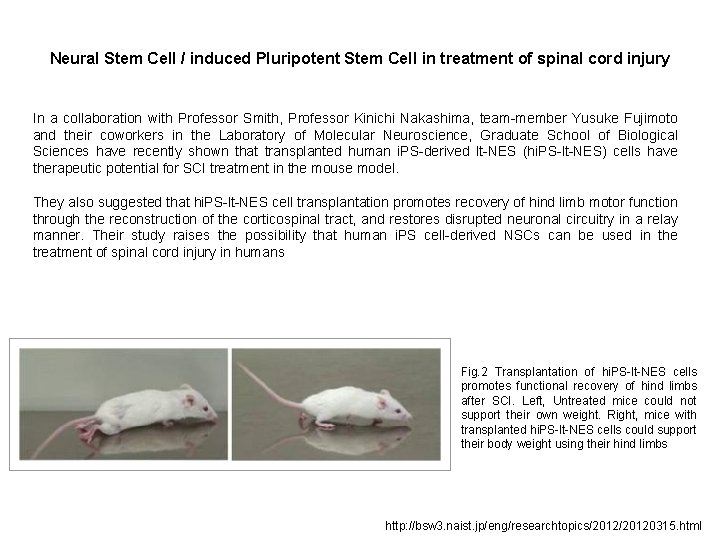
Neural Stem Cell / induced Pluripotent Stem Cell in treatment of spinal cord injury In a collaboration with Professor Smith, Professor Kinichi Nakashima, team-member Yusuke Fujimoto and their coworkers in the Laboratory of Molecular Neuroscience, Graduate School of Biological Sciences have recently shown that transplanted human i. PS-derived lt-NES (hi. PS-lt-NES) cells have therapeutic potential for SCI treatment in the mouse model. They also suggested that hi. PS-lt-NES cell transplantation promotes recovery of hind limb motor function through the reconstruction of the corticospinal tract, and restores disrupted neuronal circuitry in a relay manner. Their study raises the possibility that human i. PS cell-derived NSCs can be used in the treatment of spinal cord injury in humans Fig. 2 Transplantation of hi. PS-lt-NES cells promotes functional recovery of hind limbs after SCI. Left, Untreated mice could not support their own weight. Right, mice with transplanted hi. PS-lt-NES cells could support their body weight using their hind limbs http: //bsw 3. naist. jp/eng/researchtopics/20120315. html
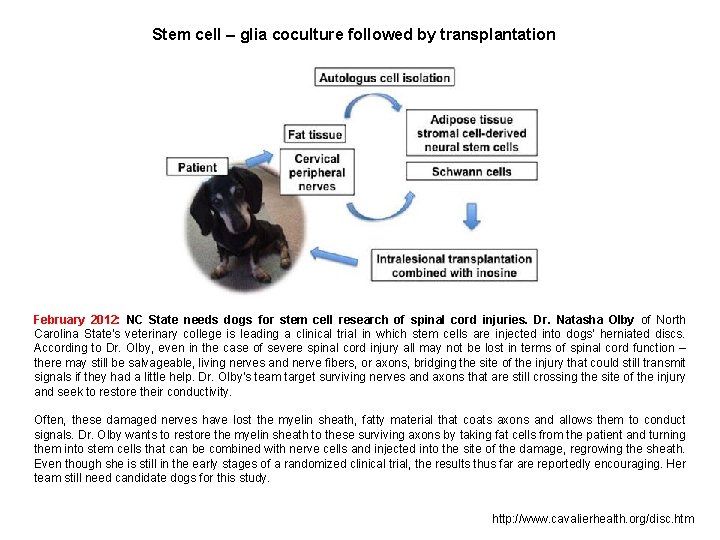
Stem cell – glia coculture followed by transplantation February 2012: NC State needs dogs for stem cell research of spinal cord injuries. Dr. Natasha Olby of North Carolina State's veterinary college is leading a clinical trial in which stem cells are injected into dogs' herniated discs. According to Dr. Olby, even in the case of severe spinal cord injury all may not be lost in terms of spinal cord function – there may still be salvageable, living nerves and nerve fibers, or axons, bridging the site of the injury that could still transmit signals if they had a little help. Dr. Olby's team target surviving nerves and axons that are still crossing the site of the injury and seek to restore their conductivity. Often, these damaged nerves have lost the myelin sheath, fatty material that coats axons and allows them to conduct signals. Dr. Olby wants to restore the myelin sheath to these surviving axons by taking fat cells from the patient and turning them into stem cells that can be combined with nerve cells and injected into the site of the damage, regrowing the sheath. Even though she is still in the early stages of a randomized clinical trial, the results thus far are reportedly encouraging. Her team still need candidate dogs for this study. http: //www. cavalierhealth. org/disc. htm

(some techniques – brief intro)
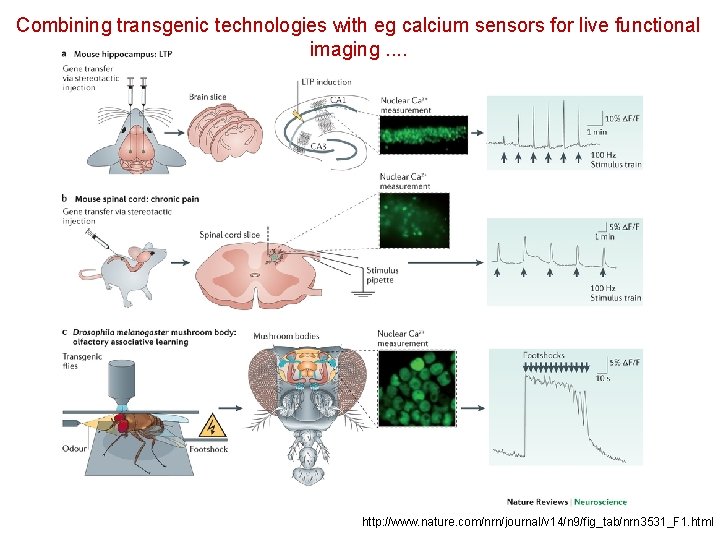
Combining transgenic technologies with eg calcium sensors for live functional imaging. . http: //www. nature. com/nrn/journal/v 14/n 9/fig_tab/nrn 3531_F 1. html
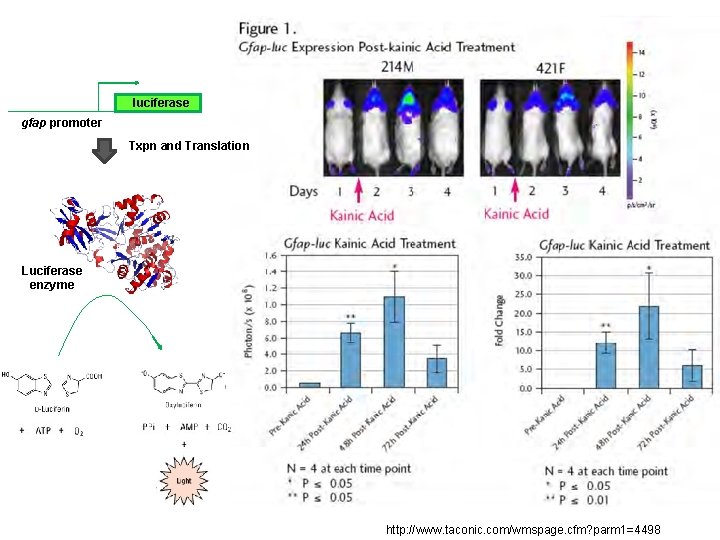
luciferase gfap promoter Txpn and Translation Luciferase enzyme http: //www. taconic. com/wmspage. cfm? parm 1=4498
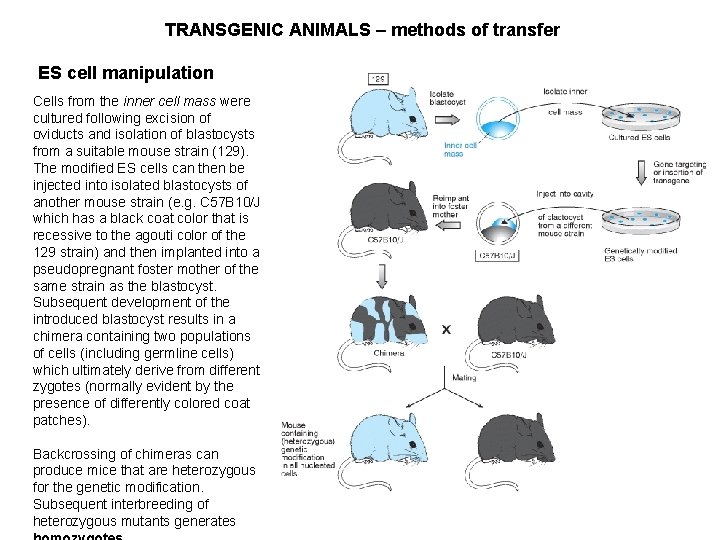
TRANSGENIC ANIMALS – methods of transfer ES cell manipulation Cells from the inner cell mass were cultured following excision of oviducts and isolation of blastocysts from a suitable mouse strain (129). The modified ES cells can then be injected into isolated blastocysts of another mouse strain (e. g. C 57 B 10/J which has a black coat color that is recessive to the agouti color of the 129 strain) and then implanted into a pseudopregnant foster mother of the same strain as the blastocyst. Subsequent development of the introduced blastocyst results in a chimera containing two populations of cells (including germline cells) which ultimately derive from different zygotes (normally evident by the presence of differently colored coat patches). Backcrossing of chimeras can produce mice that are heterozygous for the genetic modification. Subsequent interbreeding of heterozygous mutants generates
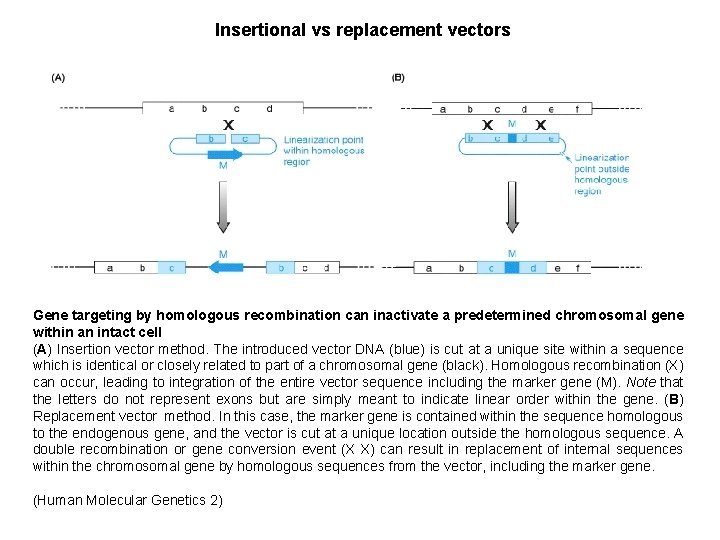
Insertional vs replacement vectors Gene targeting by homologous recombination can inactivate a predetermined chromosomal gene within an intact cell (A) Insertion vector method. The introduced vector DNA (blue) is cut at a unique site within a sequence which is identical or closely related to part of a chromosomal gene (black). Homologous recombination (X) can occur, leading to integration of the entire vector sequence including the marker gene (M). Note that the letters do not represent exons but are simply meant to indicate linear order within the gene. (B) Replacement vector method. In this case, the marker gene is contained within the sequence homologous to the endogenous gene, and the vector is cut at a unique location outside the homologous sequence. A double recombination or gene conversion event (X X) can result in replacement of internal sequences within the chromosomal gene by homologous sequences from the vector, including the marker gene. (Human Molecular Genetics 2)
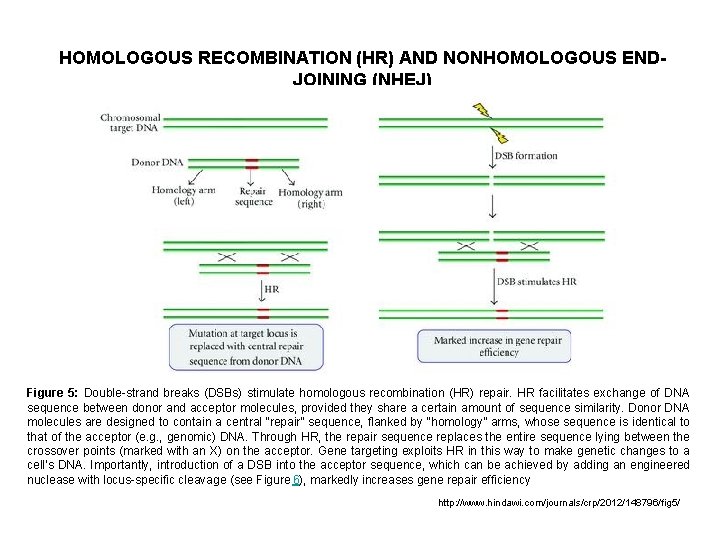
HOMOLOGOUS RECOMBINATION (HR) AND NONHOMOLOGOUS ENDJOINING (NHEJ) Figure 5: Double-strand breaks (DSBs) stimulate homologous recombination (HR) repair. HR facilitates exchange of DNA sequence between donor and acceptor molecules, provided they share a certain amount of sequence similarity. Donor DNA molecules are designed to contain a central “repair” sequence, flanked by “homology” arms, whose sequence is identical to that of the acceptor (e. g. , genomic) DNA. Through HR, the repair sequence replaces the entire sequence lying between the crossover points (marked with an X) on the acceptor. Gene targeting exploits HR in this way to make genetic changes to a cell’s DNA. Importantly, introduction of a DSB into the acceptor sequence, which can be achieved by adding an engineered nuclease with locus-specific cleavage (see Figure 6), markedly increases gene repair efficiency http: //www. hindawi. com/journals/crp/2012/148796/fig 5/
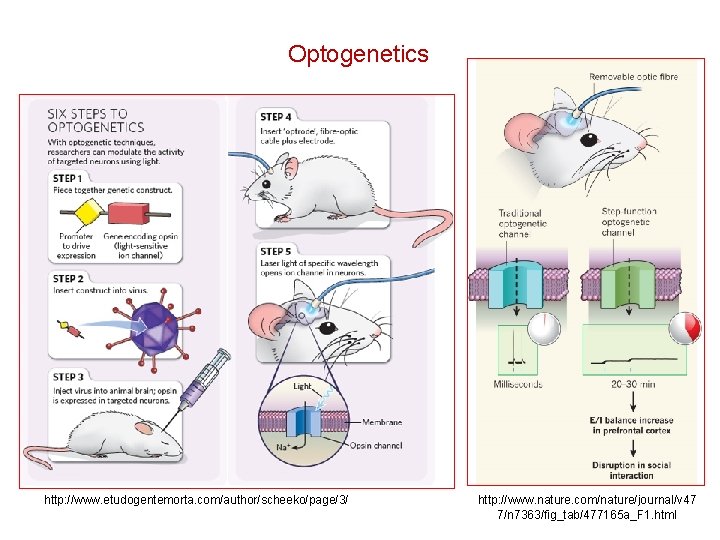
Optogenetics http: //www. etudogentemorta. com/author/scheeko/page/3/ http: //www. nature. com/nature/journal/v 47 7/n 7363/fig_tab/477165 a_F 1. html
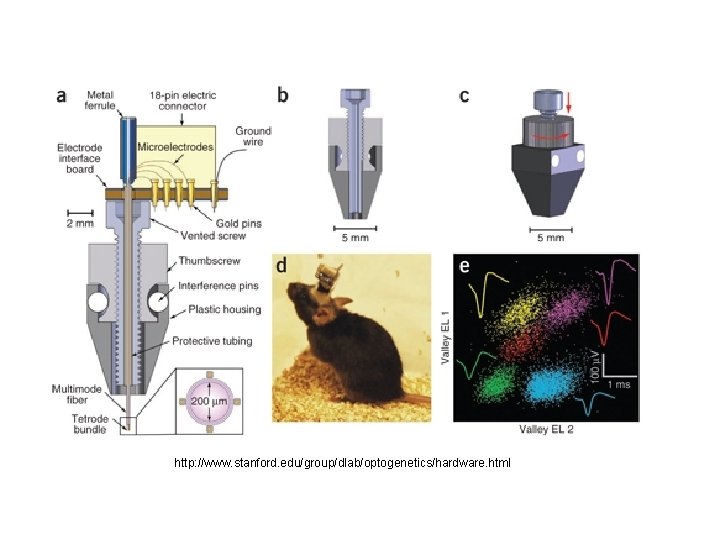
http: //www. stanford. edu/group/dlab/optogenetics/hardware. html

(some techniques – brief intro)

Stem Cell Therapy for Neurodegenerative Disorders
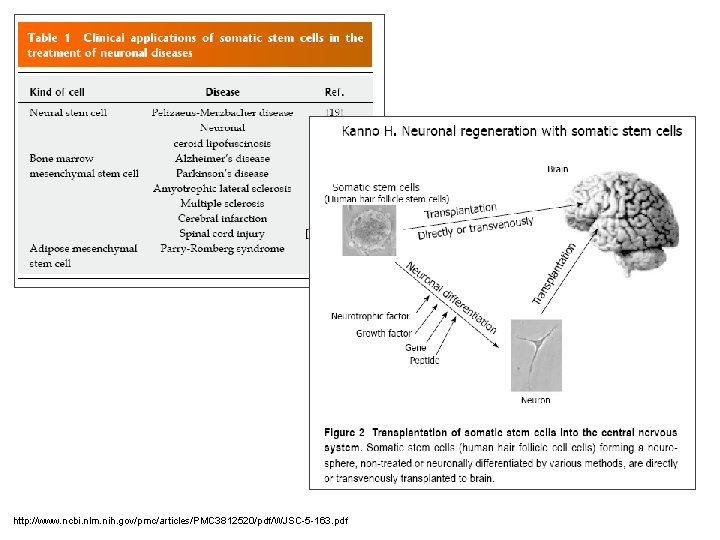
http: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 3812520/pdf/WJSC-5 -163. pdf
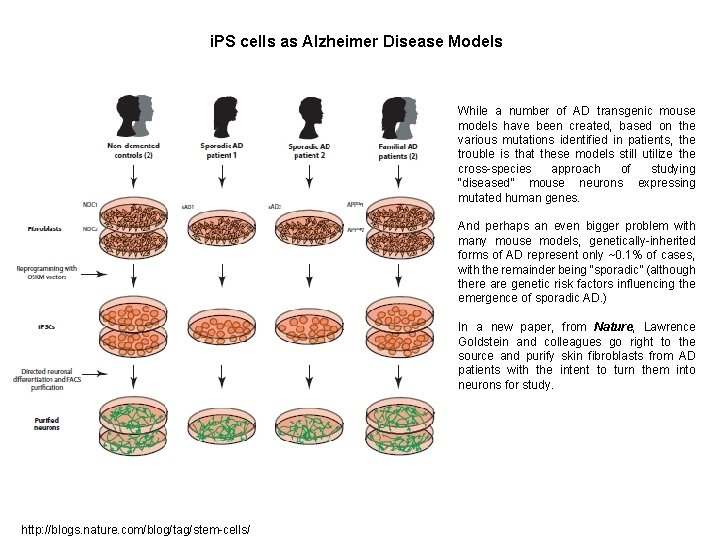
i. PS cells as Alzheimer Disease Models While a number of AD transgenic mouse models have been created, based on the various mutations identified in patients, the trouble is that these models still utilize the cross-species approach of studying “diseased” mouse neurons expressing mutated human genes. And perhaps an even bigger problem with many mouse models, genetically-inherited forms of AD represent only ~0. 1% of cases, with the remainder being “sporadic” (although there are genetic risk factors influencing the emergence of sporadic AD. ) In a new paper, from Nature, Lawrence Goldstein and colleagues go right to the source and purify skin fibroblasts from AD patients with the intent to turn them into neurons for study. http: //blogs. nature. com/blog/tag/stem-cells/
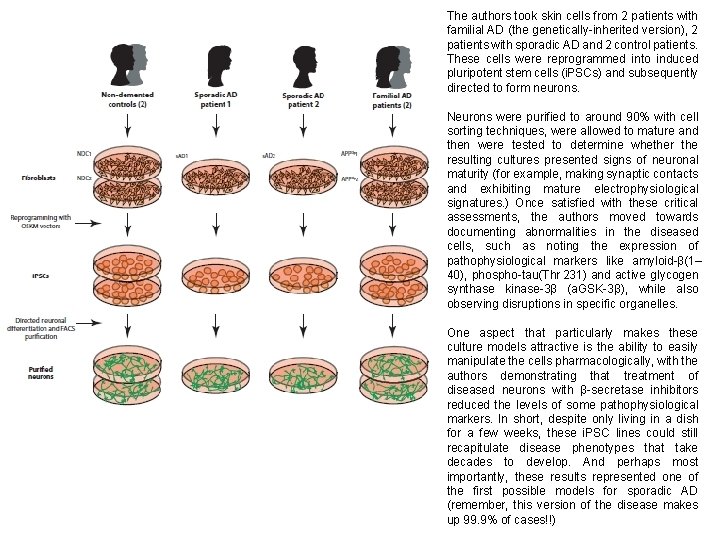
The authors took skin cells from 2 patients with familial AD (the genetically-inherited version), 2 patients with sporadic AD and 2 control patients. These cells were reprogrammed into induced pluripotent stem cells (i. PSCs) and subsequently directed to form neurons. Neurons were purified to around 90% with cell sorting techniques, were allowed to mature and then were tested to determine whether the resulting cultures presented signs of neuronal maturity (for example, making synaptic contacts and exhibiting mature electrophysiological signatures. ) Once satisfied with these critical assessments, the authors moved towards documenting abnormalities in the diseased cells, such as noting the expression of pathophysiological markers like amyloid-β(1– 40), phospho-tau(Thr 231) and active glycogen synthase kinase-3β (a. GSK-3β), while also observing disruptions in specific organelles. One aspect that particularly makes these culture models attractive is the ability to easily manipulate the cells pharmacologically, with the authors demonstrating that treatment of diseased neurons with β-secretase inhibitors reduced the levels of some pathophysiological markers. In short, despite only living in a dish for a few weeks, these i. PSC lines could still recapitulate disease phenotypes that take decades to develop. And perhaps most importantly, these results represented one of the first possible models for sporadic AD (remember, this version of the disease makes up 99. 9% of cases!!)
Stem Cells for Alzheimer Treatment ?
Stem Cells for Parkinson’s. Treatment ?
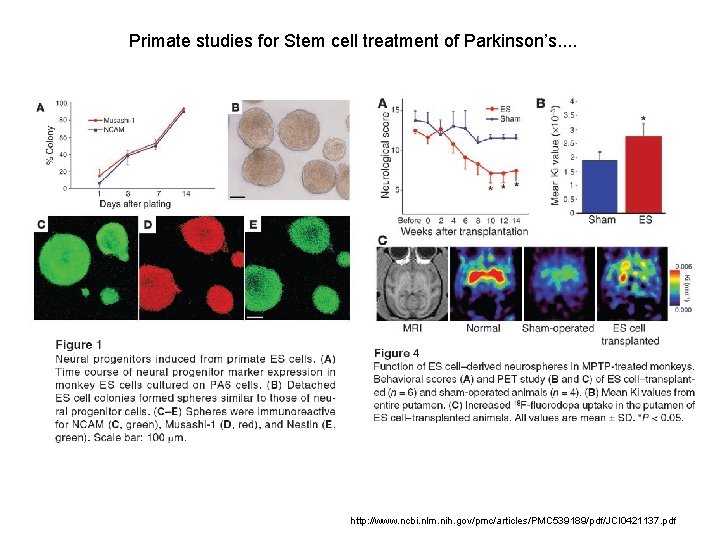
Primate studies for Stem cell treatment of Parkinson’s. . http: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 539189/pdf/JCI 0421137. pdf
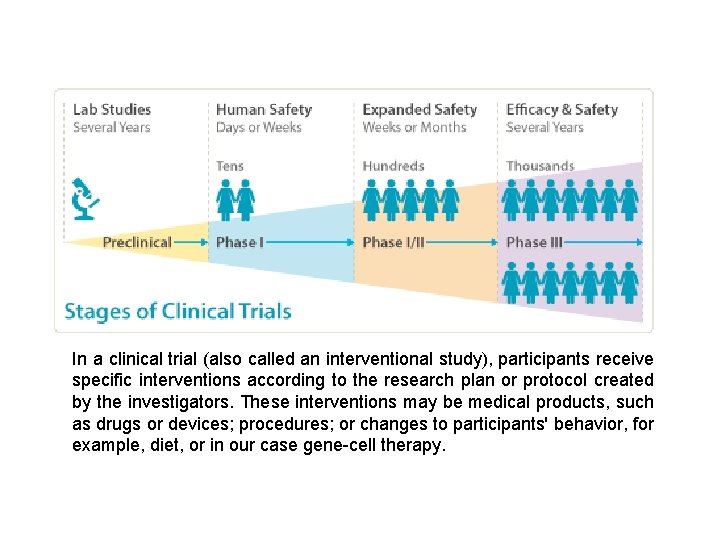
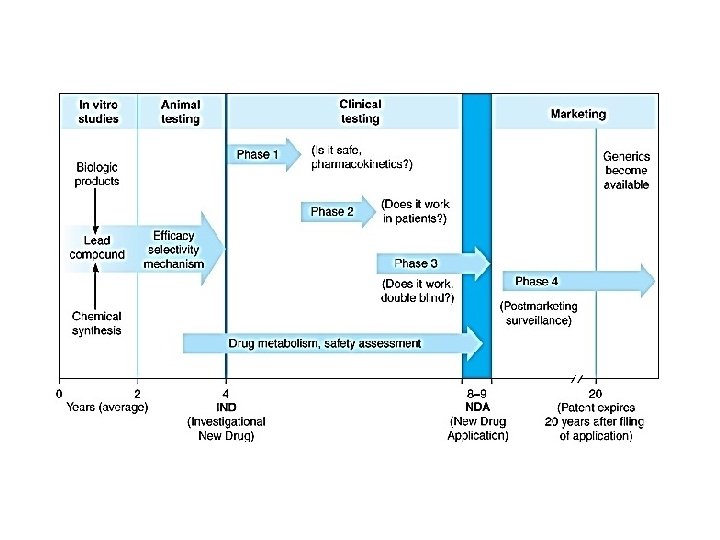
In a clinical trial (also called an interventional study), participants receive specific interventions according to the research plan or protocol created by the investigators. These interventions may be medical products, such as drugs or devices; procedures; or changes to participants' behavior, for example, diet, or in our case gene-cell therapy.
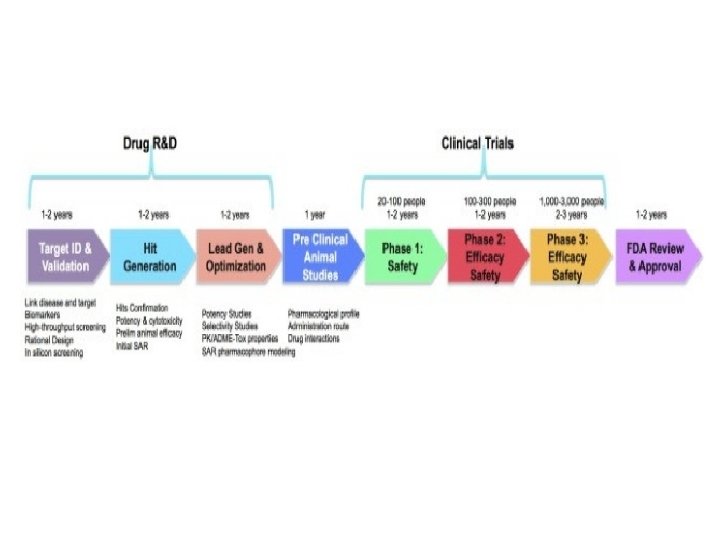

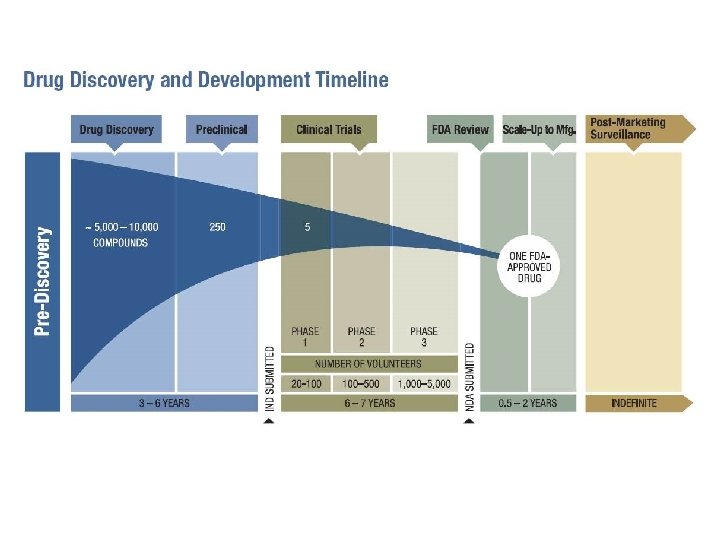
Clinical trials are conducted in a series of steps, called phases - each phase is designed to answer a separate research question: Phase I: Researchers test a new drug or treatment in a small group of people for the first time to evaluate its safety, determine a safe dosage range, and identify side effects. Phase II: The drug or treatment is given to a larger group of people to see if it is effective and to further evaluate its safety. Phase III: The drug or treatment is given to large groups of people to confirm its effectiveness, monitor side effects, compare it to commonly used treatments, and collect information that will allow the drug or treatment to be used safely. Phase IV: Studies are done after the drug or treatment has been marketed to gather information on the drug's effect in various populations and any side effects associated with long-term use. https: //www. nlm. nih. gov/services/ctphases. html

Phase 1 trials are done to find out § How much of the drug is safe to give § What the side effects are § How the body copes with the drug § If the treatment cures the disease Patients are recruited very slowly onto phase 1 trials. . . The first few patients to take part (called a cohort or group) are given a very small dose of the drug. If all goes well, the next group have a slightly higher dose. The dose is gradually increased with each group. The researchers monitor the effect of the drug until they find the best dose to give. This is called a dose escalation study. In a phase 1 trial you may have lots of blood tests because the researchers look at how the drug affects you. They also look at how your body copes with, and gets rid of the drug. They record any side effects. People taking part in phase 1 trials are often terminal patients (ie waiting to die). Phase 1 trials aim to look at doses and side effects. This work has to be done first, before we can test the potential new treatment to see if it works. http: //www. cancerresearchuk. org/about-cancer/find-a-clinical-trial/what-clinical-trials-are/phases-of-clinical-trial

Phase 2 trials Not all treatments tested in a phase 1 trial make it to a phase 2 trial. Phase 2 trials aim to find out § If the new treatment works well enough to test in a larger phase 3 trial § Which types of cancer the treatment works for § More about side effects and how to manage them § More about the best dose to use Although these treatments have been tested in phase 1 trials, you may still have side effects that the doctors don't know about. Drugs can affect people in different ways. Phase 2 trials are often larger than phase 1. There may be up to 100 or so people taking part. Sometimes in a phase 2 trial, a new treatment is compared with another treatment already in use, or with a dummy drug (placebo). If the results of phase 2 trials show that a new treatment may be as good as existing treatment, or better, it then moves into phase 3. Some phase 2 trials are randomised. This means the researchers put the people taking part into treatment groups at random. http: //www. cancerresearchuk. org/about-cancer/find-a-clinical-trial/what-clinical-trials-are/phases-of-clinical-trial

RANDOMIZED TRIALS Most phase 3 trials, and some phase 2 trials, are randomised. This means that there at least 2 different groups in the trial and people taking part are put into one or other group at random. This 'randomisation' is usually done by a computer. Certain details about you (such as your age, gender or the stage of your disease) are put into the computer first. This is to make sure the different groups in a trial are as similar as possible. Each group in the trial has a different treatment. If there are 2 groups, one group has the new treatment being tested and the other has the standard treatment they would have if they were not in the trial. People having the standard treatment are called the control group. A randomised trial that has a control group is called a randomised controlled trial. Sometimes people in the control group take a dummy treatment, called a placebo. Doctors only set up trials this way if there is no standard treatment available for the control group to have. Researchers randomise trials because they need to be sure that the results are correct and there is no bias that could distort the results. http: //www. cancerresearchuk. org/about-cancer/find-a-clinical-trial/what-clinical-trials-are/phases-of-clinical-trial
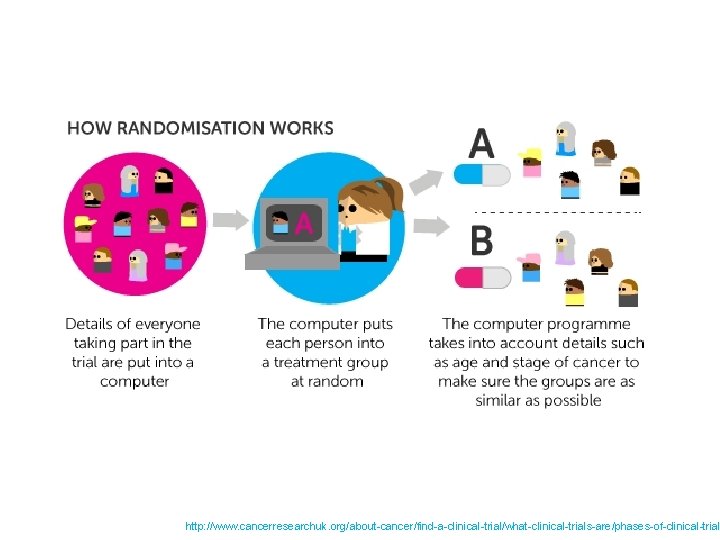
http: //www. cancerresearchuk. org/about-cancer/find-a-clinical-trial/what-clinical-trials-are/phases-of-clinical-trial

Phase 3 trials These trials compare new treatments with the best currently available treatment (the standard treatment). These trials may compare • A completely new treatment with the standard treatment • Different doses or ways of giving a standard treatment • A new way of giving radiotherapy with the standard way Phase 3 trials usually involve many more patients than phase 1 or 2. This is because differences in success rates may be small. So, the trial needs many patients to be able to show the difference. For example, if there were 50 people in the new treatment group and 50 people in the standard treatment group, there may be 3 more people in remission in the new treatment group. The 2 groups would not look that different. But if the researchers gave each treatment to 5, 000 people, there could be 300 more remissions in the new treatment group. Sometimes phase 3 trials involve thousands of patients in many different hospitals and even different countries. Most phase 3 trials are randomised. This means the researchers put the people taking part into treatment groups at random. http: //www. cancerresearchuk. org/about-cancer/find-a-clinical-trial/what-clinical-trials-are/phases-of-clinical-trial

The placebo effect Patients can also be biased. Many of us feel better if we believe we have taken something to make us feel better. Even if we've only taken a tablet made of chalk or sugar. This is called the placebo effect. Phase 3 trials could compare a new treatment with no treatment at all. But then, the people getting the new treatment might feel better, even if the new treatment didn't work. They would have the placebo effect. The people getting no treatment wouldn't feel any better, of course. So, some trials compare a new treatment with a dummy treatment called a placebo. The dummy treatment looks exactly like the new treatment – for example, the same shape or colour of pill, or size of injection. The two groups of patients then can't be biased, because they won't know if they are getting the placebo or the new treatment. A placebo is only used if there is no standard treatment available. The patients in the control group wouldn't have any treatment if they weren't in the trial. So they are not missing out on treatment they would otherwise have had. It isn't ethical to give a placebo to a group of people who really need a treatment for cancer. So the research ethics committee would not give permission for a trial designed in that way. http: //www. cancerresearchuk. org/about-cancer/find-a-clinical-trial/what-clinical-trials-are/phases-of-clinical-trial

Blind trials A blind trial is a trial where the people taking part don't know which treatment they are getting. They could be getting the new treatment. Or they could be getting standard treatment or a placebo, depending on the design of the trial. All patients receive identical injections or tablets, so they can't tell which treatment they are having. Double blind trials A double blind trial is a trial where neither the researchers nor the patients know what they are getting. The computer gives each patient a code number. And the code numbers are then allocated to the treatment groups. Your treatment arrives with your code number on it. Neither you nor your doctor knows whether it is the new treatment or not. The list of patients and their code numbers is kept secret until the end of the trial. In an emergency the researchers could find out which trial group a patient was in, but generally no one knows until the trial had finished. http: //www. cancerresearchuk. org/about-cancer/find-a-clinical-trial/what-clinical-trials-are/phases-of-clinical-trial

Phase 4 trials are done after a drug has been shown to work and has been granted a licence. Phase 4 is sometimes written as phase IV. The main reasons for running phase 4 trials are to find out § More about the side effects and safety of the drug § What the long term risks and benefits are § How well the drug works when it’s used more widely http: //www. cancerresearchuk. org/about-cancer/find-a-clinical-trial/what-clinical-trials-are/phases-of-clinical-trial
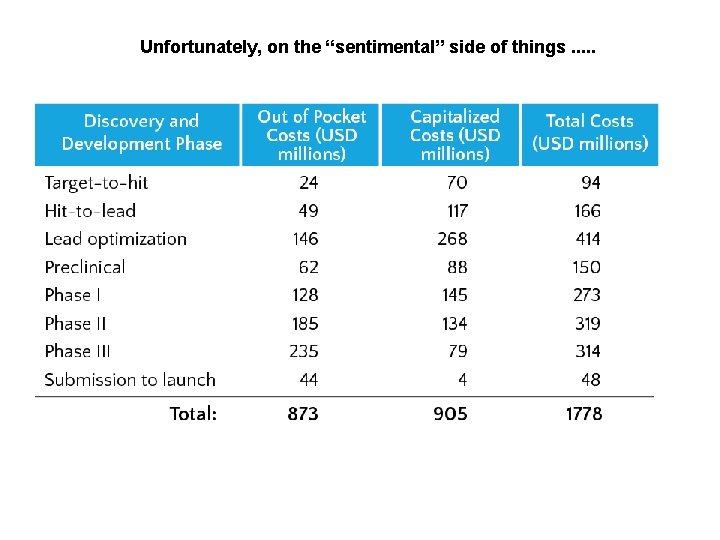
Unfortunately, on the “sentimental” side of things. . .
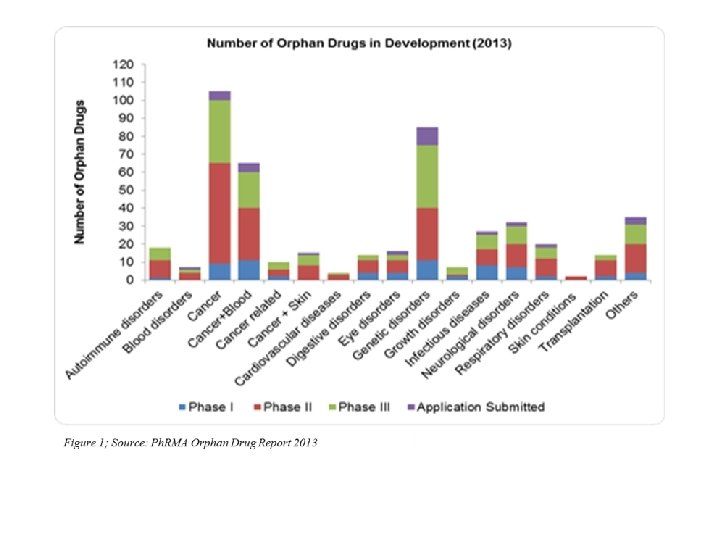
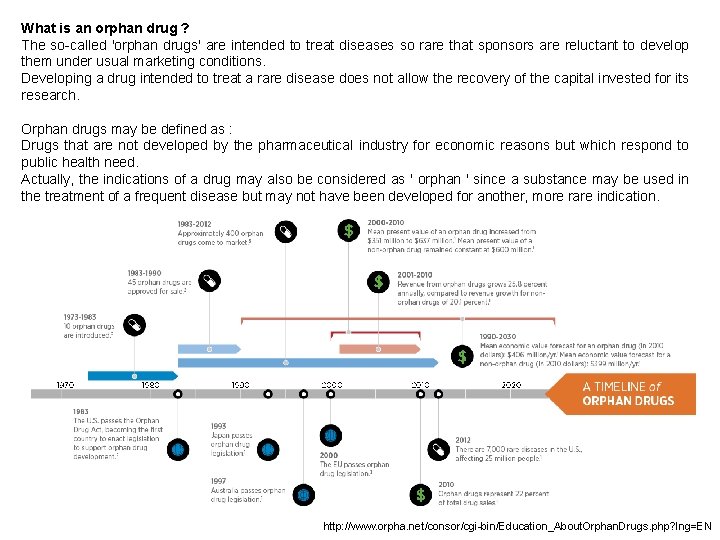
What is an orphan drug ? The so-called 'orphan drugs' are intended to treat diseases so rare that sponsors are reluctant to develop them under usual marketing conditions. Developing a drug intended to treat a rare disease does not allow the recovery of the capital invested for its research. Orphan drugs may be defined as : Drugs that are not developed by the pharmaceutical industry for economic reasons but which respond to public health need. Actually, the indications of a drug may also be considered as ' orphan ' since a substance may be used in the treatment of a frequent disease but may not have been developed for another, more rare indication. http: //www. orpha. net/consor/cgi-bin/Education_About. Orphan. Drugs. php? lng=EN
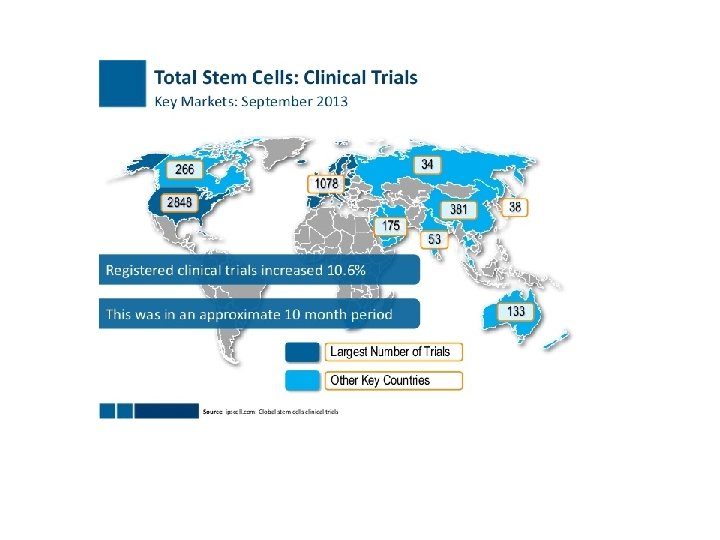
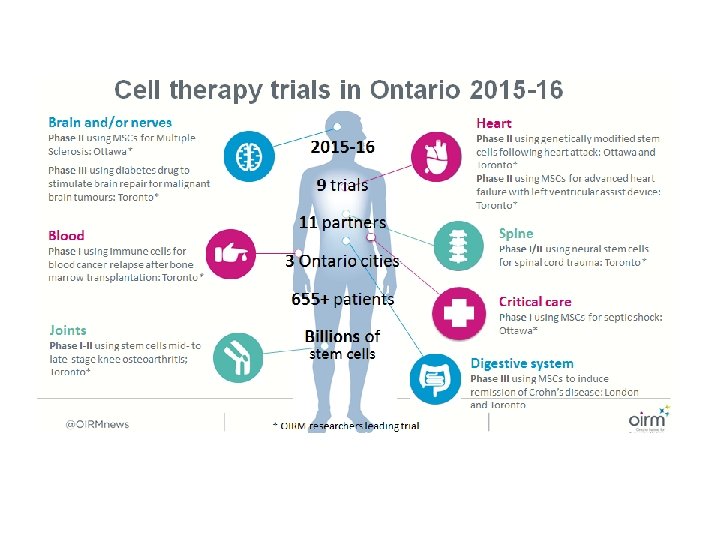
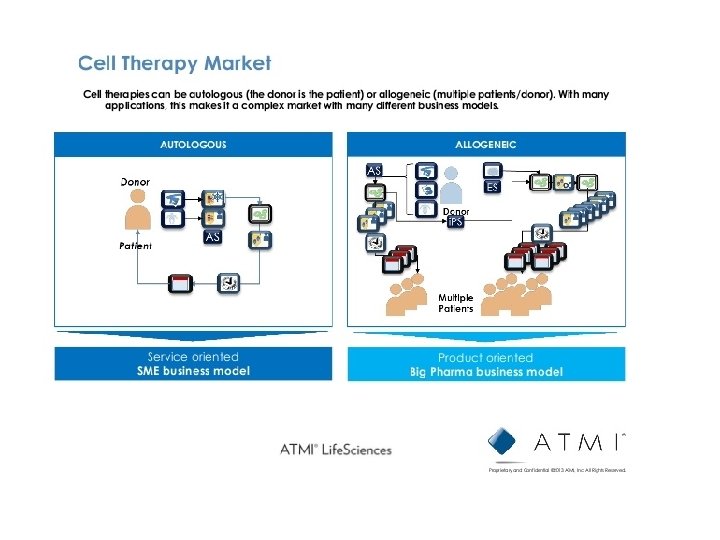
Stem cell treatment trials for ALS :

http: //www. myfoxdetroit. com/story/23283199/als-patient-walking-showingimprovement-after-stem-cell-treatment
- Slides: 67